Introduction
Colorectal cancer (CRC) is one of most common
malignant cancers and is associated with a poor prognosis and a
high mortality rate (1). Previous
studies have indicated that oncogene and tumor suppressor genes are
involved in CRC initiation and progression (2,3). However,
the molecular biological mechanism of CRC is complicated and is not
yet fully understood. A recently discovered class of
non-protein-coding small RNA, microRNA (miRNA), has extended the
understanding of the process of oncogenesis, which offers a novel
perspective to solve this problem. Over 1,000 miRNAs have been
identified in human cells and their diverse functions in normal
cell homeostasis and numerous different diseases have been
thoroughly investigated during the past decade (4–6). To date,
abnormal expression of a number of miRNAs, including miR-21
(7), miR-31 (8), and miR-137 (9), have been identified in CRC and may
contribute to the development and progression of CRC.
miRNA are a class of small molecule RNA, with a size
of ~19–22 nucleotides (10). The
miRNA maturation process is divided into 2 steps: i) The pre-miRNA
is cleaved by the RNase III enzyme Dicer-1 producing a short
doublestranded miRNA duplex, which is ii) unwound by a helicase,
creating a mature miRNA (11). In
humans, the 2 mature miRNA sequences, miR-339-5p and miR-339-3p,
are excised from opposite arms of the stem-loop pre-miR-339. The
present authors previous study indicated that miR-339-5p inhibited
tumorigenesis and metastasis of colorectal cancer through the
regulation of PRL-1 expression and activation of the ERK1/2
signalling pathway (12). However, to
the best of our knowledge, there have been no previous reports
investigating the role of miR-339-3p in CRC. Although 2 mature
miRNAs can be excised from the same stem-loop pre-miRNA, the
resulting miRNAs may have different effects. The miRNA miR-34c-3p
exhibits tumor-suppressing effects in SiHa cells through reducing
the expression of Notch pathway members, however miR-34c-5p
overexpression does not result in tumor suppressive effects in this
cell line (13). miR-28 suppresses
proliferation but activates metastasis, since the 5p and 3p forms
of miR-28 target different mRNAs. miR-28-5p altered expression of
CCND1 and HOXB3, whereas miR-28-3p bound NM23-H1 (14). The present study investigated the role
of miR-339-3p in CRC: miR-339-3p expression was detected in CRC
cells and tissue samples. Functional assays were used to analyze
the effect of miR-339-3p on cell behaviors. In addition, the
preliminary molecular mechanisms underlying the function of
miR-339-3p were investigated.
Materials and methods
Clinical specimens
A total of 30 fresh-frozen CRC tissue and matched
normal colorectal tissue samples were collected from Nanfang
Hospital of Southern Medical University (Guangzhou, China). Tumor
and non-tumor samples were confirmed by pathological examinations.
Pathological diagnoses were performed based on the system of the
International Union Against Cancer (15). The study was approved by the Ethics
Committee of Southern Medical University, and written informed
consent was obtained from all patients for the use of their
tissues. The pathological stage, grade and nodal status were
evaluated by an experienced pathologist. Clinicopathological
characteristics, including gender, age, lymph node metastasis and
differentiation were also recorded.
Cell culture and miRNA
transfection
The 293FT human embryonic kidney cell line and 6
human colorectal cancer cell lines (HCT116, HT29, LS174T, SW480,
SW620 and LOVO) with different metastatic abilities were purchased
from The American Type Culture Collection (ATCC, Manassas, VA,
USA). The cells were cultured and stored according to the ATCC's
instructions. 293FT cells were cultured in Dulbecco's modified
Eagle's medium (DMEM, Hyclone, Logan, UT, USA) supplemented with
10% fetal bovine serum (FBS, Gibco Life Technologies, Gaithersburg,
MD, USA). All CRC cells were maintained in RPMI 1640 (GE Healthcare
Life Sciences, Logan, UT, USA) supplemented with 10% FBS at a
humidity of 5% CO2 at 37°C. The miR-339-3p mimic, a
non-specific miR mimics control (miR mimics control), miR-339-3p
inhibitor (As-miR-339-3p), and a nonspecific inhibitor-negative
control (As-miR-control) were all purchased from GenePharma
(Shanghai, China). miRNAs were transfected at a working
concentration of 100 nmol/l using Lipofectamine 2000 reagent
(Invitrogen Life Technologies, Carlsbad, CA, USA), according to the
manufacturer's protocol. After 48 h, cells were harvested for
analysis of protein and RNA.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) assays
For detection of miR-339-3p expression, stem-loop
RT-qPCR was performed using an All-in-OneTM miRNA RT-qPCR Detection
Kit (GeneCopoeia, Rockville, MD, USA), as previously described.
Briefly, total RNA was extracted from cells using TRIzol reagent
(Takara Biotechnology Co., Ltd., Dalian, China) according to the
manufacturer's instructions. RT reactions were performed using 1 mg
of total RNA extracted from clinical specimens and also from cell
lines. RT-qPCR reactions were performed using the Hairpin-it TM
miRNAs qPCR kit (GeneCopoeia, Inc., Rockville, MD, USA), and U6 for
normalization to endogenous control. The thermal profile for
amplification of miR-339-3p was conducted at 95°C for 10 min,
followed by 40 cycles of 95°C for 20 sec, 60°C for 20 sec and 72°C
10 sec. For PRL-1 mRNA detection, reverse transcription was
performed using the Reverse Transcriptase System (Takara
Biotechnology Co., Ltd.). PRL-1 expression was measured by SYBR
green qPCR assay (Takara Biotechnology Co., Ltd.). The primer
sequences were as follows: PLR-1, F 5′-GACCTGGATGGGGTAAACCT-3′ and
R 5′-TGTGACTTCCACAGGAGCTG-3′; GAPDH, F 5′ACCCACTCCTCCACCTTTG-3′ and
R 5′-CACCACCCTGTTGCTGTAG-3′. The data were collected with ABI PRISM
7500 Fast Real-time PCR system (Applied Biosystems, Foster, CA,
USA). The comparative threshold cycle method was used to calculate
the amplification factor, where the threshold cycle (Ct) was
defined as the cycle number at which the fluorescence passes the
fixed threshold intensity level. The n-fold change in miR-339-3p
and PRL-1 expression was obtained using 2−ΔΔCt
method.
Cell proliferation assay
Cells were transfected with 100 nM miR-339-3p
inhibitor or mimics. 24 h later, cells were seeded in 96-well
plates (2×103 cells/well). The viability of cells was
examined by CCK8 (3-2,5-diphenyl tetrazolium bromide) assay
(Dojindo Laboratories, Kumamoto, Japan) daily for 4 days. Each well
was measured at 450 nm with a GloMaxTM96 Microplate Luminometer
(Promega Corporation, Madison, WI, USA). All experiments were
repeated 3 times.
Cell cycle analysis
A total of 1×106 cells were harvested,
washed twice with cold PBS and fixed in 70% cold ethanol. They were
then processed with a Cell Cycle Detection Kit (Nanjing KeyGen
Biotech Co., Ltd., Nanjing, China) according to the manufacturer's
instructions. The samples were analyzed by flow cytometry (BD
Biosciences, Franklin Lakes, NJ, USA).
Cell migration and invasion assay
The migration and invasion capabilities of the cells
were evaluated using Chemicon QCM 24-Well Collagen-Based Cell
Invasion assay (EMD Millipore, Billerica, MA, USA) according to the
manufacturer's instructions. For the invasion assay,
2×105 cells in serum-free medium were added into the
upper compartment of the Transwell chambers and 600 µl RPMI 1640
medium containing 10% FBS was added into the lower compartment. The
cells were incubated for 48 h at 37°C. All non-migratory cells were
then removed from the interior of the insert and the migratory
cells were stained with Wright-Giemsa (Nanjing KeyGen Biotech Co.,
Ltd.) and images were captured under the microscope (Olympus DP26;
Olympus Corporation, Tokyo, Japan) in 5 random visual fields
(magnification, ×200). For the invasion assay, the procedures were
similar, except that the cells were seeded into the top chamber,
which was pre-coated with 20% matrigel matrix (BD Biosciences, San
Jose, CA, USA).
Luciferase activity
293FT and HCT116 cells were seeded in triplicate in
24-well plates (1×105/well), respectively, and cultured
for 24 h. PRL-1 3′-UTR and mutant PRL-1 3′-UTR plasmid were
constructed in previous experiment (12). Subsequently, co-transfections of PRL-1
3′-UTR or mutant PRL-1 3′-UTR plasmid with miR-339-3p mimics into
293FT cells were accomplished using Lipofectamine 2000. The same
procedure were proceeded in HCT116 cells. Firefly and renilla
luciferase activities were measured using the Dual-LuciferaseH
Reporter Assay System (Promega Corporation) 48 h after transfection
as previously described (12).
Renilla luciferase activity was used as a transfection
normalization control.
Western blot analysis
Protein expression was assessed by immunoblot
analysis of cell lysates (20–60 µg) in RIPA buffer with 1% PMSF
(Nanjing KeyGen Biotech Co., Ltd.). Protein lysates were extracted
and separated in SDS-PAGE gels (Nanjing KeyGen Biotech Co., Ltd.),
transferred onto 0.22 µm polyvinylidene difluoride membranes (EMD
Millipore, Boston, MA, USA), and the membranes were blocked for 1 h
in Tris-buffered saline with Tween (TBST) containing 5% milk powder
(Zhongshan Golden Bridge Biotechnology Co., Ltd., Beijing, China).
The membranes were then incubated overnight at 4°C with rabbit
anti-PRL-1 monoclonal antibody (dilution, 1:800; Proteintech Group,
Inc., Chicago, IL, USA) and rabbit anti-β-tubulin antibody
(dilution, 1:2,000; Epitomics, Burlingame, CA, USA). The membranes
were washed three times with TBST and incubated with horseradish
peroxidase-conjugated goat anti-rabbit IgG secondary antibody
(dilution, 1:2,000) Zhongshan Golden Bridge Biotechnology Co.,
Ltd.) for 1 h at room temperature, prior to visualization by
enhanced chemiluminescence (Nanjing KeyGen Biotech Co., Ltd.)
according to the manufacturer's instructions. The protein
expression was normalized to β-tubulin.
Statistical analysis
The results of all the experiments are presented as
the mean ± standard deviation (SD) of ≥3 independent experiments.
All statistical analyses were performed using SPSS software,
version 13.0 (SPSS, Inc., Chicago, IL, USA) software packages. The
Shapiro-Wilk test was used to verify the clinical samples'
distribution. Differences were analyzed using the
Mann-Whitney-Wilcoxon non-parametric test. For in vitro
studies, the Student's t-test or analysis of variance were
used to compare values of test and control samples. P<0.05 was
considered to indicate a statistically significant difference.
Results
miR-339-3p is downregulated in human
colon cancer
To study the expression pattern of miR-339-3p in
CRC, RT-qPCR was used to assess miR-339-3p expression in 30 CRC
samples and the pair-matched adjacent normal colonic tissue, as
well as in 6 CRC-derived cell lines. As presented in Fig. 1A, the level of miR-339-3p expression
is modestly reduced in CRC tissues (86.7%) compared with that
observed in the adjacent normal colon mucosa tissues (P<0.05).
The association between miR-339-3p expression and
clinicopathological factors was examined in tumor tissues. The
results demonstrated that reduced levels of miR-339-3p expression
was more strongly associated with CRC samples with lymph node
metastasis compared with CRC without lymph node metastasis
(Table I, P<0.05). However,
miR-339-3p expression was not associated with the other
clinicopatholical factors assessed, including differentiation
status, gender or age. In addition, the expression of miR-339-3p
was also assessed in 6 human CRC cell lines. All 6 CRC cell lines
demonstrated a statistically significant reduction in miR-339-3p
expression, whereas the control normal colonic mucosa pooled from 3
healthy individuals (N1, N2 and N3) expressed high levels of
miR-339-3p. Notably, miR-339-3p expression was expressed at the
lowest levels in cancer cell lines SW620 and LOVO, which possess
the highest metastatic abilities compared with HCT116, HT29,
LS174T, SW480, which possess lower metastatic abilities (Fig. 1B). These results indicated that
miR-339-3p may serve a role in CRC metastasis and invasion.
 | Table I.miR-339-3p expression levels and
clinicopathological characteristics in colorectal cancer
patients. |
Table I.
miR-339-3p expression levels and
clinicopathological characteristics in colorectal cancer
patients.
| Characteristics | n | 2−ΔΔCt
(mean) | P-value |
|---|
| Differentiation |
|
| 0.204 |
| Good | 3 | 0.3287±0.0750 |
|
|
Moderate | 24 | 0.6861±0.1999 |
|
| Poor | 3 | 0.2547±0.3250 |
|
| Lymph node
status |
|
| 0.032 |
|
Negative | 18 | 0.6926±0.9219 |
|
|
Positive | 12 | 0.3591±0.4807 |
|
| Gender |
|
| 0.138 |
| Male | 18 | 0.4733±0.3651 |
|
|
Female | 12 | 0.7901±1.2156 |
|
| Age, years |
|
| 0.312 |
|
<50 | 7 | 0.8229±1.2191 |
|
| ≥50 | 23 | 0.5350±0.6731 |
|
Effect of miR-339-3p on colon cancer
cells growth, migration and invasion
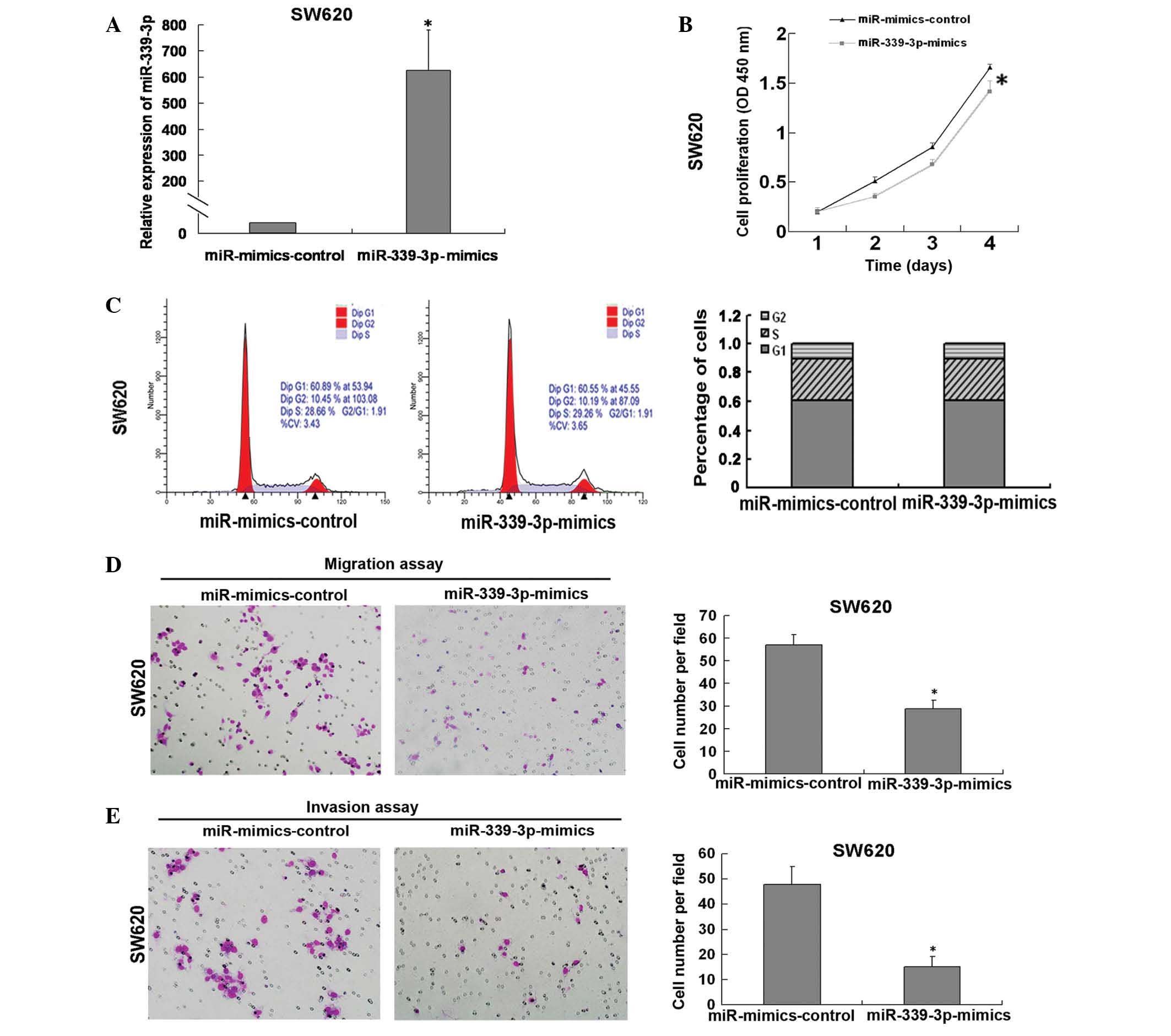
To further confirm whether miR-339-3p is involved in
regulating CRC cell growth, a proliferation assay was performed by
transfecting miR-339-3p mimics or mimics control into SW620 cells,
which were observed to express low endogenous levels of miR-339-3p
(Fig. 1B). The results of the CCK8
assay demonstrated that miR-339-3p was successfully overexpressed
in SW260 cells (Fig. 2A) and that
this resulted in inhibition of cell growth (Fig. 2B). The analysis of cell cycle
distribution demonstrated that there was no significant difference
in the percentage of cells in S and G1 phase distribution between
the miR mimics control group and miR-339-3p mimics group (Fig. 2C). To investigate the role of
miR-339-3p in the migration and invasion of tumor cells, a
Transwell assay was performed with the SW620 cells transfected with
the miR-339 mimics and control. A reduction in the migratory and
invasive activity of SW620 cells overexpressing miR-339-3p was
observed (reduced by 50.5% and 31.8%, respectively; Fig. 2D and E; both P<0.05). Inversely,
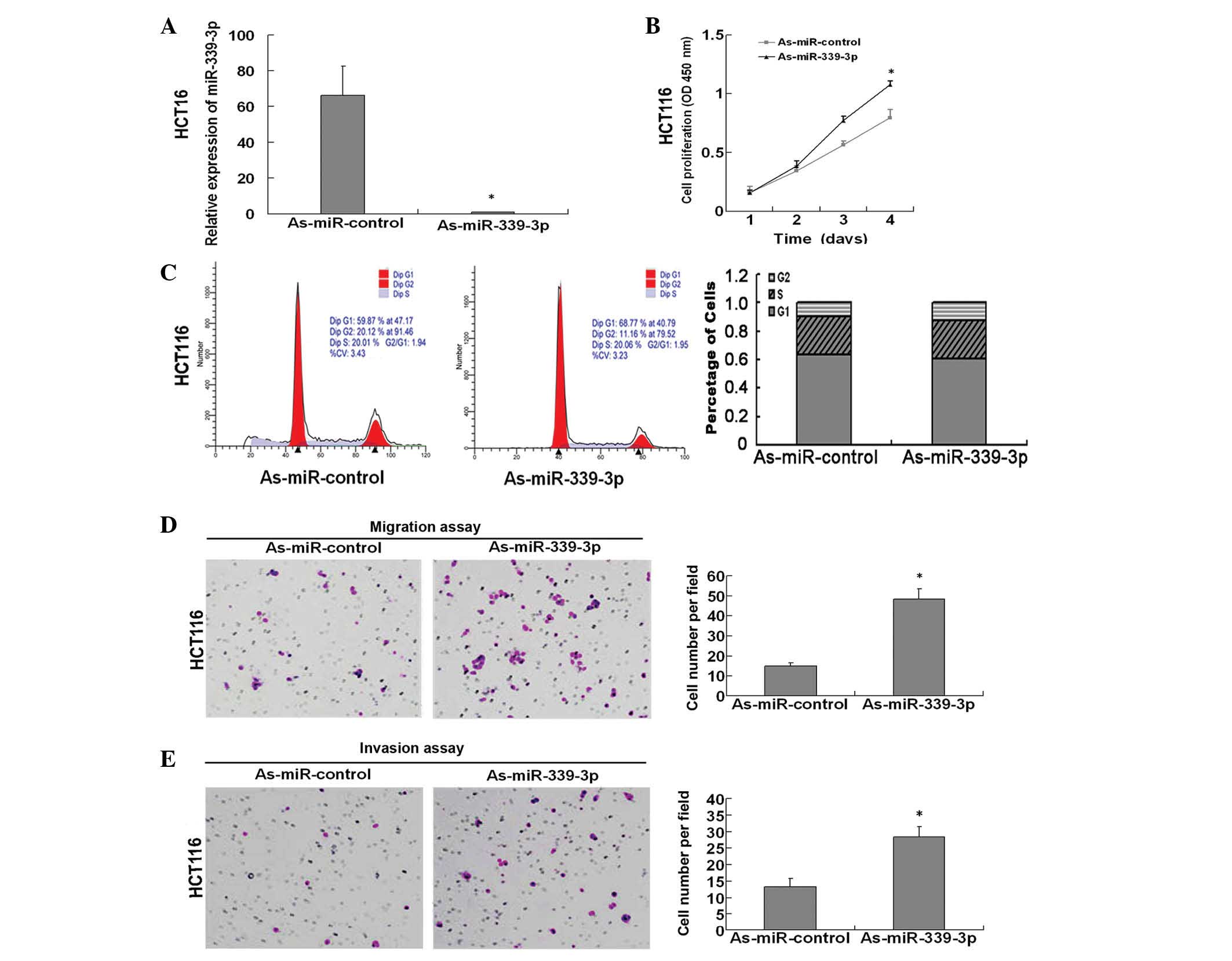
miR-339-3p was downregulated in HCT116 cells using a miR-339-3p
inhibitor (Fig. 3A), and this
resulted in increased levels of cell growth compared with the
inhibitor control (Fig. 3B). Flow
cytometry and cell cycle analysis revealed no significant changes
in the percentage of HCT116 cells treated with miR-339-3p in the G1
or S phase (Fig. 3C). In addition the
number of migratory and invasive cells transfected with the
miR-339-3p inhibitor was assessed by Transwell assay and
demonstrated that treatment with miR-339-3p inhibitor promoted
migration and invasion of the HCT116 cells by 3.23 fold and 2.15
fold, respectively (Fig. 3D and E;
both P<0.05). Taken together, these results indicated that
miR-339-3p may function as a tumor suppressor by inhibiting CRC
cells proliferation and CRC cells mobility.
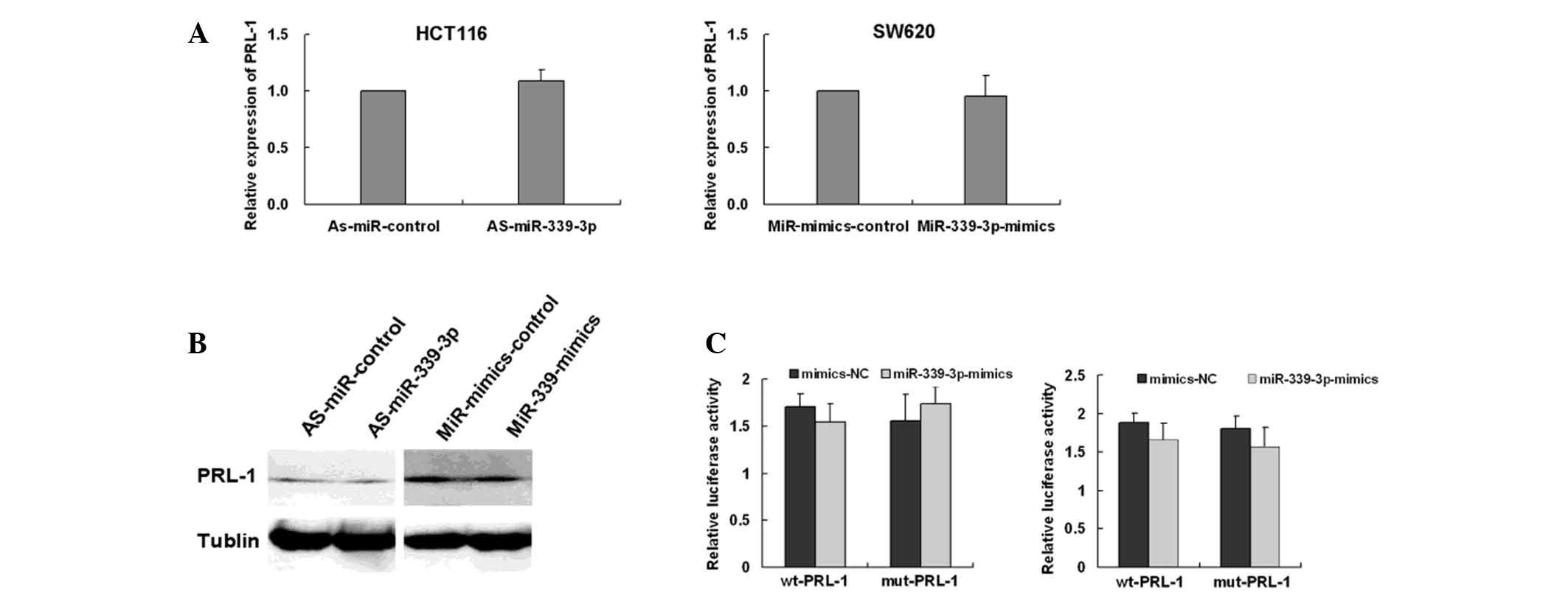
PRL-1 is not a functional target for
miR-339-3p in CRC cells
In the present authors previous study, PRL-1 was
identified as a downstream target of miR-339-5p. Since miR-5p and
miR-3p forms can target the same mRNAs (16,17), the
present study investigated whether there was an association between
miR-339-3p and PRL-1 expression. To examine this hypothesis,
miR-339-3p mimics were transfected into SW620 cells and HCT116
cells were treated with an miR-339-3p inhibitor. The mRNA and
protein expression levels were examined by RT-qPCR and western blot
analysis. The results demonstrated that miR-339-3p had no effect on
PRL-1 mRNA and protein expression in SW620 or HCT116 cells
(Fig. 4A and B; both P>0.05). To
determine whether miR-339-3p directly interacts with the 3′-UTR
region of PRL-1 luciferase reporter assays were performed.
miR-339-3p mimics and luciferase reporter constructs containing
wild type or mutant PRL-1 3′-UTR were co-transfected into 293FT
cells. The same procedure was used for HCT116 cells. Luciferase
activity of the reporter containing PRL-1 3′-UTR in 293FT and
HCT116 cells was not affected in the presence of miR-339-3p when
compared with its negative miRNA control. In addition, miR-339-3p
did not alter the activity of the mutant PRL-1 luciferase reporter
in 293FT and HCT116 cells (Fig. 4C).
These results indicated that miR-339-3p did not directly bind to
the 3′-UTR of PRL-1 under these conditions.
Discussion
Expression patterns of miRNAs are systematically
altered in a number of types of human cancer (18,19).
Previous studies have demonstrated that a number of mature miRNA
serve essential roles in cellular transformation and tumorigenesis,
angiogenesis, invasion, and metastasis (20,21).
Previous work on miR-339-5p reported that miR-339-5p may be
abnormally downregulated in colon cancer. The present authors
previous study indicated that miR-339-5p inhibits CRC cell
proliferation and migration by regulation of PRL-1 expression and
the ERK1/2 signaling pathway. Two mature miRNAs may be generated
from the same stem-loop pre-miR-339 and generate 2 different
miRNAs, miR-339-5p and miR-339-3p. Although miR-339-3p is generated
from the same primary transcript as miR-339-5p, it has a different
sequence and its expression, the modulation of its expression
profiles and the exact functional mechanism of miR-339-3p is
unknown in CRC cells.
In the present study, the potential role of
miR-339-3p was investigated in CRC cel lines and patient samples.
The results demonstrated that the downregulation of miR-339-3p was
apparent in CRC tissue and cell lines, indicating a tumor
suppressive role for miR-339-3p in CRC development. Furthermore,
the potential association between the expression level of
miR-339-3p and various clinicopathological characteristics of CRC
patients were investigated: Low miR-339-3p expression levels were
demonstrated to be significantly associated with cases with lymph
node metastases; however, reduced miR-339-3p expression was not
associated with age, gender or the differentiation status of the
tumor. In addition, to the best of our knowledge, no evidence of
the influence of miR-339-3p expression on the behavior of CRC cells
has been documented. The gain- and loss-of-function assays in the
present study demonstrated that miR-339-3p overexpression inhibited
cell proliferation and migration in vitro. However,
miR-339-3p overexpression did not affect the cell-cycle
distribution of CRC cells. miR-339-3p expression may inhibit the
proliferation of CRC cells through other ways, such as apoptosis,
but this was not examined in the present study. The results
indicated miR-339-3p serves a suppressive role in CRC
progression.
miRNAs generally exert their biological function by
suppressing their specific target genes. A single miRNA may
regulate a number of different targets. The present authors
previous study previous studies demonstrated that the suppressive
effects of miR-339-5p on CRC cells may be partially due to its
regulation of PRL-1 expression and activation of the ERK1/2
signaling pathway. Previous studies have revealed that both strands
of miR-5p and miR-3p can target the same mRNAs (16,17). As
the exact functional mechanism of miR-339-3p remains unclear, the
present study also investigated whether PRL-1 is the target of
miR-339-3p. miR-339-3p overexpression in SW620 cells and knockdown
of miR-339-3p in HCT116 cells had no effect on PRL-1 levels
respectively, at both the mRNA and protein levels. In addition,
mRNA expression of miR-339-3p and PRL-1 were not demonstrated to be
significant in CRC cells. It was also demonstrated that miR-339-3p
did not bind directly to 3′-UTR of PRL-1. Therefore, these results
indicate that miR-339-3p may function as a tumor suppressor by
regulating a gene other than PRL-1 to inhibit the progression of
CRC.
The results of the present study indicated that
miR-339-3p may participate in the modulation of malignant
biological behavior in CRC. However, PRL-1 is not regulated by
miR-339-3p in the CRC pathological processes and future experiments
should aim to identify the target gene of miR-339-3p in CRC.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant nos. 1302158 and
30871156) and the Medical Research Fund of Guangdong Province
(grant no. B2014095) and Traditional Chinese Medicine Research of
Guangdong Province (grant no. 20141166).
References
|
1
|
Brenner H, Kloor M and Pox CP: Colorectal
cancer. Lancet. 383:1490–1502. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jin R, Liu W, Menezes S, Yue F, Zheng M,
Kovacevic Z and Richardson DR: The metastasis suppressor NDRG1
modulates the phosphorylation and nuclear translocation of
β-catenin through mechanisms involving FRAT1 and PAK4. J Cell Sci.
127:3116–3130. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Miyoshi N, Yamamoto H, Mimori K, Yamashita
S, Miyazaki S, Nakagawa S, Ishii H, Noura S, Ohue M, Yano M, et al:
ANXA9 gene expression in colorectal cancer: A novel marker for
prognosis. Oncol Lett. 8:2313–2317. 2014.PubMed/NCBI
|
|
4
|
Persson H, Kvist A, Rego N, Staaf J,
Vallon-Christersson J, Luts L, Loman N, Jonsson G, Naya H, Hoglund
M, et al: Identification of new microRNAs in paired normal and
tumor breast tissue suggests a dual role for the ERBB2/Her2 gene.
Cancer Res. 71:78–86. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sasahira T, Kurihara M, Bhawal UK, Ueda N,
Shimomoto T, Yamamoto K, Kirita T and Kuniyasu H: Downregulation of
miR-126 induces angiogenesis and lymphangiogenesis by activation of
VEGF-A in oral cancer. Br J Cancer. 107:700–706. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ueno K, Hirata H, Shahryari V, Deng G,
Tanaka Y, Tabatabai ZL, Hinoda Y and Dahiya R: MicroRNA-183 is an
oncogene targeting Dkk-3 and SMAD4 in prostate cancer. Br J Cancer.
108:1659–1667. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Ferraro A, Kontos CK, Boni T, Bantounas I,
Siakouli D, Kosmidou V, Vlassi M, Spyridakis Y, Tsipras I, Zografos
G and Pintzas A: Epigenetic regulation of miR-21 in colorectal
cancer: ITGB4 as a novel miR-21 target and a three-gene network
(miR-21-ITGβ4-PDCD4) as predictor of metastatic tumor potential.
Epigenetics. 9:129–141. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun D, Yu F, Ma Y, Zhao R, Chen X, Zhu J,
Zhang CY, Chen J and Zhang J: MicroRNA-31 activates the RAS pathway
and functions as an oncogenic MicroRNA in human colorectal cancer
by repressing RAS p21 GTPase activating protein 1 (RASA1). J Biol
Chem. 288:9508–9518. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Chen DL, Wang DS, Wu WJ, Zeng ZL, Luo HY,
Qiu MZ, Ren C, Zhang DS, Wang ZQ, Wang FH, et al: Overexpression of
paxillin induced by miR-137 suppression promotes tumor progression
and metastasis in colorectal cancer. Carcinogenesis. 34:803–811.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Loedige I, Filipowicz W and Krol J: The
widespread regulation of microRNA biogenesis, function and decay.
Nat Rev Genet. 11:597–610. 2010.PubMed/NCBI
|
|
11
|
Miska EA: How microRNAs control cell
division, differentiation and death. Curr Opin Genet Dev.
15:563–568. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Zhou C, Liu G, Wang L, Lu Y, Yuan L, Zheng
L, Chen F, Peng F and Li X: MiR-339-5p regulates the growth, colony
formation and metastasis of colorectal cancer cells by targeting
PRL-1. PLoS One. 8:e631422013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
López JA and Alvarez-Salas LM:
Differential effects of miR-34c-3p and miR-34c-5p on SiHa cells
proliferation apoptosis, migration and invasion. Biochem Biophys
Res Commun. 409:513–519. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Almeida MI, Nicoloso MS, Zeng L, Ivan C,
Spizzo R, Gafà R, Xiao L, Zhang X, Vannini I, Fanini F, et al:
Strand-specific miR-28-5p and miR-28-3p have distinct effects in
colorectal cancer cells. Gastroenterology. 142:886–896. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Nishino M, Jagannathan JP, Ramaiya NH and
Van den Abbeele AD: Revised RECIST guideline version 1.1: What
oncologists want to know and what radiologists need to know. AJR Am
J Roentgenol. 195:281–289. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Uchino K, Takeshita F, Takahashi RU,
Kosaka N, Fujiwara K, Naruoka H, Sonoke S, Yano J, Sasaki H, Nozawa
S, et al: Therapeutic effects of microRNA-582-5p and −3p on the
inhibition of bladder cancer progression. Mol Ther. 21:610–619.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sakurai K, Furukawa C, Haraguchi T, Inada
K, Shiogama K, Tagawa T, Fujita S, Ueno Y, Ogata A, Ito M, et al:
MicroRNAs miR-199a-5p and −3p target the Brm subunit of SWI/SNF to
generate a double-negative feedback loop in a variety of human
cancers. Cancer Res. 71:1680–1689. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lu YX, Yuan L, Xue XL, Zhou M, Liu Y,
Zhang C, Li JP, Zheng L, Hong M and Li XN: Regulation of colorectal
carcinoma stemness, growth and metastasis by an
miR-200c-Sox2-negative feedback loop mechanism. Clin Cancer Res.
20:2631–2642. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Xu L, Li Y, Yan D, He J and Liu D:
MicroRNA-183 inhibits gastric cancer proliferation and invasion via
directly targeting Bmi-1. Oncol Lett. 8:2345–2351. 2014.PubMed/NCBI
|
|
20
|
Zhang N, Wang X, Huo Q, Sun M, Cai C, Liu
Z, Hu G and Yang Q: MicroRNA-30a suppresses breast tumor growth and
metastasis by targeting metadherin. Oncogene. 33:3119–3128. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Li J, Kong F, Wu K, Song K, He J and Sun
W: MiR-193b directly targets STMN1 and uPA genes and suppresses
tumor growth and metastasis in pancreatic cancer. Mol Med Rep.
10:2613–2620. 2014.PubMed/NCBI
|