Introduction
Glioma is the most common aggressive adult primary
tumor of the central nervous system (1). The mortality rate associated with glioma
occupies the top position among the malignant tumors worldwide
(2). During the early stages (I and
II) of disease, when the tumor is small, patients with glioma are
usually asymptomatic, whereas grade III and IV gliomas, including
glioblastoma, are aggressive and lethal malignant neoplasms
(3). Glioma, in particular
glioblastoma multiforme, is the most common malignant brain tumor
in adults (4). The median age at
diagnosis of glioblastoma patients is 65 years (5). Current treatment options include
surgical resection, radiotherapy and chemotherapy (6). However, glioma carries a particularly
poor prognosis, with survival measured in months rather than years
(7).
The treatment approaches for malignant glioma, which
is the most common type of highly aggressive primary brain tumor,
are often unsuccessful due to diffuse infiltration and poor
prognosis (8). A key problem
regarding glioma treatment is the lack of effective tumor
site-specific delivery systems available for therapeutic agents
(9). Bone marrow-derived mesenchymal
stem cells (BMSCs) have been shown to exhibit tropism for gliomas
(10). Furthermore, these cells may
be obtained easily, and may be genetically engineered and
autologously transplanted, thus providing a feasible delivery
vehicle for glioma-targeted therapy (11–17).
Previous in vivo studies have demonstrated the efficacy of
this delivery system (12,18). A number of cytokines, including
platelet-derived growth factor-BB (PDGF-BB), have been shown to
affect the migration of BMSCs (12,19–22);
however, the mechanism underlying this remains to be
elucidated.
It has been established that site-directed migration
involves interaction between multiple adhesion molecules on
migrating cells and their corresponding ligands (23,24). The
cell adhesion molecule cluster of differentiation (CD)44, which is
a BMSCs-specific transmembrane glycoprotein, is known to be
involved in intracellular interactions that affect the motility of
BMSCs (25–27). T cells migrating to inflammatory sites
express higher levels of CD44 on their cell surface, and thus are
capable of establishing more CD44-hyaluronan (HA) interactions
(28,29). Therefore, CD44 may exert certain
effects on the chemotactic migration of BMSCs to glioma cells. In
the current study, we evaluated the role of CD44 in the tropism of
BMSCs for glioma cells.
Materials and methods
Cell culture
Rat glioma C6 cells were obtained from the Key
Laboratory of Cancer Prevention and Therapy (Tianjin, China) and
cultured in serum-free low glucose-Dulbecco's modified Eagle's
medium (L-DMEM; Invitrogen; Thermo Fisher Scientific, Inc.,
Waltham, MA, USA) at 37°C in a humidified atmosphere of 5%
CO2. Cell culture plates, including 6-well plates,
24-well plates and 60-mm dishes were purchased from Nest
Biotechnology Co., Ltd. (Wuxi, China).
Ethical statement
All animal experiments were approved by the Animal
Care and Use Committee of Tianjin Medical University Cancer
Institute and Hospital (Tianjin, China), and were performed in
accordance with the National Institute of Health Guide for the Care
and Use of Laboratory Animals (30).
A total of 20 Wistar rats were purchased from Vital River
Laboratories (Beijing, China). They were housed under the specific
conditions and sacrificed immediately by cervical dislocation as
described previously by Yang et al (31).
BMSCs isolation
The rats were housed in the animal center of Tianjin
Medical University Cancer Institute and Hospital at a temperature
of 20–25 °C and relative humidity of 50–70% on a 12-h dark/light
cycle and provided a standard pelleted diet and water ad libitum.
Male rats of 4 weeks old were used, and they were individually
sacrificed by cervical dislocation. Four-week-old male Wistar rats
were used for BMSCs isolation based on the principle of their
adherence to plastic (32). Briefly,
bone marrow cells collected from the bilateral tibias and femurs of
sacrificed rats were cultured in L-DMEM supplemented with 10% fetal
bovine serum (Invitrogen; Thermo Fisher Scientific, Inc.). Three
days later, adherent cells were passaged to fresh medium to discard
non-adherent cells, and were subsequently grown to full confluence.
Next, 6,000 cells/cm2 cells were subcultured and grown to full
confluence again prior to subculturing. Cells at fourth passage
were identified as BMSCs, and used for the following experiments,
as previously described (33).
Immunocytochemistry
BMSCs were collected and seeded onto 1.5%
gelatin-coated coverslips. At 80% confluence, the C6 cells seeded
on sterilized glass slides were allowed to attach overnight.
Following fixation with 4% paraformaldehyde (Sigma-Aldrich, St.
Louis, MO, USA) for 1 h at 4°C, cells were washed with
phosphate-buffered saline (PBS; Sigma-Aldrich) three times for 20
min each, prior to incubation with PBS for 60 min at 4°C. Fixed
cells were incubated with rabbit polyclonal anti-human anti-PDGF-BB
antibody (dilution, 1:100; catalog no., ab23914; Abcam, Cambridge,
MA, USA) at 4°C overnight, followed by incubation with goat
anti-rabbit immunoglobulin G, horseradish peroxidase-conjugated
secondary antibody (dilution, 1:1,000; catalog no. 7074; Cell
Signaling Technology, Danvers, MA, USA) for 45 min at room
temperature. Next, the membranes were stained with
3,3′-diaminobenzidine (Sigma-Aldrich) and hematoxylin
(Sigma-Aldrich), and slides were mounted with 50% glycerol
(Sinopharm Chemical Reagent Co., Ltd., Shanghai, China) prior to
capturing images with a microscope (Eclipse ME600; Nikon Corp.,
Tokyo, Japan).
Immunofluorescence
BMSCs incubated in PDGF-BB-supplemented
C6-conditioned medium for 12 h were fixed in 3.7% paraformaldehyde
and permeabilized in pre-chilled acetone (Sinopharm Chemical
Reagent Co., Ltd.). BMSCs incubated with serum-free L-DMEM were
used as a negative control. Upon blocking with 5% bovine serum
albumin (Sigma-Aldrich) in PBS for 1 h, the cells were incubated
with polyclonal rabbit anti-human/mouse/rat CD44 antibody
(dilution, 1:100; catalog no., PA1021-2; Wuhan Boster Biological
Technology, Ltd., Wuhan, China) for 4 h at room temperature,
followed by incubation with rhodamine-conjugated goat anti-mouse
immunoglobulin G secondary antibody (dilution, 1:100; catalog no.,
ZF-0313; Zhongshan Golden Bridge Biotechnology Co., Ltd, Beijing,
China) for 1 h at room temperature. Images were captured using a
laser confocal microscope (TCS SP5; Leica Microsystems, Inc.,
Buffalo Grove, IL, USA).
Reverse transcription-polymerase chain
reaction (RT-PCR)
RT-PCR was performed to examine the transcriptional
levels of PDGF-BB in C6 cells and CD44 in PDGF-BB-treated BMSCs
using a 2400 GeneAmp® PCR system (Applied Biosystems; Thermo Fisher
Scientific, Inc.). BMSCs incubated with serum-free L-DMEM served as
a negative control. Total RNA was extracted from cells using TRIzol
reagent (Invitrogen; Thermo Fisher Scientific, Inc.), and cDNA was
obtained from 1 µg RNA using the ImProm-II™ Reverse Transcription
System (Promega Corporation, Madison, WI, USA) according to the
manufacturer's instructions. The primers used for PCR, synthesized
by Sangon Biotech Co., Ltd. (Shanghai, China), were as follows:
Sense, 5′-CTTTAAGAAGGCCACGGTGA-3′ and anti-sense,
5′-TCCAAGGGTCTCCTTCAGTG-3′ for PDGF-BB; sense,
5′-AAGACATCGATGCCTCAAAC-3′ and anti-sense,
5′-CTCCAGTAGGCTGTGAAGTG-3′ for CD44 (34); and sense, 5′-TATCCAGGCTGTGCTATCCC-3′
and anti-sense, 5′-CCATCTCTTGCTCGAAGTCC-3′ for β-actin. PCR was
performed under the following conditions for PDGF-BB: Denaturation
at 94°C for 46 min, followed by 40 cycles of 94°C for 15 sec, 62°C
for 1 min and 72°C for 1 min, with a final extension step at 72°C
for 7 min; PCR was performed under the following conditions for
CD44: Denaturation for 95°C for 15 min, followed by 45 cycles of
94°C for 15 sec, 55°C for 30 sec and 72°C for 30 sec. The PCR
products were separated using gel electrophoresis on a 2% agarose
gel (Sigma-Aldrich). The bands were scanned using ChemiImager 5500
version 2.03 software (Alpha Innotech, San Leandro, CA, USA).
Integrated density values were calculated using a computerized
image analysis system (Fluor Chen 2.0; Bio-Rad, Hercules, CA, USA)
and normalized to β-actin. Agarose gel, which was prepared in 1×TAE
buffer containing 40 mM Tris-acetic acid (pH 8.5; Tris-base was
purchased from Sigma Aldrich; acetic acid was from Sinopharm
Chemical Reagent Co., Ltd.) and 2 mM ethylenediaminetetraacetic
acid (Sinopharm Chemical Reagent Co., Ltd.), was supplemented with
0.5 μg/mL ethidium bromide (Sigma-Aldrich). Wide Mini-Sub Cell GT
Horizontal Electrophoresis System and PowerPac™ Universal Power
Supply (Bio-Rad Laboratories, Inc., Hercules, CA, USA) were applied
for gel electrophoresis, with voltage and time set at 100 V and 20
min, respectively. DNA fragments were visualized and quantified
using ChemiDoc MP system (Bio-Rad Laboratories, Inc.), and relative
amounts of CD44 transcripts were determined against β-actin
expression.
In vitro migration assay
The culture medium for rat glioma C6 cells was
collected following 24-h incubation. Upon centrifugation at 1,000 ×
g for 15 min at room temperature, and subsequent sterilization by
0.22-mm filtration (Thermo Fisher Scientific, Inc.), the
supernatant was identified as C6 cell-conditioned medium. For the
migration assay, BMSCs at a density of 2×105 cells/ml were seeded
in the upper chamber of a Transwell plate containing an 8-µm pore
membrane (Costar; Corning Incorporated, Corning, NY, USA), and C6
cell-conditioned medium in the presence or absence of recombinant
rat PDGF-BB (catalogue no. 220-BB-010; R&D Systems, Inc.,
Minneapolis, MN, USA) and serum-free L-DMEM containing 10, 20 or 40
µg/l PDGF-BB was added to the lower well of the Transwell plates.
Serum-free L-DMEM served as a negative control. Cells were
incubated for 24 h prior to formalin fixation and hematoxylin
staining. Images of nine randomly selected fields were captured,
and cells were counted.
To block CD44 activity, C6-conditioned medium in the
presence or absence of PDGF-BB (40 ng/ml) was incubated with mouse
monoclonal anti-rat CD44 neutralizing antibody (dilution, 1:1,000;
catalog no., OX-50; Abcam) for 3 h at room temperature, prior to
being added to the lower chamber of the Transwell plates.
Serum-free L-DMEM served as a negative control. The subsequent
procedures were performed as described above. Briefly, BMSCs were
seeded in the upper chamber, followed by an incubation of 24 h at
37°C. Migrated cells were stained prior to counting. An inverted
microscope (Zeiss Axio Vert A1 Inverted, Carl Zeiss Canada Ltd.,
North York, ON, Canada) equipped with a charge-coupled device
camera (Orca ER; Hamamatsu Photonics K.K., Hamamatsu, Japan) was
used to visualize and image stained cells, at x400
magnification.
Statistical analysis
All data were analyzed using SPSS 13.0 statistical
software (SPSS Inc., Chicago, IL, USA). Two-tailed unpaired
Student's t-test was used to determine the significance of
differences between groups. P<0.05 was considered to indicate a
statistically significant difference. All experiments were
performed at least twice, and results were expressed as the mean ±
standard deviation.
Results
Rat glioma C6 cells express
PDGF-BB
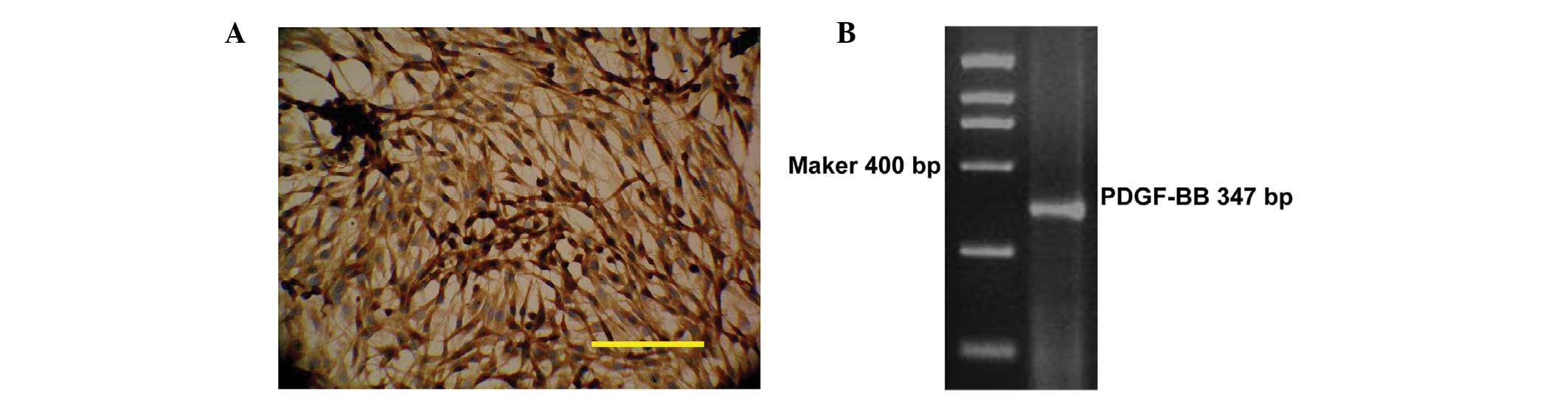
The expression levels of PDGF-BB in rat glioma C6
cells were analyzed. As shown in Fig.
1A, PDGF-BB protein was highly expressed in the cytoplasm of C6
cells (Fig. 1A). In addition, a clear
cDNA band corresponding to PDGF-BB was identified in C6 cells using
RT-PCR (Fig. 1B).
C6 cells induce chemotactic migration
of BMSCs via expression and secretion of PDGF-BB
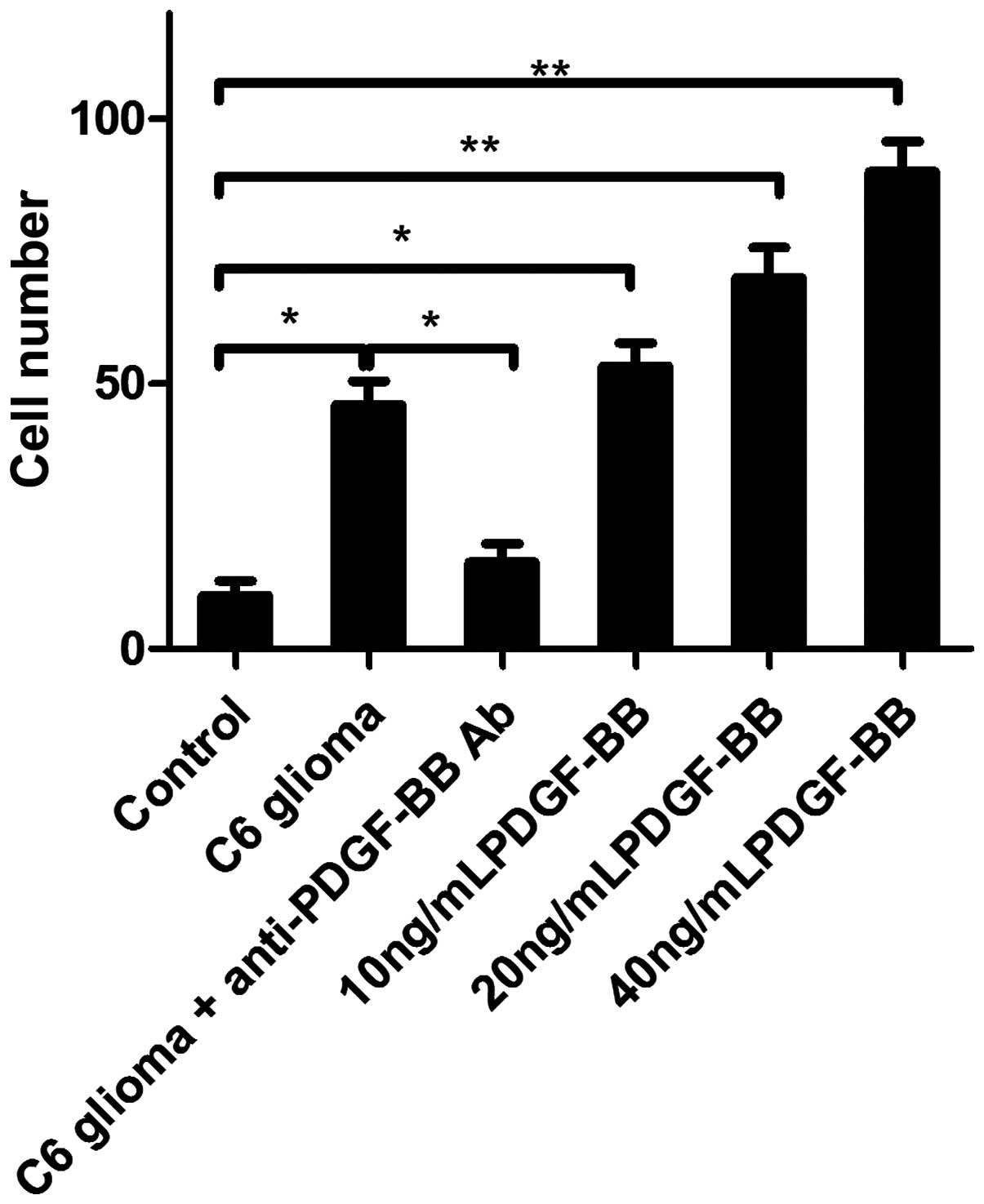
To evaluate the effect of PDGF-BB on tropism of
BMSCs toward glioma, an in vitro migration assay was
performed. As shown in Fig. 2,
increased levels of migration of BMSCs were observed in the C6
cell-conditioned medium-treated group after 24 h treatment compared
with the normal medium-treated group, which was attenuated by 4-h
pretreatment with anti-PDGF-BB antibody, indicating that C6
cell-induced chemostatic migration of BMSCs may occur as a result
of PDGF-BB secretion in the C6 cell-conditioned medium.
Additionally, supplementing C6 cell-conditioned medium with
recombinant rat PDGF-BB enhanced C6 cells-induced chemostatic
migration of BMSCs in a dose-dependent manner (Fig. 2), thus demonstrating that PDGF-BB
promotes the tropism of BMSCs.
PDGF-BB upregulates the expression of
the standard form of CD44
CD44, as a marker for BMSCs, has been reported to be
involved in the mobilization and chemostatic migration of BMSCs
(35). To evaluate the effect of
PDGF-BB on CD44 expression in BMSCs, RT-PCR and immunofluorescence
assays were performed. As shown in Fig.
3, the transcriptional and protein levels of CD44 in BMSCs were
increased in the C6 cell-conditioned medium-treated group, and
PDGF-BB augmented this effect, indicating that PDGF-BB promotes the
chemostatic migration of BMSCs toward glioma via the upregulation
of CD44 expression in BMSCs.
CD44 mediates the tropism of BMSCs for
glioma
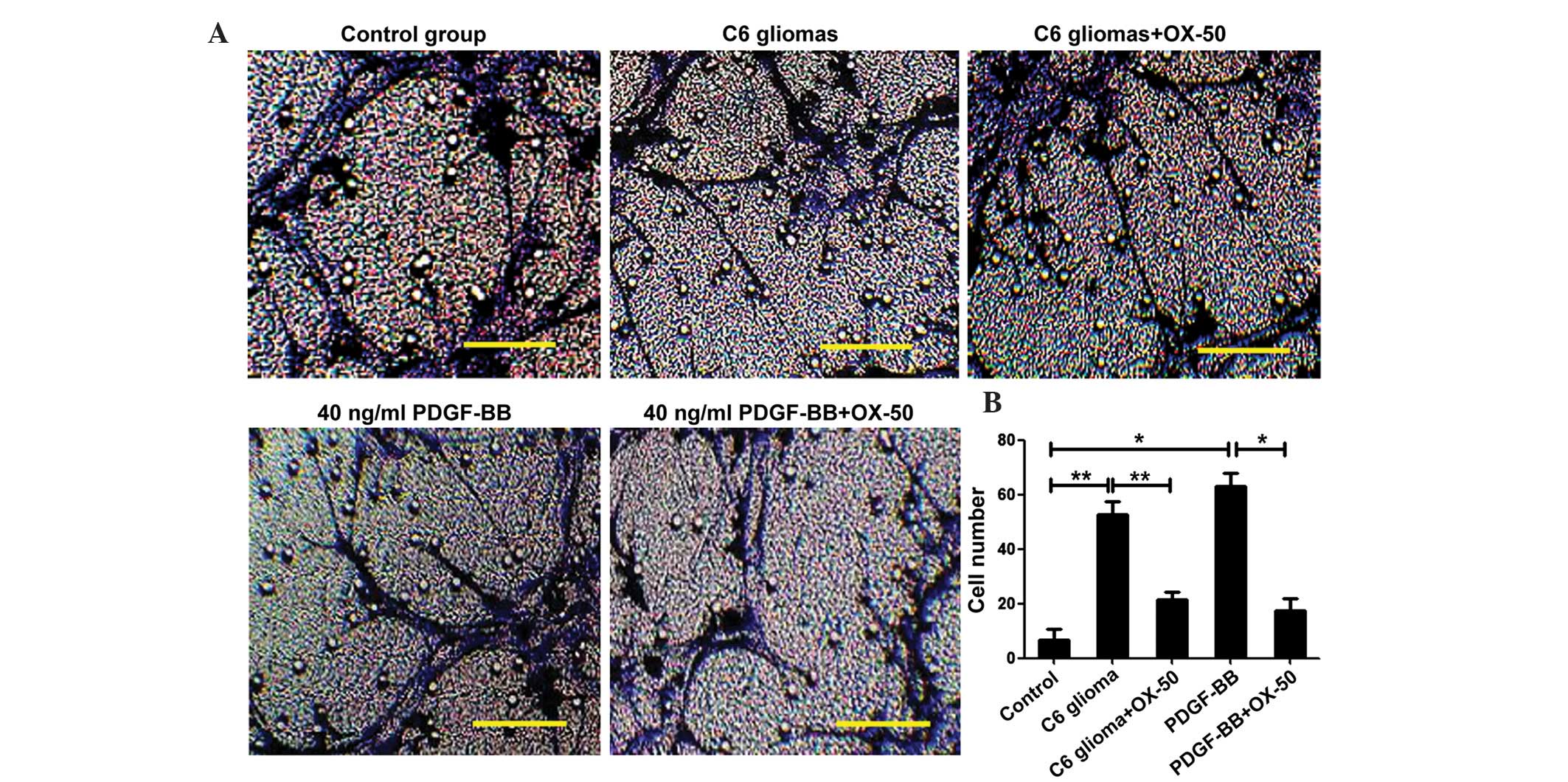
OX-50, an anti-CD44 neutralizing antibody, was used
to assess the role of CD44 in the tropism of BMSCs. As shown in
Fig. 4, pretreatment of C6
cell-conditioned medium with the anti-CD44 antibody OX-50 for 3 h
blocked the C6 cell-induced and the PDGF-BB-promoted chemostatic
migration of BMSCs, suggesting that CD44 may act as a molecular
bridge between BMSCs and glioma.
Discussion
PDGF is a strong mitogen and chemoattractant for
fibroblasts, myofibroblasts and smooth muscle cells (36,37).
PDGF-BB, a member of the PDGF family, has been demonstrated to
induce chemotactic migration of cells of mesenchymal origin
(38). A number of glioma cells
express and secrete PDGF, with high-grade gliomas expressing higher
levels of PDGF compared with low-grade gliomas (34). In the present study, rat glioma C6
cells expressed high levels of PDGF-BB, and PDGF-BB augmented the
chemostatic migration of BMSCs induced by C6 cell-conditioned
medium, indicating that PDGF-BB may mediate glioma-induced tropism
of BMSCs. However, further studies are required to corroborate
these findings.
CD44, as a unique surface antigen of BMSCs (25,26,33,39),
is involved in various cellular processes, including proliferation,
differentiation, survival and migration (40). The main function of CD44 is to
regulate the motility and chemotaxis of BMSCs (41). Previous studies have demonstrated that
CD44 is localized on the leading edge of migrating cells (42,43), and
its inhibition attenuates macrophage chemotaxis (44) and fusion (45). Additionally, loss of CD44 decreases
the migratory ability of human colon cancer cells, while
overexpression of CD44 promotes their migration (46), indicating the importance of CD44 in
such processes. The major isoform of CD44 present in MSCs is the
standard form, termed CD44s. In the present study, PDGF-BB was
observed to increase the transcriptional and protein levels of CD44
in BMSCs. In addition, C6 cell-induced and PDGF-BB-promoted
chemostatic migration of BMSCs was markedly attenuated by the
anti-CD44 neutralizing antibody OX-50, suggesting that C6 cells may
induce BMSCs tropism via the expression and secretion of PDGF-BB,
which upregulates CD44 expression in BMSCs. The CD44-HA interaction
presents a critical step required for cell migration (35), and has been reported to be involved in
the migration of CD34+ stem cells to the bone marrow, as
well as in the adhesion, motility and invasion of breast cancer
cells (47,48). However, these mechanisms require
further investigation.
In conclusion, the results of the current study
revealed that CD44 mediates the tropism of BMSCs to glioma, and
PDGF-BB promotes the migration of BMSCs toward glioma via
upregulation of CD44 expression in BMSCs. These findings suggest
CD44 inhibition may be a potential therapeutic target for the
treatment of glioma.
Acknowledgements
The present study was supported by the Doctoral
Initial Funding of the National Clinical Research Center for
Cancer, Tianjin Medical University Cancer Institute and Hospital
(Tianjin, China; grant. no. B1318) and the Young Program of Natural
Science Funding of Tianjin (grant no., 15JCQNJC44800).
References
|
1
|
Barbarin A, Seite P, Godet J, Bensalma S,
Muller JM and Chadeneau C: Atypical nuclear localization of VIP
receptors in glioma cell lines and patients. Biochem Biophys Res
Commun. 454:524–530. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Malone HR and Bruce JN: Editorial: laser
interstitial thermal therapy: an effective treatment for focally
recurrent high grade glioma. Neurosurg Focus. 37:E22014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wolking S, Lerche H and Dihne M: Episodic
itch in a case of spinal glioma. BMC Neurol. 13:1242013. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ostrom QT, Gittleman H, Farah P, Ondracek
A, Chen Y, Wolinsky Y, Stroup NE, Kruchko C and Barnholtz-Sloan JS:
CBTRUS statistical report: Primary brain and central nervous system
tumors diagnosed in the United States in 2006–2010. Neuro Oncol.
15(Suppl 2): ii1–ii56. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chakrabarti I, Cockburn M, Cozen W, Wang
YP and Preston-Martin S: A population-based description of
glioblastoma multiforme in Los Angeles County, 1974–1999. Cancer.
104:2798–2806. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Woehrer A, Bauchet L and Barnholtz-Sloan
JS: Glioblastoma survival: Has it improved? Evidence from
population-based studies. Curr Opin Neurol. 27:666–674.
2014.PubMed/NCBI
|
|
7
|
Yabroff KR, Harlan L, Zeruto C, Abrams J
and Mann B: Patterns of care and survival for patients with
glioblastoma multiforme diagnosed during 2006. Neuro Oncol.
14:351–359. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Stupp R, Mason WP, van den Bent MJ, Weller
M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn
U, et al: European Organisation for Research and Treatment of
Cancer Brain Tumor and Radiotherapy Groups; National Cancer
Institute of Canada Clinical Trials Group: Radiotherapy plus
concomitant and adjuvant temozolomide for glioblastoma. N Engl J
Med. 352:987–996. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ho IA, Toh HC, Ng WH, Teo YL, Guo CM, Hui
KM and Lam PY: Human bone marrow-derived mesenchymal stem cells
suppress human glioma growth through inhibition of angiogenesis.
Stem Cells. 31:146–155. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Hu Y, Cheng P, Xue YX and Liu YH: Glioma
cells promote the expression of vascular cell adhesion molecule-1
on bone marrow-derived mesenchymal stem cells: A possible mechanism
for their tropism toward gliomas. J Mol Neurosci. 48:127–135. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Nakamura K, Ito Y, Kawano Y, Kurozumi K,
Kobune M, Tsuda H, Bizen A, Honmou O, Niitsu Y and Hamada H:
Antitumor effect of genetically engineered mesenchymal stem cells
in a rat glioma model. Gene Ther. 11:1155–1164. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Nakamizo A, Marini F, Amano T, Khan A,
Studeny M, Gumin J, Chen J, Hentschel S, Vecil G, Dembinski J, et
al: Human bone marrow-derived mesenchymal stem cells in the
treatment of gliomas. Cancer Res. 65:3307–3318. 2005.PubMed/NCBI
|
|
13
|
Bang OY, Lee JS, Lee PH and Lee G:
Autologous mesenchymal stem cell transplantation in stroke
patients. Ann Neurol. 57:874–882. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Karussis D, Kassis I, Kurkalli BG and
Slavin S: Immunomodulation and neuroprotection with mesenchymal
bone marrow stem cells (MSCs): A proposed treatment for multiple
sclerosis and other neuroimmunological/neurodegenerative diseases.
J Neurol Sci. 265:131–135. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Liu H, Honmou O, Harada K, Nakamura K,
Houkin K, Hamada H and Kocsis JD: Neuroprotection by PlGF
gene-modified human mesenchymal stem cells after cerebral
ischaemia. Brain. 129:2734–2745. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Caplan AI and Bruder SP: Mesenchymal stem
cells: Building blocks for molecular medicine in the 21st century.
Trends Mol Med. 7:259–264. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Colter DC, Class R, DiGirolamo CM and
Prockop DJ: Rapid expansion of recycling stem cells in cultures of
plastic-adherent cells from human bone marrow. Proc Natl Acad Sci
USA. 97:3213–3218. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu X, Hu J, Zhou L, Mao Y, Yang B, Gao L,
Xie R, Xu F, Zhang D, Liu J and Zhu J: In vivo tracking of
superparamagnetic iron oxide nanoparticle-labeled mesenchymal stem
cell tropism to malignant gliomas using magnetic resonance imaging.
Laboratory investigation. J Neurosurg. 108:320–329. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Schichor C, Birnbaum T, Etminan N, Schnell
O, Grau S, Miebach S, Aboody K, Padovan C, Straube A, Tonn JC and
Goldbrunner R: Vascular endothelial growth factor A contributes to
glioma-induced migration of human marrow stromal cells (hMSC). Exp
Neurol. 199:301–310. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cheng P, Gao ZQ, Liu YH and Xue YX:
Platelet-derived growth factor BB promotes the migration of bone
marrow-derived mesenchymal stem cells towards C6 glioma and
up-regulates the expression of intracellular adhesion molecule-1.
Neurosci Lett. 451:52–56. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Hata N, Shinojima N, Gumin J, Yong R,
Marini F, Andreeff M and Lang FF: Platelet-derived growth factor BB
mediates the tropism of human mesenchymal stem cells for malignant
gliomas. Neurosurgery. 66:144–157. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Ozaki Y, Nishimura M, Sekiya K, Suehiro F,
Kanawa M, Nikawa H, Hamada T and Kato Y: Comprehensive analysis of
chemotactic factors for bone marrow mesenchymal stem cells. Stem
Cells Dev. 16:119–129. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Vicente-Manzanares M and Horwitz AR: Cell
migration: An overview. Methods Mol Biol. 769:1–24. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Misra S, Heldin P, Hascall VC, Karamanos
NK, Skandalis SS, Markwald RR and Ghatak S: Hyaluronan-CD44
interactions as potential targets for cancer therapy. FEBS J.
278:1429–1443. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Lisignoli G, Cristino S, Piacentini A,
Cavallo C, Caplan AI and Facchini A: Hyaluronan-based polymer
scaffold modulates the expression of inflammatory and degradative
factors in mesenchymal stem cells: Involvement of Cd44 and Cd54. J
Cell Physiol. 207:364–373. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Schweizer PA, Krause U, Becker R,
Seckinger A, Bauer A, Hardt C, Eckstein V, Ho AD, Koenen M, Katus
HA and Zehelein J: Atrial-radiofrequency catheter ablation mediated
targeting of mesenchymal stromal cells. Stem Cells. 25:1546–1551.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mylona E, Jones KA, Mills ST and Pavlath
GK: CD44 regulates myoblast migration and differentiation. J Cell
Physiol. 209:314–321. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
DeGrendele HC, Kosfiszer M, Estess P and
Siegelman MH: CD44 activation and associated primary adhesion is
inducible via T cell receptor stimulation. J Immunol.
159:2549–2553. 1997.PubMed/NCBI
|
|
29
|
Mohamadzadeh M, DeGrendele H, Arizpe H,
Estess P and Siegelman M: Proinflammatory stimuli regulate
endothelial hyaluronan expression and CD44/HA-dependent primary
adhesion. J Clin Invest. 101:97–108. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
National Research Council of the National
Academies: Guide for the care and use of laboratory animals (8th).
National Academies Press. USA: 112011.
|
|
31
|
Yang C, Zhou L, Gao X, Chen B, Tu J, Sun
H, Liu X, He J, Liu J and Yuan Q: Neuroprotective effects of bone
marrow stem cells overexpressing glial cell line-derived
neurotrophic factor on rats with intracerebral hemorrhage and
neurons exposed to hypoxia/reoxygenation. Neurosurgery. 68:691–704.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Geng J, Peng F, Xiong F, Shang Y, Zhao C,
Li W and Zhang C: Inhibition of myostatin promotes myogenic
differentiation of rat bone marrow-derived mesenchymal stromal
cells. Cytotherapy. 11:849–863. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Conget PA and Minguell JJ: Phenotypical
and functional properties of human bone marrow mesenchymal
progenitor cells. J Cell Physiol. 181:67–73. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hermanson M, Funa K, Hartman M,
Claesson-Welsh L, Heldin CH, Westermark B and Nistér M:
Platelet-derived growth factor and its receptors in human glioma
tissue: Expression of messenger RNA and protein suggests the
presence of autocrine and paracrine loops. Cancer Res.
52:3213–3219. 1992.PubMed/NCBI
|
|
35
|
Zhu H, Mitsuhashi N, Klein A, Barsky LW,
Weinberg K, Barr ML, Demetriou A and Wu GD: The role of the
hyaluronan receptor CD44 in mesenchymal stem cell migration in the
extracellular matrix. Stem Cells. 24:928–935. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Soma Y, Takehara K and Ishibashi Y:
Alteration of the chemotactic response of human skin fibroblasts to
PDGF by growth factors. Exp Cell Res. 212:274–277. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Trojanowska M: Role of PDGF in fibrotic
diseases and systemic sclerosis. Rheumatology (Oxford). 47(Suppl
5): v2–v4. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Rönnstrand L and Heldin CH: Mechanisms of
platelet-derived growth factor-induced chemotaxis. Int J Cancer.
91:757–762. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Stamenkovic I, Aruffo A, Amiot M and Seed
B: The hematopoietic and epithelial forms of CD44 are distinct
polypeptides with different adhesion potentials for
hyaluronate-bearing cells. EMBO J. 10:343–348. 1991.PubMed/NCBI
|
|
40
|
Naor D, Nedvetzki S, Golan I, Melnik L and
Faitelson Y: CD44 in cancer. Crit Rev Clin Lab Sci. 39:527–579.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Fanning A, Volkov Y, Freeley M, Kelleher D
and Long A: CD44 cross-linking induces protein kinase C-regulated
migration of human T lymphocytes. Int Immunol. 17:449–458. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Legg JW, Lewis CA, Parsons M, Ng T and
Isacke CM: A novel PKC-regulated mechanism controls CD44 ezrin
association and directional cell motility. Nat Cell Biol.
4:399–407. 2002. View
Article : Google Scholar : PubMed/NCBI
|
|
43
|
Thorne RF, Legg JW and Isacke CM: The role
of the CD44 transmembrane and cytoplasmic domains in co-ordinating
adhesive and signalling events. J Cell Sci. 117:373–380. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhu B, Suzuki K, Goldberg HA, Rittling SR,
Denhardt DT, McCulloch CA and Sodek J: Osteopontin modulates
CD44-dependent chemotaxis of peritoneal macrophages through
G-protein-coupled receptors: Evidence of a role for an
intracellular form of osteopontin. J Cell Physiol. 198:155–167.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Sterling H, Saginario C and Vignery A:
CD44 occupancy prevents macrophage multinucleation. J Cell Biol.
143:837–847. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Subramaniam V, Vincent IR, Gardner H, Chan
E, Dhamko H and Jothy S: CD44 regulates cell migration in human
colon cancer cells via Lyn kinase and AKT phosphorylation. Exp Mol
Pathol. 83:207–215. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Avigdor A, Goichberg P, Shivtiel S, Dar A,
Peled A, Samira S, Kollet O, Hershkoviz R, Alon R, Hardan I, et al:
CD44 and hyaluronic acid cooperate with SDF-1 in the trafficking of
human CD34+ stem/progenitor cells to bone marrow. Blood.
103:2981–2989. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Afify A, Purnell P and Nguyen L: Role of
CD44s and CD44v6 on human breast cancer cell adhesion, migration
and invasion. Exp Mol Pathol. 86:95–100. 2009. View Article : Google Scholar : PubMed/NCBI
|