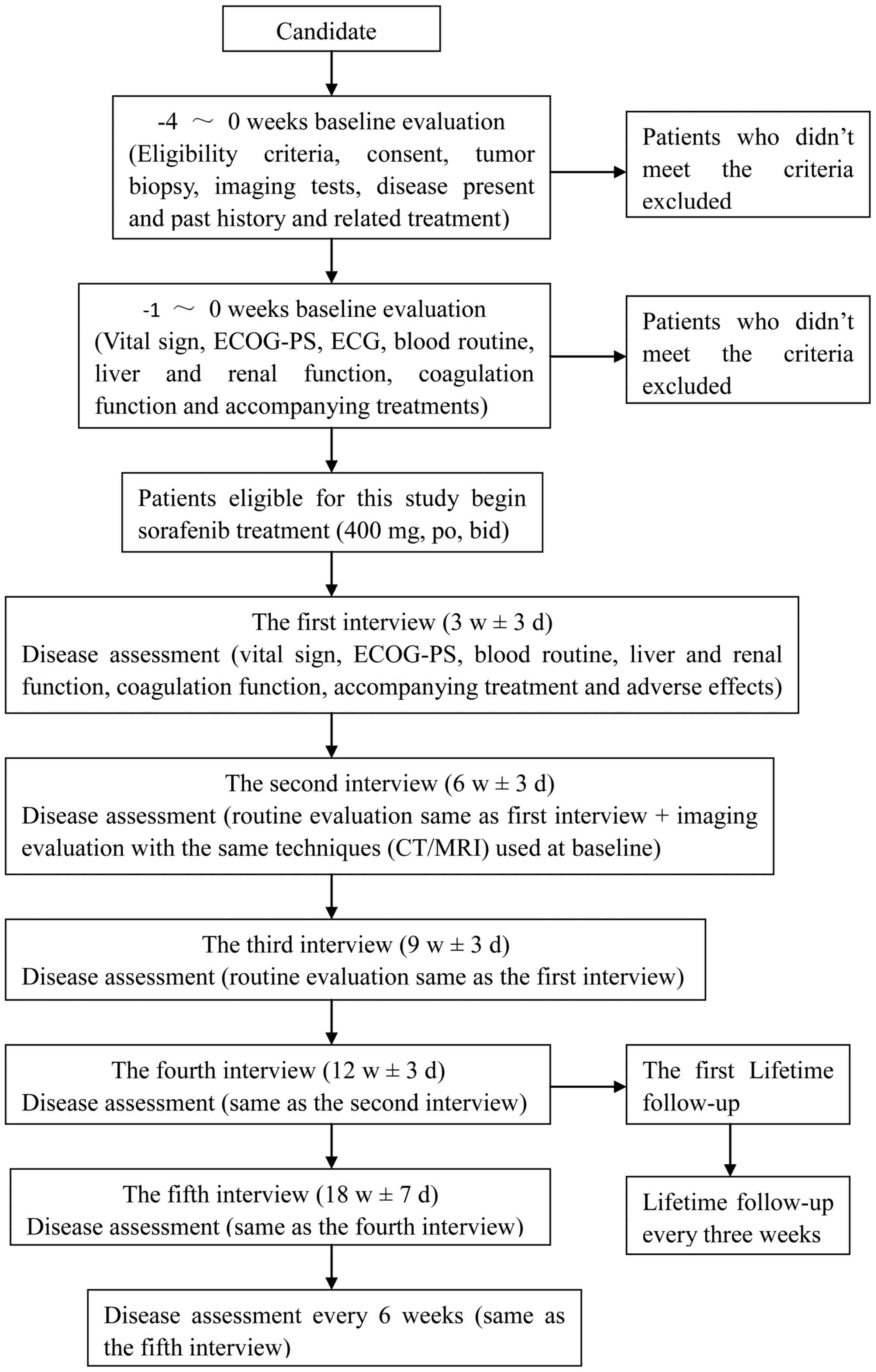
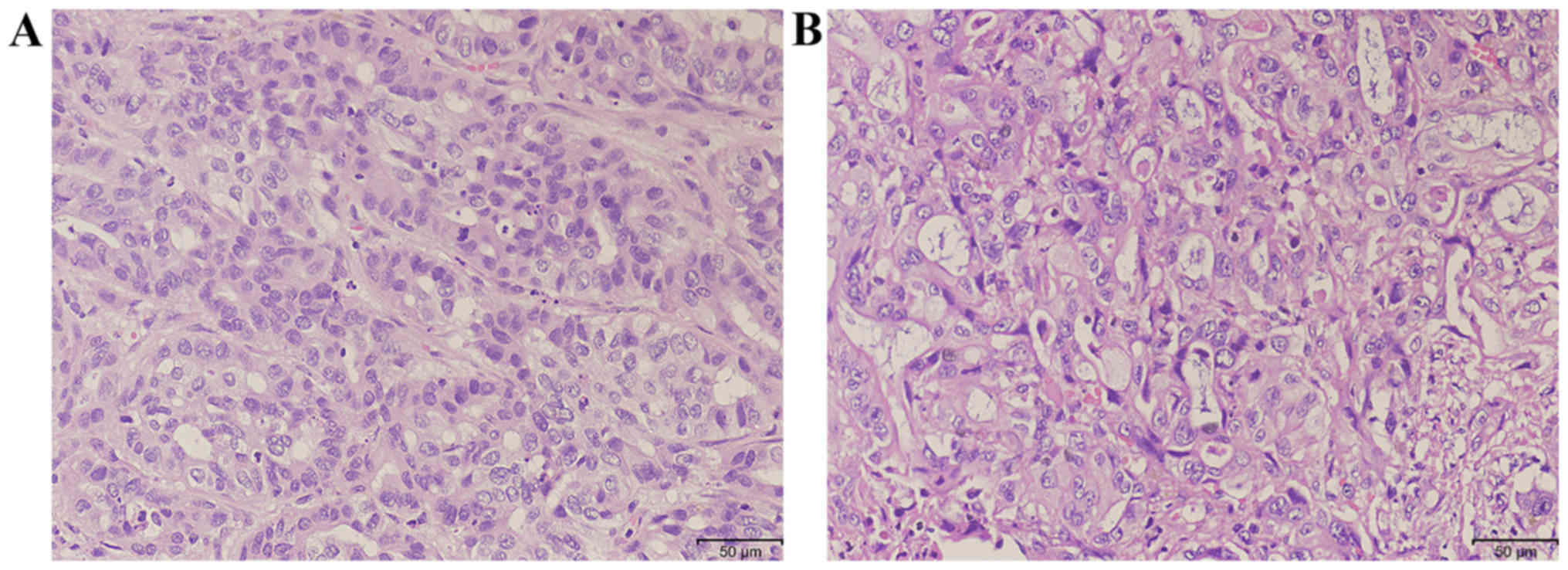
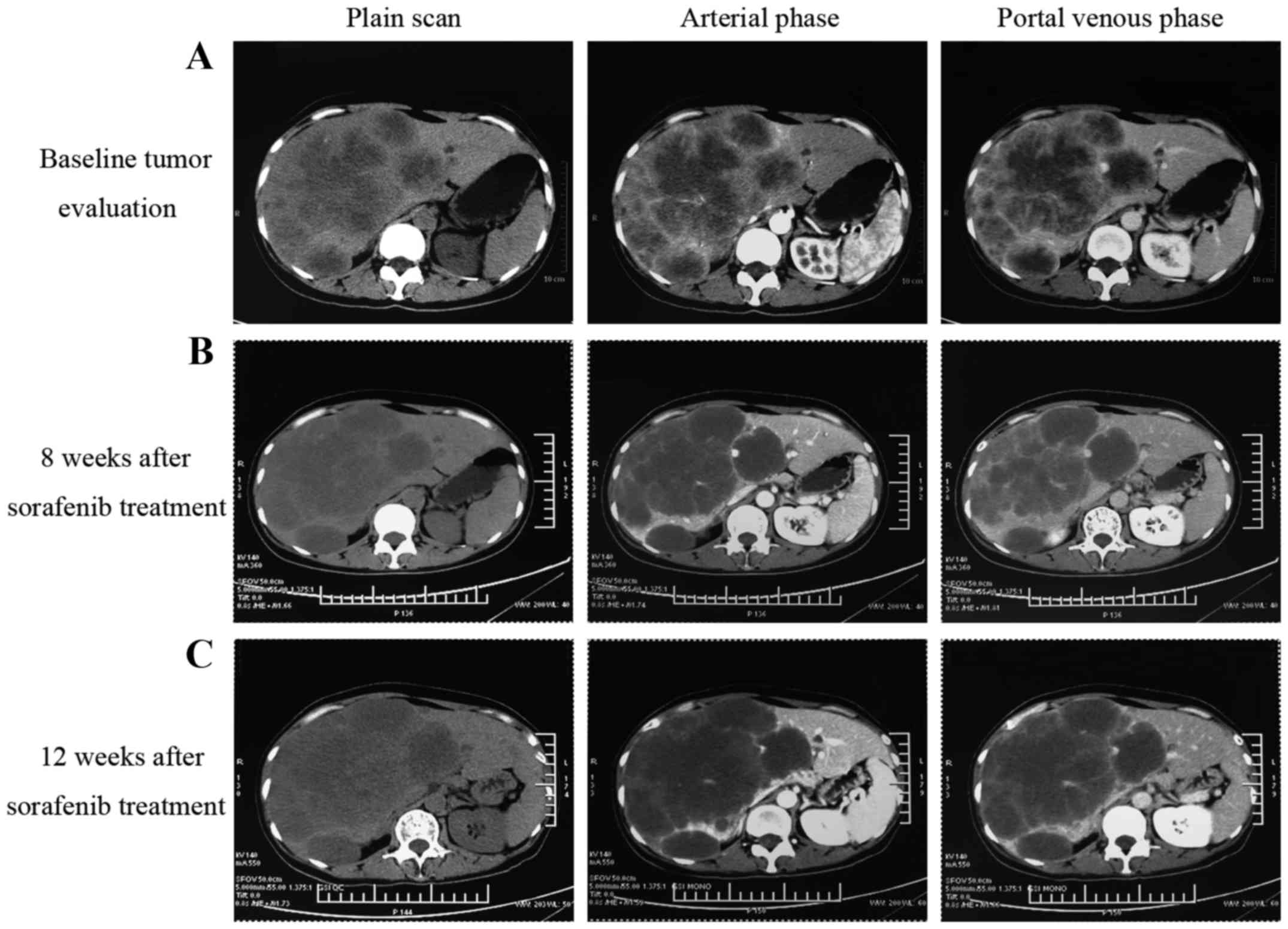
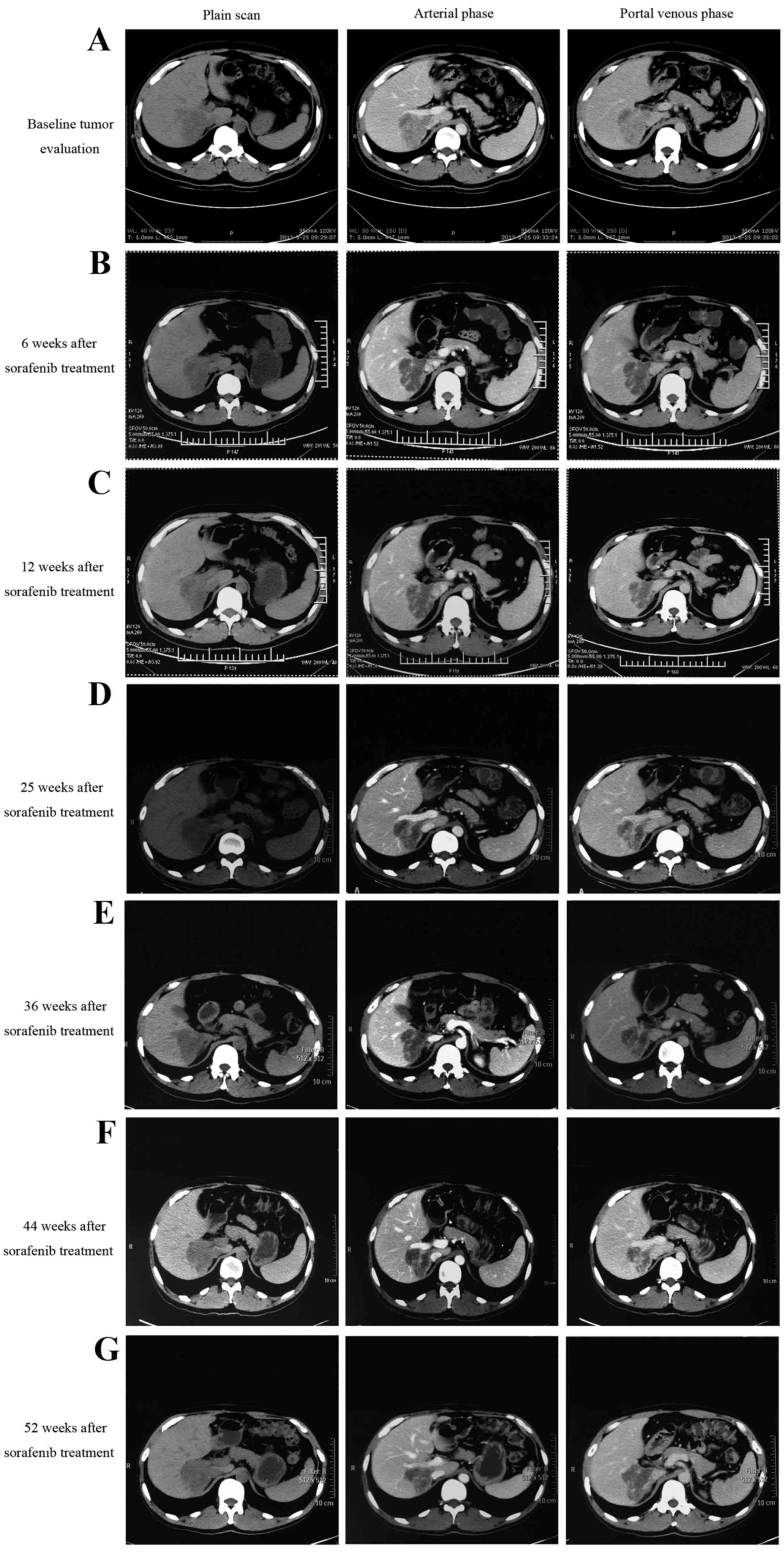
|
1
|
Casavilla FA, Marsh JW, Iwatsuki S, Todo
S, Lee RG, Madariaga JR, Pinna A, Dvorchik I, Fung JJ and Starzl
TE: Hepatic resection and transplantation for peripheral
cholangiocarcinoma. J Am Coll Surg. 185:429–436. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Morise Z, Sugioka A, Tokoro T, Tanahashi
Y, Okabe Y, Kagawa T and Takeura C: Surgery and chemotherapy for
intrahepatic cholangiocarcinoma. World J Hepatol. 2:58–64.
2010.PubMed/NCBI
|
|
4
|
Tan JC, Coburn NG, Baxter NN, Kiss A and
Law CH: Surgical management of intrahepatic cholangiocarcinoma-a
population-based study. Ann Surg Oncol. 15:600–608. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Paik KY, Jung JC, Heo JS, Choi SH, Choi DW
and Kim YI: What prognostic factors are important for resected
intrahepatic cholangiocarcinoma? J Gastroenterol Hepatol.
23:766–770. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Skipworth JR, Keane MG and Pereira SP:
Update on the management of cholangiocarcinoma. Dig Dis.
32:570–578. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Okusaka T, Ishii H, Funakoshi A, Yamao K,
Ohkawa S, Saito S, Saito H and Tsuyuguchi T: Phase II study of
single-agent gemcitabine in patients with advanced biliary tract
cancer. Cancer Chemother Pharmacol. 57:647–653. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Penz M, Kornek GV, Raderer M, Ulrich-Pur
H, Fiebiger W, Lenauer A, Depisch D, Krauss G, Schneeweiss B and
Scheithauer W: Phase II trial of two-weekly gemcitabine in patients
with advanced biliary tract cancer. Ann Oncol. 12:183–186. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hezel AF and Zhu AX: Systemic therapy for
biliary tract cancers. Oncologist. 13:415–423. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ducreux M, Van Cutsem E, Van Laethem JL,
Gress TM, Jeziorski K, Rougier P, Wagener T, Anak O, Baron B and
Nordlinger B: EORTC Gastro Intestinal Tract Cancer Group: A
randomised phase II trial of weekly high-dose 5-fluorouracil with
and without folinic acid and cisplatin in patients with advanced
biliary tract carcinoma: Results of the 40955 EORTC trial. Eur J
Cancer. 41:398–403. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Eckel F and Schmid RM: Chemotherapy in
advanced biliary tract carcinoma: A pooled analysis of clinical
trials. Br J Cancer. 96:896–902. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Chen JS, Lin YC, Jan YY and Liau CT:
Mitomycin C with weekly 24-h infusion of high-dose 5-fluorouracil
and leucovorin in patients with biliary tract and periampullar
carcinomas. Anticancer Drugs. 12:339–343. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Valle J, Wasan H, Palmer DH, Cunningham D,
Anthoney A, Maraveyas A, Madhusudan S, Iveson T, Hughes S, Pereira
SP, et al: Cisplatin plus gemcitabine versus gemcitabine for
biliary tract cancer. N Engl J Med. 362:1273–1281. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Hsu C, Shen YC, Yang CH, Yeh KH, Lu YS,
Hsu CH, Liu HT, Li CC, Chen JS, Wu CY and Cheng AL: Weekly
gemcitabine plus 24-h infusion of high-dose
5-fluorouracil/leucovorin for locally advanced or metastatic
carcinoma of the biliary tract. Br J Cancer. 90:1715–1719.
2004.PubMed/NCBI
|
|
15
|
Wilhelm SM, Carter C, Tang L, Wilkie D,
McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M, et al:
BAY 43–9006 exhibits broad spectrum oral antitumor activity and
targets the RAF/MEK/ERK pathway and receptor tyrosine kinases
involved in tumor progression and angiogenesis. Cancer Res.
64:7099–7109. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Di Marco V, De Vita F, Koskinas J, Semela
D, Toniutto P and Verslype C: Sorafenib: From literature to
clinical practice. Ann Oncol. 24:(Suppl 2). ii30–ii37. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Basch E, Reeve BB, Mitchell SA, Clauser
SB, Minasian LM, Dueck AC, Mendoza TR, Hay J, Atkinson TM,
Abernethy AP, et al: Development of the national cancer institute's
patient-reported outcomes version of the common terminology
criteria for adverse events (PRO-CTCAE). J Natl Cancer Inst.
106:pii: dju244. 2014. View Article : Google Scholar
|
|
19
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Patel T: Increasing incidence and
mortality of primary intrahepatic cholangiocarcinoma in the United
States. Hepatology. 33:1353–1357. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Altekruse SF, McGlynn KA and Reichman ME:
Hepatocellular carcinoma incidence, mortality, and survival trends
in the United States from 1975 to 2005. J Clin Oncol. 27:1485–1491.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Mosconi S, Beretta GD, Labianca R, Zampino
MG, Gatta G and Heinemann V: Cholangiocarcinoma. Crit Rev Oncol
Hematol. 69:259–270. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Anderson CD, Pinson CW, Berlin J and Chari
RS: Diagnosis and treatment of cholangiocarcinoma. Oncologist.
9:43–57. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Pattanathien P, Khuntikeo N, Promthet S
and Kamsa-Ard S: Survival rate of extrahepatic cholangiocarcinoma
patients after surgical treatment in Thailand. Asian Pac J Cancer
Prev. 14:321–324. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Jarnagin WR, Fong Y, DeMatteo RP, Gonen M,
Burke EC, Bodniewicz BSJ, Youssef BAM, Klimstra D and Blumgart LH:
Staging, resectability, and outcome in 225 patients with hilar
cholangiocarcinoma. Ann Surg. 234:517–519. 2001. View Article : Google Scholar
|
|
26
|
Kim DH, Jeong YI, Chung CW, Kim CH, Kwak
TW, Lee HM and Kang DH: Preclinical evaluation of sorafenib-eluting
stent for suppression of human cholangiocarcinomacells. Int J
Nanomedicine. 8:1697–1711. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Sugiyama H, Onuki K, Ishige K, Baba N,
Ueda T, Matsuda S, Takeuchi K, Onodera M, Nakanuma Y, Yamato M, et
al: Potent in vitro and in vivo antitumor activity of sorafenib
against human intrahepatic cholangiocarcinoma cells. J
Gastroenterol. 46:779–789. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang C, Maass T, Krupp M, Thieringer F,
Strand S, Wörns MA, Barreiros AP, Galle PR and Teufel A: A systems
biology perspective on cholangiocellular carcinoma development:
Focus on MAPK-signaling and the extracellular environment. J
Hepatol. 50:1122–1131. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Blechacz BR, Smoot RL, Bronk SF, Werneburg
NW, Sirica AE and Gores GJ: Sorafenib inhibits signal transducer
and activator of transcription-3 signaling in cholangiocarcinoma
cells by activating the phosphatase shatterproof 2. Hepatology.
50:1861–1870. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Adjei AA, Christian M and Ivy P: Novel
designs and end points for phase II clinical trials. Clin Cancer
Res. 15:1866–1872. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Bengala C, Bertolini F, Malavasi N, Boni
C, Aitini E, Dealis C, Zironi S, Depenni R, Fontana A, Del Giovane
C, et al: Sorafenib in patients with advanced biliary tract
carcinoma: A phase II trial. Br J Cancer. 102:68–72. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Lee JK, Capanu M, O'Reilly EM, Ma J, Chou
JF, Shia J, Katz SS, Gansukh B, Reidy-Lagunes D, Segal NH, et al: A
phase II study of gemcitabine and cisplatin plus sorafenib in
patients with advanced biliary adenocarcinomas. Br J Cance.
109:915–919. 2013. View Article : Google Scholar
|
|
33
|
Dal Lago L, D'Hondt V and Awada A:
Selected combination therapy with sorafenib: A review of clinical
data and perspectives in advanced solid tumors. Oncologist.
13:845–858. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
El-Khoueiry AB, Rankin CJ, Ben-Josef E,
Lenz HJ, Gold PJ, Hamilton RD, Govindarajan R, Eng C and Blanke CD:
SWOG 0514: A phase II study of sorafenib in patients with
unresectable or metastatic gallbladder carcinoma and
cholangiocarcinoma. Invest New Drugs. 30:1646–1651. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Nathan H, Pawlik TM, Wolfgang CL, Choti
MA, Cameron JL and Schulick RD: Trends in survival after surgery
for cholangiocarcinoma: A 30-year population-based SEER database
analysis. J Gastrointest Surg. 11:1488–1497. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Jarnagin WR, Klimstra DS, Hezel M, Gonen
M, Fong Y, Roggin K, Cymes K, DeMatteo RP, D'Angelica M, Blumgart
LH and Singh B: Differential cell cycle-regulatory protein
expression in biliary tract adenocarcinoma: Correlation with
anatomic site, pathologic variables, and clinical outcome. J Clin
Oncol. 24:1152–1160. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Vincenzi B, Santini D, Russo A, Addeo R,
Giuliani F, Montella L, Rizzo S, Venditti O, Frezza AM, Caraglia M,
et al: Early skin toxicity as a predictive factor for tumor control
in hepatocellular carcinoma patients treated with sorafenib.
Oncologist. 15:85–92. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Thongprasert S, Napapan S, Charoentum C
and Moonprakan S: Phase II study of gemcitabine and cisplatin as
first-line chemotherapy in inoperable biliary tract carcinoma. Ann
Onco. 16:279–281. 2005. View Article : Google Scholar
|
|
39
|
Weigt J and Malfertheiner P: Cisplatin
plus gemcitabine versus gemcitabine for biliary tract cancer.
Expert Rev Gastroenterol Hepatol. 4:395–397. 2010. View Article : Google Scholar : PubMed/NCBI
|