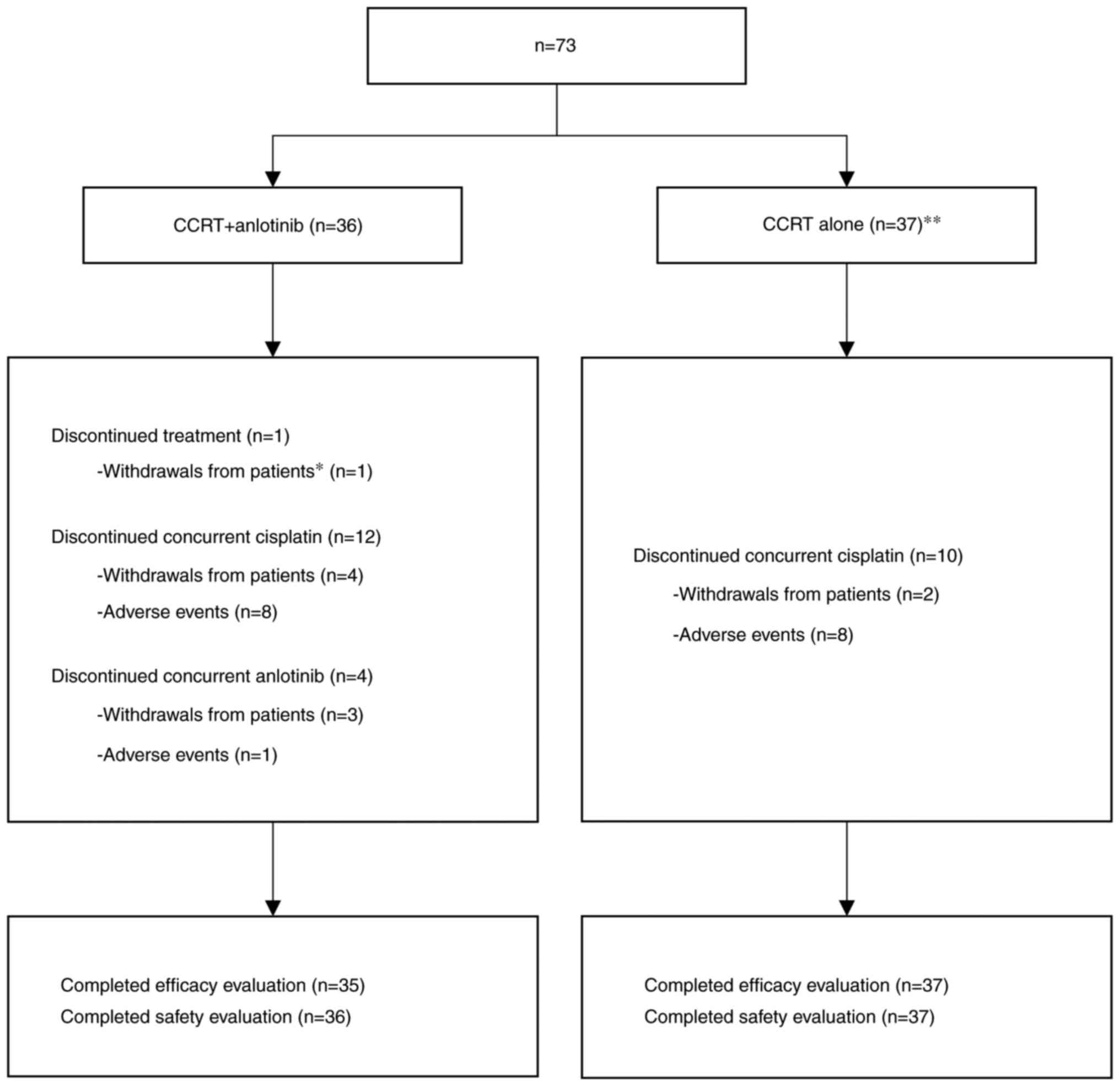
|
1
|
Sung H, Ferlay J, Siegel RL, Laversanne M,
Soerjomataram I, Jemal A and Bray F: Global cancer statistics 2020:
globocan estimates of incidence and mortality worldwide for 36
cancers in 185 countries. CA Cancer J Clin. 71:209–249. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Xia C, Yu XQ, Zheng R, Zhang S, Zeng H,
Wang J, Liao Y, Zou X, Zuo T, Yang Z and Chen W: Spatial and
temporal patterns of nasopharyngeal carcinoma mortality in China,
1973–2005. Cancer Lett. 401:33–38. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen L, Hu CS, Chen XZ, Hu GQ, Cheng ZB,
Sun Y, Li WX, Chen YY, Xie FY, Liang SB, et al: Adjuvant
chemotherapy in patients with locoregionally advanced
nasopharyngeal carcinoma: Long-term results of a phase 3
multicentre randomised controlled trial. Eur J Cancer. 75:150–158.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zhang Y, Chen L, Hu GQ, Zhang N, Zhu XD,
Yang KY, Jin F, Shi M, Chen YP, Hu WH, et al: Gemcitabine and
cisplatin induction chemotherapy in nasopharyngeal carcinoma. N
Engl J Med. 381:1124–1135. 2019. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
O'Sullivan B: Nasopharynx cancer:
Therapeutic value of chemoradiotherapy. Int J Radiat Oncol Biol
Phys. 69 (Suppl 2):S118–S121. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Huang JF, Zhang FZ, Zou QZ, Zhou LY, Yang
B, Chu JJ, Yu JH, Zhang HW, Yuan XP, Tai GM, et al: Induction
chemotherapy followed by concurrent chemoradiation and nimotuzumab
for locoregionally advanced nasopharyngeal carcinoma: Preliminary
results from a phase II clinical trial. Oncotarget. 8:2457–2465.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun Y, Niu W, Du F, Du C, Li S, Wang J, Li
L, Wang F, Hao Y, Li C and Chi Y: Safety, pharmacokinetics, and
antitumor properties of anlotinib, an oral multi-target tyrosine
kinase inhibitor, in patients with advanced refractory solid
tumors. J Hematol Oncol. 9:1052016. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Taurin S, Yang CH, Reyes M, Cho S, Coombs
DM, Jarboe EA, Werner TL, Peterson CM and Janát-Amsbury MM:
Endometrial cancers harboring mutated fibroblast growth factor
receptor 2 protein are successfully treated with a new small
tyrosine kinase inhibitor in an orthotopic mouse model. Int J
Gynecol Cancer. 28:152–160. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li D, Chi Y, Chen X, Ge M, Zhang Y, Guo Z,
Wang J, Chen J, Zhang J, Cheng Y, et al: Anlotinib in locally
advanced or metastatic medullary thyroid carcinoma: A randomized,
double-blind phase IIB trial. Clin Cancer Res. 27:3567–3575. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Han B, Li K, Wang Q, Zhang L, Shi J, Wang
Z, Cheng Y, He J, Shi Y, Zhao Y, et al: Effect of anlotinib as a
third-line or further treatment on overall survival of patients
with advanced non-small cell lung cancer: The ALTER 0303 Phase 3
randomized clinical trial. JAMA Oncol. 4:1569–1575. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Cheng Y, Wang Q, Li K, Shi J, Liu Y, Wu L,
Han B, Chen G, He J, Wang J, et al: Anlotinib vs placebo as third-
or further-line treatment for patients with small cell lung cancer:
A randomised, double-blind, placebo-controlled Phase 2 study. Br J
Cancer. 125:366–371. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ma BB, Poon TC, To KF, Zee B, Mo FK, Chan
CM, Ho S, Teo PM, Johnson PJ and Chan AT: Prognostic significance
of tumor angiogenesis, Ki 67, p53 oncoprotein, epidermal growth
factor receptor and HER2 receptor protein expression in
undifferentiated nasopharyngeal carcinoma-a prospective study. Head
Neck. 25:864–872. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lu Y, Chen D, Liang J, Gao J, Luo Z, Wang
R, Liu W, Huang C, Ning X, Liu M and Huang H: Administration of
nimotuzumab combined with cisplatin plus 5-fluorouracil as
induction therapy improves treatment response and tolerance in
patients with locally advanced nasopharyngeal carcinoma receiving
concurrent radiochemotherapy: A multicenter randomized controlled
study. BMC Cancer. 19:12622019. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Cai Q, Su N, Fang Y, Zou Q, Xia Y, Ma S,
Cai J, Liu P, Wang J and Zhang Y: Anlotinib for patients with
recurrent or metastatic nasopharyngeal carcinoma: A phase II study.
J Clin Oncol. 40 (Suppl 16):e180202022. View Article : Google Scholar
|
|
16
|
Rice TW, Patil DT and Blackstone EH: 8th
edition AJCC/UICC staging of cancers of the esophagus and
esophagogastric junction: Application to clinical practice. Ann
Cardiothorac Surg. 6:119–130. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu TT, Hu CS and LiI BS: Clinical
consensus on the treatment of locally advanced squamous cell
carcinoma of the head and neck with anti-EGFR monoclonal antibody
(2023 edition). China Oncology. 33:81–94. 2023.
|
|
18
|
Lee AW, Ng WT, Pan JJ, Poh SS, Ahn YC,
Alhussain H, Corry J, Grau C, Grégoire V, Harrington KJ, et al:
International guideline for the delineation of the clinical target
volumes (CTV) for nasopharyngeal carcinoma. Radiother Oncol.
126:25–36. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Eisenhauer EA, Therasse P, Bogaerts J,
Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S,
Mooney M, et al: New response evaluation criteria in solid tumours:
Revised RECIST guideline (version 1.1). Eur J Cancer. 45:228–247.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
US Department of Health and Human
Services, . National Institutes of Health, National Cancer
Institute: Common Terminology Criteria for Adverse Events (CTCAE)
Version 5. Published:. November 27–2017.
|
|
21
|
Li HP, Huang CY, Lui KW, Chao YK, Yeh CN,
Lee LY, Huang Y, Lin TL, Kuo YC, Huang MY, et al: Combination of
epithelial growth factor receptor blockers and CDK4/6 inhibitor for
nasopharyngeal carcinoma treatment. Cancers (Basel). 13:29542021.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Luo Y, Wang J, Wang F, Liu X, Lu J, Yu X,
Ma X, Peng X and Li X: Foxq1 promotes metastasis of nasopharyngeal
carcinoma by inducing vasculogenic mimicry via the EGFR signaling
pathway. Cell Death Dis. 12:4112021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wakisaka N, Hirota K, Kondo S,
Sawada-Kitamura S, Endo K, Murono S and Yoshizaki T: Induction of
lymphangiogenesis through vascular endothelial growth
factor-C/vascular endothelial growth factor receptor 3 axis and its
correlation with lymph node metastasis in nasopharyngeal carcinoma.
Oral Oncol. 48:703–708. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Peng QX, Han YW, Zhang YL, Hu J, Fan J, Fu
SZ, Xu S and Wan Q: Apatinib inhibits VEGFR-2 and angiogenesis in
an in vivo murine model of nasopharyngeal carcinoma. Oncotarget.
8:52813–52822. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
You R, Hua YJ, Liu YP, Yang Q, Zhang YN,
Li JB, Li CF, Zou X, Yu T, Cao JY, et al: Concurrent
chemoradiotherapy with or without Anti-EGFR-Targeted treatment for
Stage II–IVb nasopharyngeal carcinoma: Retrospective analysis with
a large cohort and long follow-up. Theranostics. 7:2314–2324. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin M, You R, Liu YP, Zhang YN, Zhang HJ,
Zou X, Yang Q, Li CF, Hua YJ, Yu T, et al: Beneficial effects of
anti-EGFR agents, Cetuximab Or Nimotuzumab, in combination with
concurrent chemoradiotherapy in advanced nasopharyngeal carcinoma.
Oral Oncol. 80:1–8. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Yin Y, Zhou Z, Li Z, Shen M, Qin Y, Yang
C, Wang R and Kang M: Efficacy of concurrent chemoradiotherapy plus
Endostar compared with concurrent chemoradiotherapy in the
treatment of locally advanced nasopharyngeal carcinoma: A
retrospective study. Radiat Oncol. 17:1352022. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Li Y, Chen QY, Tang LQ, Liu LT, Guo SS,
Guo L, Mo HY, Chen MY, Guo X, Cao KJ, et al: Concurrent
chemoradiotherapy with or without cetuximab for stage II to IVb
nasopharyngeal carcinoma: A case-control study. BMC Cancer.
17:5672017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang N, Wang K, Song F and Liu Y:
Cetuximab in combination with chemoradiotherapy for nasopharyngeal
carcinoma: A meta-analysis. Indian J Cancer. 55:196–200. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Xu T, Liu Y, Dou S, Li F, Guan X and Zhu
G: Weekly cetuximab concurrent with IMRT aggravated
radiation-induced oral mucositis in locally advanced nasopharyngeal
carcinoma: Results of a randomized phase II study. Oral Oncol.
51:875–879. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kang M, Wang F, Liao X, Zhou P and Wang R:
Intensity-modulated radiotherapy combined with endostar has similar
efficacy but weaker acute adverse reactions than IMRT combined with
chemotherapy in the treatment of locally advanced nasopharyngeal
carcinoma. Medicine (Baltimore). 97:e111182018. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
You R, Sun R, Hua YJ, Li CF, Li JB, Zou X,
Yang Q, Liu YP, Zhang YN, Yu T, et al: Cetuximab or nimotuzumab
plus intensity-modulated radiotherapy versus cisplatin plus
intensity-modulated radiotherapy for stage II–IVb nasopharyngeal
carcinoma. Int J Cancer. 141:1265–1276. 2017. View Article : Google Scholar : PubMed/NCBI
|