Introduction
Endometrial cancer (EC) represents the sixth most
common cancer in women worldwide, accounting for 7% of all types of
cancer and 4% of cancer-associated mortalities in women (1). A large proportion of patients present
with an endometrioid histology, and the 5-year overall survival
(OS) rate for patients with endometrioid type EC is >90%
(2). By contrast, 15–20% of
patients with EC exhibit non-endometroid histotypes, with a 5-year
OS rate of ~70% in uterine-confined disease (3) and 50% in cohorts including patients
with all stages (4).
The International Federation of Gynecology and
Obstetrics (FIGO) stage has been consistently identified as the
primary prognostic factor for patients with non-endometrioid ECs
(3,4). Although age, lymphovascular space
invasion (LVSI), lymph node dissection and adjuvant radiotherapy
have been reported to be associated with disease-free survival
(DFS) and/or OS, it is notable that there is considerable
inconsistency between the results of the studies (3–5). Given
that non-endometrioid ECs are more often associated with advanced
disease and poor outcomes compared with endometrioid type ECs
(5), there is a clear need to
identify novel prognostic markers in this patient population to
improve the disease outcomes.
In the past decade, numerous studies have
demonstrated the prognostic value of molecular classification in
patients with stage I to III EC (6–8).
However, a recent study has shown that molecular classification had
no effect on progression-free survival (PFS) or OS in patients with
stage IV disease, and that OS was only influenced by tumor
histotype and estrogen receptor status (9). There is growing evidence that
expression of L1 cell adhesion molecule (L1CAM) may be a predictor
of disease outcomes in patients with EC (10,11). A
systematic review and meta-analysis found that L1CAM overexpression
was associated with a worse DFS (HR, 4.11) and OS (HR, 3.62), even
in stage I EC (11). Despite the
evidence that high L1CAM expression levels are associated with a
higher FIGO grade and with older age (11), current understanding regarding the
role of L1CAM in non-endometrioid ECs is still limited. This is
partly due to the relatively small number of patients with
non-endometrioid EC included in previous analyses (10,11).
Sirtuins (SIRTs) are a family of signaling proteins
that regulate cellular functions and are encoded by the Sir2
gene (12). SIRTs utilize oxidized
nicotinamide-adenine dinucleotide as a catalyst and exhibit
mono-ADP-ribosyl transferase or deacetylase activity, thus ensuring
the proper functioning of cellular events such as metabolism,
oxidative stress, transcription, apoptosis, DNA repair and
inflammation (12). The SIRT
protein family comprises seven different isoforms (SIRT1-7), which
are distributed across various cellular compartments (13). SIRT1, SIRT6 and SIRT7 are considered
to be nuclear proteins and show different subnuclear localizations,
such as in heterochromatic regions and nucleoli. Conversely, SIRT3,
SIRT4 and SIRT5 are typically found in mitochondria (14). Although the SIRT isoforms share a
common catalytic core in terms of chemical and structural
compositions, they show minor differences in the molecular
structure of their active sites (13).
Sirtuin-1 (SIRT1) is mainly a nuclear protein but
can translocate between the cytoplasm and nucleus in response to
tissue and energy requirements (14). Several studies have reported that
SIRT1 expression is upregulated and associated with poor disease
prognosis in various types of cancer, including hepatocellular
carcinoma, non-small cell lung cancer, breast cancer, gastric
cancer, pancreatic cancer, colon cancer, prostate cancer, large
B-cell lymphoma and acute myeloid leukemia (15,16).
By contrast, a limited number of studies have investigated the
correlation between the expression of SIRT1 and types of
gynecological cancer, a large proportion of which includes cervical
and ovarian cancer (17,18).
Understanding the expression patterns of SIRT1 in
non-endometrioid EC and linking this to prognosis may help to
tailor the need for adjuvant therapy, guide the identification of
potential targeted therapies and improve patient outcomes. The
present study aimed to investigate the expression characteristics
of SIRT1 in patients with non-endometrioid type EC using
immunohistochemistry. The secondary endpoint of the present study
was to evaluate the prognostic impact of SIRT1 expression on
PFS.
Materials and methods
Study design and patients
The present study was a single-center (Saglik
Bilimleri University Antalya Training and Research Hospital;
Antalya, Turkey), retrospective cohort study that included patients
who underwent hysterectomy between June 2017 and December 2021, and
had a postoperative histopathological diagnosis of non-endometrioid
EC. Patients were excluded if they had an endometrioid histotype, a
primary synchronous malignancy, insufficient clinical data or had
poor-quality immunohistochemistry results.
Following approval from the Ethics Committee of the
Antalya Training and Research Hospital (approval no. 22/108;
Antalya, Turkey), detailed clinical and pathological data of
patients treated within the aforementioned study period were
reviewed for eligibility. A total of 51 patients with
non-endometrioid EC were identified. Of these, seven were initially
excluded from the study as four had insufficient clinical data and
three had a primary synchronous malignancy. Therefore, tissue
samples from 44 patients were extracted from the pathology archives
for immunohistochemical studies.
Immunohistochemical studies
The preparation of tissue samples for
immunohistochemistry, including fixation, dehydration, embedding
and sectioning was performed as previously described (19). Tissue sections (3 µm) were cut from
tissue blocks onto pre-coated slides. The samples were subsequently
incubated at 60°C for 60 min, deparaffinised by passage through
xylene (three times for 5 min) and rehydrated by successive
immersion in 100, 96, 90, 80 and 70% alcohol for 5 min. A
concentrated polymer-based protein-free blocking reagent (cat. no.
TA-125-UB: Lab Vision™ Ultra V Block; Thermo Fisher Scientific,
Inc.) was applied for 10 min at room temperature.
Immunohistochemical staining was carried out using an automated
platform (Shandon Pathcentre™; Thermo Fisher Scientific, Inc.) in
accordance with the manufacturer's instructions, using a monoclonal
mouse primary antibody that specifically targeted the SIRT1 protein
(cat. no. ab110304; 1:1,000; Abcam). Then, a ready-to-use
enzyme-labelled polymer secondary antibody (cat. no. TL-125-HL;
UltraVision Large Volume Detection System HRP polymer; Thermo
Fisher Scientific, Inc.) was applied for an additional 30 min at
room temperature. Finally, the sections were stained with a
diaminobenzidine (DAB) tetrahydrochloride substrate kit containing
25 ml of DAB solution and 250 ml of stable hydrogen peroxide
substrate buffer (cat. no. 34002: DAB Substrate Kit; Thermo Fisher
Scientific, Inc.) and with hematoxylin for counterstaining for 5
min each at room temperature. A gynecological pathologist, who was
blinded to the clinical and pathological data, evaluated the
staining reaction of the slides under a light microscope. The
nuclear staining reaction of SIRT1 was considered to be positive in
the presence of any percentage of nuclear staining. The cytoplasmic
staining reaction of SIRT1 was assessed using immune reactivity
scoring (IRS) system (20). The
percentage of cytoplasmic staining with SIRT1 was calculated as
follows: Number of immunopositive cells divided by the total number
of cells counted. The following criteria were used to grade the
percentage of staining: 0, no staining; 1, ≤10%; 2, 11–50%; 3,
51–80%; and 4, ≥81%. The cytoplasmic staining in the cells that
line the normal endometrial glands was used as a positive internal
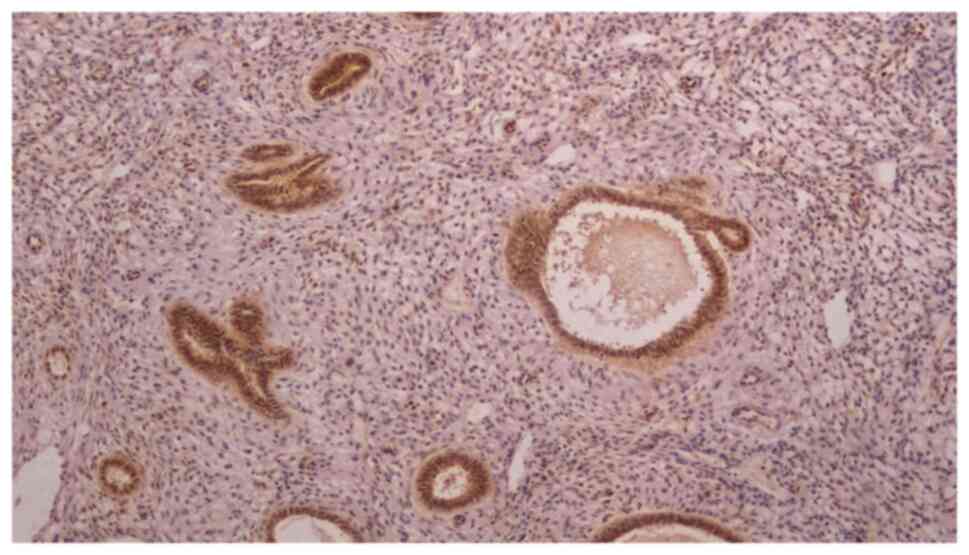
control for the intensity of the SIRT1 staining (Fig. 1). The staining intensity was graded
semi-quantitatively as follows: 0, no staining; 1, weak staining;
2, moderate staining; and 3, strong staining (Fig. 2).
The IRS values were determined by multiplying the
scores for the staining percentage and staining intensity. Scores
were graded as follows: 0–2, negative expression; 3–4, low
expression; 6–8, moderate expression; and 9–12, high
expression.
Statistical analysis
The analyses were carried out using SPSS (version
20.0; IBM Corp.) statistics software. Standard descriptive
statistics were used, with counts and frequencies being utilized
for binary variables and medians being supplemented by the range
for continuous variables. Cox proportional hazards regression
models with the enter method were used to evaluate the impact of
SIRT1 and other clinical and pathological characteristics on PFS.
The model results were presented as hazard ratios (HRs) with 95%
confidence intervals (CI). P<0.05 was considered to indicate a
statistically significant difference. Therefore, variables with a
P-value <0.05 in univariate analyses were included in the
multivariate analyses. PFS was defined as the period in months
between the date of surgery and the date of disease progression, or
relapse or mortality from any cause. The surviving patients that
were not exhibiting progression or relapse were censored at the
date they were last known to be alive according to the PFS
data.
Results
Clinical and pathological
characteristics
Of the 44 tissue samples that underwent
immunohistochemical analysis in the present study, one was excluded
from the final analyses due to poor quality immunostaining for
evaluation. Therefore, data from a total of 43 patients were
analyzed and presented.
Table I displays the
surgical and pathological characteristics of included patients. The
median age was 64 years. Of the 43 patients, six patients (16.4%)
received neoadjuvant chemotherapy. A large proportion of patients
(88.4%) had systematic lymph node dissection and 60.5% of patients
had serous tumor histotype (pure serous or mixed with other
histotypes). Deep (≥50%) myometrial invasion was observed in 60.5%
of patients, LVSI in 65.1%, cervical invasion in 25.6%, adnexal
involvement in 23.3% and lymph node metastasis in 47.4%. The
distribution of FIGO stages was as follows: Stage I, 34.9%; stage
II, 4.7%; stage III, 23.3%; and stage IV, 37.2%.
 | Table I.Surgical and pathological
characteristics of included patients. |
Table I.
Surgical and pathological
characteristics of included patients.
| Variables | Values |
|---|
| Median age, years
(range) | 64 (50–80) |
| Surgical procedure, n
(%) |
|
| TH/BSO
plus staging surgery including lymphadenectomy | 26 (60 .5) |
| TH/BSO
plus primary debulking surgery | 11 (25.6) |
|
Neoadjuvant chemotherapy
followed by interval debulking surgery | 6 (14.0) |
| Systematic lymph node
dissection | 38 (88.4) |
| Median no. of lymph
nodes removed, n (range) | 57 (16–107) |
| Median tumor size, cm
(range) | 4.4 (1–13) |
| Lymphovascular space
involvement, n (%) | 28 (65.1) |
| Deep (≥50%)
myometrial invasion, n (%) | 26 (60.5) |
| Cervical involvement,
n (%) | 11 (25.6) |
| Adnexal involvement,
n (%) | 10 (23.3) |
| Lymph node
involvement, n (%) | 18/38 (47.4) |
| Positive cytology, n
(%) | 10 (23.3) |
| FIGO stage, n
(%) |
|
| I | 15 (34.9) |
| IA | 11 (25.6) |
| IB | 4 (9.3) |
| II | 2 (4.7) |
| III | 10 (23.3) |
|
IIIC1 | 4 (9.3) |
|
IIIC2 | 6 (14.0) |
| IVB | 16 (37.2) |
| Tumor histotype |
|
|
Serous | 19 (44.2) |
| Mixed
(serous and endometrioid grade 3) | 5 (11.6) |
| Mixed
(serous and clear cell) | 2 (4.7) |
| Mixed
(endometrioid grade 3 and clear cell) | 1 (2.3) |
| Clear
cell | 1 (2.3) |
|
Carcinosarcoma | 8 (18.6) |
|
Dedifferentiated | 5 (11.6) |
|
Squamous cell | 1 (2.3) |
|
Mucinous | 1 (2.3) |
Disease outcomes of patients are summarized in
Table II. Of the included
patients, one patient succumbed to pulmonary thromboembolism on day
5 after surgery. Of the remaining patients, 95.2% received adjuvant
chemotherapy with or without external beam radiotherapy. During a
median follow-up time of 26 months, 22 of the 42 patients (52.4%)
experienced disease progression. At the time of analysis, 20
patients (46.5%) were living with no disease, 5 (11.6%) were living
with disease and 18 (41.9%) succumbed to the disease. The estimated
2-year and 3-year PFS rates were 50.0 and 43.6%, respectively,
while the estimated 2- and 3-year OS rates were 65.7 and 62.4%,
respectively.
 | Table II.Outcomes of patients. |
Table II.
Outcomes of patients.
| Variables | Values |
|---|
| 30-day
postoperative mortality, n (%) | 1 (2.3) |
| Adjuvant therapy, n
(%) | 40/42 (95.2) |
|
Brachytherapy alone | 2 (4.8) |
|
Chemotherapy alone | 16 (38.1) |
|
Chemotherapy plus EBRT | 22 (52.4) |
| Disease status
on/after first-line therapy (primary surgery +/-
adjuvant/neoadjuvant therapy), n (%) |
|
|
Complete remission | 35 (88.4) |
| Partial
remission | 6 (13.9) |
| Stable
disease | 1 (2.3) |
|
Death | 1 (2.3) |
| Recurrence in
patients at complete remission, n (%) | 15/35 (42.8) |
| Progression
(disease recurrence, progression after partial remission or stable
disease), n (%) | 22/42 (52.4) |
| Median time to
progression, months (IQR) | 7 (3–13) |
| Median follow up
time, months (IQR) | 26 (13–45) |
| Survival status, n
(%) |
|
| Alive
with no evidence of disease | 20 (46.5) |
| Alive
with disease | 5 (11.6) |
| Dead of
disease | 18 (41.9) |
| Median
progression-free survival, months (95% CI) | 31 (6.7–55.2) |
| 24,
% | 50.0 |
| 36,
% | 43.6 |
| Median overall
survival, months (95% CI) | 65 (30.3–99.6) |
| 24,
% | 65.7 |
| 36,
% | 62.4 |
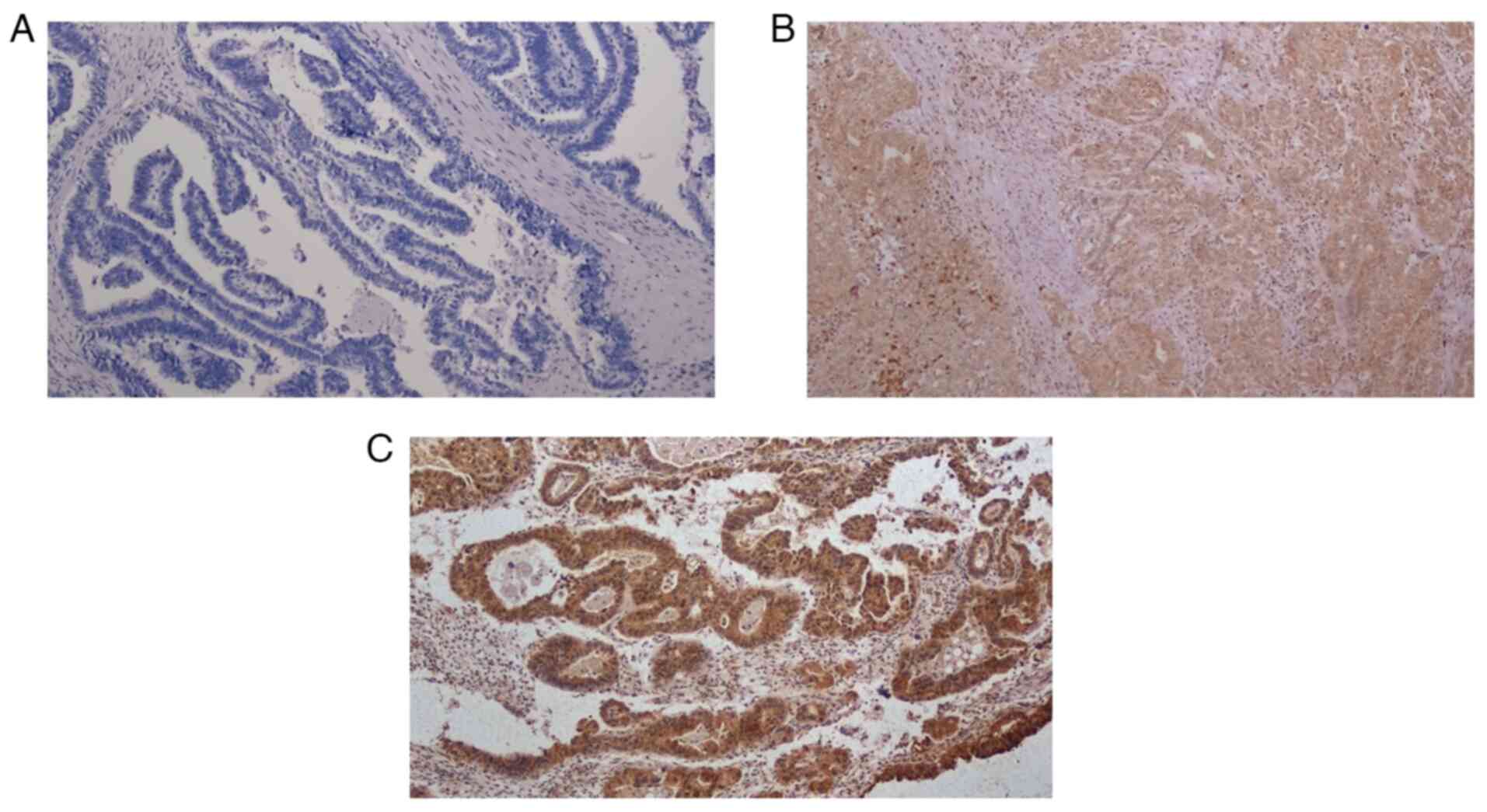
Immunohistochemistry results
The immunohistochemical staining features of tissue
samples with SIRT1 are presented in Table III. Cytoplasmic staining with
SIRT1 was detected in all samples (100%), whereas no nuclear
staining was evident in any of the tissue samples. According to the
IRS, 20.9% of samples exhibited negative cytoplasmic expression,
14.0% exhibited low expression, 37.2% exhibited moderate expression
and 27.9% exhibited high expression.
 | Table III.Immunohistochemical staining features
of tumor cells with SIRT1. |
Table III.
Immunohistochemical staining features
of tumor cells with SIRT1.
| Variables | No. of patients
(%) |
|---|
| Cytoplasmic
staining of tumor cells with SIRT1 | 43 (100) |
| Percentage of
tumor-cell staining |
|
| No
staining | - |
|
≤10 | 3 (7.0) |
|
11-50 | 10 (23.3) |
|
51-80 | 14 (32.6) |
|
≥81 | 16 (37.2) |
| Staining
intensity |
|
| No
staining | - |
|
Weak | 12 (27.9) |
|
Moderate | 18 (41.9) |
|
Strong | 13 (30.2) |
| Immune reactivity
score |
|
| 0-2
(negative expression) | 9 (20.9) |
| 3-4
(low-expression) | 6 (14.0) |
| 6-8
(moderate-expression) | 16 (37.2) |
| 9-12
(high-expression) | 12 (27.9) |
Cox regression analyses of factors
influencing PFS
In the univariate analysis, five variables were
significantly associated with PFS: Tumor size (P=0.026), deep
(≥50%) myometrial invasion (P=0.029), lymph node involvement
(P=0.001), positive peritoneal cytology (P=0.011) and FIGO stage
(P<0.001). In the multivariate analysis, however, none of these
variables remained an independent significant prognostic factor
(Table IV).
 | Table IV.Factors associated with
progression-free survival. |
Table IV.
Factors associated with
progression-free survival.
|
| Univariate | Multivariate |
|---|
|
|
|
|
|---|
| Variables | HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Age, years | 1.029 | 0.976–1.084 | 0.292 | - | - | - |
| Tumor size, cm | 1.195 | 1.022–1.397 | 0.026a | - | - | 0.615 |
| Tumor
histology |
|
| 0.965 | - | - | - |
| Serous
(pure or mixed) | 1.069 | 0.394–2.903 | 0.896 | - | - | - |
|
Carcinosarcoma | 1.014 | 0.344–2.983 | 0.980 | - | - | - |
|
Dedifferentiated | 2.040 | 0.598–6.957 | 0.255 | - | - | - |
| Lymphovascular
space involvement | 0.605 | 0.265–1.383 | 0.234 | - | - | - |
| Deep (≥50%)
myometrial invasion | 3.047 | 1.123–8.264 | 0.029a | - | - | 0.970 |
| Cervical
involvement | 1.546 | 0.654–3.652 | 0.321 | - | - | - |
| Adnexal
involvement | 2.240 | 0.937–5.357 | 0.070 | - | - | - |
| Lymph node
involvement | 8.337 | 2.388–29.107 | 0.001a | - | - | 0.915 |
| Positive peritoneal
cytology | 3.050 | 1.298–7.170 | 0.011a | - | - | 0.682 |
| FIGO stage | 2.568 | 1.591–4.145 |
<0.001a | - | - | 0.418 |
| Stage I–II vs.
III–IV | 11.772 | 2.732–50.730 | 0.001a | - | - | - |
| Adjuvant
therapy | 1.533 | 0.205–11.438 | 0.677 | - | - | - |
| No. of lymph nodes
removed | 1.015 | 0.994–1.037 | 0.154 | - | - | - |
| Staining with
sirtuin-1 |
|
|
|
|
|
|
|
Percentage of staining | 1.096 | 0.697–1.724 | 0.692 | - | - | - |
|
Intensity of staining | 1.293 | 0.728–2.296 | 0.380 | - | - | - |
| Immune
reactivity score | 1.046 | 0.934–1.172 | 0.435 | - | - | - |
Discussion
The present study investigated the expression
characteristics of SIRT1 in non-endometrioid EC using
immunohistochemistry; secondly, the effects of SIRT1 expression and
various clinicopathological variables on the PFS of patients were
examined. It was demonstrated that SIRT1 expression was found to be
cytoplasmic in non-endometrioid EC. Additionally, according to the
IRS, ~80% of cases had varying degrees of SIRT1 expression.
Furthermore, the present study demonstrated that there were no
independent factors examined that influenced PFS.
There are very few studies in the literature
investigating SIRT1 expression in EC, most of which have focused on
comparing the prevalence of SIRT1 expression between neoplastic and
non-neoplastic endometrial tissues. Lin et al (21) demonstrated that SIRT1 expression was
more prevalent in EC cells than in normal endometrial cells and
that there was an association between SIRT1 expression and the
levels of sterol regulatory element binding protein 1 (SREBP1), a
nuclear lipogenic transcription factor. This study reported that
SIRT1 knockdown could downregulate SREBP1 expression and suppress
cell proliferation, and thus new therapeutic agents targeting SIRT1
may contribute to the treatment of EC (21). Similarly, Huang et al
(22) investigated the effect of
SIRT1-mediated LC3 acetylation on autophagy and proliferation of EC
cells and reported that SIRT1 expression was higher in EC cells
than in non-neoplastic endometrium. In EC cells overexpressing
SIRT1, LC3 acetylation was inhibited, and cell proliferation was
promoted whereas knockdown of SIRT1 inhibited proliferation,
migration and invasion of EC cells (22). Bartosch et al (23) studied messenger RNA expression of
SIRT1-7 in ECs and benign endometrial tissue samples using
quantitative real-time PCR; it was found that, compared with benign
tissues, ECs showed upregulation of SIRT7, whereas SIRT1, SIRT2,
SIRT4 and SIRT5 were downregulated (23).
The number of studies investigating the relationship
between SIRT1 expression and disease outcomes in EC is limited and
the results are conflicting. Asaka et al (24) analysed SIRT1 expression in 108 cases
of endometrioid EC and found that SIRT1 overexpression
significantly increased the resistance of EC cell lines to
cisplatin and paclitaxel. The authors reported that SIRT1
overexpression was significantly aligned with poor disease outcome
(24). However, Al-Maghrabi and
Al-Maghrabi (25) reported no
association between SIRT1 overexpression and disease outcome in a
study of 66 cases of endometrioid EC and five cases of serous EC.
By contrast, Beyer et al (26) reported improved PFS and OS in cases
with SIRT1 expression in a study involving 59 patients with
endometrioid EC and six patients with clear cell EC. It was also
reported that the staining intensity of SIRT1 was significantly
higher in the endometrioid histotype in comparison to the clear
cell histotype (26).
The studies by Al-Maghrabi and Al-Maghrabi
(25) and Beyer et al
(26) both included very few cases
of non-endometrioid EC (n=5 and n=6, respectively), which is
insufficient to draw conclusions on SIRT1 expression and survival
in non-endometrioid EC. In the present study, no significant
association between SIRT1 expression and PFS was demonstrated in a
cohort consisting of only non-endometrioid ECs. To the best of our
knowledge, this is the first study to characterize the expression
patterns of SIRT1 and its association with prognosis exclusively in
non-endometrioid ECs.
In the present study, SIRT1 was found to be
expressed in the cytoplasm with no cases of nuclear expression.
Similarly, Asaka et al (24)
and Beyer et al (26)
reported the location of SIRT1 expression as the cytoplasm. By
contrast, Al-Maghrabi and Al-Maghrabi (25) reported outcomes related with nuclear
expression of SIRT1. Furthermore, the scoring systems for SIRT1
expression used in the previous studies (24–26)
differ from each other. Differences in the location of SIRT1
expression, scoring methods used and histotypes may explain the
discrepancies between the results of the aforementioned studies
(24–26).
The main strength of the present study was the
analysis of the significance of SIRT1 expression in a homogeneous
group of patients in terms of tumor histology, consisting
exclusively of non-endometrioid cases. Given the aggressive nature
of non-endometrioid ECs, it is evident that novel prognostic
molecular markers are required to improve the management of
patients with non-endometrioid EC. However, the literature on SIRT1
expression in EC consists of studies involving cases with
endometrioid histology, either alone (24) or in combination with a limited
number of non-endometrioid cases (25,26).
The analyses conducted in the present study have
certain limitations. The present study was a retrospective analysis
with a relatively small sample size. It included patients from a
tertiary referral center, which limited the generalizability of the
findings. Furthermore, the potential subjectivity in the
interpretation of immunohistochemistry results could not be
excluded due to the single center nature of the present study and
the lack of external validation. The prognostic value of SIRT1
expression was only assessed through immunohistochemical
techniques, and the results were not corroborated by molecular
analysis. In addition, molecular classification was not used, which
precludes definitive conclusions on the role of SIRT1 expression in
different molecular subgroups. Over the past decade, evidence has
emerged that there are at least four molecular subgroups of EC,
each with a different prognosis (6–8). The
subgroups included EC with a high mutation rate in the polymerase-ε
(POLE) exonuclease domain (POLE-mutated),
microsatellite-instability-high EC, EC with a low mutation rate and
low somatic copy number alteration (non-specific molecular pattern
EC, and EC with a low mutation rate but high somatic copy number
alteration rates and TP53 mutations. Therefore, the present study
should be regarded as a preliminary investigation and further
research with a larger cohort is essential to validate the
results.
In conclusion, SIRT1 expression was found to be
cytoplasmic in non-endometrioid EC. According to IRS results, ~80%
of cases exhibited varying degrees of SIRT1 expression. However,
SIRT1 expression did not significantly affect PFS.
Acknowledgements
Not applicable.
Funding
The present study was completed with the support of Education
Planning Committee of the Antalya Training and Research Hospital
(grant no. 2023/125; Antalya, Turkey).
Availability of data and materials
The data generated in this study may be requested
from the corresponding author.
Authors' contributions
NY conceptualized the study, collected, validated
and analyzed the data and wrote the original draft. HTY
conceptualized the study, designed the methodology and collected
the data. AA, MuG, and MeG collected and interpreted the data. MeG
contributed to the analysis and interpretation of the data. IU was
the project administrator, contributed to the design of the study
and critically reviewed the intellectual content. TT conceptualized
the study, designed the methodology, validated and analyzed the
data, and reviewed and edited the manuscript. NY and TT confirm the
authenticity of all the raw data. All authors read and approved the
final version of the manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of the Antalya Training and Research Hospital (approval
no. 22/108; Antalya, Turkey). Although the Ethics Committee waived
the requirement for informed consent due to the retrospective
nature of the study, written informed consent was obtained from all
patients.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
Authors' information
Dr Necim Yalcin, ORCID ID 0000000159803244;
Associate Professor Hulya Tosun Yildirim, ORCID ID
0000000249730982; Dr Aysun Alci, ORCID ID 0000000279127375; Dr
Mustafa Gokkaya, ORCID ID 000000020477157X; Dr Mehmet Goksu, ORCID
ID 0000000193306241; Professor Isin Ureyen, ORCID ID
0000000234914682;Professor Tayfun Toptas, ORCID ID
0000000267066915.
References
|
1
|
Siegel RL, Miller KD, Fuchs HE and Jemal
A: Cancer Statistics, 2021. CA Cancer J Clin. 71:7–33. 2021.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Åkesson Å, Adok C and Dahm-Kähler P:
Recurrence and survival in endometrioid endometrial cancer-a
population-based cohort study. Gynecol Oncol. 168:127–134. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Åkesson Å, Adok C and Dahm-Kähler P:
Increased survival in non-endometrioid endometrial cancer after
introducing lymphadenectomy and tailoring radiotherapy-A
population-based cohort study. Eur J Cancer. 169:54–63. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lee SJ, Kang HJ, Kwak YK, Kim M and Kim J:
Non-endometrioid endometrial cancer: Analysis of recurrence pattern
and identification of prognostic and treatment factors affecting
recurrence and survival. Strahlenther Onkol. 199:828–837. 2023.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Feng J, Zhang Y, Huang C, Li L, Liu J,
Wang J, Guo H, Yao S, Cui Z, Chen G, et al: Prognostic evaluation
of lymph-vascular space invasion in patients with endometrioid and
non-endometrioid endometrial cancer: A multicenter study. Eur J
Surg Oncol. 50:1082612024. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stelloo E, Nout RA, Osse EM,
Jürgenliemk-Schulz IJ, Jobsen JJ, Lutgens LC, van der Steen-Banasik
EM, Nijman HW, Putter H, Bosse T, et al: Improved risk assessment
by integrating molecular and clinicopathological factors in
early-stage endometrial cancer-combined analysis of the PORTEC
cohorts. Clin Cancer Res. 22:4215–4224. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Karnezis AN, Leung S, Magrill J, McConechy
MK, Yang W, Chow C, Kobel M, Lee CH, Huntsman DG, Talhouk A, et al:
Evaluation of endometrial carcinoma prognostic immunohistochemistry
markers in the context of molecular classification. J Pathol Clin
Res. 3:279–293. 2017. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
León-Castillo A, de Boer SM, Powell ME,
Mileshkin LR, Mackay HJ, Leary A, Nijman HW, Singh N, Pollock PM,
Bessette P, et al: Molecular classification of the PORTEC-3 trial
for high-risk endometrial cancer: Impact on prognosis and benefit
from adjuvant therapy. J Clin Oncol. 38:3388–3397. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Uijterwaal MH, van Dijk D, Lok CAR, De
Kroon CD, Kasius JC, Zweemer R, Gerestein CG, Horeweg N, Bosse T,
van der Marel J, et al: Prognostic value of molecular
classification in stage IV endometrial cancer. Int J Gynecol
Cancer. 34:847–854. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Vizza E, Bruno V, Cutillo G, Mancini E,
Sperduti I, Patrizi L, Certelli C, Zampa A, Giannini A and Corrado
G: Prognostic role of the removed vaginal cuff and its correlation
with L1CAM in low-risk endometrial adenocarcinoma. Cancers (Basel).
14:342021. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Giannini A, D'Oria O, Corrado G, Bruno V,
Sperduti I, Bogani G, Laganà AS, Chiantera V, Caserta D and Vizza
E: The role of L1CAM as predictor of poor prognosis in stage I
endometrial cancer: A systematic review and meta-analysis. Arch
Gynecol Obstet. 309:789–799. 2024. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wu QJ, Zhang TN, Chen HH, Yu XF, Lv JL,
Liu YY, Liu YS, Zheng G, Zhao JQ, Wei YF, et al: The sirtuin family
in health and disease. Signal Transduct Target Ther. 7:4022022.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Yuan H and Marmorstein R: Structural basis
for sirtuin activity and inhibition. J Biol Chem. 287:42428–42435.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Michishita E, Park JY, Burneskis JM,
Barrett JC and Horikawa I: Evolutionarily conserved and
nonconserved cellular localizations and functions of human SIRT
proteins. Mol Biol Cell. 16:4623–4635. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Costa-Machado LF and Fernandez-Marcos PJ:
The sirtuin family in cancer. Cell Cycle. 18:2164–2196. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Zhao E, Hou J, Ke X, Abbas MN, Kausar S,
Zhang L and Cui H: The roles of sirtuin family proteins in cancer
progression. Cancers (Basel). 11:19492019. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Saffar H, Nili F, Sarmadi S, Barazandeh E
and Saffar H: Evaluation of sirtuin1 overexpression by
immunohistochemistry in cervical intraepithelial lesions and
invasive squamous cell carcinoma. Appl Immunohistochem Mol Morphol.
31:128–131. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zeng Z, Huang Y, Li Y, Huang S, Wang J,
Tang Y and Jiang Y: Gene expression and prognosis of sirtuin family
members in ovarian cancer. Medicine (Baltimore). 99:e206852020.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Han O, Alci A, Yildirim HT, Gokkaya M,
Yalcin N, Kandemir S, Goksu M, Ureyen I and Toptas T: βcatenin
expression in endometrioid type endometrial cancer: Expression
patterns and impact on disease outcomes. Oncol Lett. 28:5802024.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Remmele W and Stegner HE: Recommendation
for uniform definition of an immunoreactive score (IRS) for
immunohistochemical estrogen receptor detection (ERICA) in breast
cancer tissue. Pathologe. 8:138–140. 1987.PubMed/NCBI
|
|
21
|
Lin L, Zheng X, Qiu C, Dongol S, Lv Q,
Jiang J, Kong B and Wang C: SIRT1 promotes endometrial tumor growth
by targeting SREBP1 and lipogenesis. Oncol Rep. 32:2831–2835. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang S, Li Y, Sheng G, Meng Q, Hu Q, Gao
X, Shang Z and Lv Q: Sirtuin 1 promotes autophagy and proliferation
of endometrial cancer cells by reducing acetylation level of LC3.
Cell Biol Int. 45:1050–1059. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bartosch C, Monteiro-Reis S, Almeida-Rios
D, Vieira R, Castro A, Moutinho M, Rodrigues M, Graça I, Lopes JM
and Jerónimo C: Assessing sirtuin expression in endometrial
carcinoma and non-neoplastic endometrium. Oncotarget. 7:1144–1154.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Asaka R, Miyamoto T, Yamada Y, Ando H,
Mvunta DH, Kobara H and Shiozawa T: Sirtuin 1 promotes the growth
and cisplatin resistance of endometrial carcinoma cells: A novel
therapeutic target. Lab Invest. 95:1363–1373. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Al-Maghrabi J and Al-Maghrabi H: SIRT1 is
overexpressed in endometrial adenocarcinoma: A tissue microarray
analysis. Eur J Gynaecol Oncol. 41:699–704. 2020. View Article : Google Scholar
|
|
26
|
Beyer S, Chen F, Meister S, Czogalla B,
Kolben TM, Hester A, Burges A, Trillsch F, Schmöckel E, Mayr D, et
al: Sirtuin1 expression and survival in endometrial and clear-cell
uterine cancer. Histochem Cell Biol. 154:189–195. 2020. View Article : Google Scholar : PubMed/NCBI
|