Introduction
Hepatitis B virus (HBV)-associated acute-on-chronic
liver failure (ACLF), characterized by acute exacerbation of
chronic hepatitis B (CHB) or liver cirrhosis (LC), is associated
with a high rate of short-term mortality (1–3). Thus, it
is important to stratify the patients with poor prognosis and
determine the degree of emergency for liver transplantation.
Although a number of scoring systems, including the Model for
End-Stage Liver Disease (MELD) (4)
and MELD-Na (5) schema, are generally
used for evaluating patients with HBV-ACLF, they have limited
accuracy in predicting prognosis, and there is currently no
effective model for predicting the prognosis of patients with ACLF
(1).
Osteopontin (OPN), a secreted phosphorylated
glycoprotein, may participate in the progression of liver injury by
promoting the inflammatory response that induces macrophage and
neutrophil activation and migration (6,7). A number
of studies have suggested that OPN may serve as a biomarker of
severe fibrosis and portal hypertension during Schistosomiasis
mansoni (8) and chronic viral
hepatitis (9,10). Nevertheless, in mice models of liver
injury induced by diethylnitrosamine or ischemia/reperfusion, OPN
exerted a protective effect by ameliorating the production of IL-6
and TNF-α in macrophages and inhibiting inflammation (11,12).
Furthermore, OPN may serve a key role in the expansion of hepatic
progenitor cells (HPC) and in promoting liver regeneration during
liver cancer (13,14) and acute liver failure (15,16).
Thus, as an acute phase protein, OPN may serve
critical roles in the inflammatory response and liver regeneration,
though is considered to exert detrimental effect in liver injury.
In addition, Arai et al (17)
demonstrated that OPN was expressed in inflammatory cells,
hepatocytes and bile ductular structures in patients with fulminant
liver failure. However, the role of OPN in ACLF is yet to be
elucidated. Therefore, the current prospective, observational study
aimed to evaluate the value of OPN in the prediction of 90-day
mortality in patients with ACLF.
Materials and methods
Patients and primary endpoint
Patients with HBV-ACLF (n=54) or CHB (n=81) were
enrolled prospectively from January 2015 to June 2017 at the
Department of Liver Diseases of the Third People's Hospital of
Changzhou (Changzhou, China). Additionally, a total of 20 healthy
individuals without HBV infection were recruited as healthy
controls (HC). The demographic characteristics of all enrolled
subjects are listed in Table I. The
diagnosis of ACLF was based on previously described criteria
(18), as follows: Acute hepatic
insult manifested as jaundice (serum bilirubin ≥5 mg/dl or 85
µmol/l) and coagulopathy [international normalized ratio (INR)
>1.5 or prothrombin activity <40%], complicated within 4
weeks by ascites and/or encephalopathy in a patient with CHB. CHB
was diagnosed according to the Chinese guidelines for the
prevention and treatment of CHB (2015 version) (19). Patients with autoimmune diseases,
immunodeficiency disorders, alcoholic liver disease, liver cancer,
drug-induced liver diseases, blood diseases or co-infection with
other hepatitis viruses were excluded. Blood samples (5 ml) were
collected at the time of diagnosis of HBV-ACLF or CHB, centrifuged
at 1,000 × g for 10 min, and preserved at −80°C. Biochemical
parameters were measured using an automatic analyzer (Hitachi 7600;
Hitachi, Ltd., Tokyo, Japan) and coagulation parameters were
determined using an STA Compact Max® analyzer
(Diagnostica Stago, Inc., Parsippany, NJ, USA). Demographic and
clinical data including sex, age, alanine transaminase (ALT),
aspartate transaminase (AST), total bilirubin (TBil), creatinine,
INR, serum HBV DNA, HBV serological markers, α-fetoprotein (AFP)
and blood cell counts were collected within the first 24 h after
the diagnosis of HBV-ACLF. The end-point of the study was 90-day
mortality or survival following the established diagnosis of
ACLF.
 | Table I.Demographic and clinical
characteristics of study subjects. |
Table I.
Demographic and clinical
characteristics of study subjects.
|
| Group |
|---|
|
|
|
|---|
| Variables | HBV-ACLF
(n=54) | CHB (n=81) | HC (n=20) |
|---|
| Age, years |
46.72±12.26a | 42.44±13.72 | 34.49±10.11 |
| Male, n (%) | 48
(88.89)a | 64 (79.01) | 13 (65.00) |
| HBeAg, positive
rate, n (%) | 10 (18.52) | 21 (25.93) | 0 |
| ALT, U/l |
480.81±604.13a,b |
256.12±78.17a | 22.17±5.29 |
| AST, U/l |
346.28±503.74a,b |
210.13±46.77a | 16.22±8.30 |
| TBil, µmol/l |
305.06±154.82a,b | 19.32±7.91 | 12.19±4.51 |
| Creatinine,
µmol/l |
75.76±21.88a,b | 61.19±16.33 | 62.78±11.39 |
| INR |
2.33±0.95b | 1.40±0.32 | N.D. |
| AFP, ng/ml | 348.31±641.87 | N.D. | N.D. |
| WBC count,
E+09/l |
8.28±4.60a,b | 5.43±1.19 | 4.26±1.63 |
| Neutrophil count,
E+09/l |
6.01±4.19a,b | 3.11±0.78 | 2.99±0.71 |
| Lymphocyte count,
E+09/l | 1.32±0.58 | 0.89±0.23 | 0.92±0.33 |
| Monocyte count,
E+09/l |
0.76±0.36a,b | 0.46±0.37 | 0.41±0.19 |
| Platelet count,
E+09/l |
90.19±51.57a,b | 136.12±21.09 | 155.19±33.10 |
| MELD |
23.07±5.79b | 5.28±2.17 | N.D. |
| MELD-Na | 21.65±11.24 | N.D. | N.D. |
| OPN, ng/ml |
8,490.96±3,750.30a,b |
6,593.82±2,555.39a |
3,880.36±1,644.27 |
The present study was anonymous, and the protocol
was approved by the Ethics Committee of the Third People's Hospital
of Changzhou according to the Declaration of Helsinki 2013. Written
informed consent was obtained from all participants prior to the
study.
ELISA
The serum concentrations of OPN were measured using
a commercial ELISA kit (R&D Systems, Inc., Minneapolis, MN,
USA) according to the manufacturer's instructions. In brief, 50 µl
diluted (1:100) serum samples were added to the ELISA plates
pre-coated with a captured OPN antibody, absorbance was measured at
450 nm using an ELISA plate reader (Synergy HT; BioTek Instruments,
Inc., Winooski, VT, USA), and a duplicate assay was performed;
consequently, a standard curve was generated.
Score systems
MELD (4) and MELD-Na
(5) scores were calculated using the
formulas described previously, as follows: MELD = 11.2 × ln(INR) +
9.6 × ln [creatinine(mg/dl)] + 3.8 × ln[total bilirubin (TBil,
mg/dl)] + 6.4; MELD-Na = MELD + 1.59 × (135-Na+).
Statistical analysis
All data were analyzed using SPSS v17.0 (SPSS, Inc.,
Chicago, IL, USA). Continuous variables were expressed as the mean
± standard deviation, and categorical values were expressed as
frequencies. The differences in variables were analyzed using
Student's t-test or one-way analysis of variance with Fisher's
least significant difference post-hoc test (for normally
distributed data), to determine the significance between the
groups. The comparisons of frequencies were performed using the
χ2 test. Pearson correlation analysis was used to
evaluate correlations between OPN and other clinical indicators.
Survival analysis was performed with Kaplan-Meier curves.
Multivariate Cox proportional hazards regression was employed to
analyze the independent risk factors of mortality. The optimal
cut-off value of OPN and its sensitivity, specificity and positive
and negative prediction values were predicted using SPSS Modeler
14.2 software (IBM Corp., Armonk, NY, USA). A two-sided P<0.05
was considered to indicate a statistically significant
difference.
Results
Characteristics of patients
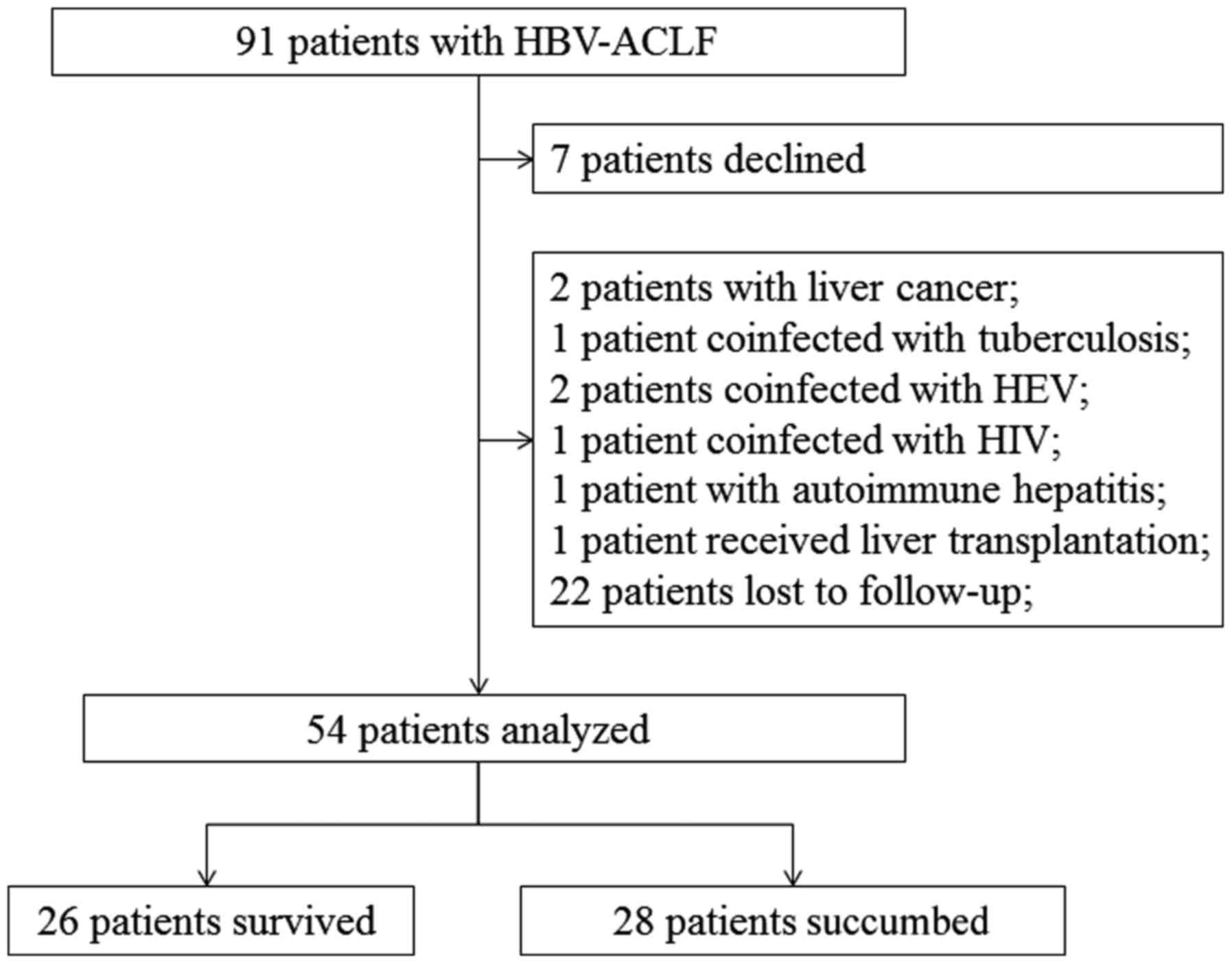
The selection process and outcomes of the patients
under study is illustrated in Fig. 1.
A total of 54 patients with ACLF, who fulfilled the diagnostic
criteria, were recruited prospectively. All of these patients
received antiviral therapy at admission due to the positive HBV DNA
level (>500 IU/ml). The demographic and clinical characteristics
of all subjects in the ACLF, CHB and HC groups are listed in
Table I. Patients with HBV-ACLF had
elevated ALT, AST, TBil, creatinine and white blood cell (WBC)
count compared with the CHB and HC groups (P<0.05). Notably,
serum OPN level in the ACLF group was significantly higher than
that in the CHB and HC groups (both P<0.01; Fig. 2A). Furthermore, serum OPN in patients
with CHB was elevated compared with that in the HC group
(P<0.01). Subsequently, the ACLF patients were divided into
cirrhotic and non-cirrhotic subgroups based on ultrasonography at
admission, and no significant difference was identified in the OPN
levels between the two subgroups (P=0.973; data not shown).
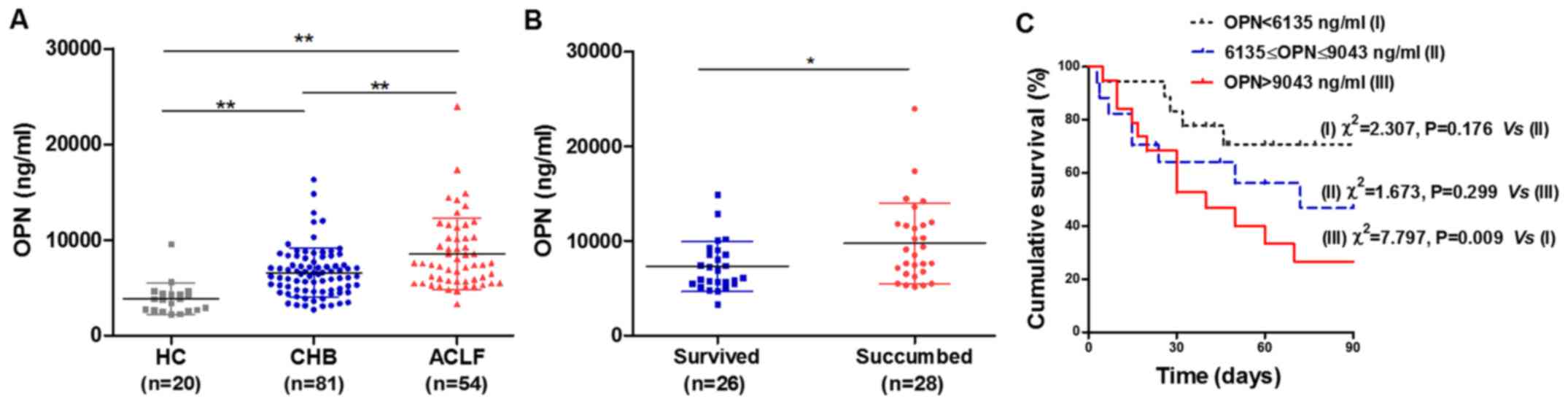 | Figure 2.High OPN level predicts poor
prognosis in HBV-ACLF. (A) Serum OPN in the ACLF group was
significantly higher than that in the CHB and HC groups. (B) OPN in
the non-survivor group was significantly higher when compared with
the surviving patients. (C) Kaplan-Meier survival curves based on
serum OPN to assess predictive value for 90-day mortality. The
90-day mortality rates were 27.78 (5/18), 52.94 (9/17) and 73.68%
(14/19) for the low (OPN<6,135 ng/ml), intermediate (6,135–9,043
ng/ml) and high (>9,043 ng/ml) mortality risk groups,
respectively, and those in the high risk group had a poorer
prognosis compared with the low risk group (P=0.009). *P<0.05
and **P<0.01 as indicated. OPN, osteopontin; ACLF,
acute-on-chronic liver failure; CHB, chronic hepatitis B; HC,
healthy controls. |
OPN in surviving and non-surviving
patients with ACLF
Among the 54 patients with ACLF, 26 patients
survived. No significant differences were observed in age, gender
ratio or LC ratio between the surviving and non-surviving groups.
Significant differences were identified regarding TBil, INR,
lymphocytes, platelet count, MELD, and MELD-Na score (Table II). Notably, OPN level in the
non-surviving group was significantly higher than that in the
surviving group (P<0.05; Fig. 2B
and Table II). Multivariable Cox
regression analysis indicated that MELD [P=0.004, odds ratio (OR)
=1.112, 95% confidence interval (CI): 1.021–1.148] and OPN
(P=0.021, OR=1.104, 95% CI: 1.003–1.116) were independent risk
factors for 90-day mortality (data not shown).
 | Table II.Comparison of the surviving and
non-surviving patients with HBV-ACLF. |
Table II.
Comparison of the surviving and
non-surviving patients with HBV-ACLF.
| Variables | Surviving
(n=26) | Non-surviving
(n=28) | P-value |
|---|
| Age, years | 44.65±13.14 | 48.64±11.28 | 0.236 |
| Male, n (%) | 22 (84.62) | 26 (92.86) | 0.299 |
| Cirrhosis, n
(%) | 14 (53.85) | 18 (64.29) | 0.435 |
| HBeAg, positive
rate, n (%) | 5 (19.23) | 5 (17.86) | 0.586 |
| ALT, U/l | 422.96±592.98 | 534.54±620.19 | 0.503 |
| AST, U/l | 290.65±409.43 | 397.93±580.65 | 0.440 |
| TBil, µmol/l | 231.48±112.32 | 373.38±159.10 | <0.001 |
| Creatinine,
µmol/l | 79.22±21.97 | 72.56±21.69 | 0.268 |
| INR | 1.89±0.53 | 2.72±1.08 | 0.001 |
| AFP, ng/ml | 310.90±388.93 | 383.04±816.13 | 0.684 |
| WBC count,
E+09/l | 7.59±4.59 | 8.91±4.59 | 0.296 |
| Neutrophil count,
E+09/l | 5.31±4.18 | 6.84±4.13 | 0.181 |
| Lymphocyte count,
E+09/l | 1.54±0.66 | 1.12±0.41 | 0.017 |
| Monocyte count,
E+09/l | 0.67±0.28 | 0.85±0.40 | 0.062 |
| Platelet count,
E+09/l | 105.65±61.28 | 75.82±36.06 | 0.032 |
| MELD | 20.39±5.19 | 25.55±5.25 | 0.001 |
| MELD-Na | 17.02±8.91 | 25.94±11.61 | 0.003 |
| OPN, ng/ml |
7,283.32±2,638.39 |
9,730.02±4,276.14 | 0.015 |
The ACLF patients were stratified into three groups
according to serum OPN levels (low mortality risk: <6,135 ng/ml;
intermediate risk: 6,135–9,043 ng/ml; and high risk: >9,043
ng/ml), predicted using SPSS Modeler 14.2 software. For the low,
intermediate and high risk groups, the 90-day mortality rate was
27.78 (5/18), 52.94 (9/17) and 73.68% (14/19), respectively, and
those in the high risk group had a poorer prognosis compared with
the low risk group (P=0.009; Fig.
2C). With an optimal cut-off value of 9,043 ng/ml, OPN achieved
50.00% sensitivity, 80.77% specificity, 73.68% positive prediction
value and 60.00% negative predictive value regarding prediction of
high mortality risk.
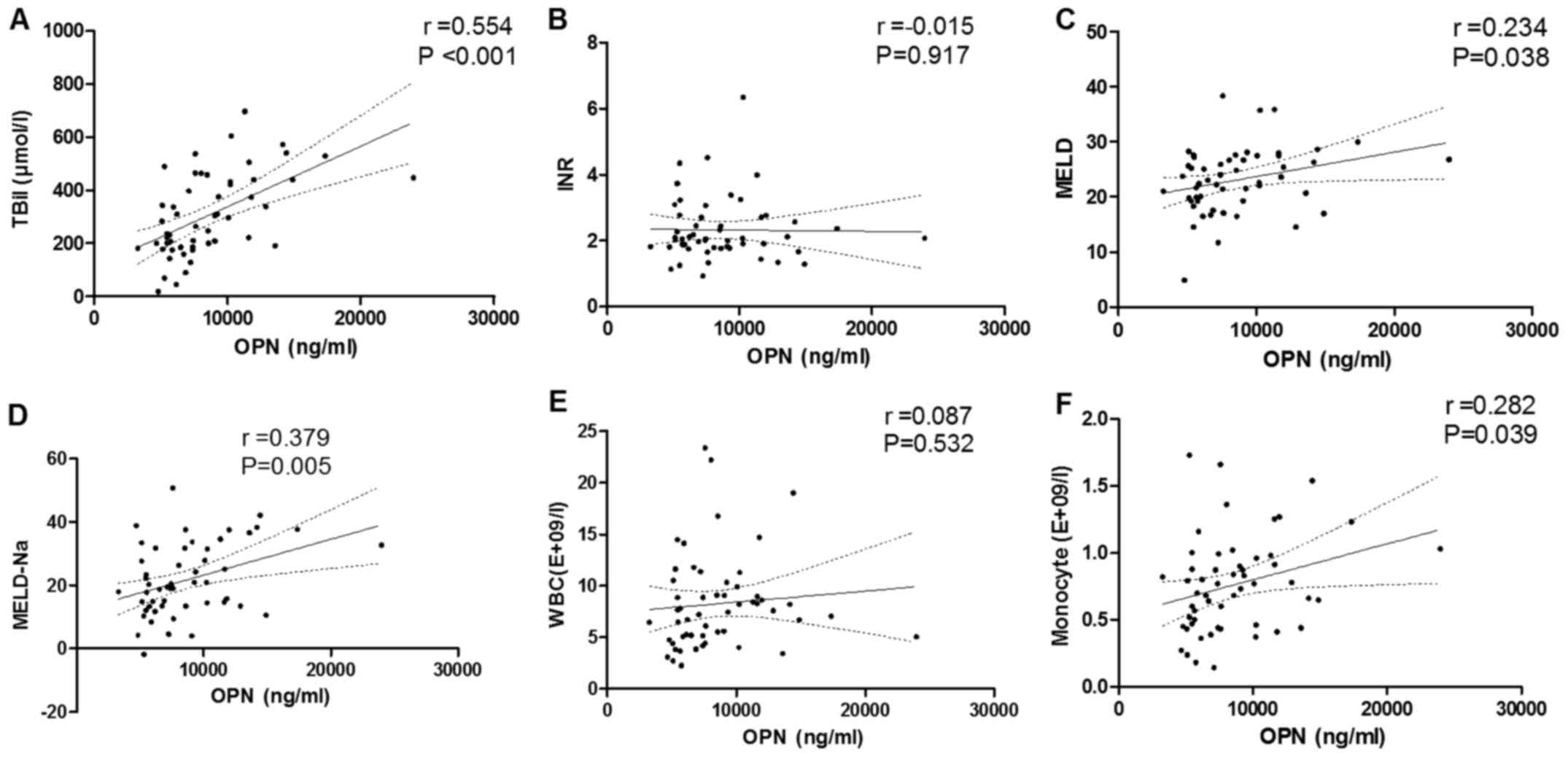
Correlations between OPN and liver
injury indicators
TBil, INR, MELD and MELD-Na scores were indicated as
markers of liver injury severity in ACLF patients (Table II); thus, the correlations of OPN
with these indicators were determined. Serum OPN level was
positively correlated with serum TBil (r=0.554, P<0.001;
Fig. 3A), though not with plasma INR
value (Fig. 3B). A significant
positive correlation was observed between OPN and MELD score
(r=0.234, P=0.038; Fig. 3C), as well
as MELD-Na score (r=0.379, P=0.005; Fig.
3D). Although OPN was not associated with WBC count (Fig. 3E), it was positively correlated with
monocyte count in peripheral blood (r=0.282, P=0.039; Fig. 3F). AFP level and neutrophil,
lymphocyte and platelet counts were not significantly correlated
with OPN (data not shown).
Discussion
In the present study, it was demonstrated that OPN
was significantly higher in HBV-ACLF patients, particularly in
those who succumbed to mortality. Additionally, OPN was positively
correlated with TBil level, MELD and MELD-Na score, and was
implicated as an independent risk factor for prognosis of ACLF.
Furthermore, OPN >9,043 ng/ml predicted a poor prognosis.
As described previously, ACLF is a syndrome
associated with a high rate of short-term mortality, and patients
are typically diagnosed at a late stage when effective treatments
are not applicable except for liver transplantation (1,3).
Considering the high mortality rate, it is important to identify
those at high risk of mortality and refer these patients for liver
transplantation. While evaluation of ACLF patients is recommended,
the procedure is not optimal, owing to inadequate criteria.
Recently, Graupera et al (20)
reported that OPN levels were associated with the 3-month
probability of readmission in patients with liver cirrhosis; thus,
the current study hypothesized and validated that OPN may be
associated with the exacerbation of CHB or LC. Serum OPN level was
higher in ACLF patients, and was positively correlated with MELD
and MELD-Na scores. According to the survival analysis, ACLF
patients with high OPN level (>9,043 ng/ml) should be referred
for liver transplantation at the earliest possible time, due to the
high 90-day mortality rate. Collectively, these results suggested
that OPN and MELD were complementary for the prediction of
prognosis in ACLF patients.
OPN is involved in cell migration and inflammatory
signaling, and previous studies have demonstrated that OPN was
associated with hepatic inflammation and invasiveness in
hepatocellular carcinoma (HCC) (8,9,21). In line with these experimental
observations, an association of circulating OPN levels and HCC
incidence has previously been demonstrated in a population cohort
of more than 520,000 subjects from ten Western European countries
(21). As cell proliferation is among
the key mechanisms in HCC and liver repair (8,22–24), high levels of OPN may suggest tumor
progression or liver regeneration. However, the present study
demonstrated that high levels of OPN predicted poor prognosis in
ACLF. Therefore, it would be premature to conclude that OPN is
protective or beneficial to liver regeneration in ACLF
patients.
Furthermore, Urtasun et al (25) demonstrated that OPN was expressed by
hepatic stellate cells (HSCs), and upregulated collagen I
production in oxidant stress, ultimately leading to fibrogenesis.
Wang et al (26) identified a
role of OPN in HPC activation, and demonstrated that HPC-driven
ductular reaction was a critical mechanism underlying the
progression of liver fibrosis. Another study demonstrated that OPN
neutralization suppressed liver fibrosis in mice (27). As a compensatory mechanism of liver
injury repair, liver fibrosis is exacerbated in ACLF (27,28); thus,
OPN may be a potential therapeutic target during ACLF development.
Additionally, Coombes et al (27) indicated a role of OPN in HPC expansion
and OPN deficiency, which reduced the response of HPCs, thereby
affecting the proliferation ability of hepatocytes. However,
downregulation of OPN may inhibit liver regeneration, which is
critical for the recovery of liver failure. Thus, further studies
are required to identify the role of OPN in experimental models of
liver failure.
Previous studies have demonstrated that two forms of
OPN coexist: An intracellular form (iOPN) and a secreted form
(sOPN) (15,29,30).
Although sOPN was measured in the present study, further studies
are imperative to investigate the role of iOPN in lymphocytes and
dendritic cells. Furthermore, as sOPN may be produced by many cell
types, including hepatocytes, cholangiocytes, HSCs, macrophages, T
lymphocytes and natural killer T cells, identifying the primary
sources of OPN in liver failure is challenging (6,28,31). In the present study, Pearson's
correlation analysis indicated that OPN levels were not associated
with WBC or lymphocyte count; however, a positive correlation was
established with monocyte count in the peripheral blood. This
result suggests that monocytes may contribute to the production of
sOPN in ACLF patients. Thus, immunohistochemistry may be useful for
investigating the role iOPN and the origin of sOPN in future
studies.
Nonetheless, the present study had several
limitations. First, although OPN was associated with prognosis in
ACLF, the mechanisms underlying the role of OPN were not
investigated. Second, the dynamic changes in serum OPN should be
determined during the progression of ACLF, along with an evaluation
of long-term prognostic value. Finally, multicenter studies are
also required for confirmation of the current findings.
In conclusion, serum OPN was significantly higher in
patients with ACLF and correlated with indicators of liver injury.
Additionally, it was an independent risk factor associated with
prognosis. Collectively the present findings suggest that OPN may
be a predictor of prognosis in patients with HBV-ACLF.
Acknowledgements
The present study was supported by the Chinese
Foundation for Hepatitis Prevention and Control-Tianqing Liver
Disease Research Fund Subject (grant nos. TQGB201700139 and
TQGB20150006), the Major Projects of Changzhou Municipal Health and
Family Planning Commission (grant no. ZD2011002) and the Projects
of Changzhou Science and Technology Commission (grant no.
CJ20160024). Yuan Xue conceived and designed the study. Longgen Liu
and Jianchun Lu performed the experiments. Chunyan Ye, Lin Lin,
Shuqin Zheng, Hongyu Zhang and Qing Lan collected and confirmed the
data. Yuan Xue, Longgen Liu and Jianchun Lu performed the analysis
and drafted the manuscript. All authors read and approved the final
manuscript.
Glossary
Abbreviations
Abbreviations:
|
HBV
|
hepatitis B virus
|
|
ACLF
|
acute-on-chronic liver failure
|
|
LC
|
liver cirrhosis
|
|
CHB
|
chronic hepatitis B
|
|
OPN
|
osteopontin
|
|
HPC
|
hepatic progenitor cell
|
|
HCC
|
hepatocellular carcinoma
|
|
HSC
|
hepatic stellate cell
|
|
MELD
|
model for end-stage liver disease
|
|
ALT
|
alanine aminotransferase
|
|
AST
|
aspartate aminotransferase
|
|
TBil
|
total bilirubin
|
|
INR
|
international normalized ratio
|
|
WBC
|
white blood cell
|
|
AFP
|
α-fetoprotein
|
|
r
|
correlation coefficient
|
|
CI
|
confidence interval
|
|
HC
|
healthy control
|
|
OR
|
odds ratio
|
References
|
1
|
Hernaez R, Solà E, Moreau R and Ginès P:
Acute-on-chronic liver failure: An update. Gut. 66:541–553. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Arroyo V and Jalan R: Acute-on-chronic
liver failure: Definition, diagnosis, and clinical characteristics.
Semin Liver Dis. 36:109–116. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Arroyo V and Moreau R: Diagnosis and
prognosis of acute on chronic liver failure (ACLF) in cirrhosis. J
Hepatol. 66:451–453. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kamath PS, Wiesner RH, Malinchoc M,
Kremers W, Therneau TM, Kosberg CL, D'Amico G, Dickson ER and Kim
WR: A model to predict survival in patients with end-stage liver
disease. Hepatology. 33:464–470. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Biggins SW, Kim WR, Terrault NA, Saab S,
Balan V, Schiano T, Benson J, Therneau T, Kremers W, Wiesner R, et
al: Evidence-based incorporation of serum sodium concentration into
MELD. Gastroenterology. 130:1652–1660. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Diao H, Iwabuchi K, Li L, Onoe K, Van Kaer
L, Kon S, Saito Y, Morimoto J, Denhardt DT, Rittling S, et al:
Osteopontin regulates development and function of invariant natural
killer T cells. Proc Natl Acad Sci USA. 105:pp. 15884–15889. 2008;
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Diao H, Kon S, Iwabuchi K, Kimura C,
Morimoto J, Ito D, Segawa T, Maeda M, Hamuro J, Nakayama T, et al:
Osteopontin as a mediator of NKT cell function in T cell-mediated
liver diseases. Immunity. 21:539–550. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pereira TA, Syn WK, Pereira FE,
Lambertucci JR, Secor WE and Diehl AM: Serum osteopontin is a
biomarker of severe fibrosis and portal hypertension in human and
murine schistosomiasis mansoni. Int J Parasitol. 46:829–832. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Matsue Y, Tsutsumi M, Hayashi N, Saito T,
Tsuchishima M, Toshikuni N, Arisawa T and George J: Serum
osteopontin predicts degree of hepatic fibrosis and serves as a
biomarker in patients with hepatitis C virus infection. PLoS One.
10:e01187442015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhao L, Li T, Wang Y, Pan Y, Ning H, Hui
X, Xie H, Wang J, Han Y, Liu Z, et al: Elevated plasma osteopontin
level is predictive of cirrhosis in patients with hepatitis B
infection. Int J Clin Pract. 62:1056–1062. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fan X, He C, Jing W, Zhou X, Chen R, Cao
L, Zhu M, Jia R, Wang H, Guo Y, et al: Intracellular Osteopontin
inhibits toll-like receptor signaling and impedes liver
carcinogenesis. Cancer Res. 75:86–97. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Patouraux S, Rousseau D, Rubio A,
Bonnafous S, Lavallard VJ, Lauron J, Saint-Paul MC, Bailly-Maitre
B, Tran A, Crenesse D, et al: Osteopontin deficiency aggravates
hepatic injury induced by ischemia-reperfusion in mice. Cell Death
Dis. 5:e12082014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Liu Y, Cao L, Chen R, Zhou X, Fan X, Liang
Y, Jia R, Wang H, Liu G, Guo Y, et al: Osteopontin Promotes Hepatic
Progenitor Cell Expansion and Tumorigenicity via Activation of
β-Catenin in Mice. Stem Cells. 33:3569–3580. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lee SH, Park JW, Woo SH, Go DM, Kwon HJ,
Jang JJ and Kim DY: Suppression of osteopontin inhibits chemically
induced hepatic carcinogenesis by induction of apoptosis in mice.
Oncotarget. 7:87219–87231. 2016.PubMed/NCBI
|
|
15
|
Srungaram P, Rule JA, Yuan HJ, Reimold A,
Dahl B, Sanders C and Lee WM: Acute Liver Failure Study Group:
Plasma osteopontin in acute liver failure. Cytokine. 73:270–276.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tajiri T, Tate G, Kunimura T, Endo Y,
Inoue K, Mitsuya T, Morohoshi T and Yoshiba M: Osteopontin
expression in proliferated bile ductules: The correlation with
liver damage in fulminant hepatitis. Dig Dis Sci. 50:188–195. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Arai M, Yokosuka O, Kanda T, Fukai K,
Imazeki F, Muramatsu M, Seki N, Miyazaki M, Ochiai T, Hirasawa H,
et al: Serum osteopontin levels in patients with acute liver
dysfunction. Scand J Gastroenterol. 41:102–110. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sarin SK, Kedarisetty CK, Abbas Z,
Amarapurkar D, Bihari C, Chan AC, Chawla YK, Dokmeci AK, Garg H,
Ghazinyan H, et al: APASL ACLF Working Party: Acute-on-chronic
liver failure: Consensus recommendations of the Asian Pacific
Association for the Study of the Liver (APASL) 2014. Hepatol Int.
8:453–471. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Tang CM, Yau TO and Yu J: Management of
chronic hepatitis B infection: Current treatment guidelines,
challenges, and new developments. World J Gastroenterol.
20:6262–6278. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Graupera I, Solà E, Fabrellas N, Moreira
R, Solé C, Huelin P, de la Prada G, Pose E, Ariza X, Risso A, et
al: Urine Monocyte Chemoattractant Protein-1 Is an Independent
Predictive Factor of Hospital Readmission and Survival in
Cirrhosis. PLoS One. 11:e01573712016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Duarte-Salles T, Misra S, Stepien M,
Plymoth A, Muller D, Overvad K, Olsen A, Tjønneland A, Baglietto L,
Severi G, et al: Circulating osteopontin and prediction of
hepatocellular carcinoma development in a large European
population. Cancer Prev Res (Phila). 9:758–765. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rastogi A, Bihari C, Maiwall R, Ahuja A,
Sharma MK, Kumar A and Sarin SK: Hepatic stellate cells are
involved in the pathogenesis of acute-on-chronic liver failure
(ACLF). Virchows Arch. 461:393–398. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shih SC, Ho TC, Chen SL and Tsao YP:
Pigment epithelium-derived factor (PEDF) peptide promotes the
expansion of hepatic stem/progenitor cells via ERK and
STAT3-dependent signaling. Am J Transl Res. 9:1114–1126.
2017.PubMed/NCBI
|
|
24
|
Wang G, Zhao C, Chen S, Li X, Zhang L,
Chang C and Xu C: A preliminary in vivo study of the effects of OPN
on rat liver regeneration induced by partial hepatectomy. Mol Biol
Rep. 43:1371–1382. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Urtasun R, Lopategi A, George J, Leung TM,
Lu Y, Wang X, Ge X, Fiel MI and Nieto N: Osteopontin, an oxidant
stress sensitive cytokine, up-regulates collagen-I via integrin
α(V)β(3) engagement and PI3K/pAkt/NFκB signaling. Hepatology.
55:594–608. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Wang X, Lopategi A, Ge X, Lu Y, Kitamura
N, Urtasun R, Leung TM, Fiel MI and Nieto N: Osteopontin induces
ductular reaction contributing to liver fibrosis. Gut.
63:1805–1818. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Coombes JD, Swiderska-Syn M, Dollé L, Reid
D, Eksteen B, Claridge L, Briones-Orta MA, Shetty S, Oo YH, Riva A,
et al: Osteopontin neutralisation abrogates the liver progenitor
cell response and fibrogenesis in mice. Gut. 64:1120–1131. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Strazzabosco M, Fabris L and Albano E:
Osteopontin: A new player in regulating hepatic ductular reaction
and hepatic progenitor cell responses during chronic liver injury.
Gut. 63:1693–1694. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Uede T: Osteopontin, intrinsic tissue
regulator of intractable inflammatory diseases. Pathol Int.
61:265–280. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Inoue M and Shinohara ML: Intracellular
osteopontin (iOPN) and immunity. Immunol Res. 49:160–172. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Nagoshi S: Osteopontin: Versatile
modulator of liver diseases. Hepatol Res. 44:22–30. 2014.
View Article : Google Scholar : PubMed/NCBI
|