Introduction
Otitis media is reported as one of the most common
diseases due to viral, and fungal pathogens and bacterial infection
(1). The pathophysiology,
progression, as well as pathogenesis of otitis media are affected
by various factors, including pathogenicity, reactive oxygen
species (ROS) generation and inflammatory response (2,3).
For instance, the gram-negative bacillus Nontypeable Haemophilus
influenzae (NTHi) is a main cause, leading to one third of
otitis media. Presently, ~10–20% children experience recurrence and
persistence of otitis media with long-term loss of hearing
(4,5). Now, finding effective therapy is
urgent for clinical treatment due to the large use of antibiotics,
causing resistance for otitis media treatment.
Inflammatory response has been reported to be of
great importance in otitis media formation. Otitis media is
characterized by inflammation (6). The formation of otitis media,
epithelial cells play a vital role in defending numorous stresses,
including pro-inflammatory cytokine secretion (7). Appropriate inflammation is essential
for removing different pathogen. However, overexpression of
pro-inflammatory cytokines results in cell or tissue injury
(8). The chemokine CXC receptor 4
(CXCR4) is mainly activated by stromal cell-derived factor
(SDF-1α). CXCR4 may be a part of a lipopolysaccharide (LPS) sensing
for co-clustering complex that regulates TLRs signaling pathway to
suppress inflammation (9,10). Previously, CXCR4 in mediating
inflammatory response has been widely suggested, the mechanism
indicating CXCR4 expression in NHTi-induced otitis media in
vitro and in vivo are not known.
Quercetin, an important flavonoid antioxidant, found
in red apples as well as broccoli, rutin and green tea, helps blood
pressure balance, fight asthma and allergies, and prevents
angiocardiopathy and tumor progress (11,12). Quercetin as a well-known flavonoid
with various biological effects has been widely used and evidenced
in many disease models. Due to the tolerability and non-toxicity of
quercetin, it could be used for prolonged periods in the absence of
any side effects (13,14). However, though it is well known in
suppressing inflammation response, the effect of quercetin on
otitis media improvement through CXCR4 modulation is unknown. In
the study, we explored the possible molecular mechanism of
NTHi-stimulated CXCR4 expression. NTHi increases CXCR4 expression
through IKKα and p38 MAPK signaling pathways activation. In
addition, quercetin inhibited CXCR4 expression induced by NTHi in
human middle ear epithelial cells as well as in tissue samples.
Quercetin inhibited CXCR4 expression through inactivating IKKα and
p38 MAPK phosphorylation, providing effective therapeutic strategy
for otitis media treatment by inhibiting CXCR4 expression.
Materials and methods
Treatment of animals
Seventy-five male, 6–8-week-old C57BL/6 mice
weighing 20–22 g, were purchased from Shanghai Laboratory. Animal
Research Center (Shanghai, China). The mice were housed in a
constant temperature of 22±2°C and relative humidity of 60±10%
environment under 12 h light/dark cycles. The mice were then
inoculated with NTHi trans-tympanically at a dose of
5×107 CFU in each mouse. In the control group, saline
was inoculated. For quercetin studies in vivo, experimental
mice were then intraperitoneally (i.p) injected with quercetin (20,
40 and 80 mg/kg) purchased from Shaanxi Huike Botanical Development
Co., Ltd. (Shaanxi, China) 2 h after NTHi inoculation for 6 h. For
mRNA analysis, mice were sacrificed 6 h post-NTHi inoculation.
Total RNA was extracted from the dissected mouse middle ear.
Finally, all mice were sacrificed and the eyeball blood was
collected and centrifuged at 15,000 × g for 20 min prepared for
following research. Middle ear were then isolated immediately on
ice and stored at −80°C for further research. All animal studies
here were performed in accordance with the guidelines of, and were
approved by, the First Affiliated Hospital of Jinan University.
Cell and bacterial culture and
treatment
Human middle ear epithelial cells (HMEECs), renal
epithelial cells (HK2), and mouse lung epithelial cells (MLE-12)
were purchased from American Type Culture Collection (ATCC,
Manassas, VA, USA). The human liver epithelial cells (L02) and
human lung epithelial cells (BEAS-2B) were purchased from the
KeyGen Biotech (Nanjing, China). HMEECs, L02 and BEAS-2B cells were
routinely cultured in RPMI-1640 medium, containing 10% fetal bovine
serum (FBS) (both from Gibco, Grand Island, NY, USA), 1%
penicillin/streptomycin. The cell lines HK2 and MLE-12 were
cultured in DMEM (Gibco) supplemented with 10% FBS, 100 U/ml
penicillin, and 100 µg/ml streptomycin. The cells were kept
in a humidified atmosphere with 5% CO2 and 95% humidity
at 37°C in an incubator.
Clinical isolates of NTHi strains 2019, 12 and 2866
were included in the present study (15,16). NTHi was cultured on chocolate agar
plate under 5% CO2 atmosphere for 16 h, which is
followed by culture overnight in the brain heart infusion (BHI)
broth supplemented with 10 µg/ml hemoglobin and 3.5
µg/ml NAD (BD Biosciences, Franklin Lakes, NJ, USA). Next,
bacteria were then subcultured in fresh BHI broth (5 ml) and the
growth situation was monitored by assessment of optical density
(OD). Bacteria in log phase were collected, washed and suspended in
medium for experiments in vitro and isotonic saline for
experiments in vivo. For in vitro experiments across
our study the cells were cultured with NTHi at a multiplicity of
infection (MOI) of 50. Cells were induced with NTHi for 6 h, or as
indicated in our study. For suppression study, cells were
pretreated with the specific inhibitor for 2 h ahead of NTHi
induction. For post-treatment studies cells were treated with
quercetin at different concentrations (40, 80 and 120 µM) 2
h after NTHi stimulation. NF-κB inhibitor, PDTC, and IKKα
inhibitor, MRT67307 as well as p38 inhibitor SB203580 were
purchased from Biovision (Milpitas, CA, USA), MedChem Express
(Monmouth Junction. NJ, USA) and Beyotime (Shanghai, China)
respectively.
Enzyme-linked immunosorbent assay (ELISA)
method analysis
Concentrations of CXCR4 in the middle ear effusions
of mice were determined by the ELISA with the mouse enzyme
immunoassay sets (R&D Systems, Inc., Minneapolis, MN, USA)
following the manufacturer's instructions. The samples were
performed in duplicate.
Transfection, plasmids and luciferase
analysis
The expression plasmid investigation, for mutations
(Mut) of TLR3, TLR4, MyD88, IRAK1, TAK1, TRAF6, p38, and
constitutively active form of NF-κB have been described previously
(17). NF-κB luciferase reporter
vector was purchased from Promega (Madison, WI, USA). All transient
transfections were carried out with Lipofectamine™ 2000
Transfection Reagent (Lipo 2000; Invitrogen, Carlsbad, CA, USA)
according to the manufacturer's protocol. HMEECs were analyzed
after transfection for 48 h. The empty vector was transfected as a
control. The pSV-β-galactosidase vector was performed as a control
for luciferase activity assay. The luciferase activity as well as
pSV-β-galactosidase activity was determined with luciferase assay
system and pSV-β-galactosidase (both from Promega). NF-κB
luciferase activity was normalized regarding
pSV-β-galactosidase.
Gene knockdown treatment
Human siRNA (TLR3, TLR4, MyD88, IRAK1, TRAF6 and
TAK1; IKKα; NF-κB) were obtained from Generay Biotech Co., Ltd.
(Shanghai, China). HMEECs were transfected with 20 nM siRNA with
Lipofectamine™ 2000 transfection reagent (Lipo 2000; Invitrogen)
following the manufacturer's protocol.
Western blot assays
The HEMMCs and the middle ear tissues were
harvested. Proteins were extracted from the middle ear tissue
samples using T-PER tissue protein extraction reagent kit (Thermo
Fisher Scientific, Waltham, MA, USA) according to the
manufacturer's instructions. Protein concentrations were determined
by BCA protein assay kit, and equal amounts of protein were loaded
per well on a 10% sodium dodecyl sulphate-polyacrylamide (SDS) gel.
Subsequently, proteins were transferred onto polyvinylidene
difluoride (PVDF) membrane. The resulting membrane was blocked with
Tris-buffered saline containing 0.05% Tween-20 (TBS-T),
supplemented with 5% skim milk (Sigma) at room temperature for 2 h
on a rotary shaker, and followed by TBS-T washing 5 times. The
specific primary antibody, diluted in TBST, was incubated with the
membrane at 4°C overnight. Subsequently, the membrane was washed
with TBS-T followed by incubation with the peroxidase-conjugated
secondary antibody at room temperature for 2 h. The immunoactive
proteins were detected by using an enhanced chemiluminescence
western blot detection kit. Western blot bands were observed using
GE Healthcare ECL western blotting analysis system and exposed to
Kodak X-ray film. The primary antibodies used are shown in Table I.
 | Table IPrimary antibodies for western blot
analysis. |
Table I
Primary antibodies for western blot
analysis.
| Primary
antibodies | Dilution ratio | Corporation |
|---|
| Rabbit
anti-p-IKKα | 1:1,000 | Abcam |
| Rabbit
anti-IKKα | 1:1,000 | Abcam |
| Rabbit
anti-p-p38 | 1:1,000 | Cell Signaling
Technology |
| Rabbit
anti-p38 | 1:1,000 | Cell Signaling
Technology |
| GAPDH | 1:200 | Santa Cruz
Biotechnology |
Real-time (RT) quantitative PCR (Q-PCR)
analysis
Total RNA from the middle ear tissue samples and
cells under different conditions were isolated using TRIzol
(Invitrogen) following the manufacturer's instructions. The cDNA
was synthesized using SuperScript II reverse transcriptase (Thermo
Fisher Scientific). Quantitative PCR was performed with SYBR-Green
Real-Time PCR Master mix (Thermo Fisher Scientific). The
quantitative expression data were collected and analyzed by a 7900
Real-time PCR system (Applied Biosystems, Foster City, CA, USA).
Primers were designed to determine endogenous genes and
glyceraldehyde 3-phosphate dehydrogenase (GAPDH) using as the
endogenous control. Human (h) and mouse (m) primer sequences were:
forward hCXCR4, (5′-3′) TTA CCT ATA TTC TCG GCG TGG ACA G and
reverse primers, (5′-3′) CTC GAT GGT CAT GAC TAA GTG TTC; forward
mCXCR4, (5′-3′) GGA TAC TTG TAC GAG CAA GC and reverse primers,
(5′-3′) GGA GTG GTG CTT GAG TTG ATT; forward GAPDH, (5′-3′) GAC GGA
GCT GAG AAC ATG T and reverse primers, (5′-3′) TGT CCG CGT ATT ATG
AGA TG.
Immunofluorescence analysis
For fluorescent analysis, the mouse middle ear
tissue samples were carefully isolated and fixed in 4%
paraformaldehyde for 16 h after cold 4% paraformaldehyde perfusion.
Then, optimum cutting temperature (OCT) package tissues were cut to
20–30 µm sections. The tissues were incubated with primary
antibodies (p-IKKα and p38) at 4°C overnight after deparaffinized
and rehydrated. Fluorophore-conjugated secondary antibodies were
treated 1 h at 25°C. The Alexa Fluor 488 labeled anti-rabbit or
anti-mouse secondary antibodies (Invitrogen) were used. Sections
were subjected to immunofluorescence staining via epifluorescence
microscopy (Sunny Co., Beijing, China). Leica TCS SP5 confocal
microscope (Leica, Richmond Hill, ON, Canada) was used to obtain
images in a blinded manner with respect to treatment groups.
Histochemical assays
Histopathologic evaluation was performed on mice.
Mouse middle ear tissue samples were fixed with 10% buffered
formalin, imbedded in paraffin, and sliced. After hematoxylin and
eosin (H&E) staining, pathological changes of the tissues were
observed under a light microscope.
Statistical analysis
Data are expressed as means ± SEM Treated tissues
and the corresponding controls were compared using GraphPad PRISM
(version 6.0; GraphPad Software, La Jolla, CA, USA) by a one-way
ANOVA with Dunn's least significant difference tests. Differences
between groups were considered significant at p<0.05.
Results
CXCR4 is activated by NTHi in epithelial
cells of middle ear
Epithelial cells are reported to be of great
importance against various injuries under different stresses
through inflammation response regulation (18,19). Hence, here we attempted to
calculate if NTHi could induce CXCR4 activation in epithelial cells
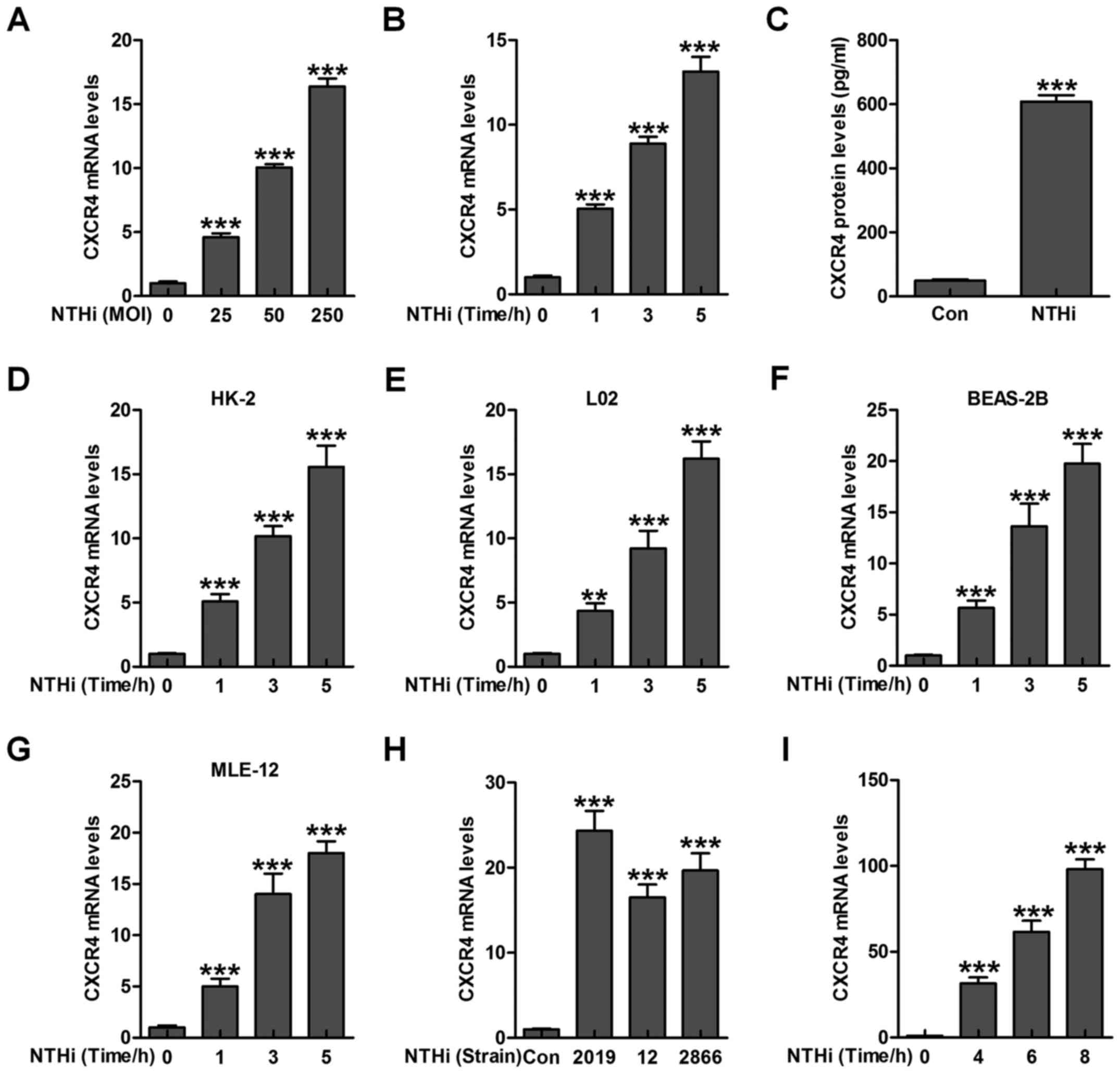
from human middle ear (HMEEC). As shown in Fig. 1A and B, CXCR4 expression was
highly activated after NTHi stimulation, shown in a dose- and
time-dependent manner. In addition, CXCR4 protein levels in HMEEC
cells were significantly upregulated due to NTHi exposure through
ELISA method (Fig. 1C). Moreover,
CXCR4 levels in human renal epithelial HK-2 cells, human liver
epithelial L02 cells, human lung epithelial BEAS-2B cells as well
as in the mouse lung epithelial MLE-12 cells were detected by
RT-qPCR assays. CXCR4 gene levels were highly upregulated in HMEECs
after NTHi stimulation in a time-dependent manner (Fig. 1D–G). Another two stains of NTHi,
12 and 2866, reported to be effective in inducing otitis media,
were also used to investigate whether CXCR4 could be upregulated.
As shown in Fig. 1H, increased
CXCR4 mRNA levels were observed in NTHi-induced HMEECs compared to
the Con group. Consistently, CXCR4 mRNA levels were also stimulated
in the mouse middle ear tissue samples induced by NTHi (Fig. 1I). Collectively, the data above
indicated that CXCR4 may be of essential importance in NTHi-induced
otitis media.
NTHi-stimulated CXCR4 activation relies
on TLR3/MyD88 signaling pathway
TLRs are well known as vital cell surface receptors,
playing an essential role in regulating inflammation response
against various pathogens under different conditions (20). Up until now, ~13 TLRs have been
reported. Among these receptors, TLR3 and TLR4 are significant in
realizing LPS, which is a Gram-negative bacteria characteristic
(21). TLRs are involved in
NTHi-induced inflammatory response, which may be also related to
CXCR4 activation. Thus, here the HMEECs were transfected with TLR3
and TLR4 mutants. Of note, TLR3 mutant in high expression
apparently reduced NTHi-induced CXCR4 expression at the gene levels
(Fig. 2A). However, no
significant difference related to CXCR4 expression was observed in
TLR4-mutation cells after NTHi stimulation compared to the
TLR3-Mut−/TLR4-Mut− group. The TLRs
down-streaming signals, MyD88/IRAK1, and TRAF6/TAK1 were also
investigated. As shown in Fig. 2B and
C, we found that both MyD88 and IRAK1 mutations downregulated
CXCR4 mRNA levels in NTHi-treated cells. Also, TRAF6/TAK1 mutations
dramatically decreased CXCR4 mRNA expression through RT-qPCR
analysis. Moreover, depletion of TLR3 with TLR3 siRNA also
decreased CXCR4 expression from the gene levels. In the results
above, TLR4 knockdown showed no significant difference on CXCR4
gene expression (Fig. 2D).
Furthermore, MyD88 and IRAK1 silence through specific RNA knockdown
reduced CXCR4 expression in HMEECs after NTHi induction (Fig. 2E). Finally, in order to further
confirm TRAF6 and TAK1 in CXCR4 regulation, we found that TRAF6 and
TAK1 silence considerably reduced CXCR4 gene expression levels
(Fig. 2F). The data above
indicated that TLR3/MyD88/IRAK1/TRAF6/TAK1 signaling pathway was,
at least partly, involved in NTHi-induced otitis media.
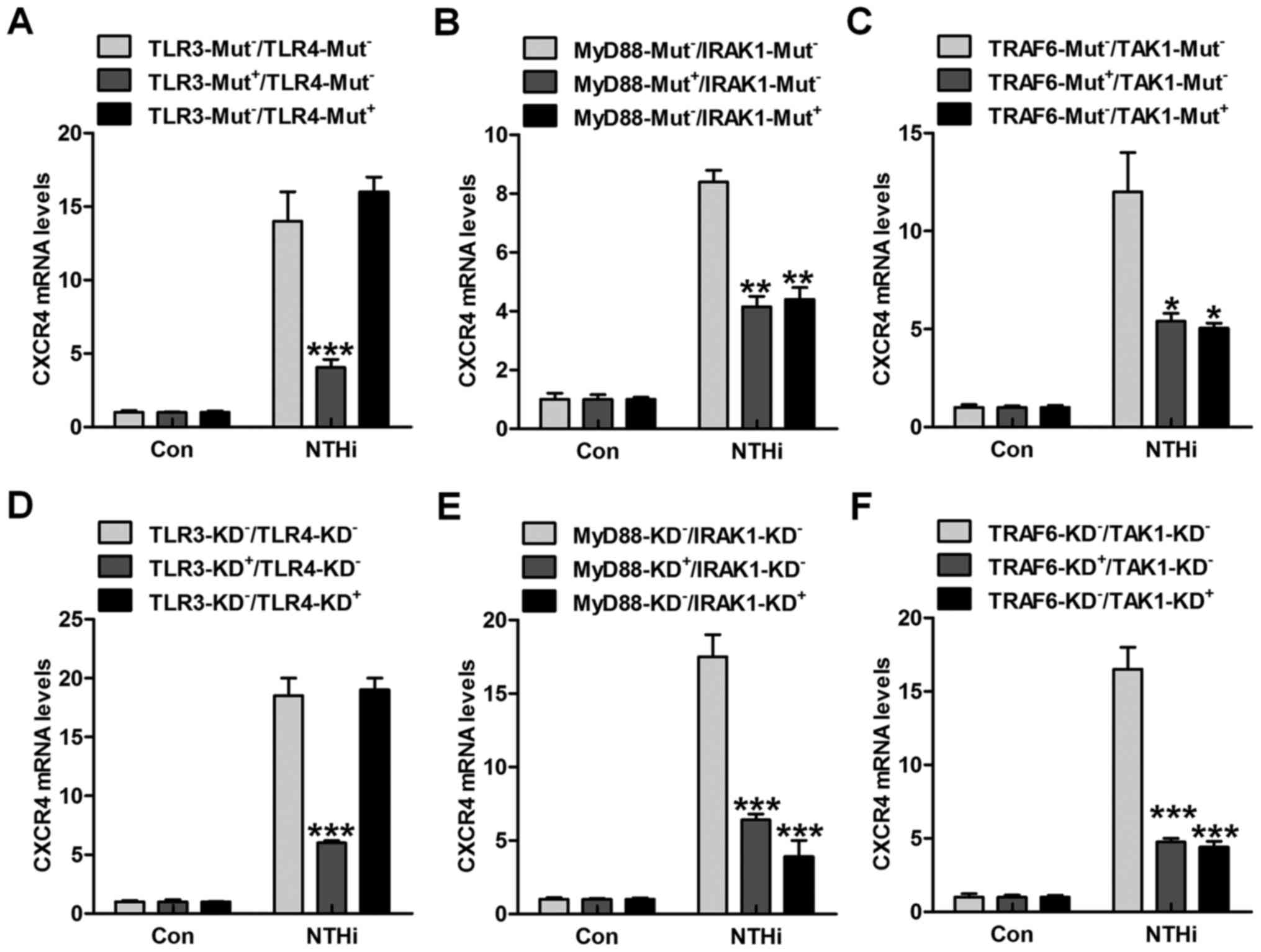 | Figure 2Nontypeable Haemophilus
influenzae (NTHi)-stimulated chemokine CXC receptor 4 (CXCR4)
activation relies on TLR3/MyD88 signaling pathway. human middle ear
epithelial cells (HMEECs) were transfected with the control, (A)
TLR3-, TLR4-, (B) MyD88-, IRAK1-, (C) TRAF6- and TAK1-mutant
plasmids. Then, the cells were exposed to NTHi for 6 h. The CXCR4
mRNA levels were then measured through RT-qPCR analysis. HMEECs
were transfected with the Control siRNA, (D) TLR3-, TLR4-, (E)
MyD88-, IRAK1-, (F) TRAF6- and TAK1-siRNA for knockdown. CXCR4 gene
levels in cells were evaluated via RT-qPCR analysis. Representative
data are shown as SEM. *p<0.05,
**p<0.01 and ***p<0.001 vs. the group
in the absence of gene mutants or silence. |
NTHi-induced CXCR4 activation is related
to IKKα and p38 MAPK activity
According to previous studies, IKKα and p38 MAPK are
two important signaling pathways in regulating otitis media
progression induced by NTHi (22). IKKα is considered as an essential
molecule, activated by TLRs/MyD88 signaling pathway, contributing
to inflammation response (23).
As shown in Fig. 3A, we found
that IKKα phosphorylation was upregulated due to NTHi stimulation.
Next, IKKα inhibitor MRT67307, was used to suppress IKKα
expression, and with the increasing of MRT67307 concentration,
CXCR4 mRNA levels were reduced, which was comparable to the
NTHi-treated group in the absence of MRT67307 (Fig. 3B). Moreover, IKKα mutation
significantly reduced CXCR4 mRNA levels in HMEECs stimulated by
NTHi (Fig. 3C). Similarly, IKKα
knockdown indicated that CXCR4 mRNA levels were decreased after
NTHi stimulation (Fig. 3D).
Fig. 3E suggested that IKKα was
successfully silenced for knockdown to inhibit its activation. In
contrast, IKKα activator was used to improve its phosphorylation,
leading to CXCR4 upregulation in a dose-dependent manner (Fig. 3F). Additionally, p38
phosphorylated levels were evaluated after NTHi stimulation in
HMEECs. p38 phosphorylation was also upregulated with the
increasing time of NTHi induction (Fig. 3G). Further, p38 inhibitor SB203580
usage reduced CXCR4 mRNA levels in a dose-dependent manner
(Fig. 3H). Finally, p38 mutation
was included to decrease CXCR4 mRNA levels through RT-qPCR analysis
(Fig. 3I). The data above
illustrated that IKKα and p38 signaling pathways were included in
NTHi-induced otitis media.
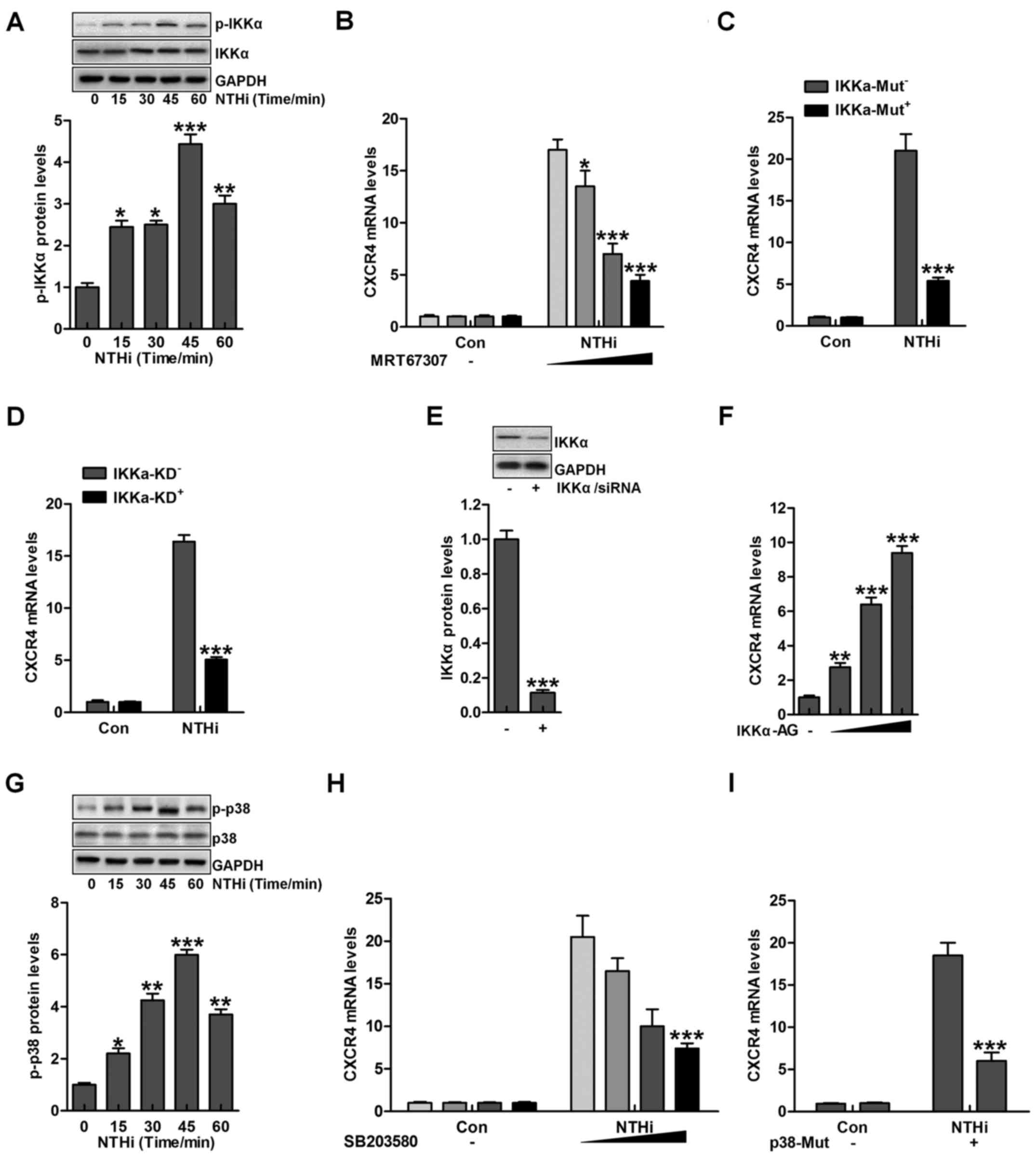 | Figure 3Nontypeable Haemophilus
influenzae (NTHi)-induced chemokine CXC receptor 4 (CXCR4)
activation is related to IKKα and p38 MAPK activity. (A) Human
middle ear epithelial cells (HMEECs) were exposed to NTHi for
different times ranging from 0 to 60 min. Western blot analysis was
used to investigate IKKα phosphorylation. *p<0.05,
**p<0.01 and ***p<0.001 vs. the control
group. (B) HMEECs were administered to IKKα inhibitor MRT67307 at
various concentrations (0, 0.5, 1 and 2 µM) for 2 h,
followed by CXCR4 gene level measurement through RT-qPCR analysis.
*p<0.05, **p<0.01 and
***p<0.001 vs. the NTHi-treated group in the absence
of MRT67307 treatment. (C) HMEECs were transfected with the
Control, and IKKα-mutant plasmid. CXCR4 expression from the gene
levels was determined by the use of RT-qPCR analysis.
***p<0.001 vs. the NTHi-treated group without IKKα
mutation. (D) HMEECs were transfected with the Control, and
IKKα-silence for knockdown. CXCR4 mRNA expression levels were
detected by RT-qPCR assays. ***p<0.001 vs. the
NTHi-treated group without IKKα silence. (E) After IKKα knockdown,
western blot analysis was carried out to calculate CXCR4 expression
from the protein levels. ***p<0.001 vs. the control
group. (F) HMEECs were treated with or without IKK activator at
different concentrations (0, 0.5, 1 and 2 µM) for 12 h.
Next, CXCR4 mRNA levels were determined by RT-qPCR analysis.
**p<0.01 and ***p<0.001 vs. the control
group without any treatment. (G) HMEECs were exposed to NTHi for
different times (0 to 60 min). Then, western blot analysis was
performed to calculate phosphorylated p38 activation.
*p<0.05, **p<0.01 and
***p<0.001 vs. the control group. (H) HMEECs were
pretreated with p38 inhibitor at various concentrations (0, 0.5, 1
and 2 µM) for 2 h, followed by CXCR4 gene level measurement
through RT-qPCR analysis. ***p<0.001 vs. the
NTHi-treated group without SB203580 treatment. (I) HMEECs were
transfected with the Control, and p38-mutants plasmid. CXCR4
expression levels were calculated by RT-qPCR assays.
***p<0.001 vs. the NTHi-treated group without p38
mutation. Representative data are shown as SEM. |
NF-κB phosphorylation is involved in
NTHi-induced CXCR4 expression dependent on IKKα and p38 MAPK
activity
The findings above revealed that IKKα and p38 had a
close relationship with otitis media (24). NF-κB is known as an essential
signal in regulating inflammation response, which is regulated by
IKKα activity (25). As shown in
Fig. 4A and B, CXCR4 gene and
protein levels were significantly reduced for MRT67307 and SB203580
treatment alone, especially in combination. Of note, NF-κB
inhibitor, PDTC, was used here to suppress NF-κB activation. CXCR4
mRNA levels were highly reduced, particularly in the highest
concentration of PDTC (Fig. 4C).
Pretreatment with PDTC markedly abolished NTHi-triggered NF-κB
promoter-induced luciferase activity (Fig. 4D). Depletion of NF-κB by the use
of NF-κB siRNA, reduced CXCR4 mRNA expression levels (Fig. 4E). Notably, Fig. 4F overexpression of NF-κB further
promoted NTHi-triggered CXCR4 mRNA expression. In oder to calculate
if p38 triggers CXCR4 expression through NF-κB, HMEECs were
transfected with NF-κB combined with SB203580 or not, prior to
stimulation by NTHi. SB203580 reduced CXCR4 mRNA expression in
NF-κB-transfected HMEECs (Fig.
4G). In addition, pretreatment with SB203580 apparently reduced
NTHi-stimulated NF-κB promoter-driven luciferase activity, which as
shown in Fig. 4H. p38 mutation
decreased NF-κB promoter activity induced by NTHi (Fig. 4I). The data above indicated that
p38-regulated CXCR4 expression induced by NTHi relied on NF-κB
signaling pathway.
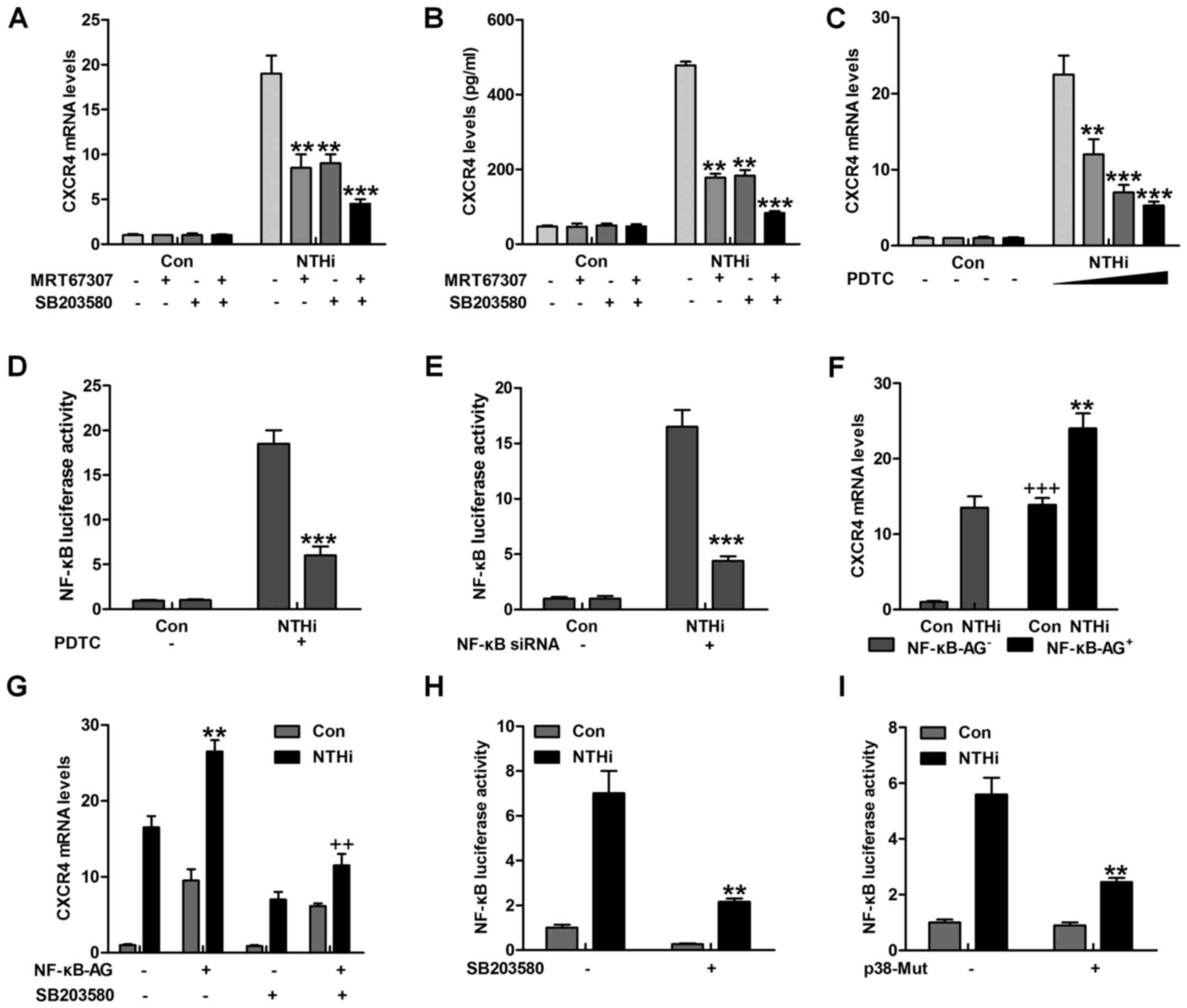 | Figure 4Nuclear factor-κB (NF-κB)
phosphorylation is involved in nontypeable Haemophilus
influenzae (NTHi)-induced chemokine CXC receptor 4 (CXCR4)
expression dependent on IKKα and p38 MAPK activity. (A) Human
middle ear epithelial cells (HMEECs) were pretreated with IKKα and
p38 MAPK inhibitors, MRT67307 (0.5 µM) and SB203580 (0.5
µM), for 2 h, followed by NTHi exposure for 6 h. Then, the
mRNA levels of CXCR4 were calculated by RT-qPCR analysis. (B)
HMEECs were pretreated with IKKα and p38 MAPK inhibitors, MRT67307
and SB203580, for 2 h, followed by NTHi exposure for 6 h. Then, the
CXCR4 protein levels were calculated via enzyme-linked
immunosorbent assay (ELISA) method. *p<0.05,
**p<0.01 and ***p<0.001 vs. the
NTHi-treated group in the absence of MRT67307 and SB203580
treatment. (C) HMEECs were pretreated with NF-κB inhibitor, PDTC
(0.5 µM), for 2 h, followed by NTHi exposure for 6 h. Then,
CXCR4 gene levels were calculated by RT-qPCR analysis. (D) HMEECs
were first transfected with the vector of NF-κB luciferase. Then,
the cells were pretreated with PDTC (0.5 µM) for 2 h,
followed by NTHi induction for 6 h. Finally, NF-κB promoter
activity was detected through luciferase analysis.
*p<0.05, **p<0.01 and
***p<0.001 vs. the NTHi-treated group without PDTC
treatment. (E) HMEECs were transfected with siRNA control or siRNA
NF-κB. Cells were then stimulated with NTHi for another 6 h, and
NF-κB promoter activity was evaluated via luciferase analysis.
***p<0.001 vs. the NTHi-treated group without NF-κB
silence. (F) HMEECs were transfected with NF-κB. Cells were then
stimulated using NTHi for 6 h, and the CXCR4 mRNA levels were
calculated. **p<0.01 vs. the NTHi-treated group
without NF-κB transfection; +++p<0.001 vs. the
control group without NF-κB transfection. (G) HMEECs were treated
with NF-κB transfection or SB203580 alone or in combination,
followed by CXCR4 mRNA level measurement. **p<0.01
vs. the NTHi-treated group without any treatment;
++p<0.001 vs. the NTHi-treated group after NF-κB
transfection in the absence of SB203580. (H) NF-κB luciferase
vector was transfected to HMEECs combined with SB203580, followed
by NF-κB promoter activity. **p<0.01 vs. the
NTHi-treated group without SB203580. (I) HMEECs were transfected
with the control, and p38-mutants plasmid after NF-κB luciferase
transfection. Then, the NF-κB promoter activity was evaluated via
luciferase analysis. The representative data are shown as SEM. |
Quercetin inhibits NTHi-triggered CXCR4
activation
The findings above indicated the possible molecular
mechanism by which otitis media was induced for NTHi, which was
related to CXCR4 expression levels. Quercetin has been reported
before to have an essential role in suppressing inflammation
response through various signaling pathways, including TLR4/NF-κB
and MAPKs (26). Thus, we
attempted to explore if quercetin could improve NTHi-induced otitis
media in vivo and in vitro through targeting CXCR4.
As shown in Fig. 5A and B, after
NTHi stimulation, overexpressed CXCR4 gene and protein levels were
reduced for quercetin administration in a dose-dependent manner.
Also, in other NTHi strains, 2019, 12 and 2866, CXCR4 high
expression was significantly downregulated for quercetin treatment
under different concentrations in HMEECs stimulated by NTHi
(Fig. 5C–E). Finally, in in
vivo study, we found that CXCR4 mRNA levels in the middle ear
of mice were upregulated by NTHi induction, which was downregulated
for quercetin treatment in a dose-dependent manner (Fig. 5F). The histologic sections and the
mucosa thickness in the mouse middle ear were observed through
H&E staining (Fig. 5G). By
assessment, the mucosa in the roof of NTHi-treated mice with otitis
media was much thicker compared to the control ones, which was
reduced for quercetin treatment at different concentrations. The
data above indicated that quercetin has potential value for
ameliorating NTHi-induced inflammation seen in otitis media.
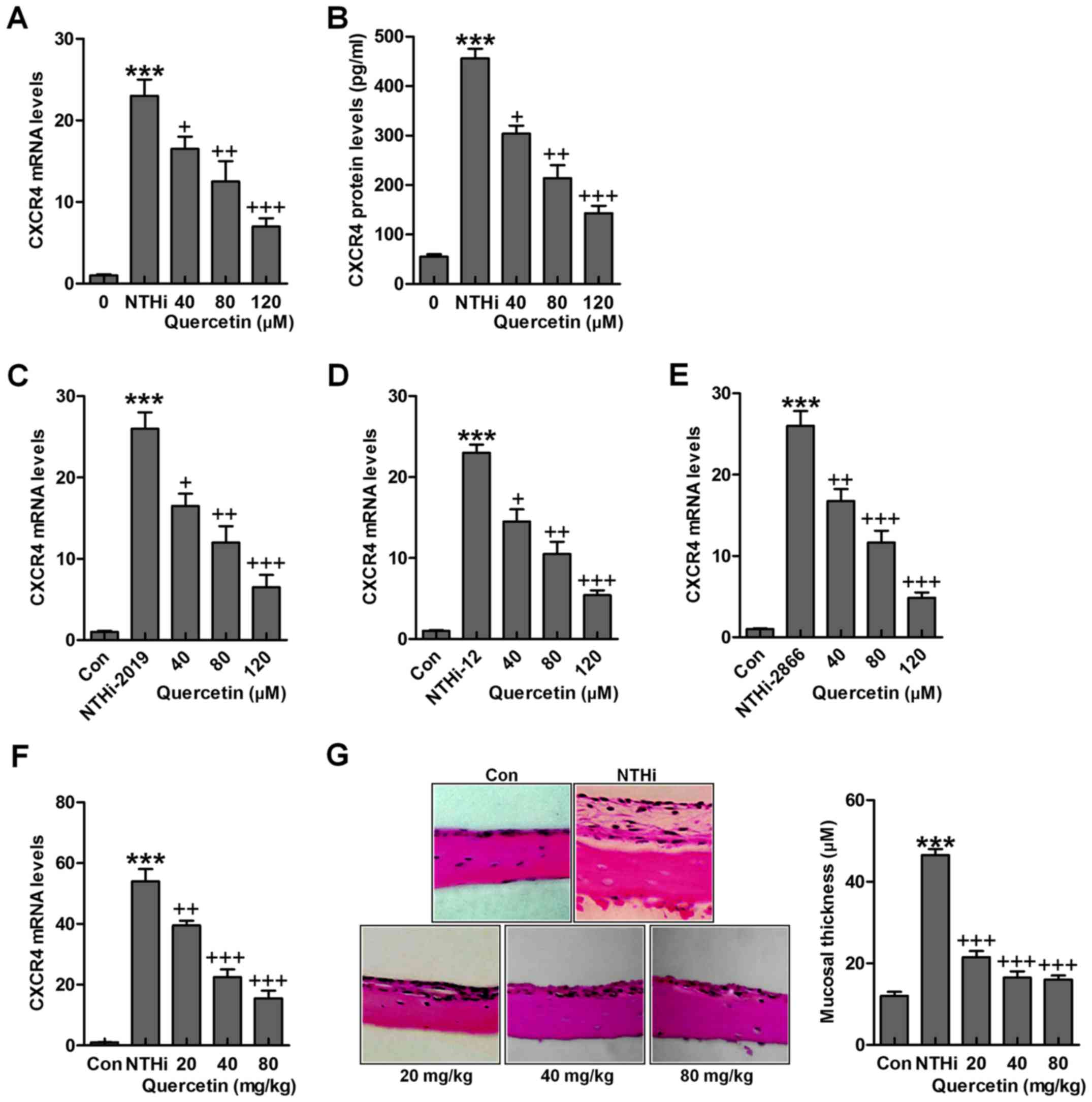 | Figure 5Quercetin inhibits NTHi-triggered
chemokine CXC receptor 4 (CXCR4) activation. Human middle ear
epithelial cells (HMEECs) were stimulated with nontypeable
Haemophilus influenzae (NTHi) for 6 h, followed by quercetin
treatment at different concentrations (40, 80 and 120 µM)
for 2 h. Then, (A) CXCR4 mRNA levels (B) and protein levels were
evaluated by RT-qPCR analysis and enzyme-linked immunosorbent assay
(ELISA) method, respectively. HMEECs were stimulated with NTHi
strains (C) 2019, (D) 12 and (E) 2866 for 6 h. Quercetin was
administered to cells under different conditions for 2 h. Then,
RT-qPCR assay was carried out to investigate CXCR4 mRNA levels. (F)
Mice were pretreated with 5×107 CFU NTHi for 6 h,
followed by quercetin administration through i.p. at 20, 40 and 80
mg/kg for 2 h. CXCR4 mRNA levels in the dissected middle ear tissue
samples were calculated. (G) The representative images of middle
ear histophathology in NTHi-treated mice with or without quercetin
administration exhibited by H&E staining. The quantification of
mucosa thickness is shown. The representative data are shown as
SEM. *p<0.05, **p<0.01 and
***p<0.001 vs. the control (Con) group.
+p<0.05, ++p<0.01 and
+++p<0.001 vs. the NTHi group. |
Quercetin inhibits NTHi-stimulated CXCR4
activation through IKKα and p38 MAPK suppression
Next, we attempted to investigate the molecular
mechanism by which quercetin suppressed CXCR4 expression. IKKα and
p38 signaling pathway activation has been revealed to be related
with NTHi-stimulated CXCR4 expression, we calculated the role of
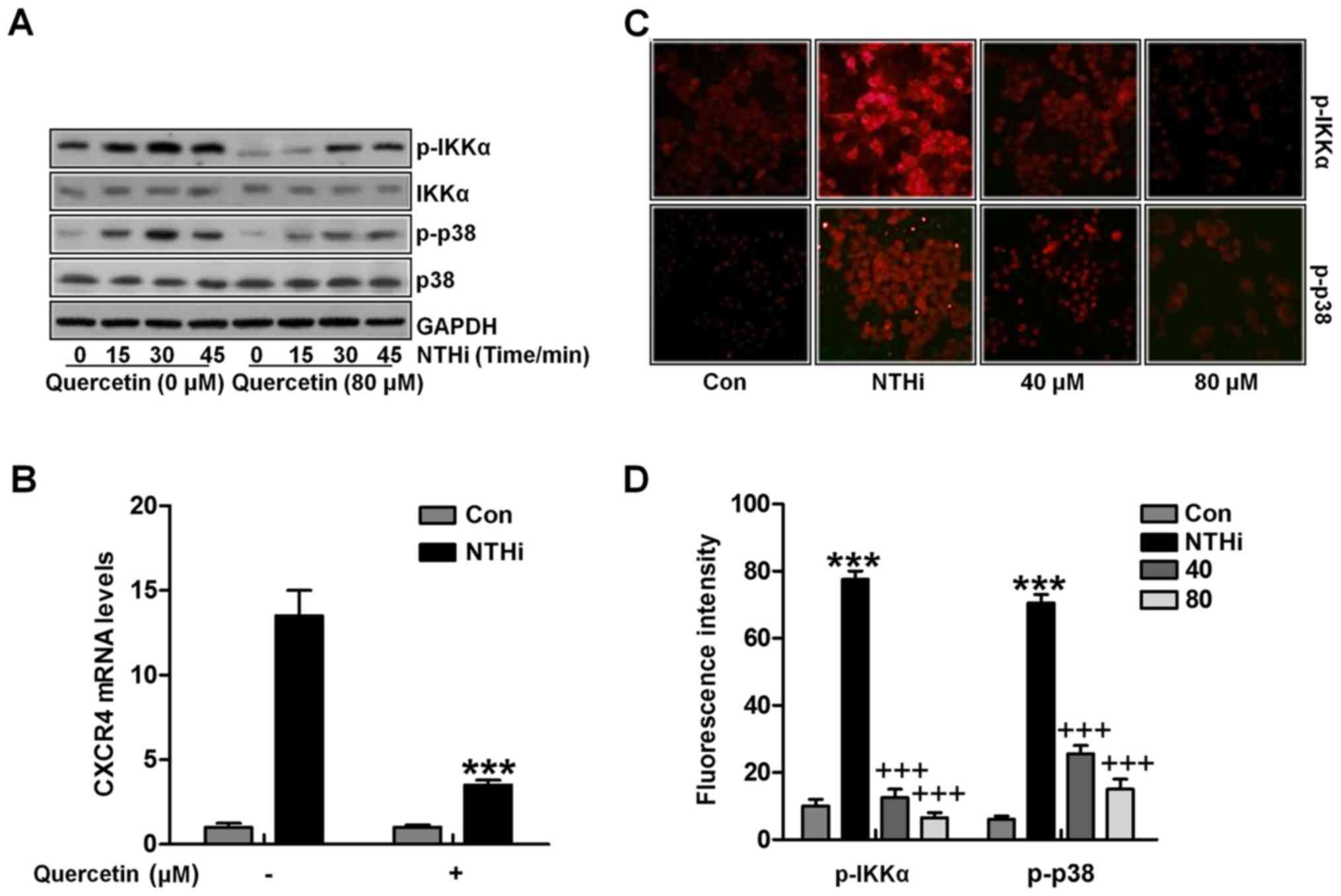
quercetin in the two signaling pathways. Quercetin abolished
NTHi-induced IKKα and p38 phosphorylation through western blot
analysis (Fig. 6A). Additionally,
quer-cetin suppressed IKKα activator-induced CXCR4 expression in
vitro (Fig. 6B). Finally,
fluorescent analysis indicated that IKKα and p38 phosphorylated
levels induced by NTHi were apparently reduced for quercetin
treatment, which was in a dose-dependent manner in vivo
(Fig. 6C and D). Collectively,
the data above indicated that quercetin suppressed NTHi-induced
CXCR4 expression and activation by inhibiting IKKα and p38 MAPK
signaling pathways.
Discussion
Otitis media is reported as one of the most common
infectious diseases for children. Acute otitis media could develop
into chronic otitis media, leading to hearing loss (1–3,27).
Severely, complications include language disability and
intellectual impairment (28).
However, up until now the specific molecular mechanism of otitis
media is scarsly known, and finding effective therapeutic strategy
is urgently required. Quercetin has been reported to be effective
in anti-inflammation through various signaling pathways (29). Further, previous study, otitis
media development has a close relationship with inflammatory
response (30). In the present
study, we show that quercetin suppressed NTHi-induced CXCR4
upregulation in otitis media model both in in vitro and
in vivo studies. Here, we found that NTHi stimulation
increased CXCR4 expression through IKKα and p38 MAPK signaling
pathways activation, which was dependent on NF-κB translocation.
Quercetin could inhibit the IKKα signaling pathway as well as p38
MAPK activation, contributing to CXCR4 expression suppression.
Therefore, the present study supplied a novel molecular mechanism
revealing the close regulation of CXCR4 in the progression and
pathogenesis of NTHi-induced otitis media, and notably, the
potential value of quercetin was found in treating NTHi-induced
otitis media.
Presently, otitis media is a leading cause,
contributing to hearing loss for childhood, which is highly related
to NTHi infection. Otitis media is characterized by inflammatory
response in the middle ear (31,32). Therapeutic strategies for otitis
media treatment are highly advanced. For instance, antibiotics
usage help many patients suffering from the disease, but generate
drug-resistance (33). However,
side effects still exist. Thus, finding novel treatment is also
necessary. In addition, identifying the possible molecular
mechanism causing the inflammatory response is essential for
development of new strategies. Chemokines are reported as a
superfamily of chemoattractant proteins, inducing cytoskeletal
rearrangement, firming adhesion to specific cells as well as
directional migration via interacting with receptors for cognation
(34). Chemokines have an
important role in recruiting leukocytes to inflammatory sites
(35). CXCR4 serves as a key
factor for inflammation formation in various diseases, regulating
cellular processes, including cell migration and proliferation
(36). Accumulating evidence
indicates that chemokines are considered as useful targets for new
drug investigation (37). In this
study, quercetin treatment effectively suppressed CXCR4 expression,
which has a close relevance in otitis media regulation through
modulating inflammation response. Quercetin is isolated from
natural plants, which has been well investigated for its medicinal
properties with little side effects (38). Quercetin could interact with
various signaling pathways, such as protein kinases, cytokines,
transcription factors and growth factors (39). Hence, it possesses potential
against a variety of diseases. In the present study, we further
evidenced an effective role of quercetin in CXCR4 expression
inhibition.
Activation of TLR signaling which stimulates
inflammation is the key point in the pathogenesis of otitis media
in mice induced under different conditions (40). Previous studies indicated that
recognition of TLRs to pathogen-linked molecular patterns could
initiate various protective immune responses. Also, investigations
of TLR signaling pathway have indicated that TLRs result in IRAKs
recruitment and activation (41).
Once binding to TLRs/MyD88 complex, IRAKs could been
phosphorylated, regulating the TRAF6 recruitment (42). The dissociation from the receptor
complex, IRAK/TRAF6 complex could interact with TAK1 for
activation. The phosphorylated TAK1 results in NF-κB activation
subsequently, leading to proinflammatory cytokine transcription
(43). In our study, we found
that CXCR4 expression was highly upregulated for TLR3/MyD88
signaling pathway activation, contributing to NF-κB phosphorylation
and inflammation eventually. Quercetin could suppress NF-κB
activation, which was related to TLR3/MyD88 signaling pathway
inactivation in line with previous studies (42).
Mitogen-activated protein kinases (MAPKs), involving
p38, extracellular signal-regulated kinase (ERK), as well as c-Jun
N-terminal kinase (JNK), are important signaling molecules, which
could transduce extracellular stimulus into intracellular
transcriptional or post-translational information (44). Numerous studies before have
indicated that MAPK members could be phosphorylated after the
activation of chemokine receptors, including CXCR1, CXCR2 as well
as CXCR4 (45). Previous study
has indicated that suppressing CXCR4/MAPKs signaling pathways could
ameliorate pain hyperalgesia, which is associated with inflammatory
response (46). In line with the
results above, we found that CXCR4 high expression relied on p38
phosphorylation, and notably, quercetin showed suppressive role in
p38 phosphorylation, which may be a possible mechanism by which
otitis media was ameliorated in quercetin treatment.
Collectively, this study indicated that quercetin
may be a potential suppressor for CXCR4, contributing to
inflammation blocking through TLR3/MyD88 and p38 MAPK signaling
pathway inhibition, which may have potential applications for
otitis media treatment in clinic.
Acknowledgments
The authors would like to thank all the members of
the Department of Otorhinolaryngology for helping in this work.
References
|
1
|
Morris LM, DeGagne JM, Kempton JB, Hausman
F and Trune DR: Mouse middle ear ion homeostasis channels and
intercellular junctions. PLoS One. 7:e390042012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Topcuoglu N, Keskin F, Ciftci S, Paltura
C, Kulekci M, Ustek D and Kulekci G: Relationship between oral
anaerobic bacteria and otitis media with effusion. Int J Med Sci.
9:256–261. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Bluestone CD and Klein JO: Physiology,
pathophysiology and pathogenesis Otitis Media in Infants and
Children. 4th edition. BC Decker; pp. 41–42. 2007
|
|
4
|
Post JC: Direct evidence of bacterial
biofilms in otitis media. Laryngoscope. 111:2083–2094. 2001.
View Article : Google Scholar
|
|
5
|
Darrow DH, Dash N and Derkay CS: Otitis
media: Concepts and controversies. Curr Opin Otolaryngol Head Neck
Surg. 11:416–423. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ting PJ, Lin CH, Huang FL, Lin MC, Hwang
KP, Huang YC, Chiu CH, Lin TY and Chen PY: Epidemiology of acute
otitis media among young children: A multiple database study in
Taiwan. J Microbiol Immunol Infect. 45:453–458. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Cayé-Thomasen P, Hermansson A, Bakaletz L,
Hellstrøm S, Kanzaki S, Kerschner J, Lim D, Lin J, Mason K and
Spratley J: Panel 3: Recent advances in anatomy, pathology, and
cell biology in relation to otitis media pathogenesis. Otolaryngol
Head Neck Surg. 148(Suppl): E37–E51. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Movahedan A, Majdi M, Afsharkhamseh N,
Sagha HM, Saadat NS, Shalileh K, Milani BY, Ying H and Djalilian
AR: Notch inhibition during corneal epithelial wound healing
promotes migration. Invest Ophthalmol Vis Sci. 53:7476–7483. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Burns JM, Summers BC, Wang Y, Melikian A,
Berahovich R, Miao Z, Penfold ME, Sunshine MJ, Littman DR, Kuo CJ,
et al: A novel chemokine receptor for SDF-1 and I-TAC involved in
cell survival, cell adhesion, and tumor development. J Exp Med.
203:2201–2213. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Balabanian K, Lagane B, Infantino S, Chow
KY, Harriague J, Moepps B, Arenzana-Seisdedos F, Thelen M and
Bachelerie F: The chemokine SDF-1/CXCL12 binds to and signals
through the orphan receptor RDC1 in T lymphocytes. J Biol Chem.
280:35760–35766. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Lu DY, Tang CH, Yeh WL, Wong KL, Lin CP,
Chen YH, Lai CH, Chen YF, Leung YM and Fu WM: SDF-1alpha
upregulates interleukin-6 through CXCR4, PI3K/Akt, ERK, and
NF-kappaB-dependent pathway in microglia. Eur J Pharmacol.
613:146–154. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ganesan S, Faris AN, Comstock AT, Wang Q,
Nanua S, Hershenson MB and Sajjan US: Quercetin inhibits rhinovirus
replication in vitro and in vivo. Antiviral Res. 94:258–271. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Choi HJ, Song JH and Kwon DH: Quercetin
3-rhamnoside exerts antiinfluenza A virus activity in mice.
Phytother Res. 26:462–464. 2012.
|
|
14
|
Slusarz A, Shenouda NS, Sakla MS,
Drenkhahn SK, Narula AS, MacDonald RS, Besch-Williford CL and
Lubahn DB: Common botanical compounds inhibit the hedgehog
signaling pathway in prostate cancer. Cancer Res. 70:3382–3390.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Casey JR, Kaur R, Friedel VC and
Pichichero ME: Acute otitis media otopathogens during 2008 to 2010
in Rochester, New York. Pediatr Infect Dis J. 32:805–809.
2013.PubMed/NCBI
|
|
16
|
Roier S, Leitner DR, Iwashkiw J,
Schild-Prüfert K, Feldman MF, Krohne G, Reidl J and Schild S:
Intranasal immunization with nontypeabl Haemophilus influenzae
outer membrane vesicles induces cross-protective immunity in mice.
PLoS One. 7:e426642012. View Article : Google Scholar
|
|
17
|
Nakajima F, Aratani S, Fujita H, Yagishita
N, Ichinose S, Makita K, Setoguchi Y and Nakajima T: Synoviolin
inhibitor LS-102 reduces endoplasmic reticulum stress-induced
collagen secretion in an in vitro model of stress-related
interstitial pneumonia. Int J Mol Med. 35:110–116. 2015. View Article : Google Scholar
|
|
18
|
Jiang GX, Cao LP, Kang PC, Zhong XY, Lin
TY and Cui YF: Interleukin 6 induces epithelial mesenchymal
transition in human intrahepatic biliary epithelial cells. Mol Med
Rep. 13:1563–1569. 2016. View Article : Google Scholar
|
|
19
|
Jiang HY, Wang F, Chen HM and Yan XJ:
κ-carrageenan induces the disruption of intestinal epithelial
Caco-2 monolayers by promoting the interaction between intestinal
epithelial cells and immune cells. Mol Med Rep. 8:1635–1642. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Takeda K and Akira S: Toll-like receptors
in innate immunity. Int Immunol. 17:1–14. 2005. View Article : Google Scholar
|
|
21
|
Alexopoulou L, Holt AC, Medzhitov R and
Flavell RA: Recognition of double-stranded RNA and activation of
NF-kappaB by Toll-like receptor 3. Nature. 413:732–738. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang X, Chen Q and Xing D: Focal adhesion
kinase activates NF-κB via the ERK1/2 and p38MAPK pathways in
amyloid-β25–35--induced apoptosis in PC12 cells. J Alzheimers Dis.
32:77–94. 2012. View Article : Google Scholar
|
|
23
|
Sloane JA, Blitz D, Margolin Z and
Vartanian T: A clear and present danger: Endogenous ligands of
Toll-like receptors. Neuromolecular Med. 12:149–163. 2010.
View Article : Google Scholar :
|
|
24
|
Euba B, Moleres J, Segura V, Viadas C,
Morey P, Moranta D, Leiva J, de-Torres JP, Bengoechea JA and
Garmendia J: Genome expression profiling-based identification and
administration efficacy of host-directed antimicrobial drugs
against respiratory infection by nontypeabl Haemophilus influenzae.
Antimicrob Agents Chemother. 59:7581–7592. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Kyo Y, Kato K, Park YS, Gajghate S,
Umehara T, Lillehoj EP, Suzaki H and Kim KC: Antiinflammatory role
of MUC1 mucin during infection with nontypeabl Haemophilus
influenzae. Am J Respir Cell Mol Biol. 46:149–156. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lai WW, Hsu SC, Chueh FS, Chen YY, Yang
JS, Lin JP, Lien JC, Tsai CH and Chung JG: Quercetin inhibits
migration and invasion of SAS human oral cancer cells through
inhibition of NF-κB and matrix metalloproteinase-2/-9 signaling
pathways. Anticancer Res. 33:1941–1950. 2013.PubMed/NCBI
|
|
27
|
Zhang J, Xu M, Zheng Q, Zhang Y, Ma W and
Zhang Z: Blocking macrophage migration inhibitory factor activity
alleviates mouse acute otitis media in vivo. Immunol Lett.
162:101–108. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Chu TG, Cachola DR III, Regal MA, Llamas
AC, Martinez NV and Santos WR: Pneumococcal conjugate vaccine
(non-typeable haemophilus influenzae (NTHi) protein D, diphtheria
or tetanus toxoid conjugates) in prevention of acute otitis media
in children: A Cohort Study. Philipp J Otolaryngol Head Neck Surg.
31:13–15. 2016.
|
|
29
|
Borska S, Chmielewska M, Wysocka T,
Drag-Zalesinska M, Zabel M and Dziegiel P: In vitro effect of
quercetin on human gastric carcinoma: Targeting cancer cells death
and MDR. Food Chem Toxicol. 50:3375–3383. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Slusarz A, Shenouda NS, Sakla MS,
Drenkhahn SK, Narula AS, MacDonald RS, Besch-Williford CL and
Lubahn DB: Common botanical compounds inhibit the hedgehog
signaling pathway in prostate cancer. Cancer Res. 70:3382–3390.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Cohen R, Bingen E, Levy C, Thollot F,
Boucherat M, Derkx V and Varon E: Nasopharyngeal flora in children
with acute otitis media before and after implementation of 7 valent
pneumococcal conjugate vaccine in France. BMC Infect Dis.
12:522012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Pichichero ME, Kaur R, Casey JR, Xu Q,
Almudevar A and Ochs M: Antibody response to Streptococcus
pneumoniae proteins PhtD, LytB, PcpA, PhtE and Ply after
nasopharyngeal colonization and acute otitis media in children. Hum
Vaccin Immunother. 8:799–805. 2012. View
Article : Google Scholar : PubMed/NCBI
|
|
33
|
Pumarola F, Marès J, Losada I, Minguella
I, Moraga F, Tarragó D, Aguilera U, Casanovas JM, Gadea G, Trías E,
et al: Microbiology of bacteria causing recurrent acute otitis
media (AOM) and AOM treatment failure in young children in Spain:
Shifting pathogens in the post-pneumococcal conjugate vaccination
era. Int J Pediatr Otorhinolaryngol. 77:1231–1236. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Vindrieux D, Escobar P and Lazennec G:
Emerging roles of chemokines in prostate cancer. Endocr Relat
Cancer. 16:663–673. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Liang JJ, Zhu S, Bruggeman R, Zaino RJ,
Evans DB, Fleming JB, Gomez HF, Zander DS and Wang H: High levels
of expression of human stromal cell-derived factor-1 are associated
with worse prognosis in patients with stage II pancreatic ductal
adenocarcinoma. Cancer Epidemiol Biomarkers Prev. 19:2598–2604.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Castellone MD, Guarino V, De Falco V,
Carlomagno F, Basolo F, Faviana P, Kruhoffer M, Orntoft T, Russell
JP, Rothstein JL, et al: Functional expression of the CXCR4
chemokine receptor is induced by RET/PTC oncogenes and is a common
event in human papillary thyroid carcinomas. Oncogene.
23:5958–5967. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Torregrossa L, Giannini R, Borrelli N,
Sensi E, Melillo RM, Leocata P, Materazzi G, Miccoli P, Santoro M
and Basolo F: CXCR4 expression correlates with the degree of tumor
infiltration and BRAF status in papillary thyroid carcinomas. Mod
Pathol. 25:46–55. 2012. View Article : Google Scholar
|
|
38
|
Su L, Zhang J, Xu H, Wang Y, Chu Y, Liu R
and Xiong S: Differential expression of CXCR4 is associated with
the metastatic potential of human non-small cell lung cancer cells.
Clin Cancer Res. 11:8273–8280. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nguyen TT, Tran E, Nguyen TH, Do PT, Huynh
TH and Huynh H: The role of activated MEK-ERK pathway in
quercetin-induced growth inhibition and apoptosis in A549 lung
cancer cells. Carcinogenesis. 25:647–659. 2004. View Article : Google Scholar
|
|
40
|
Meisgen F, Xu Landén N, Wang A, Réthi B,
Bouez C, Zuccolo M, Gueniche A, Ståhle M, Sonkoly E, Breton L, et
al: MiR-146a negatively regulates TLR2-induced inflammatory
responses in keratinocytes. J Invest Dermatol. 134:1931–1940. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Blasius AL and Beutler B: Intracellular
Toll-like receptors. Immunity. 32:305–315. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Lombardo E, DelaRosa O, Mancheño-Corvo P,
Menta R, Ramírez C and Büscher D: Toll-like receptor-mediated
signaling in human adipose-derived stem cells: Implications for
immunogenicity and immunosuppressive potential. Tissue Eng Part A.
15:1579–1589. 2009. View Article : Google Scholar
|
|
43
|
Cassatella MA, Mosna F, Micheletti A, Lisi
V, Tamassia N, Cont C, Calzetti F, Pelletier M, Pizzolo G and
Krampera M: Toll-like receptor-3-activated human mesenchymal
stromal cells significantly prolong the survival and function of
neutrophils. Stem Cells. 29:1001–1011. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Kim KN, Ham YM, Moon JY, Kim MJ, Jung YH,
Jeon YJ, Lee NH, Kang N, Yang HM, Kim D, et al: Acanthoic acid
induces cell apoptosis through activation of the p38 MAPK pathway
in HL-60 human promyelocytic leukaemia. Food Chem. 135:2112–2117.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Hyun MS, Hur JM, Mun YJ, Kim D and Woo WH:
BBR induces apoptosis in HepG2 cell through an
Akt-ASK1-ROS-p38MAPKs-linked cascade. J Cell Biochem. 109:329–338.
2010.
|
|
46
|
Liu N, Tian J, Cheng J and Zhang J:
Migration of CXCR4 gene-modified bone marrow-derived mesenchymal
stem cells to the acute injured kidney. J Cell Biochem.
114:2677–2689. 2013. View Article : Google Scholar : PubMed/NCBI
|