Introduction
The human T cell leukemia virus type 1 (HTLV-1) is a
complex human retrovirus that causes an aggressive leukemia known
as adult T cell leukemia (ATL) and HTLV-1-associated
myelopathy/tropical spastic paraparesis (HAM/TSP) (1–3). The
HTLV-1-encoded oncoprotein Tax alters the expression of numerous
genes associated with cell proliferation, immune and inflammatory
response, apoptosis and other biological processes through the
activation of NF-κB pathway (4–6). The
pathophysiology of ATL and other neurological disorders is largely
attributed to the persistent activity of the NF-κB factors. Tax
promotes the nuclear translocation of the NF-κB factors through
dissociation from the IκB proteins and then this oncoprotein links
p65 to the co-activators to enhance the transcriptional activity
(7). The target genes include
several IκB family members such as p100, IκBζ and
bcl-3(8). p100 and p52 are
strongly expressed in numbers of Tax-positive HTLV-transformed
cells, with Tax is regarded as the inducer. The aberrant
expressions of p100 and p52 were also observed in the Tax-negative
ATL cells (9), indicating that the
noncanonical NF-κB pathway is also involved in the development of
ATL since p52 is a primary member in that pathway.
Bcl-3 (B-cell chronic leukemia protein 3), an
oncoprotein, is also a member of the IκB family of NF-κB inhibitors
characterized by a conserved central ankyrin repeat domain
(10). Aberrant Bcl-3 leads to
increased cell proliferation, cell survival and malignant potential
(11–13). Bcl-3 is a nuclear protein that is
specifically associated with the homodimers of p50 and p52
(14) and functions as a
co-activator by binding to p50 and p52 to regulate the
transcription of target genes because of its two cooperative
transactivation domains (15–17).
This protein was also reported to inhibit DNA binding of p50 and
p52, thereby negatively regulating gene transcription (16,18–20).
Early (21) and recent reports
(22) have also shown that Bcl-3
increases the p50 homodimer binding to NF-κB sites without causing
co-activation. Bcl-3 increases the p50 homodimer NF-κB site
occupancy, thereby indirectly repressing the NF-κB target gene
transcription. Interestingly, Bcl-3 can repress its own
transcription by forming a complex with p50 at the NF-κB sites
within the Bcl-3 promoter and intronic enhancer HS3 (23). Thus, Bcl-3 is a multifaceted
modulator of the NF-κB activity and plays multiple functions.
Previous studies have shown that Tax
transcriptionally upregulates the Bcl-3 expression mainly through
NF-κB pathway (24) and Bcl-3 is
constitutively expressed in many HTLV-1-infected T cell lines
(25). Given the importance of
NF-κB activation and the strong correlation between Bcl-3 and
HTLV-1, we investigated the function of Bcl-3 in Tax-induced NF-κB
activation. Both knockdown and overexpression of Bcl-3 inhibit
NF-κB activation in Tax-positive cells. Ectopic expression of Bcl-3
dramatically suppresses Tax-induced NF-κB activation through a
mechanism associated with reversing Tax-mediated p65 nuclear
translocation. Bcl-3 overexpression reduces the NF-κB/DNA binding
activity. These data reveal pleiotropic functions of Bcl-3 in
Tax-induced NF-κB activation.
Materials and methods
Plasmid construction
Flag-tagged human Bcl-3-expressing plasmid
(pCMV-entry-Bcl-3) was obtained from Origene (China). To construct
a plasmid expressing full-length Bcl-3 without tag, the CDs of
Bcl-3 was subcloned from pCMV-entry-Bcl-3 into the EcoRI and
XhoI sites of the pcDNA3.0 vector (Invitrogen, USA). Bcl-3
shRNA and p65 shRNA plasmids were purchased from Santa Cruz
Biotechnology (USA). pNF-κB-luc, pCMVBam-Tax and pCMV-Bam plasmids
were kindly provided by Haojiang Luan and Shoji Yamaoka,
respectively.
Western blot analysis
Whole cell lysates were extracted from cells
suspended in radio immune precipitation buffer supplemented with 1
mM PMSF (Beyotime, China). The cytoplasmic and nuclear extracts
were obtained using the Cytoplasmic and Nuclear Extract kit
(Beyotime) according to the manufacturer’s instructions. The
lysates were resolved by electrophoresis on polyacrylamide gels
containing 0.1% SDS (SDS-PAGE) and then transferred to the
nitrocellulose membranes. The blots were incubated with the
appropriate primary antibody diluted by TBST and then exposed to
the appropriate second antibody conjugated with horseradish
peroxidase after being washed with TBST. The bands on the membrane
were visualized and captured using the ECL reagent (Beyotime) and
X-ray films (Kodak, USA). The pixel densities of proteins were
quantitated using ImageJ 1.44 software (National Institute of
Health). Graphs represent the pixel density for different proteins
normalized to β-actin.
Luciferase reporter assay
The cells were transfected using pNF-κB-luc,
pSV-β-Gal plasmids and the appropriate plasmid. The enzymatic
activities were assayed using the Luciferase Assay System (Promega,
USA) and 20/20n Luminometer (Turner BioSystems, USA)
according to the manufacturer’s instructions. The luciferase
activity was expressed as the fold of the relevant control of each
experiment. The β-galactosidase Enzyme Assay System (Promega) was
used to detect β-galactosidase activity, which normalized the
transfection efficiency. All reporter assays were performed in
triplicate and repeated in 3 independent experiments.
Electrophoretic mobility shift assay
(EMSA)
The biotin-labeled NF-κB consensus double stranded
oligonucleotides (5′-AGT TGAGGGGACTTTCCCAGGC-3′) were purchased
from Beyotime. The DNA-protein complexes were resolved on a 4%
non-denaturing polyacrylamide gel. The gels were subsequently
transferred to nylon membranes and blocked with 5% bovine serum
albumin (BSA) in TBST. The membranes were incubated with the
appropriate streptavidin conjugated with horseradish peroxidase.
The bands were visualized and captured using the ECL reagent
(Beyotime) and X-ray film (Kodak).
Immunofluorescence staining
The cells were fixed with chilled 95% ethanol. The
fixed cells were washed with TBS and transferred to a slide. The
cells were locked with TBS containing 5% BSA and washed twice with
TBS containing 0.1% Triton X-100. The cells were incubated
overnight with TBS containing anti-p65 rabbit pAb at 4°C. The cells
were incubated with TBS containing FITC-conjugated goat anti-rabbit
IgG. The nuclei were stained with 10 μg/ml DAPI. Finally,
the cells were washed twice with PBS and observed using the Olympus
FluoView™ FV1000 microscope. The data were analyzed using the
FV10-ASW1.6 Olympus software.
Data analysis
Statistical significance for the luciferase reporter
assays was determined using the Student’s t-test or one-way ANOVA
and a p<0.05 was regarded as statistically significant. The
standard errors were demonstrated by the bar in the figures.
Results
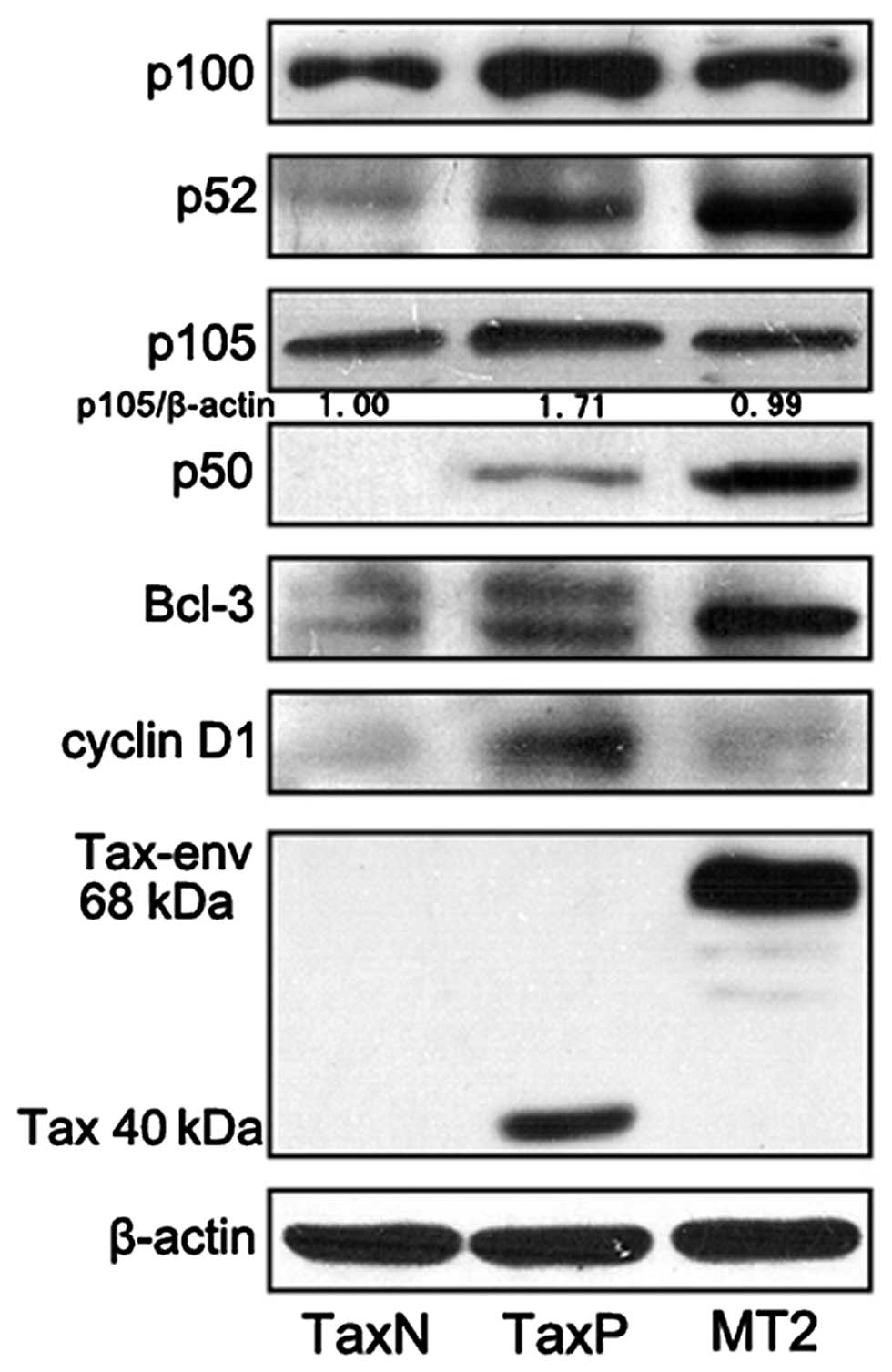
Tax induces the expression of p105, p100,
Bcl-3 and cyclin D1
A Tax-positive subline of Jurkat (TaxP) was
established (26), which stably
expresses Tax and a Tax-negative subline of Jurkat (TaxN) was
established as the control to investigate the role of Tax in
carcinogenesis. We analyzed the expression of Bcl-3, p100/p52,
p105/50 and cell cycle-associated protein cyclin D1 in TaxN, TaxP
and a HTLV-1-infected cell line MT2 (Fig. 1) to understand the transcriptional
targets of Tax. Tax could be detected in the cell line TaxP and a
known fusion between the envelope and the Tax-coding sequence
(Tax-env) (27,28) was observed in MT2 cells.
Interestingly, the Bcl-3 protein was abundantly expressed in MT2
cells and only slightly increased Bcl-3 was detected in TaxP cells
compared with that in TaxN cells. As a transcriptional target of
Bcl-3 and p52, cyclin D1 was expressed in lower levels in MT2
cells, which expressed high levels of Bcl-3 and p52 proteins. TaxP
cells, which expressed a low level of Bcl-3, expressed a higher
level of cyclin D1. Similarly, the p100 and p105 protein levels
expressed in TaxP cells are higher than those in the other two cell
lines, suggesting that p100 and p105 are also induced by Tax alone.
Western blot analysis also showed the elevated p52 and p50 protein
in TaxP and MT2 cells.
Bay 11-7082 downregulates the expression
of cyclin D1 and Bcl-3
An NF-κB inhibitor Bay 11-7082 was introduced to
determine the roles of NF-κB pathway in Tax-induced Bcl-3
expression. Bay 11-7082, an IκBα phosphorylation inhibitor, blocks
the nuclear translocation of NF-κB factors and suppresses the
activation of the genes containing NF-κB binding site. The effect
of NF-κB inhibition by Bay 11-7082 (5 μM) was assessed using
western blot analysis to detect the protein expression of Bcl-3,
p100/p52, p105/p50 and cyclin D1 after treatment in TaxP (Fig. 2A) and MT2 cells (Fig. 2B). Increased Bcl-3 expression was
observed after 1 h of Bay 11-7082 treatment (Fig. 2, compare lanes 1 and 2), followed
by a gradual decrease in the Bcl-3 expression after 24-h treatment
in both TaxP and MT2 cells. Similarly, increased levels of
phosphorylated IκBα (Ser32) were also found after 1-h treatment for
in TaxP cells (Fig. 2A, compare
lanes 1 and 2). The phosphorylation of IκBα was significantly
inhibited by Bay 11-7082 after 3, 6 and 24 h of treatment (Fig. 2A, lanes 4, 6 and 8). The expression
levels of p100, another NF-κB target gene and p52 protein were not
remarkably changed in the presence of 5 μM Bay 11-7082. By
contrast, cyclin D1 was significantly downregulated by Bay 11-7082
in the both Tax-positive cell lines. The p105 and p50 protein
levels were not significantly changed after treatment in MT2 cells.
These results suggest that the NF-κB pathway is involved in the
Tax-induced Bcl-3 expression in HTLV-1-infected cells. The Bay
11-7082-induced downregulation of cyclin D1 was accompanied by the
decrease of Bcl-3.
Knockdown of Bcl-3 suppresses the
Tax-induced NF-κB activation
Unlike other IκB family members, Bcl-3 can either
promote or inhibit NF-κB activation (12). Bcl-3 expression was remarkably
reduced in TaxP and MT2 cells after transfection of Bcl-3 shRNA
plasmid (Fig. 3A). The activity of
pNF-κB-luc reporter was lower in the Bcl-3-knocked down cells
compared with the control shRNA-transfected cells regardless of the
presence of Tax (Fig. 3B).
Bcl-3-knockdown induced the inhibition of Tax-induced NF-κB
activation in a dose-dependent manner. A total of 200 ng Bcl-3
shRNA inhibited the NF-κB activation in the three cell lines
(Fig. 3C), confirming that Bcl-3
is involved in the Tax-induced NF-κB activation and plays a
positive role although Bcl-3 shRNA failed to inhibit the
Tax-induced NF-κB activation specifically. Bcl-3 promotes NF-κB
activity mainly through the non-canonical pathway and Tax induces
NF-κB activation through both non-canonical and canonical pathways
(29–32). We investigated the effect on NF-κB
activation in TaxP and MT2 cells after the knockdown of p65
(Fig. 3A), which is a central
member of the canonical NF-κB pathway, to characterize the role of
canonical NF-κB pathway in Tax-induced NF-κB activation. The
analysis of the luciferase activity indicated that p65 knockdown
greatly inhibits the Tax-induced NF-κB activation (Fig. 3C). These results demonstrate the
important positive role of Bcl-3 in NF-κB pathway and confirm that
Tax promotes NF-κB activity through both non-canonical and
canonical pathways.
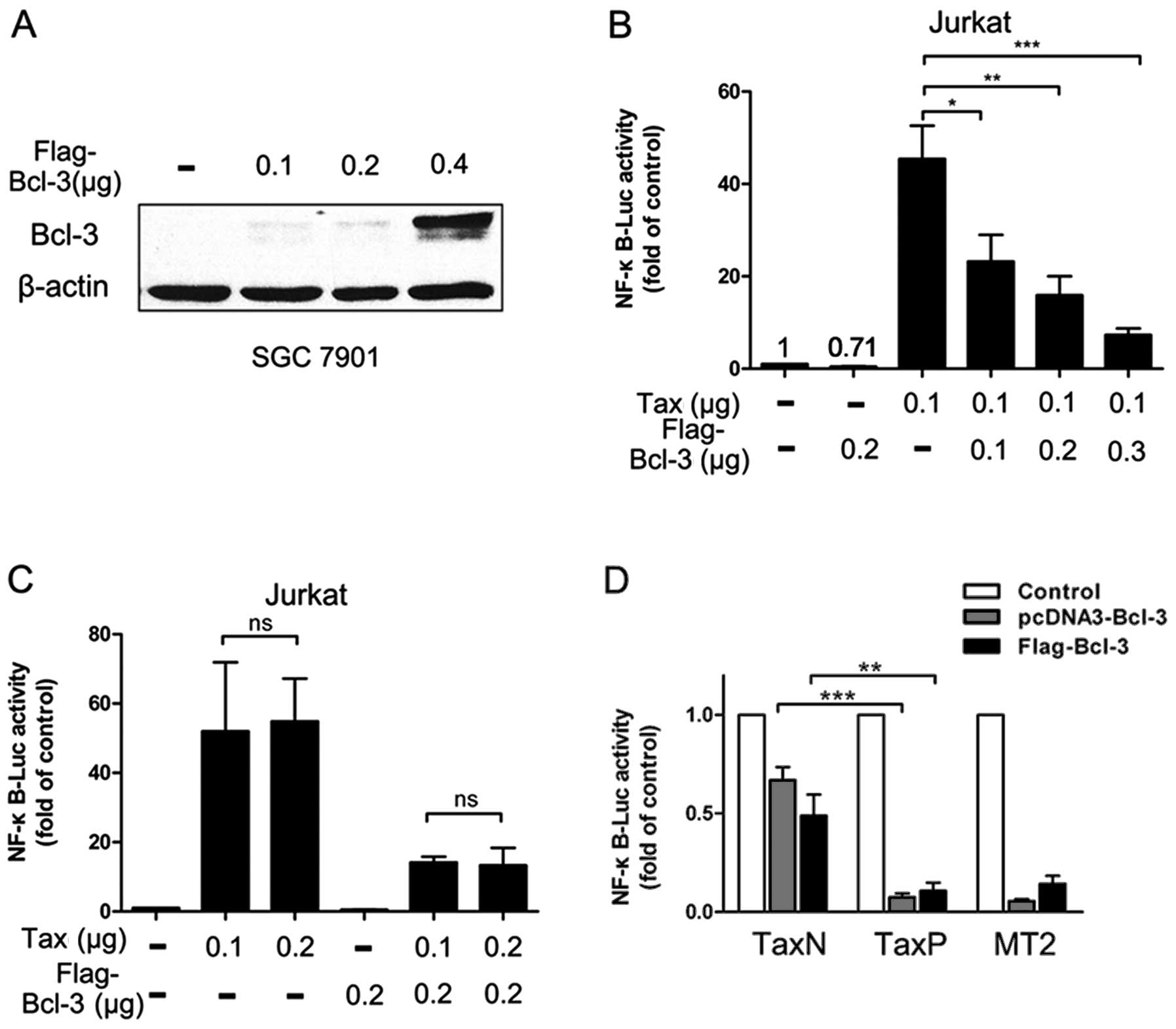
Bcl-3 overexpression dramatically
inhibits the Tax-induced NF-κB activation
Tax-induced Bcl-3 promotes the NF-κB activation
because the knockdown of Bcl-3 suppresses the NF-κB activity. We
analyzed the effect of Bcl-3 overexpression on NF-κB activation in
Tax positive cells to determine whether this condition further
enhances the Tax-induced NF-κB activation. Unexpectedly,
Tax-induced NF-κB activation was significantly repressed by Bcl-3
overexpression in Jurkat cells (Fig.
4B). The redundant Bcl-3 depressed the NF-κB activity in the
absence of Tax and inhibited the NF-κB activity in a dose-dependent
manner in Jurkat cells in its presence. Excess Bcl-3 led to the
inhibition of NF-κB activation when co-transfected with more Tax
plasmids (Fig. 4C). The NF-κB
activity in TaxP and MT2 cells was dramatically decreased by excess
Bcl-3 compared with that in TaxN cells (Fig. 4D). Similarly, the expression
vectors for Bcl-3 without tag of Flag (pcDNA3-Bcl-3) led to the
same inhibitory action (Fig. 4D).
These data indicate that the exogenous Bcl-3 regulates negatively
the NF-κB activity and can specifically inhibit Tax-induced NF-κB
activation.
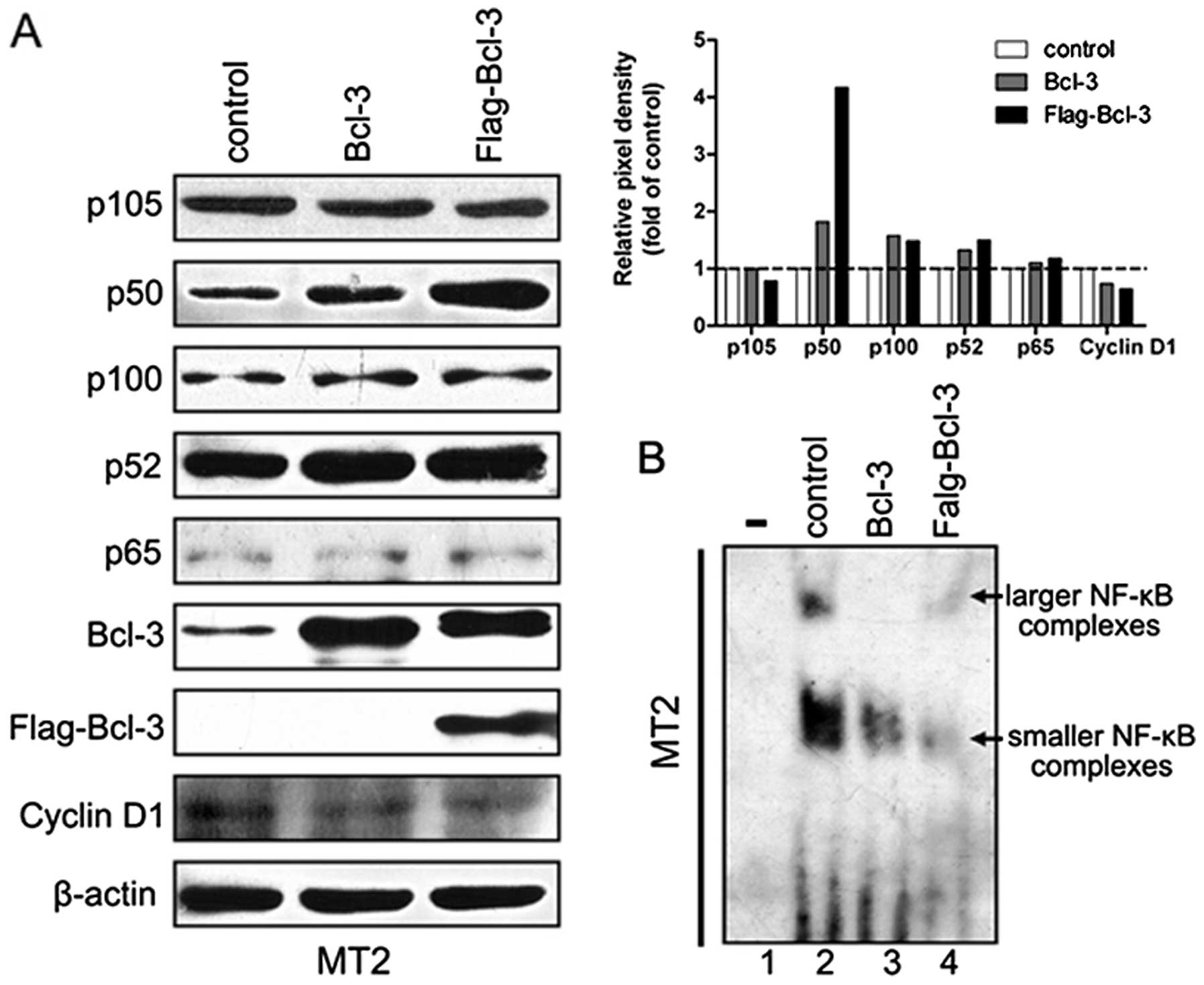
Excess Bcl-3 stabilizes p50 and reduces
NF-κB/DNA binding activity in MT2 cells
We examined the production of NF-κB factors, p50 and
p52, which were associated with Bcl-3 by western blot analysis to
investigate the molecular mechanism through which excess Bcl-3
inhibited the Tax-induced NF-κB activity. The production of p50 was
elevated significantly in the presence of exogenous Bcl-3 in MT2
cells, whereas its precursor, p105, was not significantly changed
(Fig. 5A). By contrast, the p100
and p52 proteins slightly increased in the Bcl-3-transfected MT2
cells. Cyclin D1 expression was weakly inhibited in MT2 cells,
which expressed high levels of exogenous Bcl-3. The redundant Bcl-3
almost did not influence the p65 expression (Fig. 5A). These data indicate that
excessive levels of Bcl-3, while stabilizing p50, may play a
negative role in the regulation of cyclin D1.
We performed EMSA to clarify the mechanisms of NF-κB
inhibition induced by excess Bcl-3 to assess the effect of
exogenous Bcl-3 on NF-κB/DNA binding activity in MT2 cells. The
Bcl-3-transfected MT2 cells displayed lower NF-κB/DNA binding
compared with the control MT2 cells (Fig. 5B, compare lanes 2 and 3),
suggesting that overexpression of Bcl-3 reduces the binding of
NF-κB with DNA to inhibit the NF-κB transcriptional activity.
Bcl-3 expression promotes p65
translocation from nucleus to cytoplasm
NF-κB activation is always related to the nuclear
translocation of NF-κB factors. Considering that Bcl-3
overexpression inhibits Tax-induced NF-κB activation, we analyzed
the distribution of NF-κB factors in the cytoplasm and nucleus by
western blot analysis after transfection (Fig. 6A). Bcl-3 was detected in both
nuclear and cytoplasmic extracts. However, compared with the
control, excess Bcl-3 was located in the cytoplasm, suggesting that
the redundant Bcl-3 plays a negative role on NF-κB activity in the
cytoplasm. Cytoplasmic p50 and p52 levels were enhanced slightly
after the Bcl-3 transfection, whereas the p50 and p52 levels in the
nucleus were not altered. We then examined the p65 nuclear and
cytoplasmic levels. Strikingly, the nuclear p65 was significantly
reduced in the presence of excessive Bcl-3 levels. By contrast, the
p65 expression in whole cell extracts was not significantly changed
after the transfection of pcDNA3-Bcl-3, suggesting that the excess
Bcl-3-induced inhibition of NF-κB activation is due to the blockage
of p65 nuclear translocation.
We immunostained MT2 cells transfected with Bcl-3 or
empty vectors to confirm that p65 nuclear translocation is
inhibited by excess Bcl-3 (Fig.
6B). p65 was mainly located in the nucleus in the control MT2
cells, but p65 was observed mainly in the cytoplasm after Bcl-3
transfection. These data strongly indicate that Bcl-3
overexpression inhibits p65 translocation in HTLV-1-infected
cells.
Discussion
HTLV-1 Tax protein exerts an essential role in cell
transformation and Tax-induced activation of NF-κB is critical for
the immortalization and survival of HTLV-1-infected T cells. Tax
activates the NF-κB pathway mainly through direct recruitment of
p65 from the cytoplasm to the nucleus (7,29).
As an IκB family member and a co-activator, Bcl-3 has been reported
to either promote or inhibit the NF-κB activation with p50 and p52
homodimers. Elevated levels of Bcl-3 have been detected in many
HTLV-1-infected cell lines (28)
and are mainly regulated by the Tax-NF-κB pathway. The protein
levels of p100, p105 and cyclin D1 were found to be upregulated by
Tax alone, in contrast to their slight decrease in HTLV-1-positive
cells. Bcl-3 has several intermediate phosphoforms in different
cell lines and migrates in SDS-PAGE as several bands (14). Bcl-3 exists mainly in one form in
MT2 cells and two forms were found in TaxN and TaxP cells,
indicating that Bcl-3 plays multiple roles in different forms in
different cells. Bay 11-7082 has been reported to inhibit
specifically the Tax-induced NF-κB activation and expression of
cyclin D1 (33), a cell cycle
regulator, which can be regulated by Bcl-3 and p52. Bay 11-7082
decreases the expression of cyclin D1 and Bcl-3 in the TaxP and MT2
cells. Therefore, Tax-induced Bcl-3 is regulated by the NF-κB
pathway and the downregulated cyclin D1 is at least partially
attributable to the inhibition of Bcl-3.
The knockdown of Bcl-3 was found to inhibit NF-κB
activity in the Tax-expressing cells and HTLV-1-positive cells,
suggesting that Bcl-3 plays a positive role in the Tax-induced
NF-κB activation and a Bcl-3 positive feedback is confirmed in the
Tax-positive cells. The positive regulation of Bcl-3 in the NF-κB
pathway may be caused by its co-activator function with p50 and
p52. Bcl-3 overexpression was found to inhibit enormously the NF-κB
activation in the Tax-positive and virus-positive cells. Both
knockdown and overexpression of Bcl-3 inhibit the Tax-induced NF-κB
activation, suggesting the pleiotropic roles of Bcl-3 in the
presence of Tax. Tax-induced p65 nuclear translocation has been
reported to act as a major cause in the increased NF-κB activity
(34,35). Reduced p65 nuclear translocation
was observed after the forced Bcl-3 expression in
HTLV-1-transformed cells and the p65 nuclear distribution is
dramatically decreased by the excess Bcl-3. The ankyrin repeat
domain of Bcl-3 can interacts with Tax (24) and excessive Bcl-3 were located in
the cytoplasm. With respect to the importance of Tax in p65 nuclear
translocation, Bcl-3 possibly interacts with Tax in the cytoplasm
and blocks Tax nuclear translocation. Thus, p65 nuclear
translocation pathway is blocked by Bcl-3.
Hence, the downregulation and upregulation of Bcl-3
inhibit Tax-induced NF-κB activation. Exogenous Bcl-3 inhibits
Tax-induced NF-κB activation by reducing the NF-κB/DNA binding
activity and blocking the Tax-induced nuclear translocation of p65.
Tax induces Bcl-3 overexpression by activating the NF-κB pathway
and the elevated Bcl-3 leads to increased cell proliferation, cell
survival and malignant potential. Tax-induced NF-κB activation is
critical in the HTLV-1 malignant progression. We found that Bcl-3
modulates Tax-induced NF-κB activation both positively and
negatively. Positive regulation of NF-κB activation by Tax-induced
Bcl-3 would promote cell proliferation and inhibit apoptosis.
Excessive Bcl-3 would inhibit the Tax-induced NF-κB activation to
modulate other NF-κB target gene transcription. Thus, Tax-induced
Bcl-3 may contribute to the pathogenic process of ATL through the
regulation of NF-κB activation. The present findings contributed to
a better understanding of the interaction between Bcl-3 and
Tax-induced NF-κB activation. The data also highlight the
pleiotropic functions of Bcl-3 in the Tax-induced NF-κB activation.
A balance in the aberrant Bcl-3 expression may be established by
Tax to act as a modulator of the NF-κB pathway and to play an
important role in the maintenance of proliferation, as well as
anti-apoptosis, in HTLV-1-infected and ATL cells. Further studies
should explore the potential stabilization of NF-κB factors by
Bcl-3 in HTLV-1-infected and ATL cells. The other functions of
Bcl-3 in the pathologic process of ATL should be investigated
further to improve the understanding of the oncogenic potential of
this protein with Tax and other NF-κB factors.
Acknowledgements
We are most grateful to Haojiang Luan
and Shoji Yamaoka for the plasmids as well as to Ms. Wen Zhang, Mr.
Jiqiang Guo and Xiaofei Zhu for the technical assistance. We thank
all laboratory staff members for the critical reading of the
manuscript. We would like to acknowledge Dr Tomas Ondrejcak, School
of Medicine, Trinity College Dublin, for the English language
improvement. The present study was supported by a grant from the
National Natural Science Foundation of China (grant no.
30972755).
References
|
1.
|
Gessain A, Barin F, Vernant JC, et al:
Antibodies to human T-lymphotropic virus type-I in patients with
tropical spastic paraparesis. Lancet. 2:407–410. 1985. View Article : Google Scholar : PubMed/NCBI
|
|
2.
|
Yoshida M, Miyoshi I and Hinuma Y:
Isolation and characterization of retrovirus from cell lines of
human adult T-cell leukemia and its implication in the disease.
Proc Natl Acad Sci USA. 79:2031–2035. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
3.
|
Osame M, Usuku K, Izumo S, et al: HTLV-I
associated myelopathy, a new clinical entity. Lancet. 1:1031–1032.
1986. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Harhaj E and Harhaj N: Mechanisms of
Persistent NF-κB Activation by HTLV-I Tax. Int Union Biochem Mol
Biol: Life. 57:83–91. 2005.
|
|
5.
|
Shvarzbeyn J and Huleihel M: Effect of
propolis and caffeic acid phenethyl ester (CAPE) on NFκB activation
by HTLV-1 Tax. Antiviral Res. 90:108–115. 2011.
|
|
6.
|
Yasunaga J, Lin FC, Lu X and Jeang KT:
Ubiquitin-specific peptidase 20 targets TRAF6 and human T cell
leukemia virus type 1 tax to negatively regulate NF-κB signaling. J
Virol. 85:6212–6219. 2011.PubMed/NCBI
|
|
7.
|
Azran I, Jeang K-T and Aboud M: High
levels of cytoplasmic HTLV-1 Tax mutant proteins retain a
Tax-NF-κB-CBP ternary complex in the cytoplasm. Oncogene.
24:4521–4530. 2005.PubMed/NCBI
|
|
8.
|
Hayden MS, West AP and Ghosh S: SnapShot:
NF-κB signaling Pathways. Cell. 127:1286–1287. 2006.
|
|
9.
|
Hironaka N, Mochida K, Mori N, Maeda M,
Yamamoto N and Yamaoka S: Tax-independent constitutive IκB kinase
activation in adult T-cell leukemia cells. Neoplasia. 6:266–278.
2004.
|
|
10.
|
Michel F, Soler-Lopez M, Petosa C, Cramer
P, Siebenlist U and Muller CW: Crystal structure of the ankyrin
repeat domain of Bcl-3: a unique member of the IkappaB protein
family. EMBO J. 20:6180–6190. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Courtois G and Gilmore TD: Mutations in
the NF-kappaB signaling pathway: implications for human disease.
Oncogene. 25:6831–6843. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12.
|
Palmer S and Chen YH: Bcl-3, a
multifaceted modulator of NF-κB-mediated gene transcription.
Immunol Res. 42:210–218. 2008.PubMed/NCBI
|
|
13.
|
McKeithan TW, Takimoto GS, Ohno H, et al:
BCL3 rearrangements and t(14;19) in chronic lymphocytic leukemia
and other B-cell malignancies: a molecular and cytogenetic study.
Genes Chromosomes Cancer. 20:64–72. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Bundy DL and McKeithan TW: Diverse effects
of BCL3 phosphorylation on its modulation of NF-kappaB p52
homodimer binding to DNA. J Biol Chem. 272:33132–33139. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
Fujita T, Nolan GP, Liou HC, Scott ML and
Baltimore D: The candidate proto-oncogene bcl-3 encodes a
transcriptional coactivator that activates through NF-kappa B p50
homodimers. Genes Dev. 7:1354–1363. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Franzoso G, Bours V, Azarenko V, et al:
The oncoprotein Bcl-3 can facilitate NF-kappa B-mediated
transactivation by removing inhibiting p50 homodimers from select
kappa B sites. EMBO J. 12:3893–3901. 1993.PubMed/NCBI
|
|
17.
|
Bours V, Franzoso G, Azarenko V, et al:
The oncoprotein Bcl-3 directly transactivates through kappa B
motifs via association with DNA-binding p50B homodimers. Cell.
72:729–739. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Franzoso G, Bours V, Park S,
Tomita-Yamaguchi M, Kelly K and Siebenlist U: The candidate
oncoprotein Bcl-3 is an antagonist of p50/NF-kappa B-mediated
inhibition. Nature. 359:339–342. 1992. View
Article : Google Scholar : PubMed/NCBI
|
|
19.
|
Inoue J, Takahara T, Akizawa T and Hino O:
Bcl-3, a member of the I kappa B proteins, has distinct specificity
towards the Rel family of proteins. Oncogene. 8:2067–2073.
1993.PubMed/NCBI
|
|
20.
|
Nolan GP, Fujita T, Bhatia K, et al: The
bcl-3 proto-oncogene encodes a nuclear I kappa B-like molecule that
preferentially interacts with NF-kappa B p50 and p52 in a
phosphorylation-dependent manner. Mol Cell Biol. 13:3557–3566.
1993.PubMed/NCBI
|
|
21.
|
Caamano JH, Perez P, Lira SA and Bravo R:
Constitutive expression of Bc1-3 in thymocytes increases the DNA
binding of NF-kappaB1 (p50) homodimers in vivo. Mol Cell Biol.
16:1342–1348. 1996.PubMed/NCBI
|
|
22.
|
Kabuta T, Hakuno F, Cho Y, et al: Insulin
receptor substrate-3, interacting with Bcl-3, enhances p50 NF-κB
activity. Biochem Biophys Res Commun. 394:697–702. 2010.PubMed/NCBI
|
|
23.
|
Brocke-Heidrich K, Ge B, Cvijic H, et al:
BCL3 is induced by IL-6 via Stat3 binding to intronic enhancer HS4
and represses its own transcription. Oncogene. 25:7297–7304. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
24.
|
Kim YM, Sharma N and Nyborg JK: The
proto-oncogene Bcl3, induced by Tax, represses Tax-mediated
transcription via p300 displacement from the human T-cell leukemia
virus type 1 promoter. J Virol. 82:11939–11947. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Saito K, Saito M, Taniura N, Okuwa T and
Ohara Y: Activation of the PI3K-Akt pathway by human T cell
leukemia virus type 1 (HTLV-1) oncoprotein Tax increases Bcl3
expression, which is associated with enhanced growth of
HTLV-1-infected T cells. Virology. 403:173–180. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
26.
|
Xia YSY, Gao Y, Guo JQ, Song XF and Wang
H: Establishment of the tax gene expression cell lines and their
biological activity. Zhongguo Shi Yan Xue Ye Xue Za Zhi.
16:1425–1429. 2008.PubMed/NCBI
|
|
27.
|
Kress AK, Schneider G, Pichler K, Kalmer
M, Fleckenstein B and Grassmann R: Elevated cyclic AMP levels in T
lymphocytes transformed by human T-cell lymphotropic virus type 1.
J Virol. 84:8732–8742. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Ishikawa C, Nakachi S, Senba M, Sugai M
and Mori N: Activation of AID by human T-cell leukemia virus Tax
oncoprotein and the possible role of its constitutive expression in
ATL genesis. Carcinogenesis. 32:110–119. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
29.
|
Boxus M, Twizere J-C, Legros S, Dewulf
J-F, Kettmann R and Willems L: The HTLV-1 Tax interactome.
Retrovirology. 5:762008. View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Sun SC and Ballard DW: Persistent
activation of NF-kappaB by the tax transforming protein of HTLV-1:
hijacking cellular IkappaB kinases. Oncogene. 18:6948–6958. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Li XH and Gaynor RB: Mechanisms of
NF-kappaB activation by the HTLV type 1 tax protein. AIDS Res Hum
Retroviruses. 16:1583–1590. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
32.
|
Kfoury Y, Nasr R, Hermine O, de The H and
Bazarbachi A: Proapoptotic regimes for HTLV-I-transformed cells:
targeting Tax and the NF-kappaB pathway. Cell Death Differ.
12(Suppl 1): 871–877. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Mori N, Yamada Y, Ikeda S, et al: Bay
11-7082 inhibits transcription factor NF-kappaB and induces
apoptosis of HTLV-I-infected T-cell lines and primary adult T-cell
leukemia cells. Blood. 100:1828–1834. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
34.
|
O’Mahony AM: Human T-cell lymphotropic
virus type 1 Tax induction of biologically active NF-κB requires
IκB kinase-1-mediated phosphorylation of RelA/p65. J Biol Chem.
279:18137–18145. 2004.
|
|
35.
|
Sun SC, Elwood J, Beraud C and Greene WC:
Human T-cell leukemia virus type I Tax activation of NF-kappa B/Rel
involves phosphorylation and degradation of I kappa B alpha and
RelA (p65)-mediated induction of the c-rel gene. Mol Cell Biol.
14:7377–7384. 1994.PubMed/NCBI
|