Introduction
Pulmonary sclerosing hemangioma (PSH) is a
relatively rare benign tumor. Due to its rarity, the symptoms and
natural course of the tumor are not well understood. In the present
study, a case is presented of a 35-year-old woman who underwent
intermittent fevers for more than one year, and features of a rare
symptom and an aggressive biological tendency are described.
Case report
A 35-year-old woman started to get fever two months
prior to admission to our hospital (West China Hospital, Sichuan
University, Chengdu, China). During these two months, she had
intermittent fevers daily, which usually worsened during the day
and improved at night, without night sweats. The highest
temperature reached was 39.3°C. Simultaneously, the patient
experienced productive cough, dyspnea and right-sided chest pain.
The patient had no history of shaking chills, headache, myalgia or
arthralgia. The patient did not smoke or use illicit drugs. The
patient had no history of exposure to cats or other animals, or
exposure to risk factors for pulmonary tuberculosis or human
immunodeficiency virus (HIV). Physical examination of the patient
revealed entirely normal results. Routine blood and urine specimens
were normal. Serological tests for syphilis and HIV were negative.
Tuberculosis antibody testing and purified protein derivative
(tuberculin) skin testing yielded positive results. Bacterial
culture was performed, revealing that no infectious bacteria grew.
Immune marker and anti-neutrophil cytoplasmic antibody testing also
proved negative. Cytomegalovirus, rubella virus and herpes simplex
virus immunoglobulin G (IgG) tests were revealed to be positive,
although the patient was negative for immunoglobulin M (IgM).
Inflammatory factors, including procalcitonin, serum amyloid
protein, C- reactive protein and interleukin-6, were also
negative.
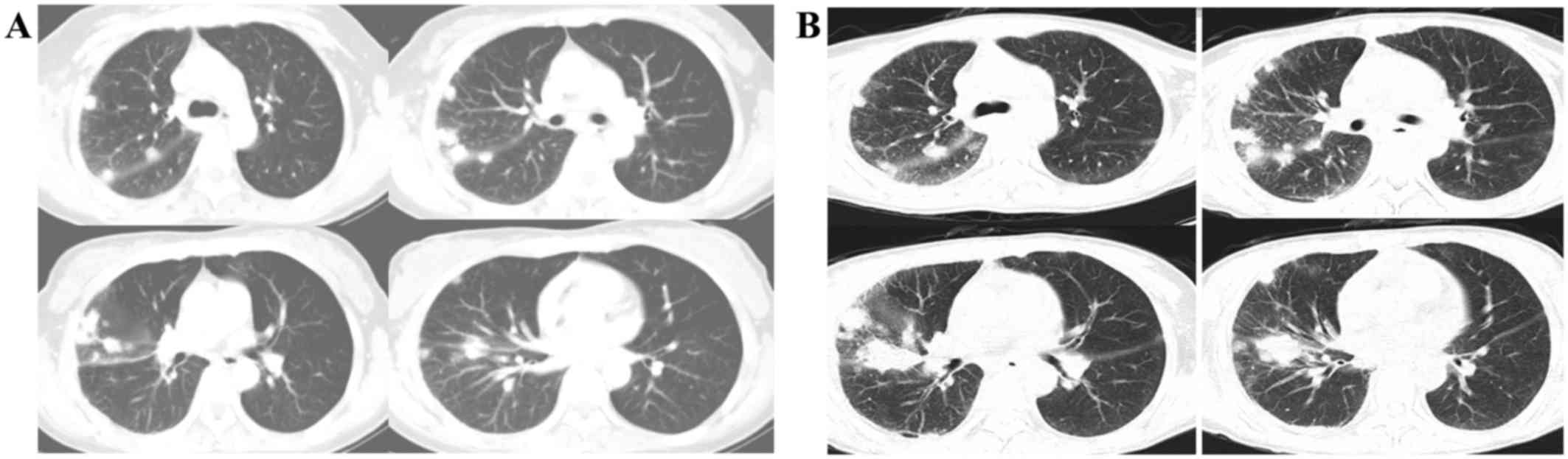
A contrast-enhanced computed tomography (CT)
examination of the chest on admission of the patient, performed on
an Aquilion™ 64 CT Scanner (Toshiba Medical Systems, Otawara,
Japan), revealed innumerable lesions that varied from several
millimeters to 3 cm in diameter in all lobes of the right lung,
although none were present in the left lung. Each lesion was a
round-shaped nodule with a distinct margin and homogeneous density.
The CT scan revealed enlarged mediastinal and right hilar lymph
nodes (Fig. 1A). In the two months
following admission, the patient received anti-infection and
anti-tuberculosis treatments. Clindamycin, moxifloxacin,
piperacillin and tazobactam were administered, without any
alleviation of the symptoms. Subsequently, the patient was
administered with anti-tuberculosis treatments, including
rifampicin, armazide, ethambutol and Levofloxacin, although the
symptoms remained. The CT scan was subsequently re-examined. This
revealed that the lesions had grown larger, and several lesions had
coalesced. The mediastinal and right hilar lymph nodes, however,
showed no further signs of increasing (Fig. 1B).
The bronchoscope and lung biopsy through
percutaneous paracentesis yielded negative results. Finally, the
patient underwent a thoracoscopic lung biopsy. The nodules in the
right pleural cavity were biopsied. No specific bacterial infection
was found in the bacterial culture of the nodules. On the basis of
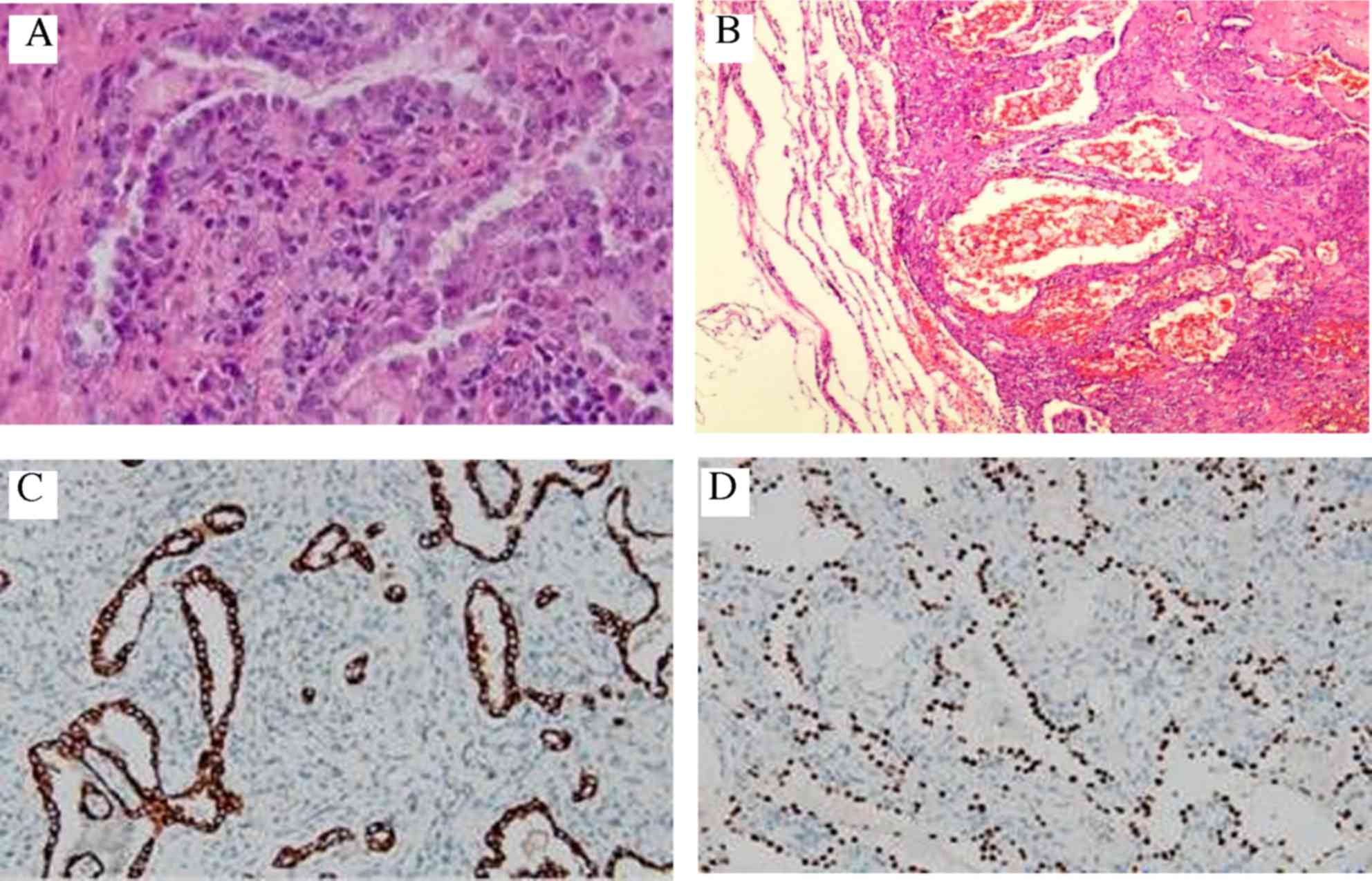
postoperative pathological diagnosis of paraffin-embedded tissue
samples, these lesions were diagnosed as multiple sclerosing
hemangiomas of the lung (i.e., as PSH), in which papillary
(Fig. 2A), solid and sclerotic
(Fig. 2B) patterns appeared.
Immunohistochemical evaluation of the lesions revealed positive
staining for thyroid transcription factor 1 (TTF1) (Fig. 2C), epithelial membrane antigen (EMA)
(Fig. 2D), pancytokeratin (PCK) and
cytoskeleton 7 (CK7). Staining proved to be negative for CD56,
synaptophysin (Syn) and chromogranin A (CgA). The Ki-67 labeling
index was <5%.
Since the patient's general state of health was
poor, she was unable to tolerate a further operation. Therefore,
the patient was discharged from hospital ten days after the
thoracoscopic lung biopsy. The patient did not receive any further
adjuvant therapy (either chemotherapy or radiotherapy). During the
follow-up for 1 year, the patient had recurring, intermittent
fevers each day. The highest temperature recorded reached 39.3°C.
However, the CT scans did not reveal any major changes during this
year. In March 2016, the patient underwent lobectomy of the middle
lobe of the right lung in a different hospital. These lesions
continued to be diagnosed as multiple sclerosing hemangiomas of the
lung. Following the operation, the patient did not have a fever for
one month (up to the time of writing).
Discussion
PSH is a relatively rare benign tumor, which was
first described by Liebow and Hubell in 1956 (1). PSH is categorized as a ‘miscellaneous
tumor’ according to the 2004 World Health Organization
classification of lung tumors. However, as they are extremely rare
manifestations, the natural course of PSHs has yet to be fully
elucidated. The etiology and origin of PSH remains a controversial
topic in the literature.
Concerning the clinical features, PSH of the lung is
most frequently encountered in middle-aged women, with a peak
incidence in 50-year-old women. The female to male ratio is 5:1 in
this patient group (2).
Most individuals with PSH are asymptomatic, with the
lesion being discovered incidentally on chest radiographs performed
for other reasons, but presentations with cough, hemoptysis, chest
pain, dyspnea and pleuritic pain have also been reported (3). The symptom of fever had not been
mentioned previously in the literature; however, it was the
predominant symptom for the patient in the present case report.
This symptom cannot be explained by any of the other findings;
perhaps fever may be one of the symptoms of PSH, although it is
less likely to be present.
Most PSHs arise in the peripheral parenchyma,
particularly in a subpleural location. It has been reported to be
more common on the right side, although cases with multiple lesions
in both lungs have also been described (4). Very rarely, PSH may originate from the
mediastinum (5).
Devouassoux-Shisheboran et al (6) studied 100 cases of PSH which were
presented, and 96 lesions (96%) were solitary and unilateral. The
tumors ranged in size from 0.3–7 cm (mean: 2.6 cm). Nine PSHs
(9.5%) were 1 cm or smaller, whereas seven PSHs (7.4%) were 5 cm or
larger. The majority (73.7%) measured <3 cm. PSH is almost
always benign, with only ~2–4% of the PSHs having nodal metastases
that otherwise do not appear to affect patient prognosis (3).
The patient in the present case study has multiple
nodules in the right lung. The CT scan revealed enlarged
mediastinal and right hilar lymph nodes. However, it has yet to be
determined whether the enlarged lymph node suggests a progressive
clinical course or not. Compared with previous reports (2,3), the
difference was that these lesions became larger, and several
lesions even coalesced in the space of two months. This case
suggests that the biological behavior of PSH may become aggressive
in certain cases.
The radiological features were typically
non-specific. Tumors may be closely associated with the pleural
surfaces, including the fissures (7). PSH is usually a solitary,
well-circumscribed nodule. Calcification is seldom seen, cystic
formation is rare (although it does occur), and cavitation does not
occur (8). There is a typical sign
in PSHs, which is termed the ‘air meniscus sign’: This is a lucent
zone surrounding PSHs of the lung on plain films (9). This sign, particularly in a non-smoker,
may be relatively specific, although it is not seen in the majority
of PSHs, and it has also been reported in aspergilloma,
tuberculoma, hamartoma and lung carcinoma (10).
Concerning the pathological features,
macroscopically, a PSH is typically between 1 and 4 cm, is
well-defined, and is often hemorrhagic, with a variegated tan or
white appearance (3). All the tumors
with adjacent lung parenchyma that have been reported in the
literature were well-circumscribed, although not encapsulated
(5).
Microscopically, PSH is predominantly composed of
two types of cells: Cuboidal surface cells that tend to
differentiate into type II pneumocytes, and polygonal stromal cells
that have considerable multiple differentiation potential (11). Surface cells are cuboidal and
resemble reactive type II pneumocytes, whereas stromal cells are
somewhat larger, with fine chromatin and inconspicuous nucleoli.
PSH is histologically characterized by the presence of hemorrhagic,
papillary, solid or sclerotic areas (3). The different histological patterns
change abruptly from one area to another. The majority of the
tumors exhibit at least three of these histological
characteristics, as was also identified in the present case study.
The papillae and tubular patterns were composed of uniform cuboidal
cells with the morphology of bronchiolar epithelium and activated
type II pneumocytes. The sclerotic pattern was composed of dense
hyaline collagen tissue around the hemorrhagic areas, within the
stalk of the papillae or within the solid areas. The solid pattern
was composed of a sheet-like proliferation of round to polygonal
cells with pale cytoplasm. In the hemorrhagic pattern, red blood
cells had accumulated within cystic spaces composed of cuboidal
surface cells (5).
An immunohistochemical examination revealed that the
tumor cells were immunopositive for TTF1 and EMA. That TTF1 and EMA
are the two epithelial markers that have been found to stain the
surface cells and stromal cells of PSH most consistently in >90%
of cases not only indicates that these antibodies are the most
useful for diagnostic purposes, but also provides insights into the
cellular differentiation of these lesions (5). Besides TTF1 and EMA, other epithelial
markers, such as PCK and CK7, were immunopositive. This provided
further proof that PSH was endothelial in origin.
More recent studies (8,11) have
offered a variety of proposed etiologies regarding the cell line of
origin, including mesothelial, mesenchymal and neuroendocrine.
However, in the present case study, CD56, Syn and CgA were all
negative. This eliminated the possibility of there being
neuroendocrine cells within the PSH in our case. The presence of
neuroendocrine cells within PSH in certain cases could represent
either differentiation of primitive respiratory cells towards a
neuroendocrine phenotype, as proposed by Noguchi et al
(12) or it could result from a
secondary neuroendocrine cell hyperplasia induced by PSH (5).
The Ki-67 nuclear antigen is associated with cell
proliferation, and is detectable in the nuclei of cells undergoing
cell cycling (the G1, S, G2, and M phases), although it is absent
in resting (G0 phase) cells. It is considered that biologically
active tumors express high levels of Ki-67 nuclear antigen
(13). The Ki-67 labeling index was
determined to be <5% in the present case study. These results
indicate that patients with PSH do not exhibit a malignant
phenotype; however, this result could not explain why the lesions
became enlarged in a two-month period in our case study. Further
follow-up is required to explain this phenomenon.
In conclusion, in the present case study, a patient
with the rare symptom of fever lasting for more than one year has
been described. Fever may be one of the symptoms of PSH, although
it is less likely to occur. Another feature of the patient was that
the tumor grew quickly in two months. It may be surmised that the
biological behavior of PSH could be aggressive in certain cases.
Further follow-up and pathological studies will be performed on the
patient.
Acknowledgements
The present study was supported by the Science
Foundation of Chengdu Municipal Science and Technology Bureau
(grant no. 2015-ofHM01-00254-SF).
References
|
1
|
Liebow AA and Hubbell DS: Sclerosing
hemangioma (histiocytoma, xanthoma) of the lung. Cancer. 9:53–75.
1956. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kuo KT, Hsu WH, Wu YC, Huang MH and Li WY:
Sclerosing hemangioma of the lung: An analysis of 44 cases. J Chin
Med Assoc. 66:33–38. 2003.PubMed/NCBI
|
|
3
|
Miyagawa-Hayashino A, Tazelaar HD, Langel
DJ and Colby TV: Pulmonary sclerosing hemangioma with lymph node
metastases: Report of 4 cases. Arch Pathol Lab Med. 127:321–325.
2003.PubMed/NCBI
|
|
4
|
Sugio K, Yokoyama H, Kaneko S, Ishida T
and Sugimachi K: Sclerosing hemangioma of the lung: Radiographic
and pathological study. Ann Thorac Surg. 53:295–300. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Sakamoto K, Okita M, Kumagiri H, Kawamura
S, Takeuchi K and Mikami R: Sclerosing hemangioma isolated to the
mediastinum. Ann Thorac Surg. 75:1021–1023. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Devouassoux-Shisheboran M, Hayashi T,
Linnoila RI, Koss MN and Travis WD: A clinicopathologic study of
100 cases of pulmonary sclerosing hemangioma with
immunohistochemical studies: TTF-1 is expressed in both round and
surface cells, suggesting an origin from primitive respiratory
epithelium. Am J Surg Pathol. 24:906–916. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Im JG, Kim WH, Han MC, Han YM, Chung JW,
Ahn JM and Do YS: Sclerosing hemangiomas of the lung and interlobar
fissures: CT findings. J Comput Assist Tomogr. 18:34–38. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Neuman J, Rosioreanu A, Schuss A, Turi G,
Yung E, Trow TK, Williams L and Katz DS: Radiology-pathology
conference: Sclerosing hemangioma of the lung. Clin Imaging.
30:409–412. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bahk YW, Shinn KS and Choi BS: The air
meniscus sign in sclerosing hemangioma of the lung. Radiology.
128:27–29. 1978. View
Article : Google Scholar : PubMed/NCBI
|
|
10
|
Nam JE, Ryu YH, Cho SH, Lee YJ, Kim HJ,
Lee DY, Choe KO and Kim SJ: Air-trapping zone surrounding
sclerosing hemangioma of the lung. J Comput Assist Tomogr.
26:358–361. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang Y, Dai S and Wang E: Differential
gene expressions of polygonal cells and cuboidal cells in so-called
pulmonary sclerosing hemangioma. Zhongguo Fei Ai Za Zhi.
10:466–470. 2007.(In Chinese). PubMed/NCBI
|
|
12
|
Noguchi M, Morikawa A, Kawasaki M, Matsuno
Y, Yamada T, Hirohashi S, Kondo H and Shimosato Y: Small
adenocarcinoma of the lung. Histologic characteristics and
prognosis. Cancer. 75:2844–2852. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Cattoretti G, Becker MH, Key G, Duchrow M,
Schlüter C, Galle J and Gerdes J: Monoclonal antibodies against
recombinant parts of the Ki-67 antigen (MIB 1 and MIB 3) detect
proliferating cells in microwave-processed formalin-fixed paraffin
sections. J Pathol. 168:357–363. 1992. View Article : Google Scholar : PubMed/NCBI
|