Introduction
Colorectal carcinoma (CRC) is one of the most common
malignancies worldwide and the most common cause of
cancer-associated mortality in China (1). Ulcerative colitis (UC) is an
idiopathic colorectal inflammatory disorder that has a high risk of
developing into CRC if not effectively treated, at which point it
cannot be resolved (2–5). As an adenoma-carcinoma sequence,
UC-CRC undergoes a series of inflammatory processes and dysplasia
in order to develop into carcinoma; this is known as the
‘inflammation-cancer link’ (6).
The development of UC-CRC occurs through a multi-factorial and
multi-step evolution of molecular events, that includes the
activities of nuclear factor (NF)-κB and signal transducer and
activator of transcription-3 signaling pathways (7–10).
However, numerous key questions regarding the molecular mechanisms
and the involvement of subpopulations of immune cells, remain
unanswered, particularly regarding the microenvironment during
UC-CRC carcinogenesis.
UC is an autoimmune disease that affects numerous
organs, as well as the digestive system, and the development of
UC-CRC is associated with immune system malfunction (11). Macrophages have an indispensable
role in inflammation and carcinoma. Activated macrophages can mount
specific functions, which may be either pro-inflammatory or
anti-inflammatory, thereby contributing to local tissue
destruction, or regeneration and wound healing. These mechanisms
are dependent on the polarized phenotypes that the macrophages
acquire, within that particular microenvironment (12). Polarized macrophages are mainly
classified into two groups: Classically activated macrophages, M1;
and alternatively activated macrophages, M2 (13). M1 macrophages are characterized by
the secretion of pro-inflammatory cytokines, including interleukin
(IL)-1β, IL-6, IL-12, and tumor necrosis factor (TNF)-α.
Conversely, M2 cells induce a weak immune response and reduce
pro-inflammatory responses (14).
However, the mechanisms responsible for the changes in M1/M2
polarization, and their functions in UC-CRC carcinogenesis, have
yet to be elucidated.
Previous studies have shown that macrophage
infiltration is greater in patients with UC, as well as CRC, as
compared with healthy patients (15–17).
However, little is currently known about the dynamic and sequential
changes that occur in macrophages during the transition from UC to
CRC (18–21). In the present study, the variable
roles of colorectal macrophages and subpopulations were
investigated in the changing microenvironment during UC-CRC
carcinogenesis. M1/M2 macrophages were purified and sorted from a
chemically-induced mouse model, in order to evaluate the dynamic
changes of their expression and functions during the various phases
of the inflammation-cancer process.
Materials and methods
Animals and experimental procedure
All of the procedures in the present study involving
mice were performed in accordance with institutional guidelines and
the Ethics Committee Concerning the Care and Use of Experimental
Animals provided by the Institutional Review Board of The Third
Xiangya Hospital (Changsha, China). An azoxymethane (AOM) and
dextran sodium sulfate (DSS)-induced colitis-associated carcinoma
mouse model was used in the present study, which has been described
by previous methods (22–24). A total of 96 six-week-old male
Crj:CD-1 (ICR) mice, purchased from Hunan Slack King of Laboratory
Animals Co., Ltd., (Changsha, China) were randomized into either
the AOM/DSS group or the control group. The Disease Activity Index
(DAI) was used to evaluate disease development, by scoring body
weight loss, stool characterization and hematochezia, as previously
described (25). The two groups of
mice were randomly, sequentially sacrificed by exsanguination at
the end of week 0 (before treatment), and at weeks 2, 4, 7, 10, 16,
24 and 56 post-treatment. The mice were anesthetized with a cotton
ball dipped in ~0.5–1 ml ethyl ether in a sealed chamber, and were
sacrificed by exsanguination, through eye enucleation. Spontaneous
activity and the corneal reflex disappeared and no significant
contraction was observed in the limb muscles on stimulation.
Histopathology and immunohistochemistry
(IHC)
The mice were anaesthetized and sacrificed by
exsanguination, as detailed above. The colon was then incised
longitudinally along the main axis and washed twice with 1X
phosphate-buffered saline (PBS). Following careful macroscopic
inspection, half of the distal colon and rectum was cut
longitudinally, fixed with 4% paraformaldehyde, embedded in
paraffin, and subsequently cut into 5 μm sections. The sections
were then stained with hematoxylin and eosin, and assessed by two
investigators in a blind manner to provide a consensus on staining
patterns.
Immunohistochemical staining was performed using a
rabbit anti-mouse CD68 (cat.no BA3638; 1:100 dilution; Boster
Biological Technology, Inc., Wuhan, China) primary antibody, at 4°C
overnight. The sections were then incubated with biotin-conjugated
goat anti-rabbit secondary antibody (cat.no. BA1003; 1:2,000
dilution; Boster Biological Technology, Inc.). A 3,
3′-diaminobenzidine (DAB; Boster Biological Technology, Inc.) kit
was used for staining, at a 1:20 dilution. Omission of the primary
antibody was used as a negative control. The images were captured
using a BX51 microscope (Olympus America Inc., Center Valley, PA,
USA).
Macrophage isolation and sorting by
fluorescence-activated cell sorting (FACS)
Single-cell suspensions from the colonic tissue
samples were generated. The colonic tissue samples were minced into
1 mm3 pieces using opthalmic scissors and digested in
serum-free Dulbecco’s modified Eagle’s medium (DMEM), supplemented
with 1 mg/ml collagenase IV (Invitrogen Life Technologies,
Carlsbad, CA, USA), 1% hyaluronidase (Sigma-Aldrich, St Louis, MO,
USA), and 0.25% DNase I (Merck Millipore, Darmstadt, Germany). The
samples were incubated at 37°C for enzyme digestion, with
oscillation every 10–15 min. The normal colon samples were digested
for 2–2.5 h and the carcinoma samples were digested for 1–1.5 h,
until the tissues were dispersed into cell clusters or single
cells. The samples were then passed through a 70 μm cell strainer
(BD Biosciences, Franklin Lakes, NJ, USA), and the resulting cell
suspension was centrifuged at 500 g for 5 min, and subsequently
resuspended in 1X PBS.
The single-cell suspensions were incubated, at 4°C
for 30 min, with the following rat anti-mouse antibodies:
APC/Cy7-conjugated F4/80 (clone CI:A3–1), APC-conjugated CD68
(clone, FA-11), PerCP/Cy5.5-conjugated CD11b (clone, M1/70),
phycoerythrin-conjugated CD16/32 (clone 93) and fluorescein
isothiocyanate (FITC)-conjugated CD206 (clone, C068C2).
FITC-conjugated rat anti-mouse immunoglobulin G was used as a
negative control (clone poly4060). All of the antibodies were
purchased from Biolegend (San Diego, CA, USA), and used according
to the manufacturer’s instructions. The cells were washed twice
with stroke-physiological saline solution of mice (0.75%) and then
sorted and assessed by FACS, using a MoFloTM XDP
High-Performance Cell Sorter (Beckman Coulter, Brea, CA, USA). The
data were acquired and analyzed using Summit v5.2 software (Beckman
Coulter).
Purified cell cultures and ELISA
Following cell sorting by FACS, the purified cells
were cultured for 24 h, at 37°C in a 5% CO2 humidified
atmosphere, in DMEM (Life Technologies, Grand Island, NY, USA)
supplemented with 100 U/ml penicillin, 100 μg/ml streptomycin, and
10% fetal bovine serum (Life Technologies). The cells were
centrifuged at 700 g for 10 min and aliquots of the supernatant
were frozen at −80°C, until further use. Commercially available
ELISA kits for TNF-α, IL-12, and IL-10 were purchased from R&D
Systems, Inc. (Minneapolis, MN, USA), and were used according to
the manufacturer’s instructions. The results of the ELISA were
examined using the ParadigmTM Detection Platform
(Beckman Coulter).
RNA extraction and quantitative
polymerase chain reaction (qPCR)
Sorted colorectal M1/M2 macrophages were collected
following a 24 h culture. Total RNA was extracted using the RNeasy
Micro kit (Qiagen, Hilden, Germany) and cDNA synthesis was
conducted using the SuperScriptTM III First-Strand
Synthesis system (Invitrogen Life Technologies), according to the
manufacturer’s instructions.
A qPCR analysis was performed using the GoTaq qPCR
Master Mix (Promega Corp., Madison, WI, USA). The primers used were
GAPDH, CXCR4, GCSF, GMCSF, ICAM-1, IL-1β, IL-6, IL-10, IL-12,
TGFβ, TNFα, and VEGF, the sequences for which are shown
in Table I. The qPCR was performed
using a Bio-Rad CFK96TM Real-Time System (Bio-Rad
Laboratories, Inc., Hercules, CA, USA). The cycle conditions were
as follows: 2 mins at 95°C, 15 sec at 95°C and 1 min at 60°C for 59
cycles, followed by 10 sec at 95°C with a melt curve of 65–95°C and
increments of 0.5°C. The data were analyzed using Bio-Rad CFK
Manager 2.0 software. The relative gene expression levels were
quantified based on the cycle threshold value, and normalized to
the housekeeping gene GAPDH.
 | Table IPrimers used for quantitative
polymerase chain reaction analysis. |
Table I
Primers used for quantitative
polymerase chain reaction analysis.
| Gene | Forward primers
(5′→3′) | Reverse primers
(5′→3′) |
|---|
| CXCR4 |
aaagctagccgtgatcctca |
caccatttcaggctttggtt |
| G-CSF |
ccttcacttctgccttccag |
gctcaggtctaggccaagtg |
| GM-CSF |
atgcctgtcacgttgaatga |
ccgtagaccctgctcgaata |
| ICAM-1 |
agggctggcattgttctcta |
cttcagaggcaggaaacagg |
| IL-1β |
accccaaaagatgaagggctgctt |
tgcctgcctgaagctcttgttgat |
| IL-10 |
ggcagagaagcatggcccagaa |
ttcacctgctccactgccttgc |
| IL-12 |
gccggctatccagacaatta |
ggccaaactgaggtggttta |
| IL-6 |
ccggagaggagacttcacag |
cagaattgccattgcacaac |
| TGF-β |
ttgcttcagctccacagaga |
tggttgtagagggcaaggac |
| TNF-α |
tatggctcagggtccaactc |
ctccctttgcagaactcagg |
| VEGF |
tgatctgctccctccctcta |
aatgctttctccgctctgaa |
| GAPDH |
gggtgaggccggtgctgagtatg |
ggcagaaggggcggagatgatg |
Statistical analyses
All of the statistical analyses were performed using
SPSS version 17.0 software (SPSS Inc., Chicago, IL, USA). The
distributions of continuous variables were described by means and
standard errors. Statistical significance was determined using the
Independent Samples t-test. A P<0.05 was considered to indicate
a statistically significant difference.
Results
Establishment of the AOM/DSS mouse model
showing the sequence of inflammation, inflammatory hyperplasia,
dysplasia, and carcinoma to metastasis transition
Morphological and pathological analyses of the
colonic mucosa revealed a sequential process of inflammation by
week 2, inflammatory hyperplasia by week 4, dysplasia by week 7,
and carcinoma at weeks 10, 16, 24, and 56. Furthermore, peritoneal
metastasis was observed by week 56 in the mice of the AOM/DSS
group. Conversely, the control mice exhibited normal mucosa between
weeks 2 and 10, and mild inflammation between weeks 16 and 56
(Fig. 1). The morphological and
pathological analyses showed that the progression observed was
concordant with the UC-CRC process, including metastasis. These
results verify the successful establishment of the UC-CRC
carcinogenesis transition in vivo, using an AOM/DSS mouse
model that mimicked the development pattern observed in humans.
 | Figure 1Establishment of
inflammation-carcinoma metastasis sequence using the
azoxymethane/sextran sodium sulfate (AOM/DSS) mouse model. (A) The
Disease Activity Index (DAI) of the AOM/DSS group was higher, as
compared with the control group. DAI = (weight loss score + stool
characteristic score + hematochezia score)/3,
*P<0.05. (B) Morphological observation of the distal
colon and rectum cut longitudinally. Colonic mucosa with erosions
and ulcers was observed in weeks 2, 4, and 7. Neoplasms were
observed from week 10–56 in the AOM/DSS mice, as compared with the
control mice. (C) Histopathology of colonic mucosa by hematoxylin
and eosin staining (magnification, ×200) In the AOM/DSS group, the
colonic mucosa was examined for (a) inflammation at week 2, (b)
inflammatory hyperplasia at week 4 and (c) dysplasia at week 7.
(d-g) Carcinoma and (o and p) peritoneal metastasis through the
intestinal wall was observed at week 56. No metastasis was observed
in the (q-s) celiac lymph node, liver, or lung. (h-k) Normal mucosa
and (l-n) mild inflammation were observed in the control group
mice. |
Distribution of colorectal macrophages
and M1/M2 subpopulations, during UC-CRC carcinogenesis
To investigate the expression pattern of macrophages
in the colonic mucosa during the UC-CRC sequential process, IHC was
performed using an antibody specifically targeting the
macrophage-specific marker CD68. The percentage of CD68+
macrophages increased sequentially from the normal mucosa to
inflammatory hyperplasia, dysplasia, and carcinoma transition in
the mice of the AOM/DSS group. In addition, a greater number of
CD68+ cells were observed in the mucosa adjacent to the
carcinoma, as compared with the carcinoma tissue itself (Fig. 2A).
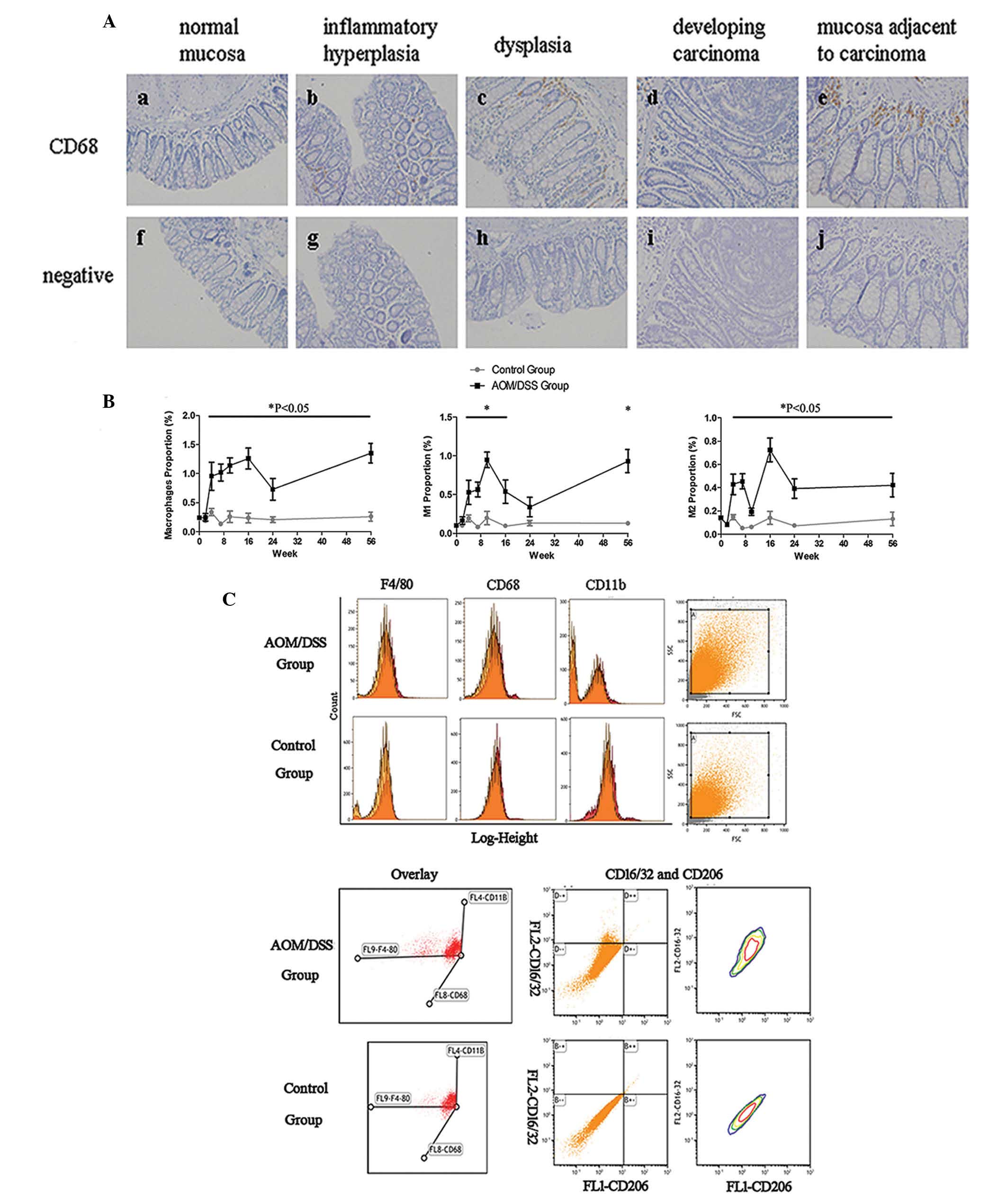 | Figure 2Expression pattern of macrophages and
M1/M2 macrophage subpopulations in colonic tissue during ulcerative
colitis-colorectal carcinoma (UC-CRC) carcinogenesis. (A) The
expression pattern of macrophages was assessed by
immunohistochemistry using an anti-CD68 antibody. CD68 was
localized to the cell membranes. Hematoxylin was used to stain the
nuclei. The expression of CD68+ cells increased
sequentially from (a) normal mucosa to (b) inflammatory
hyperplasia, (c) dysplasia, and (d) carcinoma. (e) The
CD68+ cells were also observed in the mucosa adjacent to
carcinoma tissue. All of the samples were compared to the (f-j)
negative control. (B) Colorectal macrophages were evaluated and
purified by fluorescence-activated cell sorting, using the
indicated surface markers. Further sorting generated the M1 and M2
macrophage subpopulations. *P<0.05. (C) Flow
cytometric description of colorectal macrophages and M1/M2
macrophage subpopulations isolated from azoxymethane/sextran sodium
sulfate (AOM/DSS) mice at week 4 post-induction. Single-parameter
histograms show the expression of macrophage markers F4/80, CD68,
or CD11b (red), and negative control (orange), for the two groups.
A three dimensional coordinate system shows
F4/80+CD68+CD11b+ cells by x, y,
and z axes. Two-parameter histograms show
F4/80+CD68+CD11b+ cells stained
with anti-CD16/32 in the FL2-phycoerythrin channel and anti-CD206
in the FL1-fluorescein isothiocyanate channel. CD16/32 and CD206
were markers for M1 and M2 macrophages, respectively, based on
F4/80, CD68, and CD11b staining. All of the samples were compared
to the immunoglobulin G isotype control. |
To further evaluate the changes in the presence of
macrophages and the M1/M2 macrophage subpopulations in the colonic
tissue during UC-CRC transition, M1 and M2 macrophages were
examined using antibodies targeting distinct cellular markers.
CD68, F4/80, and CD11b are widely recognized as surface markers of
macrophages in mice. Colorectal macrophages were initially
identified in the AOM/DSS mice, by FACS, at week 2 post-induction,
and continued to week 10 post-induction, which was the time at
which the carcinoma tissue first appeared. The highest macrophage
percentage was observed in inflammatory hyperplasia at week 4,
which was concordant with the results obtained from the
CD68+ IHC staining. The percentage of M1 macrophages
present in the colon mucosa was similar to that of the total number
of macrophages present during this process, but exhibited a marked
increase during the transition from dysplasia to carcinoma, between
weeks 7 and 10. Conversely, the percentage of M2 macrophages
remained constant during this process. The mice in the control
group maintained low levels of both M1 and M2 macrophages in the
colonic tissue, during the entire experimental period (Fig. 2B and C).
The expression pattern of M1 and M2 macrophages
changed as the carcinoma progressed to peritoneal metastasis, from
week 10 post-induction. A decrease in the number of M1 and M2
macrophages was observed as the carcinoma progressed in situ
from week 16–24 post-induction, and an earlier decrease in the
number of cells was observed for the M1 subpopulation after week
10, as compared with the M2 subpopulation, which markedly increased
between weeks 10 and 16. This increase was followed by an immediate
decrease between weeks 16 and 24, suggesting that the expression of
M2 macrophages is most likely correlated with the early stages of
carcinoma development. As the carcinoma invaded through the colonic
mucosa, resulting in peritoneal metastasis, the total number of
macrophages markedly increased between weeks 24 and 56. This was
similar to the M1 macrophage subpopulation and most likely
associated with carcinomatous peritonitis, as a result of the
metastasis. Furthermore, a moderate increase in the number of M2
macrophages was maintained during this process. These data suggest
that marked changes occur in the M1 and M2 macrophage
subpopulations during UC-CRC carcinogenesis transition,
particularly during inflammatory hyperplasia. These results
indicate that this period during UC-CRC may be a key event in the
progression of inflammation to carcinoma.
Functional changes in the M1/M2
subpopulations during the UC-CRC process
The results of the present study indicate that the
inflammatory hyperplasia step, during the UC-CRC process, may be a
key point where marked changes occur in the expression patterns of
total macrophages, and M1 and M2 subpopulations. These changes are
then followed by dysplasia, carcinoma, and metastasis. To evaluate
the roles of M1/M2 macrophages in the pro- and anti-inflammatory
balance of the tumor microenvironment during this process, the mRNA
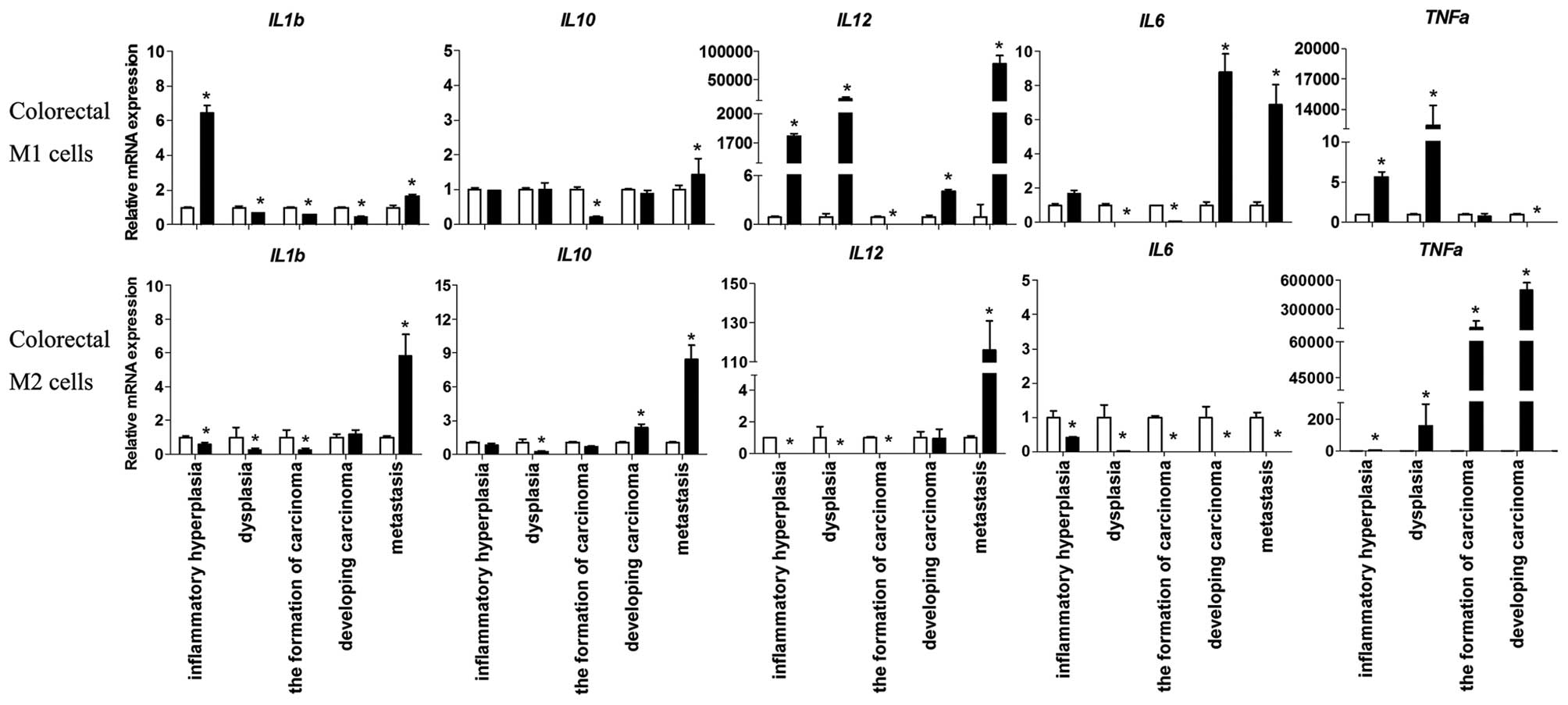
expression levels of various secreted cytokines, including IL-1β,
IL-6, IL-10, IL-12, and TNF-α, were determined by qPCR.
Pro-inflammatory cytokines were expressed by colorectal M1
macrophages during inflammatory hyperplasia (IL-1β, IL-6) and
dysplasia (IL-12, TNF-α), thus indicating that MI macrophages have
a positive role during the process of carcinoma. In addition, high
expression levels of the pro-inflammatory factors IL-1β, IL-6, and
IL-12, as well as the anti-inflammatory cytokine IL-10, were
detected during metastasis. The suppression of anti-inflammatory
IL-10 was observed in the colorectal M2 macrophages, which occurred
concomitantly with low expression levels of pro-inflammatory
cytokines, prior to carcinoma formation. Conversely, high
expression levels of pro- and anti-inflammatory cytokines were
detected during metastasis (Fig.
3).
To investigate further, ELISAs were performed to
measure the release of cytokines from the purified M1/M2
macrophages, following a 24 h in vitro culture. Concordant
with the mRNA expression data, M1-induced IL-12 and TNF-α secretion
was markedly upregulated in the inflammatory hyperplasia and
metastasis colonic samples from the AOM/DSS mice. Furthermore,
there was a concomitant release of IL-10. The suppression of
M2-induced IL-10, IL-12, and TNF-α secretion was observed during
the transition from inflammatory hyperplasia to carcinoma, but
these cytokines were again expressed at high levels during
metastasis, which was concordant with the mRNA expression data
(Figs. 3 and 4).
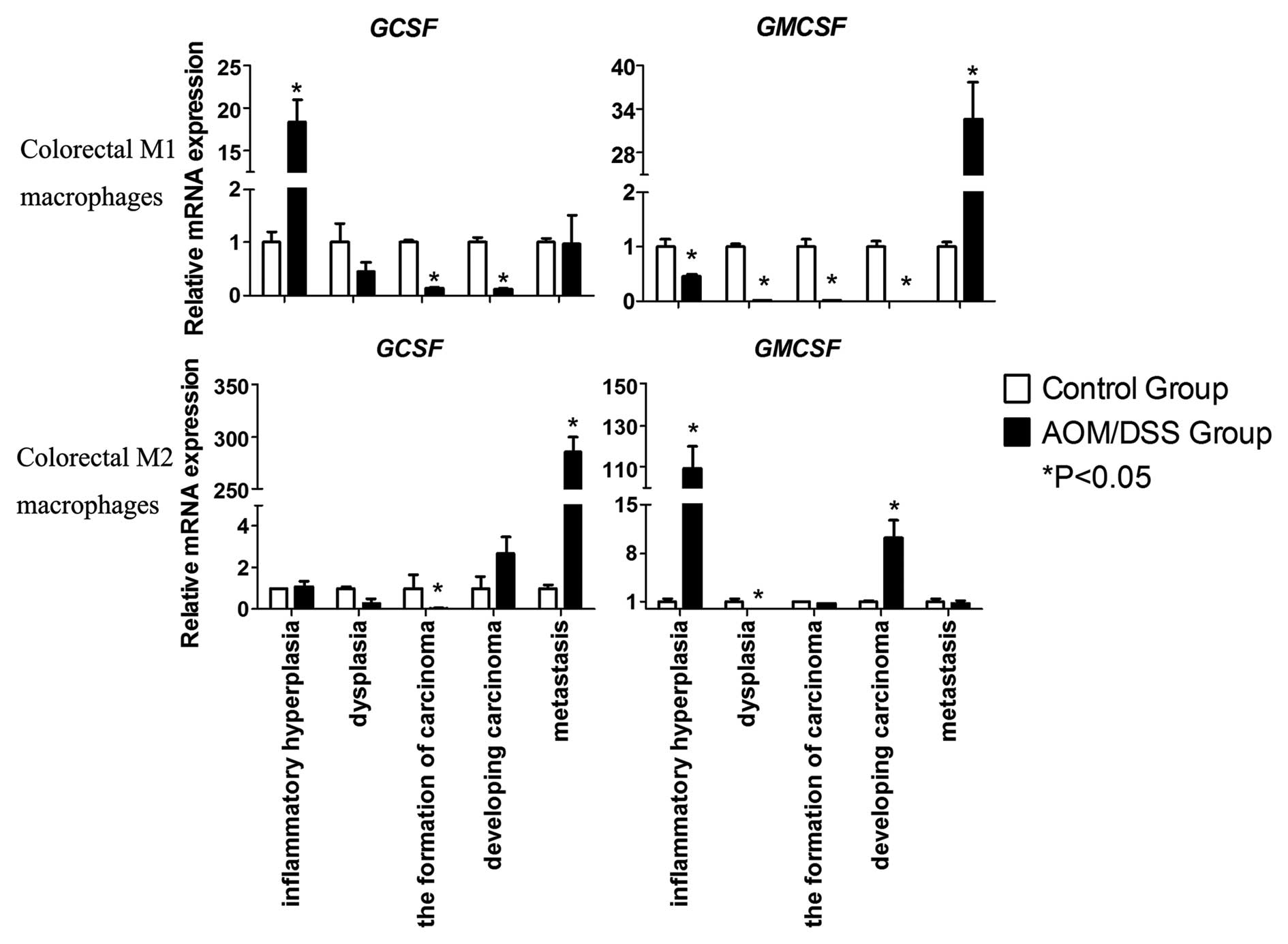
To determine the recruitment and migration of
macrophages in CRC and peritoneal metastasis through autocrine
regulation, granulocyte colony-stimulating factor (G-CSF) and
granulocyte macrophage colony-stimulating factor (GM-CSF)
expression levels were determined. G-CSF expression levels were
markedly increased in the M1 macrophages from the inflammatory
hyperplasia samples and M2 macrophages from the metastasis samples
(Fig. 5). Notably, the expression
levels of GM-CSF were high in the M2 macrophages isolated from the
inflammatory hyperplasia samples and the M1 macrophages from the
metastasis samples, as compared with the other stages of the UC-CRC
transition (Fig. 5). These results
were concordant with the flow cytometric data.
C-X-C chemokine receptor type 4 (CXCR4) expression
levels were elevated in the M2 macrophages isolated from dysplastic
and metastatic tissue samples from the AOM/DSS mice, as well as in
the M1 macrophages from the inflammatory hyperplasia samples.
Furthermore, vascular endothelial growth factor (VEGF) expression
levels were markedly elevated in the M1 and M2 macrophages isolated
from the carcinoma tissues (Fig.
6). These results indicate that coordinated CXCR4 and VEGF
enhancement in colorectal macrophages may contribute to invasion
and metastasis of carcinoma, especially in the M1 macrophages
during the later stages of the UC-CRC transition. Transforming
growth factor (TGF)-β expression levels were markedly elevated in
the colorectal macrophages during carcinoma formation, especially
in the M2 macrophages (Fig. 6);
however, this elevation was not sustained in the later stages of
the UC-CRC transition. These results indicate that TGF-β may have
an essential role in the proliferation of CRC cells in the earlier
stages of carcinogenesis.
The mRNA expression levels of intercellular adhesion
molecule-1 (ICAM-1) were also assessed in the M1 and M2
macrophages, as this is a factor known to have a role in the
transmigration of monocytes from the circulation to tissue. ICAM-1
mRNA expression levels were markedly increased in the M2
macrophages isolated from the developing carcinoma and metastatic
tissues of the AOM/DSS mice, as compared with the M1 macrophages.
These results indicate that M2 macrophages may have a greater
effect on attracting circulating monocytes, as compared with the M1
macrophages, during the later stages of the UC-CRC transition
(Fig. 6).
Discussion
CRC is associated with a marked increase in the
number of macrophages, particularly M2 macrophage polarization,
which has previously been shown to correlate with metastasis and
poor prognosis of the disease (11–14,26,27).
The tumor microenvironment is affected by sequential changes and
pathological progression, which may lead to changes in macrophage
populations and macrophage-dependent inflammatory activity.
Therefore, it is critically important to genetically define the
expression pattern and function of M1/M2 macrophages during
inflammation, and in the tumor microenvironment.
The present study used the AOM/DSS mouse model of
UC-CRC carcinogenesis. The expression of macrophages markedly
increased as the disease progressed from normal mucosa to
inflammatory hyperplasia, dysplasia, and carcinoma, as detected by
IHC. These results were concordant with previous studies (22–24),
and further confirmed the sequential changes between pathological
stages and macrophage expression patterns. The M1/M2 macrophage
expression pattern is complex and diverse during CRC and peritoneal
metastasis transitions; therefore, it remains unclear whether M1/M2
polarization alone may explain the various functional transitions
during the UC-CRC process. The most substantial changes in M1/M2
macrophage expression occurred during the period between
inflammatory hyperplasia and formation of carcinoma in the colonic
tissue. These results indicate that inflammatory hyperplasia is a
key transition event between inflammation and the development of
carcinoma.
The expression pattern of colorectal M1 macrophages
was similar to that of total macrophages. Notably, the expression
of colorectal M2 macrophages markedly increased in the carcinoma
stage, between weeks 10 and 16; however, it hardly changed during
the other stages of the disease. These data indicate that the
subpopulation of M2 macrophages is relatively stable during CRC
development, and is involved in the early development of carcinoma,
induced by a changing microenvironment. It may be hypothesized,
that the changes to the expression levels of the described factors
in M1/M2 macrophages, may correlate with the functional changes
during the UC-CRC process.
Macrophage activation can be either pro- or
anti-inflammatory in an adaptive immune response, depending on the
M1/M2 phenotype (15,28,29).
Macrophage-mediated inflammation has a differential effect in
different stages of colon cancer and the present study demonstrated
that the pro- and anti-inflammatory axis is unbalanced in
colorectal M1/M2 macrophages. The present study demonstrated a loss
of anti-inflammatory IL-10, during the transition from inflammation
to carcinoma formation, and the coordinated promotion of pro- and
anti-inflammatory cytokines in the development of carcinoma and
metastasis by M2 macrophages. These results confirm that these
cells have an important role in the promotion of CRC through
inhibition of the pro- and anti-inflammatory response.
Previous studies have shown that macrophages in CRC
are pro-inflammatory in function, and inhibit the proliferation of
tumor cells through an anti-tumor type-1 inflammatory response
(30). The results of the present
study suggest that M1 macrophages conferred some, but not all, of
the pro-inflammatory responses during carcinoma development in the
UC-CRC process. Whereas, the M2 macrophages had a weak
anti-inflammatory response and expressed a strong pro-tumor
influence. Furthermore, the aberrant enhancement of the pro- and
anti-inflammatory axis involved in the migration and invasion of
carcinoma during metastasis, by both M1 and M2 macrophages,
suggested that functional changes in M1 macrophages occurred during
the transition of carcinoma in situ to metastasis with an
unchanged polarization phenotype.
Notably, the expression levels of G-CSF and GM-CSF
in colorectal macrophages were markedly upregulated during
inflammatory hyperplasia and metastasis, respectively. These
results suggest a potential reason as to why there is a marked
recruitment of colorectal macrophages, during the UC-CRC process,
which is concordant with flow cytometric data. Conversely, other
studies have shown that macrophage infiltration is an obligatory
step for tumor cell migration, invasion, and metastasis in both CRC
and breast cancer (31,32,33).
Previous studies have shown that CXCR4, VEGF, TGF-β, ICAM-1 are
associated with various roles of macrophages during the process of
carcinoma invasion and metastasis (34–35).
The present study also assessed the expression levels of CXCR4,
VEGF, and TGF-β, in order to define the role of M1/M2 macrophages
in migration and invasion. The results of the present study
indicate that a coordinated enhancement of CXCR4 and VEGF
expression in colorectal macrophages, especially M2 macrophages,
contributed to invasion and peritoneal metastasis of CRC. The
expression levels of TGF-β were markedly elevated during carcinoma
development in colorectal M2 macrophages, indicating a potential
role for TGF-β at this stage of the disease, as compared with the
other stages. In addition, CXCR4, VEGF, and TGF-β expression levels
were much higher in colorectal M2 macrophages, as compared with the
M1 macrophages isolated from the various stages of the UC-CRC
process. These results suggest that M2 macrophages have a critical
role in the initiation, promotion, and metastasis of CRC, which is
concordant with the changes observed in inflammatory cytokine
expression. ICAM-1 expression levels were also markedly upregulated
in colorectal M2 macrophages, as compared with the M1 macrophages
at all stages of the disease process, thus indicating that M2
macrophages may impart a stronger effect on metastasis than M1
macrophages. The ICAM-1 expression analysis also confirmed the
pro-tumor role of colorectal M2 macrophages in carcinoma
progression and metastasis.
The percentage of colorectal M2 macrophages was
markedly increased as the pathological states progressed in the
UC-CRC process. Furthermore, colorectal M2 cells not only broke the
balance of the pro- and anti-inflammatory axis, but also enhanced
the expression levels of CXCR4, VEGF, TGF-β, and ICAM-1, which are
all known to promote CRC and metastasis. Previous studies have
shown opposite results and have indicated that M1 macrophages
impart some effects on CRC development similar to those of M2
macrophages (29–32). However, the pro-tumor effects were
not as severe as the effects demonstrated by M2 macrophages during
UC-CRC carcinogenesis in the present study. In addition, functional
changes also occurred in M1 macrophages without any change to the
phenotype during the development of carcinoma and metastasis, but
not during the stages of inflammation, dysplasia, or carcinoma.
Functional changes in these cells, particularly those associated
with inflammation and migration, occurred earlier in the process
than polarization, during UC-associated carcinogenesis of the
colon.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (nos. 81172300, 81272975,
81272736), the Key Project of Hunan Provincial Natural Science
Foundation (no. 12JJ2044), the Hunan Provincial Innovation
Foundation for Postgraduate (no. CX2011B073), and the Mittal
Student Innovation Project (no. 11MX25). The authors of the present
study would also like to acknowledge the members of their lab,
Professor Songqing Fan and Lei Shi (The Second Xiangya Hospital),
and the technical support of Beckman Coulter and Medjaden
Bioscience Limited.
References
|
1
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global Cancer Statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Nathan C and Ding A: Nonresolving
inflammation. Cell. 140:871–882. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Lakatos L, Mester G, Erdelyi Z, et al:
Risk factors for ulcerative colitis-associated colorectal cancer in
a Hungarian cohort of patients with ulcerative colitis: results of
a population-based study. Inflamm Bowel Dis. 12:205–211. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lakatos PL and Lakatos L: Risk for
colorectal cancer in ulcerative colitis: changes, causes and
management strategies. World J Gastroenterol. 14:3937–3947. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Gong W, Lv N, Wang B, et al: Risk of
ulcerative colitis-associated colorectal cancer in China: a
multi-center retrospective study. Dig Dis Sci. 57:503–507. 2012.
View Article : Google Scholar
|
|
6
|
Zisman TL and Rubin DT: Colorectal cancer
and dysplasia in inflammatory bowel disease. World J Gastroenterol.
14:2662–2669. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Karin M: Nuclear factor-kappaB in cancer
development and progression. Nature. 441:431–436. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Pikarsky E, Porat RM, Stein I, et al:
NF-kappaB functions as a tumour promoter in inflammation-associated
cancer. Nature. 431:461–466. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bollrath J, Phesse TJ, von Burstin VA, et
al: gp130-mediated Stat3 activation in enterocytes regulates cell
survival and cell-cycle progression during colitis-associated
tumorigenesis. Cancer Cell. 15:91–102. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Grivennikov S, Karin E, Terzic J, et al:
IL-6 and Stat3 are required for survival of intestinal epithelial
cells and development of colitis-associated cancer. Cancer Cell.
15:103–113. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Baumgart DC and Sandborn WJ: Inflammatory
bowel disease: clinical aspects and established and evolving
therapies. Lancet. 369:1641–1657. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Mosser DM and Edwards JP: Exploring the
full spectrum of macrophage activation. Nat Rev Immunol. 8:958–969.
2008. View
Article : Google Scholar : PubMed/NCBI
|
|
13
|
Lawrence T and Natoli G: Transcriptional
regulation of macrophage polarization: enabling diversity with
identity. Nat Rev Immunol. 11:750–761. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Martinez FO, Sica A, Mantovani A and
Locati M: Macrophage activation and polarization. Front Biosci.
13:453–461. 2008. View
Article : Google Scholar
|
|
15
|
Siveen KS and Kuttan G: Role of
macrophages in tumour progression. Immunol Lett. 123:97–102. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Xie J and Itzkowitz SH: Cancer in
inflammatory bowel disease. World J Gastroenterol. 14:378–389.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bar-On L, Zigmond E and Jung S: Management
of gut inflammation through the manipulation of intestinal
dendritic cells and macrophages? Semin Immunol. 23:58–64. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Sica A, Schioppa T, Mantovani A and
Allavena P: Tumor-associated macrophages are a distinct M2
polarised population promoting tumor progression: potential targets
of anti-cancer therapy. Eur J Cancer. 42:717–727. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Bailey C, Negus R, Morris A, et al:
Chemokine expression is associated with the accumulation of tumour
associated macrophages (TAMs) and progression in human colorectal
cancer. Clin Exp Metastasis. 24:121–130. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Sica A, Allavena P and Mantovani A: Cancer
related inflammation: the macrophage connection. Cancer Lett.
267:204–215. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Green CE, Liu T, Montel V, et al:
Chemoattractant signaling between tumor cells and macrophages
regulated cancer cell migration, metastasis and neovascularization.
PLoS One. 4:e67132009. View Article : Google Scholar
|
|
22
|
Wirtz S, Neufert C, Weigmann B and Neurath
MF: Chemically induced mouse models of intestinal inflammation. Nat
Protoc. 2:541–546. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Clapper ML, Cooper HS and Chang WC:
Dextran sulfate sodium-induced colitis-associated neoplasia: a
promising model for the development of chemopreventive
interventions. Acta Pharmacol Sin. 28:1450–1459. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Lai CS, Tsai ML, Cheng AC, et al:
Chemoprevention of colonic tumorigenesis by dietary hydroxylated
polymethoxyflavones in azoxymethane-treated mice. Mol Nutr Food
Res. 55:278–290. 2011. View Article : Google Scholar
|
|
25
|
Cooper HS, Murthy SN, Shah RS and
Sedergran DJ: Clinicopathologic study of dextran sulfate sodium
experimental murine colitis. Lab Invest. 69:238–249.
1993.PubMed/NCBI
|
|
26
|
Forssell J, Oberg A, Henriksson ML,
Stenling R, Jung A and Palmqvist R: High macrophage infiltration
along the tumor front correlates with improved survival in colon
cancer. Clin Cancer Res. 13:1472–1479. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Mantovani A, Sica A and Locati M:
Macrophage polarization comes of age. Immunity. 23:344–346. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Mantovani A: Inflammation and cancer: the
macrophage connection. Medicina. 67:32–34. 2007.
|
|
29
|
Mantovani A: Tumor-associated macrophages
in cancer-related inflammation. Immunotherapy. 3:21–22. 2011.
View Article : Google Scholar
|
|
30
|
Ong SM, Tan YC, Beretta O, et al:
Macrophages in human colorectal cancer are pro-inflammatory and
prime T cells towards an anti-tumour type-1 inflammatory response.
Eur J Immunol. 42:89–100. 2012. View Article : Google Scholar
|
|
31
|
Condeelis J and Pollard JW: Macrophages:
obligate partners for tumor cell migration, invasion, and
metastasis. Cell. 124:263–266. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhou Q, Peng RQ, Wu XJ, et al: The density
of macrophages in the invasive front is inversely correlated to
liver metastasis in colon cancer. J Transl Med. 8:132010.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Solinas G, Germano G, Mantovani A and
Allavena P: Tumor-associated macrophages (TAM) as major players of
the cancer-related inflammation. J Leukoc Biol. 86:1065–1073. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Imtiyaz HZ, Williams EP, Hickey MM, et al:
Hypoxia-inducible factor 2alpha regulates macrophage function in
mouse models of acute and tumor inflammation. J Clin Invest.
120:2699–2714. 2010. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
Low-Marchelli JM, Ardi VC, Vizcarra EA,
van Rooijen N, Quigley JP and Yang J: Twist1 induces CCL2 and
recruits macrophages to promote angiogenesis. Cancer Res.
73:662–671. 2013. View Article : Google Scholar : PubMed/NCBI
|