Introduction
Breast cancer (BC) is the most common type of cancer
among females worldwide and is the second leading cause of
cancer-associated mortality, accounting for 39,520 fatalities among
US females during 2011 (1). Recent
molecular classification of BC identified subtypes with diverse
histopathological features, clinical outcomes and therapeutic
implications. Generally, BC is classified into two subgroups, those
with tumors that are estrogen receptor-α (ER)-positive; and those
that are with ER-negative (2).
Triple-negative BC (TNBC) represents a heterogeneous group of
tumors that account for 20–25% of all cases of BC. In TNBC, ER,
progesterone receptor and human epidermal growth factor 2 (HER2)
expression is absent and the cancer is basal-like (expresses basal
epithelial cell genes, such as those encoding cytokeratins 5 and
14) (3). Collectively, TNBC
represents an aggressive subtype of BC, which is often associated
with a high risk of local recurrence, distant metastasis and
mortality during the first 3–5 years of follow up (4–6). At
present, there is no targeted therapy for TNBC and patients often
respond poorly to routine treatment with conventional third
generation combination chemotherapy, which is commonly complicated
by local recurrence, distant metastasis, frequent recurrence and
high mortality rates (4,7). Therefore, there is an increasing
demand for novel biomarkers and biological targeted therapies to
improve clinical outcome and patient prognosis (8).
A number of circulating tumor proteins have been
suggested as prognostic and predictive biomarkers that may be used
to assess patients with BC at any stage of the disease, one of
which is transforming growth factor-β (TGF-β) (9). Overexpression of TGF-β1 in breast
tumors is commonly associated with late-stage disease and/or poor
outcome, with frequent occurrence of bone and lung metastases. A
study has demonstrated that elevated levels of TGF-β1 and vascular
endothelial growth factor (VEGF) promote angiogenesis and stimulate
the production of extracellular matrix in the vicinity of the tumor
cells. This provides a scaffold for cell proliferation and
migration, which facilitates tumor metastasis (10). In addition, treatment with TGF-β1
neutralizing antibodies or receptor kinase inhibitors has been
indicated to strongly prevent the development of lung and bone
metastases in mouse models of TNBC or basal-like BC. This was
attributed to the inhibition of angiogenesis, derepression of
antitumor immunity or the reversal of the mesenchymal, motile and
invasive phenotypes characteristic of basal-like and HER2-positive
BC cells (11).
High serum levels of VEGF-A alone have been commonly
associated with unfavorable clinical outcomes, including disease
progression, poor response to treatment and reduced survival rates
in patients with BC. Therefore, VEGF-A is considered as a
prognostic marker and a candidate for targeted therapy in BC, in
addition to other solid and hematological malignancies (12,13).
Insulin-like growth factor I (IGF-I) is an important
regulator of growth, survival, migration and invasion and is
clearly implicated in BC (14).
IGF-I stimulation contributes to BC progression via its mitogenic
and anti-apoptotic effects on the mammary epithelial cells
(15) and additionally protects BC
cells from the toxic effects of radio- and chemotherapy (16,17).
IGF-IR increases angiogenesis/lymphangiogensis and induces
alterations in the integrins and cell adhesion complexes, leading
to an increase in cancer cell metastasis (18). Aberrant expression and activity of
IGF-I/IGF-IR has been previously detected in proliferative breast
tissues in conjunction with significant alterations in cellular
morphology, which are associated with cancer progression (15).
Therefore, in the current study the prognostic and
predictive values of aberrant serum expression of TGF-β1, VEGF-A
and IGF-I/IGF-IR were investigated in patients with TNBC and
non-TNBC. To achieve this, the expression levels of the markers in
the two groups were correlated with the standard prognostic factors
for BC, the response to treatment and the survival rates. It was
hypothesized that the results may aid in the further elucidation of
the function of these interrelated proteins in the development and
progression of TNBC. Additionally, the current study aimed to
identify a group of serological biomarkers that may be used to
monitor patients with BC during the course of the disease.
Materials and methods
Study cohort
The present study involved 96 recently diagnosed
patients with BC who attended the National Cancer Institute (NCI)
at Cairo University, (Cairo, Egypt) between September 2009 and
October 2012. All patients were clear of distant metastasis at the
initial diagnosis. Based on histological and immunophenotypical
assessment of tumor samples, patients were separated into the
following groups: i) TNBC, n=43, mean age=51.91±12.34 years,
range=30–78 years; and ii) non-TNBC, n=53, mean age=52.77±12.13
years, range=27–81 years. Twenty healthy females who were
age-matched (mean age=35±13.94 years; range, 22–64 years) were also
included in the study as the controls.
Written informed consent was obtained from all
participants prior to enrollment in the study. The Institutional
Review Board of the NCI approved the study protocol, which was in
accordance with the 2007 Declaration of Helsinki.
Inclusion criteria
Patients enrolled in the study were ≥18 years old;
presented with histologically-confirmed BC (TNBC or non-TNBC); had
an Eastern Cooperative Oncology Adequate performance: ≤2 (19); and exhibited adequate bone marrow
(WBC count, ≥3.0×109/l; ANC, ≥1.5×109/l;
platelet count, ≥100×109/l; hemoglobin level, ≥9 g/l),
liver (serum bilirubin, <1.5×ULN; ALT and AST levels, <3
times normal values) and kidney (plasma creatinine level, <1.5
times normal value) function. Exclusion criteria included
pregnancy, breast-feeding, an active secondary malignancy or
involvement in another clinical trial.
Treatment and follow-up
Patients received FEC100 as follows:
Cyclophosphamide (Baxter, Deerfield, IL, USA) 500 mg/m2
IV diluted in 50 ml of normal saline as a 5- to 10-minute
intravenous infusion on day 1; Epirubicin (Pfizer, New York, NY,
USA) 100 mg/m2 IV diluted in 50 ml of normal saline as a 5- to
10-minute intravenous infusion on day 1; Fluorouracil (Ebewe Pharm,
Unterach, Austria) 500 mg/m2 IV D1 as a bolus
intravenous injection adjuvant for three cycles followed by 75
mg/m2 docetaxel (Sanofi-Aventis, Paris, France) for four
cycles every 21 days with standard pre-medication (anti-emetics,
anti-allergins and proton pump inhibitors). Radiotherapy was
administered when indicated subsequent to the completion of
chemotherapy (50 Gy in 2.0 Gy daily fractions) followed by hormonal
therapy whenever indicated, according to the hormonal status of the
tumor in ER and/or PR positive types of tumor. In the cases with
metastasis, responses to treatment were assessed using the Response
Evaluation Criteria in Solid Tumors system (20) and accordingly, patients were
categorized into the following groups: i) Complete response (CR),
complete disappearance of disease confirmed at 4 weeks; ii) partial
response (PR), 30% reduction; iii) stable disease (SD), neither CR
nor PR criteria were fulfilled; and iv) progressive disease (PD)
presence of metastasis and/or recurrence observed as a 20% increase
in tumor measurements or appearance of new lesions. The median
follow-up period was 33 months. Local recurrence and distant
metastases were assessed; and disease-free survival (DFS) and
overall survival (OS) were calculated.
Blood acquisition and serum
preparation
A total of 5 ml venous blood was collected in a
sterile 15 ml plastic Falcon tube (Becton-Dickinson, Franklin
Lakes, NJ, USA), was left to clot and was then centrifuged at
11,000 × g for 15 min. Serum samples were stored at −80°C until
they were required for the assay.
Detection of VEGF-A, TGF-β1, IGF-I and
IGF-IR expression in sera
Quantitative enzyme-linked immunosorbent assay
(ELISA) kits were used to assess the levels of human IGF-I (cat.
no. EIA-4140; DRG Instruments GmbH, Marburg, Germany), IGF-IR (cat.
no. # OK-0226; Uscn Life Sciences Inc., Missouri, TX, USA), VEGF-A
(cat. no. BMS277/2; eBioscience Bender Medsystems GmbH, Vienna,
Austria) and TGF-β1 (cat. no. EIA-1864; DRG Instruments GmbH)
according to the manufacturer’s instructions. A total of 100 μl
(for VEGF-A, IGF-I and IGF-IR) or 200 μl (for TGF-β1) of prediluted
sera was added to micro-titer wells precoated with anti-human IGF-I
monoclonal, anti-human IGF-IR monoclonal, anti-human VEGF-A
polyclonal and anti-human TGF-B1 polyclonal antibodies followed by
a biotin-conjugated anti-human IGF-I, IGF-IR, VEGF-A, mouse
anti-TGF-β1 antibodies and streptavidin-horseradish peroxidase.
Color was developed using a tetramethyl benzidine-hydrogen dioxide
mixture and terminated with sulfuric acid. The absorbance of each
well was determined using a spectrophotometer (PR 3100 TSC
Microplate Reader, Bio-Rad, Hercules, TX, USA).
Statistical analysis
SPSS, version 20.0 (IBM SPSS, Armonk, NY, USA) was
used for statistical analysis and data were expressed as the mean
rank or mean ± standard deviation for continuous variables.
Categorical variables were assessed using the χ2 test
when appropriate. All P-values are two-tailed and P<0.05 was
considered to indicate a statistically significant difference.
Comparisons of the markers between different groups were performed
using the Mann-Whitney U test or Kruskal Wallis one-way analysis of
variance. The associations between TNBC, clinicopathologic
variables and the significance of markers were examined using the
χ2 test. The association with survival was analyzed
using Kaplan-Meier analysis and curves were compared using the
log-rank test and Cox regression analysis to adjust for other
prognostic indicators. The Receiver Operating Characteristic (ROC)
curve was used to determine the appropriate cut-off level of
markers in the diagnosis of TNBC.
Results
Patient characteristics
In general, the patients with TNBC presented with
more aggressive tumors compared with those with non-TNBC, with a
higher incidence of lymph nodes metastasis (P<0.001), recurrence
(P<0.001) and distant metastasis (P<0.001). Patients with
TNBC exhibited significantly lower response rates compared with the
patients with non-TNBC (P<0.001) (Table I).
 | Table IClinicopathological features of the
patients with triple-negative and non-triple-negative breast cancer
(descriptive statistics). |
Table I
Clinicopathological features of the
patients with triple-negative and non-triple-negative breast cancer
(descriptive statistics).
| Patient
characteristic | Total no. | Triple-negative, %
(n) total=43 |
Non-triple-negative, % (n) total=53 | Statistics |
|---|
| Age at diagnosis
(years) |
| ≤35 | 8 | 9.3 (4) | 7.55 (4) |
χ2=0.32 |
| 36–49 | 34 | 32.56 (14) | 37.74 (20) | P=0.85 |
| ≥50 | 54 | 58.14 (25) | 54.71 (29) | |
| Tumor size
(cm) |
| <4 | 52 | 44.2 (19) | 62.30 (33) |
χ2=3.13 |
| ≥4 | 44 | 55.8 (24) | 37.70 (20) | P=0.071 |
| Family history |
| No | 85 | 95.30 (41) | 83.02 (44) |
χ2=3.40 |
| Yes | 11 | 4.70 (2) | 16.98 (9) | P=0.065 |
| Menopausal
status |
|
Pre-menopausal | 44 | 44.19 (19) | 47.17 (25) |
χ2=0.085 |
|
Post-menopausal | 52 | 55.81 (24) | 52.83 (28) | P=0.71 |
| Tumor stage |
| Early (I-II) | 55 | 60.50 (26) | 54.70 (29) |
χ2=0.50 |
| Late (III-IV) | 41 | 39.50 (17) | 45.30 (24) | P=0.48 |
| Tumor grade |
| 1 | 2 | 0.00 (0) | 3.80 (2) |
χ2=3.13 |
| 2 | 79 | 79.07 (34) | 84.90 (45) | P=0.28 |
| 3 | 15 | 20.90 (9) | 11.30 (6) | |
| Nodal status |
| Negative | 26 | 6.98 (3) | 43.40 (23) |
χ2=15.90 |
| Positive | 70 | 93.02 (40) | 56.70 (30) | P<0.001 |
| Metastasis |
| No | 51 | 30.77 (12/39) | 76.47 (39/51) |
χ2=18.80 |
| Yes | 39 | 69.23 (27/39) | 23.53 (12/51) | P<0.001 |
| Estrogen receptor
status |
| Positive | 38 | 0.00 (0) | 71.70 (38) |
χ2=51.03 |
| Negative | 58 | 100 (43) | 28.30 (15) | P<0.001 |
| Progesterone
receptor status |
| Positive | 31 | 0.00 (0) | 58.50 (31) |
χ2=37.15 |
| Negative | 65 | 100 (43) | 41.50 (22) | P<0.001 |
| HER2 status |
| Positive | 18 | 0.00 (0) | 33.96 (18) |
χ2=17.97 |
| Negative | 78 | 100 (43) | 66.04 (35) | P<0.001 |
| Breast cancer
recurrence |
| No | 53 | 31.70 (13/41) | 81.60 (40/49)) |
χ2=22.98 |
| Yes | 37 | 68.30 (28/41) | 18.40 (9/49) | P<0.001 |
| Mortality |
| No | 50 | 36.60 (15/41) | 71.40 (35/49) |
χ2=9.20 |
| Yes | 40 | 63.40 (26/41) | 28.60 (14/49) | P=0.002 |
| Response to
treatment |
| Responsive
(CR+PR) | 14 | 14.81 (4/27) | 83.33 (10/12) |
χ2=19.80 |
| Nonresponsive
(SD+PD) | 25 | 85.19 (23/27) | 16.67 (2/12) | P<0.001 |
Marker expression in the sera of patients
and controls
The serum level mean ranks for VEGF-A were 90.49,
44.73 and 26.22; for IGF-I were 90.72, 46.80 and 20.23; for IGF-IR
were 90.20, 50.17 and 12.40; and for TGF-β1 were 68.01, 68.76 and
10.85, in the TNBC, non-TNBC and NC groups, respectively. The
differences between the serum levels of VEGF-A, IGF-I and IGF-IR in
the three groups were all statistically significant (P<0.001).
The level of serum TGF-β1 differed significantly between the
control group and the other two groups (P<0.001), but not
between TNBC and non-TNBC (P=0.282) (Fig. 1 and Table II).
 | Table IISerum levels of VEGF-A, IGF-I, IGF-IR
and TGF-β1. |
Table II
Serum levels of VEGF-A, IGF-I, IGF-IR
and TGF-β1.
| Group | VEGF-A
| IGF-I
| IGF-IR
| TGF-β1
|
|---|
| Mean rank | P-value | Mean rank | P-value | Mean rank | P-value | Mean rank | P-value |
|---|
| TNBC | 90.5ab | | 90.7ab | | 90.2ab | | 68.0a | |
| Non-TNBC | 44.7a | <0.001 | 46.8a | <0.001 | 50.2a | <0.001 | 68.8a | <0.001 |
| Control | 26.2 | | 20.2 | | 12.4 | | 10.9 | |
The cutoffs identified by ROC curve analysis that
were able to differentiate between the patients with TNBC and
non-TNBC were 106.96 ng/ml for serum IGF-I (93% sensitivity and
86.3% specificity), 10.09 ng/ml for serum IGF-IR (100% sensitivity
and 89% specificity) and 412.54 pg/ml for serum VEGF-A (97.7%
sensitivity and 94.2% specificity) (Fig. 2). The positivity rates of IGF-I
(93%), IGF-IR (100%) and VEGF-A (92.7%) were significantly higher
in the TNBC group compared with non-TNBC group(P<0.001; Fig. 3).
Correlation between the markers
Significant correlations were identified between the
serum levels of IGF-IR and IGF-I (r=0.645; P<0.001), VEGF-A
(r=0.594; P<0.001) and TGF-β1 (r=0.307; P=0.001). Serum VEGF-A
and IGF-I were also positively correlated (r=0.511; P<0.001)
(Table III).
 | Table IIIPearson bivariate analysis r-values
of serum levels of VEGF-A, IGF-I, IGF-IR and TGF-β1. |
Table III
Pearson bivariate analysis r-values
of serum levels of VEGF-A, IGF-I, IGF-IR and TGF-β1.
| VEGF-A | IGF-I | IGF-IR | TGF-β1 |
|---|
| VEGF-A | 1 | 0.511 | 0.594 | 0.116 |
| P-value | | <0.001 | <0.001 | 0.22 |
| IGF-I | 0.511 | 1 | 0.645 | 0.121 |
| P-value | <0.001 | | <0.001 | 0.20 |
| IGF-IR | 0.594 | 0.645 | 1 | 0.307 |
| P-value | <0.001 | <0.001 | | 0.001 |
| TGF-β1 | 0.116 | 0.121 |
0.307b | 1 |
| P-value | 0.22 | 0.22 | 0.001 | |
Correlations between serological markers
and clinicopathological features of the patients
A significant association was identified between
high serum levels of VEGF-A and large tumor size (P=0.02) whereas
high TGF-β1 was associated with positive HER2 expression (P=0.043).
High levels of serum VEGF-A, IGF-I and IGF-IR were significantly
associated with the presence of lymph nodes metastasis (P=0.007,
P=0.007 and P=0.03, respectively) and incidence of mortality
(P<0.001, P=0.009 and P=0.03, respectively). High serum levels
of VEGF-A, IGF-I, IGF-IR and TGF-β1 were significantly associated
with a high incidence of recurrence (P<0.001, P<0.001,
P<0.001 and P=0.02, respectively), the presence of distant
metastasis (P=0.026, P<0.001, P<0.001 and P=0.02,
respectively) in TNBC and non-TNBC patients and lower rate of
response (P=0.004, P<0.001, P=0.003 and P<0.001) in
metastatic BC patients (Table
IV). In the TNBC group, high serum levels of TGF-β1 were
associated with development of distant metastasis (P=0.009),
recurrence (P=0.003) and poor response to treatment (P= 0.01),
whereas, high serum levels of IGF-I were associated with metastasis
(P=0.026), recurrence (P=0.01), mortality rate (P=0.03) and
impaired response (P= 0.037; Table
V).
 | Table IVCorrelation of the mean rank of
markers to different clinicopathological parameters. |
Table IV
Correlation of the mean rank of
markers to different clinicopathological parameters.
| Characteristic | Total no. | VEGF-A mean
rank | Statistics | IGF-I mean
rank | Statistics | IGF-IR mean
rank | Statistics | TGF-β1 mean
rank | Statistics |
|---|
| Age at diagnosis
(years) |
| ≤35 | 8 | 42.81 |
χ2=0.66 | 49.81 |
χ2=0.19 | 45.69 |
χ2=0.12 | 51.00 |
χ2=0.23 |
| 36–49 | 34 | 51.06 | P=0.72 | 46.84 | P=0.91 | 48.10 | P=0.94 | 49.76 | P=0.89 |
| ≥50 | 54 | 47.73 | | 49.35 | | 49.17 | | 47.33 | |
| Tumor size
(cm) |
| <4 | 52 | 40.56 |
χ2=5.47 | 42.8 |
χ2=2.1 | 42.52 |
χ2=2.60 | 45.57 |
χ2=0.08 |
| ≥4 | 44 | 53.57 | P=0.02 | 50.90 | P=0.15 | 51.24 | P=0.12 | 47.61 | P=0.72 |
| Family history |
| No | 85 | 48.53 |
χ2=7.70 | 47.63 |
χ2=3.2 | 47.23 |
χ2=3.40 | 45.78 |
χ2=0.05 |
| Yes | 11 | 22.94 | P=0.006 | 31.11 | P=0.07 | 34.78 | P=0.18 | 48.00 | P=0.81 |
| Menopausal
status |
|
Pre-menopausal | 44 | 47.72 |
χ2=0.07 | 47.57 |
χ2=0.09 | 48.90 |
χ2=0.017 | 48.22 |
χ2=0.005 |
|
Post-menopausal | 52 | 49.16 | P=0.8 | 49.29 | P=0.76 | 48.16 | P=0.9 | 48.74 | P=0.93 |
| Tumor stage |
| Early (I–II) | 55 | 47.99 |
χ2=1.60 | 45.77 |
χ2=0.093 | 43.56 |
χ2=0.38 | 46.31 |
χ2=0.32 |
| Late (III–IV) | 41 | 40.80 | P=0.2 | 43.92 | P=0.74 | 47.03 | P=0.53 | 43.16 | P=0.57 |
| Tumor grade |
| 1 | 2 | 39.25 |
χ2=0.24 | 21.50 |
χ2=3.96 | 46.50 |
χ2=2.59 | 77.5 |
χ2=1.58 |
| 2 | 79 | 46.60 | P=0.61 | 46.28 | P=0.13 | 45.58 | P=0.27 | 47.80 | P=0.21 |
| 3 | 15 | 53.71 | | 58.04 | | 58.32 | | 41.54 | |
| Nodal status |
| Negative | 26 | 31.54 |
χ2=8.20 | 31.50 |
χ2=8.21 | 33.72 |
χ2=4.8 | 48.96 |
χ2=1.57 |
| Positive | 70 | 47.87 | P=0.007 | 47.88 | P=0.007 | 47.07 | P=0.03 | 41.51 | P=0.22 |
| Metastasis |
| No | 51 | 40.14 |
χ2=4.60 | 36.75 |
χ2=10.80 | 36.96 |
χ2=11.60 | 40.02 |
χ2=5.20 |
| Yes | 39 | 52.51 | P=0.026 | 56.94 | P<0.001 | 56.67 | P<0.001 | 52.67 | P=0.02 |
| Estrogen receptor
status |
| Positive | 38 | 26.89 |
χ2=27.40 | 26.26 |
χ2=28.20 | 27.53 |
χ2=24.80 | 47.44 |
χ2=0.32 |
| Negative | 58 | 57.35 | P<0.001 | 57.75 | P<0.001 | 56.94 | P<0.001 | 44.26 | P=0.57 |
| Progesterone
receptor status |
| Positive | 31 | 24.07 |
χ2=28.99 | 27.98 |
χ2=20.31 | 28.68 |
χ2=19.10 | 46.71 |
χ2=0.06 |
| Negative | 65 | 55.18 | P<0.001 | 53.41 | P<0.001 | 53.10 | P<0.001 | 44.95 | P=0.77 |
| HER2 status |
| Positive | 18 | 32.60 |
χ2=4.76 | 26.40 |
χ2=9.30 | 27.67 |
χ2=18.70 | 57.00 |
χ2=4.09 |
| Negative | 78 | 48.08 | P=0.03 | 49.32 | P=0.002 | 49.07 | P<0.001 | 43.20 | P=0.043 |
| Breast cancer
recurrence |
| No | 53 | 35.11 |
χ2=13.50 | 34.41 |
χ2=15.80 | 35.47 |
χ2=12.40 | 40.00 |
χ2=5.20 |
| Yes | 37 | 55.15 | P=<0.001 | 56.13 | P=<0.001 | 54.65 | P=<0.001 | 53.00 | P=0.02 |
| Mortality |
| No | 50 | 37.98 |
χ2=12.20 | 40.14 |
χ2=6.77 | 41.20 |
χ2=4.70 | 46.49 |
χ2=0.001 |
| Yes | 40 | 57.58 | P=<0.001 | 54.76 | P=0.009 | 53.38 | P=0.03 | 46.51 | P=0.99 |
| Response to
treatment |
| Responsive
(CR+PR) | 14 | 13.10 |
χ2=8.30 | 11.90 |
χ2=11.1 | 12.80 |
χ2=8.70 | 11.07 |
χ2=13.4 |
| Nonresponsive
(SD+PD) | 25 | 23.80 | P=0.004 | 24.70 | P=0.001 | 24.01 | P=0.003 | 25.00 | P<0.001 |
| Disease
progression |
| Progressive
(PD) | 24 | 23.60 |
χ2=8.30 | 24.60 |
χ2=11.1 | 24.00 |
χ2=8.70 | 24.90 |
χ2=13.4 |
| Non-progressive
(CR+PR+SD) | 15 | 13.30 | P=0.004 | 11.95 | P=0.001 | 12.90 | P=0.003 | 11.09 | P<0.001 |
 | Table VCorrelation of serum levels of
markers in triple-negative breast cancer patients (n=43). |
Table V
Correlation of serum levels of
markers in triple-negative breast cancer patients (n=43).
| Characteristic | Total no. | VEGF | Statistics | IGF-I | Statistics | IGF-IR | Statistics | TGF-β1 | Statistics |
|---|
| Age at diagnosis
(years) |
| ≤35 | 4 | 769.6±161.5 | F=2.05 | 429.8±223.6 | F=2.8 | 11.78±1.4 | F=0.24 | 11499.5±5341.8 | F=0.41 |
| 36–49 | 14 | 1172.2±534.3 | P=0.14 | 268.8±75.8 | P=0.07 | 12.3±2.5 | P=0.79 | 9910±3023.3 | P=0.67 |
| ≥50 | 25 | 932.7±377.4 | | 275.4±131 | | 11.88±1.4 | | 9947.4±3086.4 | |
| Tumor size
(cm) |
| <4 | 19 | 814.5±317 | F=6.7 | 284.2±153 | F=0.02 | 11.6±1.1 | F=1.7 | 9779.6±3012.4 | F=029 |
| ≥4 | 24 | 1138.8±467.4 | P=0.013 | 290.3±115.1 | P=0.88 | 12.3±2.2 | P=0.2 | 10317.1±3461.2 | P=0.6 |
| Menopausal
status |
|
Pre-menopausal | 19 | 10528±499.6 | F=0.7 | 303.4±130.3 | F=0.48 | 12.12±2.2 | F=0.14 | 10295.4±3437. | F=0.15 |
|
Post-menopausal | 24 | 945.5±379.9 | P=0.4 | 275.1±133.8 | P=0.49 | 11.9±1.4 | P=0.71 | 9908.8±3146.6 | P=0.7 |
| Tumor stage |
| Early (I-II) | 26 | 946.2±485 | F=0.8 | 283.2±136.4 | F=0.07 | 11.8±1.4 | F=0.81 | 9986.1±3108.5 | F=0.053 |
| Late (III) | 17 | 1070.9±345.3 | P=0.36 | 294.4±127.4 | P=0.79 | 12.3±2.3 | P=0.37 | 10222.6±3533.7 | P=0.82 |
| Tumor grade |
| 1 | 0 | | F=3.7 | | F=0.02 | | F=0.007 | | F=0.24 |
| 2 | 34 | 931.8±387.6 | P=0.06 | 286.1±123.3 | P=0.89 | 11.98±1.9 | P=0.9 | 9953±3018 | P=0.63 |
| 3 | 9 | 1236.1±539.8 | | 293.4±167.5 | | 12.04±1.3 | | 10558±4164.4 | |
| Nodal status |
| Negative | 3 | 760.7±335.8 | F=0.94 | 315.4±101.5 | F=0.14 | 12.8±2.1 | F=0.01 | 9789.4±2965.6 | F=0.22 |
| Positive | 40 | 1013.1±439.8 | P=0.34 | 285.5±134 | P=0.71 | 11.8±1.7 | P=0.8 | 10227.2±3487 | P=0.65 |
| Metastasis |
| No | 27 | 1045.2±416 | F=0.89 | 322.9±140.1 | F=5.2 | 11.94±1.9 | F=0.18 | 10892.8±3496.2 | F=7.5 |
| Yes | 12 | 900.2±500.1 | P=0.35 | 219.7±104.2 | P=0.026 | 11.7±1.2 | P=0.68 | 7951.4±1897.3 | P=0.009 |
| Breast cancer
recurrence |
| No | 13 | 880.7±501.4 | F=0.7 | 196.5±113.1 | F=7.48 | 12.4±2.8 | F=1.65 | 7478±1698.6 | F=10.5 |
| Yes | 28 | 1013.3±400.3 | P=0.41 | 321.4±121.3 | P=0.01 | 11.6±1.03 | P=0.21 | 11092.2±3319.4 | P=0.003 |
| Mortality |
| No | 15 | 879.1±434.8 | F=1.36 | 235.6±129.8 | F=5.1 | 12.6±2.5 | F=3.3 | 9063.1±2857.6 | F=3.42 |
| Yes | 26 | 1035.4±408.2 | P=0.25 | 328.5±123.1 | P=0.03 | 11.5±1.0 | P=0.08 | 10926.4±3316.3 | P=0.07 |
| Response to
treatment |
| Responsive
(CR+PR) | 4 | 923.7±477.3 | F=0.31 | 213.8±111.7 | F=4.69 | 11.6±1.0 | F=2.7 | 7887.7±1687.6 | F=7.34 |
| Nonresponsive
(SD+PD) | 23 | 1009.2±401.4 | P=0.58 | 308.9±122.8 | P=0.037 | 12.6±2.8 | P=0.11 | 10861.2±3308.7 | P=0.01 |
Response to treatment
During the period of follow-up, 90 (41 TNBC and 49
non-TNBC) out of the 96 cases were assessed for disease
progression. At the conclusion of the study (median follow up, 33
months; range, 2–85 months), 41/90 (45.6%) of the patients
exhibited disease progression (presence of metastasis and/or
recurrence). Local recurrence occurred in 28/41 (68.3%) of the TNBC
group compared with 9/49 (18.40%) of the non-TNBC group
(P<0.001) (Table I). BC
recurrence was associated with large tumor size 21/37 (56.80%;
P=0.046), lymph node metastasis 35/37 (94.60%; P<0.001) and
distant metastasis 34/37 (91.90%; P<0.001). The overall response
rate was 35.9% (14/39, 95% CI, 20.5–51.3%) and was significantly
lower in patients with TNBC (Table
I). Patients with PD exhibited higher serum levels of VEGF-A,
IGF-I, IGF-IR and TGF-β1, compared with those who responded or had
SD (P=0.004, P=0.001, P=0.003 and P<0.001, respectively;
Table IV).
Survival analysis
A total of 40/90 patients (44.4%) succumbed to the
disease over the course of the study and of these, 63.40% were
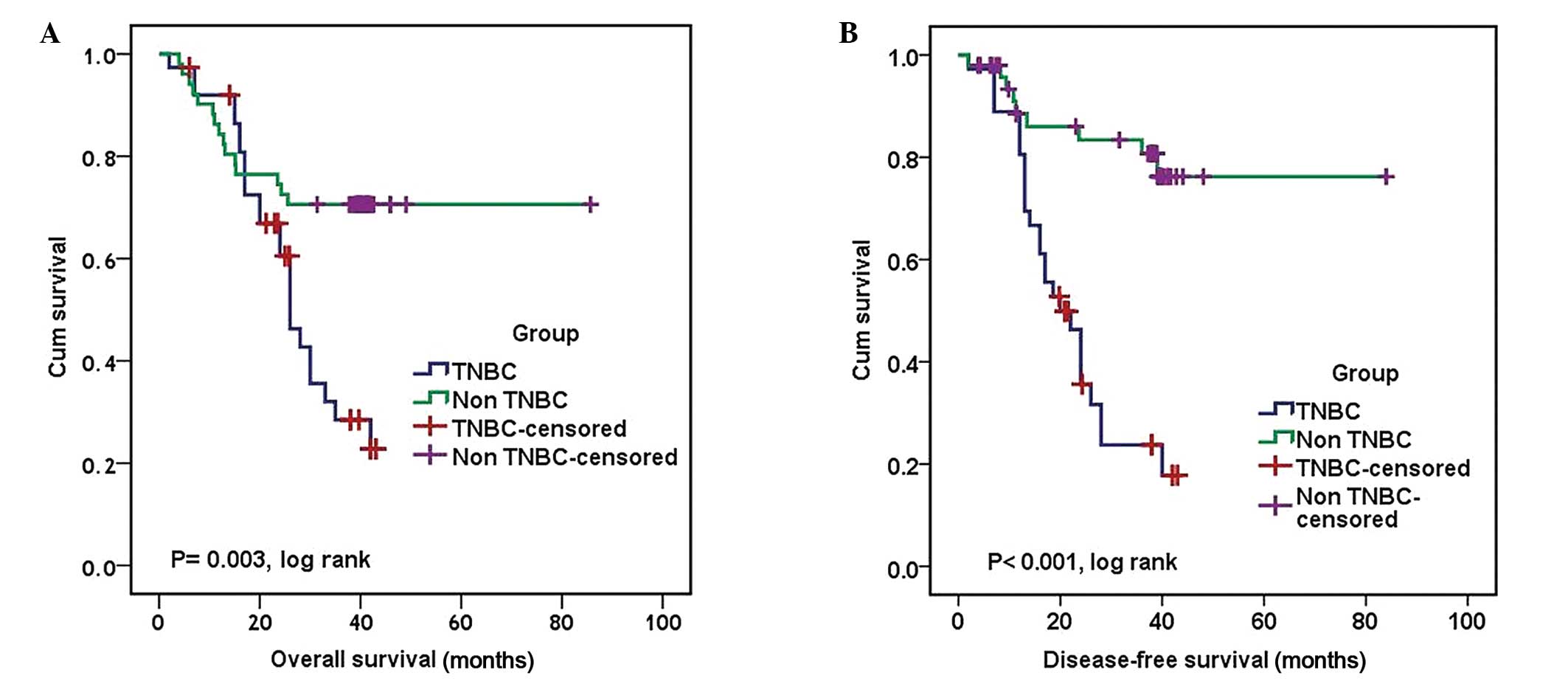
classified as TNBC and 28.60% were non-TNBC patients (Table II). TNBC patients showed poor
prognosis in terms of OS (P= 0.003, log rank) and DFS (P<0.001,
log rank) compared to non-NTNBC patients (Fig. 4). The median OS and DFS for
patients with TNBC were 26 and 20 months, respectively (95% CI,
22.09–29.91 and 14.52–24.75 months, respectively). The median OS
and DFS for patients with non-TNBC could not be defined as >50%
of patients were alive and free of recurrence at the conclusion of
the study (P=0.003 and P<0.001, log rank, respectively)
(Table VI and Fig. 4).
 | Table VIUnivariate and multivariate cox
regression for disease-free survival and overall survival
analysis. |
Table VI
Univariate and multivariate cox
regression for disease-free survival and overall survival
analysis.
| Factor | Overall survival
| Disease-free
survival
|
|---|
| HR | 95% CI | P-value | HR | 95% CI | P-value |
|---|
| Univariate
factor | | | | | | |
| Age group | 0.69 | 0.5–1.09 | 0.12 | 0.65 | 0.4–1.04 | 0.08 |
| Tumor size | 2.44 | 1.27–4.68 | 0.007 | 2.5 | 1.26–4.88 | 0.008 |
| Menopausal
status | 0.69 | 0.37–1.3 | 0.25 | 0.56 | 0.28–1.1 | 0.09 |
| Tumor grade | 1.83 | 0.83–4 | 0.13 | 2.8 | 1.28–6.11 | 0.01 |
| Tumor stage | 1.41 | 0.74–2.67 | 0.296 | 1.83 | 0.94–3.6 | 0.08 |
| LN status | 18.2 | 2.55–133 | 0.004 | 19.4 | 2.6–142.6 | 0.004 |
| Tumor group (TN vs
NTN) | 2.6 | 1.37–5 | 0.003 | 5.2 | 2.4–11.3 | <0.001 |
| TGF-β1 | 1.87 | 0.95–3.7 | 0.07 | 4.1 | 1.79–9.5 | <0.001 |
| VEGF-A | 2.73 | 1.39–5.36 | 0.003 | 4.8 | 2.16–10.75 | <0.001 |
| IGF-I | 1.86 | 0.97–3.59 | 0.03 | 3.2 | 1.49–6.85 | 0.003 |
| IGF-IR | 2.15 | 1.1–4.2 | 0.02 | 3.85 | 1.85–8.02 | <0.001 |
| Multivariate
factor | | | | | | |
| Tumor size | 1.95 | 0.9–3.79 | 0.06 | 2.4 | 1.13–4.9 | 0.02 |
| LN status | 14.68 | 1.8–104.6 | 0.01 | 14.7 | 1.8–118 | 0.01 |
| Group | 1.8 | 0.84–3.74 | 0.13 | 1.5 | 0.09–24.4 | 0.77 |
| TGF-β1 | | | | 4.3 | 1.7–10.96 | 0.002 |
| VEGF-A | 1.001 | 0.2–1.14 | 0.005 | 2.6 | 0.4–16.9 | 0.3 |
| IGF-I | 2.0 | 1.019–3.4 | 0.044 | 0.52 | 0.06–4.4 | 0.54 |
| IGF-IR | 1.98 | 1.03–3.84 | 0.048 | 1.3 | 0.3–5.5 | 0.73 |
Patients with positive expression of VEGF-A, IGF-I
and IGF-IR experienced poorer outcomes in terms of OS (P=0.001,
P=0.012 and P=0.012, log rank, respectively) and those with
positive expression of four studied markers were associated with
reduced DFS (Fig. 5 and Fig. 6) A univariate analysis demonstrated
that reduced OS was associated with TNBC (P=0.003), high serum
levels of VEGF-A (P=0.003), IGF-I (P=0.03), IGF-IR (P=0.02), large
tumor size (P=0.007), and lymph nodes metastasis (P=0.004) in the
overall population (Table VI).
Reduced DFS was significantly associated with high serum levels of
VEGF-A, TGF-β1, IGF-IR (P<0.001) and IGF-I (P=0.003), high tumor
grade (P=0.01; tumor grade indicates the aggression of the tumor),
lymph nodes metastasis (P=0.004) and large tumor size (P=0.008;
Table VI).
In the multivariate analysis, only positive lymph
nodes (P=0.01; HR, 14.68; 95% CI, 1.8–104.6) and high serum levels
of VEGF-A (P=0.005; HR, 1.001; 95% CI, 0.2–1.14), IGF-I (P=0.044;
HR, 2.0; 95% CI, 1.019–3.4) and IGF-IR (P=0.048; HR, 1.98; 95% CI,
1.03–3.84) were independent factors for OS. Tumor size, lymph nodes
invasion and TGF-B1 were independent predictors for DFS.
Discussion
The prognostic outcome of TNBC should be discussed
with regard to specific molecular subgroups. Previous studies have
focused on the prognostic and predictive values of circulating
tumor-associated markers due to the use of these biomarkers being
cheap, non-invasive and non-specific to disease stage (21).
In the current study, compared with patients with
non-TNBC, TNBC patients were observed to present with more
aggressive tumors with lymph nodes invasion, distant metastases,
low response to treatment and a high incidence of early recurrence
and cancer-associated mortality, which was consistent with previous
studies (22–24). The serum levels of VEGF-A, IGF-I
and IGF-IR, but not TGF-β1 were identified at specific cutoffs as
potential surrogate markers to differentiate between TNBC and
non-TNBC, and to better sub-classify TNBC into prognostic
subgroups, since their expression was significantly higher in the
TNBC group compared with non-TNBC groups and healthy controls
(P<0.001).
The current study demonstrated that high serum
levels of TGF-β1 were significantly associated with a high
incidence of metastasis, recurrence and a poor response to
treatment, which was consistent with previous studies (21,25).
It was demonstrated that although BC tumor tissue exhibits higher
levels of expression of TGF-β1 than the corresponding normal
tissues, the association of TGF-β1 with cancer is strongest in the
advanced stages of the disease. One possible explanation is that
TGF-β1 signaling has dual tumor suppressive and metastatic roles in
BC. In the early stages, TGF-β1 suppresses tumor development by
maintaining the balance between cell renewal and cell
differentiation or loss (26).
Loss of this homeostatic function occurs early in carcinogenesis
(27) then, two important
alterations occur during tumor development. The first alteration is
associated with a global loss of receptor signaling, resulting in a
reduction in the tumor suppressive activity of TGF-β1, whereas the
second is associated with overproduction of bioactive TGF-β1,
resulting in the activation of a pro-invasive, -angiogenic and
-metastatic TGF-β1-regulated gene expression program, thus inducing
a tumor cell phenotype that is mesenchymal and highly metastatic
(11). This hypothesis provides an
explanation for the association between TGF-β1 overexpression and
the increased incidence of metastasis and recurrence reported in
the current study. A reduction in the expression of the TGF-β1
receptor (TGF-β1-R) is associated with an increase in the levels of
TGF-β1 in the tumor microenviroment and abrogates the
tumor-suppressive effects of TGF-β1 and the invasive phenotype in
the majority of cases of BC.
It was reported that ER negative BC cells expressed
TGF-βR which were undetectable in ER positive cells. Moreover, It
has been shown that, the differentiated ER positive luminal cells
are unresponsive to TGF-β, since the TGF-βR-II gene is
transcriptionally silent in these cells (28). They respond to estrogen via down
regulation of TGF-β, whereas anti-estrogens act by upregulating
TGF-β1 signal transduction pathway (29). Thus, the inhibition of TGF-β1
signaling results in the differ entiation of mammary stem cells
into ductal cells. Accordingly, the TGF-β1 antagonists convert
basal-like or HER2-positive tumor cells into epithelioid,
non-proliferating and non-metastatic cells, which makes them
candidates for targeted therapy in the TNBC cases (11).
TGF-β1 signaling also induces macro-metastases,
particularly in the bones and lungs (20–32).
In a mouse model of TNBC or basal-like BC, treatment with TGF-β1
neutralizing antibodies or receptor kinase inhibitors strongly
inhibited the development of distant metastases. This was via the
derepression of antitumor immunity, inhibition of angiogenesis or
the reversal of the mesenchymal, invasive phenotype characteristic
of HER2-positive and basal-like BC (11). Additionally, TGF-β1 may
autonomously promote metastasis, as the expression of a
dominant-negative mutant of TGF-βR-II in the TNBC cell line
MDA-MB-231 was reported to inhibit experimental bone metastases,
whereas the overexpression of constitutively active TGF-βR-I
increased the production of parathyroid hormone-related protein by
the tumor cells and enhanced bone metastases (33). In addition, VEGF-A, -B and -C were
observed to be upregulated in bone and bone marrow metastases
compared with those in the brain or lung (34).
The prognostic impact of TGF-β1 in BC remains
controversial. Certain studies have demonstrated that TGF-β1
overexpression is significantly higher in patients with favorable
outcomes compared with those with a poor prognosis (35), whereas others indicated the reverse
(36,37). The results of the current study
regarding the poor prognostic impact of TGF-β1 overexpression are
consistent with those of Ivanović et al (21) and Dave et al (38), who observed increased levels of
plasma TGF-β1 in locally advanced BC (stages III and IV). In
addition to the observation by Dave et al (38) who reported a correlation between
low serum TGF-β1 levels and pathological CR and prolonged DFS
In the present study, VEGF-A was observed to be
significantly overexpressed in TNBC compared with non-TNBC. It was
also associated with aggressive tumors, lymph nodes invasion, a
high incidence of metastasis, poor response to treatment and
reduced survival. These observations are comparable to those of
previous studies on metastatic (39) and non-metastatic (40,41)
TNBC in which VEGF-A was demonstrated to be important in the
progression of TNBC. As a key mediator of angiogenesis, VEGF-A
stimulates the proliferation and migration of epithelial cells,
inhibits apoptosis of endothelial tissues and increases vascular
permeability and vasodilation (42). In accordance with this, the current
study reported low VEGF-A levels in tumors that were responsive (CR
and PR) compared with those that were nonresponsive (SD and PD)
(P=0.004) to chemotherapy, and this was also associated with
prolonged survival. Similar results were reported previously by
Björndahl et al (43), who
suggested that IGF-IR is able to induce metastasis via the
regulation of tumor cell survival and proliferation in secondary
sites, in addition to the promotion of angiogenesis and
lymphangiogenesis either through direct action on the endothelial
cells or by transcriptional regulation of VEGF-A and -C.
IGF-IR, a member of a transmembrane receptor
tyrosine kinase family, is expressed on the cell surface of cells
in the majority of tissues. Together with its ligand (IGF-I), it is
important in the regulation of cell cycle progression, cell
survival and apoptosis (16,17,44–47).
Although several multi-center studies have demonstrated that serum
IGF-I predicts the outcome of patients with BC (48–50)
and others (51,52) observed the correlation between high
IGF-I mRNA levels and longer OS and DFS in cases of BC, this was
not assessed in TNBC. Thus, to the best of our knowledge, this is
the first study to investigate these factors in TNBC.
High levels of IGF-IR were detected in 100% of the
TNBC cases. Previous studies reported IGF-IR expression in 29–36%
of TNBC (53) and in certain
studies IGF-IR overexpression in TNBC was attributed to either
mutations in tumor suppressor genes, including p53 and BRCA1, which
repress the IGF-IR promoter (54),
or to the amplification of IGF-IR in basal or HER-2 positive BC.
However, these were not assessed in the current study. A
significant correlation between IGF-I/IGFR-IR and VEGF-A expression
was demonstrated in the current study, and the contribution of
these markers to an aggressive BC phenotype was confirmed. Serum
IGF-IR levels were demonstrated to be significantly lower in
patients who experienced complete and partial responses compared
with those with PD and SD (P=0.003). In addition, high serum
IGF-I/IGF-IR levels were significantly associated with reduced OS,
independent of other clinicopathological features. Concerning this
observation, Haffner et al (51) demonstrated that the IGF-I mRNA
level was an independent predictor of OS and DFS in 89
lymph-node-negative cases of BC. Additionally, Shin et al
(52) measured IGF-I and IGF-IR
mRNA levels in 508 breast tumors and adjacent tissues, and observed
that patients in the highest tertile of tumor IGF-I mRNA levels
exhibited a longer DFS and OS compared with those in the lower
tertile.
One hypothesis is that although a number of studies
regarding cancer cell lines have indicated that IGF-I stimulation
leads to aggressive, fast growing, metastasizing tumors, other
studies demonstrate that IGF-I is also able to increase cell
differentiation in certain cancer cell lines that are associated
with less aggressive types of cancer and hence improved prognosis.
One explanation is that IGF-I expression may be a by-product of
another cellular process that results in a less aggressive
phenotype. An additional possibility is that unlike serum IGF-I
levels, the expression of IGF-I in breast tissue may not be in
large enough quantities to result in an increase in stimulation of
the IGF-IR signaling pathway (16,55).
However, until verified by larger studies, IGF-I expression in
tissue and the serum should be used as an intermediate prognostic
marker, rather than a potential therapeutic target.
By contrast, Munagala et al (56) demonstrated that IGF-I and TGF-β1
act synergistically to induce epithelial-mesenchymal transition in
BC cells, leading to metastasis. It was identified that IGF-I
transmits signals via the phosphoinositide 3-kinase and
mitogen-activated protein kinase pathways resulting in the
transcription of unknown but specific genes. The products of these
genes result in the extracellular activation of IGF-IR, which
stimulate cancer cell proliferation and survival, and confer
resistance to cytotoxic, hormonal and targeted therapies in BC. In
accordance with this, Munagala et al concluded that IGF-I
and TGF-β1 are promising molecular targeted therapies in BC.
Shimizu et al (57) also observed IGF-IR overexpression
in BC biopsies while Pizon et al (58) and Munagala et al (56) demonstrated that IGF-IR may serve an
important role in determining how aggressive circulating tumor
cells (CTCs) are and their ability to grow subsequent to adhesion
to form metastatic deposits. Patients with high CTC counts were
demonstrated to commonly exhibit high IGF-IR expression on the
tumor cells and in the CTCs. In addition, a significant linear
correlation was reported between IGF-IR expression and the presence
of VEGFR-2 on the isolated CTCs.
Through the multivariate analysis, serum IGF-I,
IGF-IR and VEGF-A levels were identified as independent predictors
for OS together with lymph nodes invasion and TNBC, whereas only
TGF-β1, large tumor size and lymph nodes metastasis were
independent predictors for DFS. Similar results were obtained by
Grau et al (59), who
observed that patients with the highest circulating levels of
TGF-β1 often have reduced survival independent of the disease
stage, whereas additional studies demonstrated that TGF-β1 levels
were significantly higher in patients with a favorable outcome
(36,37). These discrepancies may be due to
the small sample sizes and the relatively short follow-up periods
in the majority of studies.
In conclusion, to the best of our knowledge, the
current study was the first to simultaneously assess serum IGF-I,
IGF-IR, VEGF-A and TGF-β1 levels and their interrelations in two
well defined groups of patients with BC (TNBC and non-TNBC). The
results of the present study confirm that high expression of these
proteins is more common in patients with TNBC and is usually
associated with an aggressive tumor phenotype with higher incidence
of recurrence, poor response to treatment and reduced survival.
Therefore, TGF-β1, IGF-I/IGF-IR and VEGF-A may be promising
surrogate prognostic markers and possibly candidates for targeted
therapy, particularly in patients with TNBC. However, large
clinical trials are required in order to verify the results of the
current study.
References
|
1
|
Siegel R, Ward E, Brawley O and Jemal A:
Cancer statistics, 2011: the impact of eliminating socioeconomic
and racial disparities on premature cancer deaths. CA Cancer J
Clin. 61:212–236. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Litzenburger BC, Creighton CJ, Tsimelzon
A, et al: High IGF-IR activity in triple-negative breast cancer
cell lines and tumorgrafts correlates with sensitivity to
anti-IGF-IR therapy. Clin Cancer Res. 17:2314–2327. 2011.
View Article : Google Scholar :
|
|
3
|
Rakha EA, Reis-Filho JS and Ellis IO:
Basal-like breast cancer: a critical review. J Clin Oncol.
26:2568–2581. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Dent R, Trudeau M, Pritchard KI, Hanna WM,
Kahn HK, Sawka CA, Lickley LA, Rawlinson E, Sun P and Narod SA:
Triple-negative breast cancer: clinical features and patterns of
recurrence. Clin Cancer Res. 13:4429–4434. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Liedtke C, Mazouni C, Hess KR, Andre F,
Tordai A, Mejia JA, Symmans WF, Gonzalez-Angulo AM, Hennessy B,
Green M, et al: Response to neoadjuvant therapy and long-term
survival in patients with triple-negative breast cancer. J Clin
Oncol. 26:1275–1281. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brouckaert O, Wildiers H, Floris G and
Neven P: Update on triple-negative breast cancer: prognosis and
management strategies. Int J Womens Health. 4:511–520.
2012.PubMed/NCBI
|
|
7
|
Cao H, Yan M, Wang S, Zheng T, Xu R, Chen
Y and Liu Y: Clinical characteristics and prognosis of
triple-negative breast cancer. Chin-Ger J Clin Oncol. 8:471–475.
2009. View Article : Google Scholar
|
|
8
|
Oakman C, Moretti E, Galardi F, Biagioni
C, Santarpia L, Biganzoli L and Di Leo A: Adjuvant systemic
treatment for individual patients with triple negative breast
cancer. Breast. 20(Suppl 3): S135–S141. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Kashiwagi S, Yashiro M, Takashima T,
Nomura S, Noda S, Kawajiri H, Ishikawa T, Wakasa K and Hirakawa K:
Significance of E-cadherin expression in triple-negative breast
cancer. Br J Cancer. 103:249–255. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Dumont N, Bakin AV and Arteaga CL:
Autocrine transforming growth factor-beta signaling mediates
Smad-independent motility in human cancer cells. J Biol Chem.
278:3275–3285. 2003. View Article : Google Scholar
|
|
11
|
Tan AR, Alexe G and Reiss M: Transforming
growth factor-beta signaling: emerging stem cell target in
metastatic breast cancer? Breast Cancer Res Treat. 115:453–495.
2009. View Article : Google Scholar :
|
|
12
|
Adams J, Carder PJ, Downey S, Forbes MA,
MacLennan K, Allgar V, Kaufman S, Hallam S, Bicknell R, Walker JJ,
et al: Vascular endothelial growth factor (VEGF) in breast cancer:
comparison of plasma, serum, and tissue VEGF and microvessel
density and effects of tamoxifen. Cancer Res. 60:2898–2905.
2000.PubMed/NCBI
|
|
13
|
George ML, Eccles SA, Tutton MG, Abulafi
AM and Swift RI: Correlation of plasma and serum vascular
endothelial growth factor levels with platelet count in colorectal
cancer: clinical evidence of platelet scavenging? Clin Cancer Res.
6:3147–3152. 2000.PubMed/NCBI
|
|
14
|
Pollak MN, Schernhammer ES and Hankinson
SE: Insulin-like growth factors and neoplasia. Nat Rev Cancer.
4:505–518. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hankinson SE: Circulating levels of sex
steroids and prolactin in premenopausal women and risk of breast
cancer. Adv Exp Med Biol. 617:161–169. 2008.PubMed/NCBI
|
|
16
|
Reiss K, Wang JY, Romano G, Furnari FB,
Cavenee WK, Morrione A, Tu X and Baserga R: IGF-I receptor
signaling in a prostatic cancer cell line with a PTEN mutation.
Oncogene. 19:2687–2694. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Valentinis B, Navarro M, Zanocco-Marani T,
Edmonds P, McCormick J, Morrione A, Sacchi A, Romano G, Reiss K and
Baserga R: Insulin receptor substrate-1, p70S6K, and cell size in
transformation and differentiation of hemopoietic cells. J Biol
Chem. 275:25451–25459. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Guerra FK, Eijan AM, Puricelli L, Alonso
DF, Bal de Kier Joffé E, Kornblihgtt AR, Charreau EH and Elizalde
PV: Varying patterns of expression of insulin-like growth factors I
and II and their receptors in murine mammary adenocarcinomas of
different metastasizing ability. Int J Cancer. 65:812–820. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the Eastern Cooperative Oncology Group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Therasse P, Arbuck SG, Eisenhauer EA, et
al: New guidelines to evaluate the response to treatment in solid
tumors European Organization for Research and Treatment of Cancer,
National Cancer Institute of the United States, National Cancer
Institute of Canada. J Natl Cancer Inst. 92:205–216. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Iva nović V, To dorović-Ra ković N, D
emajo M, Nesković-Konstantinović Z, Subota V,
Ivanisević-Milovanović O and Nikolić-Vukosavljević D: Elevated
plasma levels of transforming growth factor-beta 1 (TGF-beta 1) in
patients with advanced breast cancer: association with disease
progression. Eur J Cancer. 39:454–461. 2003. View Article : Google Scholar
|
|
22
|
Jones C, Nonni AV, Fulford L, Merrett S,
Chaggar R, Eusebi V and Lakhani SR: CGH analysis of ductal
carcinoma of the breast with basaloid/myoepithelial cell
differentiation. Br J Cancer. 85:422–427. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Sorlie T, Tibshirani R, Parker J, Hastie
T, Marron JS, Nobel A, Deng S, Johnsen H, Pesich R, Geisler S, et
al: Repeated observation of breast tumor subtypes in independent
gene expression data sets. Proc Natl Acad Sci USA. 100:8418–8423.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wang X, Chao L, Ma G, Chen L, Tian B, Zang
Y and Sun J: Increased expression of osteopontin in patients with
triple-negative breast cancer. Eur J Clin Invest. 38:438–446. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Sheen-Chen SM, Chen HS, Sheen CW, Eng HL
and Chen WJ: Serum levels of transforming growth factor beta1 in
patients with breast cancer. Arch Surg. 136:937–940. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Massagué J, Cheifetz S, Laiho M, Ralph DA,
Weis FM and Zentella A: Transforming growth factor-beta. Cancer
Surv. 12:81–103. 1992.PubMed/NCBI
|
|
27
|
Reiss M: TGF-beta and cancer. Microbes
Infect. 1:1327–1347. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shipitsin M, Campbell LL, Argani P,
Weremowicz S, Bloushtain-Qimron N, Yao J, Nikolskaya T,
Serebryiskaya T, Beroukhim R, Hu M, et al: Molecular definition of
breast tumor heterogeneity. Cancer Cell. 11:259–273. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Buck M, von der Fecht J and Knabbe C:
Antiestrogenic regulation of transforming growth factor beta
receptors I and II in human breast cancer cells. Ann NY Acad Sci.
963:140–143. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kang Y, He W, Tulley S, Gupta GP,
Serganova I, Chen CR, Manova-Todorova K, Blasberg R, Gerald WL and
Massagué J: Breast cancer bone metastasis mediated by the Smad
tumor suppressor pathway. Proc Natl Acad Sci USA. 102:13909–13914.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Yin JJ, Selander K, Chirgwin JM, Dallas M,
Grubbs BG, Wieser R, Massagué J, Mundy GR and Guise TA: TGF-beta
signaling blockade inhibits PTHrP secretion by breast cancer cells
and bone metastases development. J Clin Invest. 103:197–206. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Tang B, Vu M, Booker T, Santner SJ, Miller
FR, Anver MR and Wakefield LM: TGF-beta switches from tumor
suppressor to prometastatic factor in a model of breast cancer
progression. J Clin Invest. 112:1116–1124. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Kingsley LA, Fournier PG, Chirgwin JM and
Guise TA: Molecular biology of bone metastasis. Mol Cancer Ther.
6:2609–2617. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kareddula A, Zachariah E, Notterman D and
Reiss M: Transforming growth factor-b signaling strength determines
target gene expression profile in human keratinocytes. J Epithel
Biol Pharmaco. 1:40–94. 2008. View Article : Google Scholar
|
|
35
|
Marrogi AJ, Munshi A, Merogi AJ, Ohadike
Y, El-Habashi A, Marrogi OL and Freeman SM: Study of tumor
infiltrating lymphocytes and transforming growth factor-beta as
prognostic factors in breast carcinoma. Int J Cancer. 74:492–501.
1997. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Gorsch SM, Memoli VA, Stukel TA, Gold LI
and Arrick BA: Immunohistochemical staining for transforming growth
factor beta 1 associates with disease progression in human breast
cancer. Cancer Res. 52:6949–6952. 1992.PubMed/NCBI
|
|
37
|
Desruisseau S, Palmari J, Giusti C, Romain
S, Martin PM and Berthois Y: Determination of TGFbeta1 protein
level in human primary breast cancers and its relationship with
survival. Br J Cancer. 94:239–246. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dave H, Shah M, Trivedi S and Shukla S:
Prognostic utility of circulating transforming growth factor beta 1
in breast cancer patients. Int J Biol Markers. 27:53–59. 2012.
View Article : Google Scholar
|
|
39
|
Taha FM, Zeeneldin AA, Helal AM, Gaber AA,
Sallam YA, Ramadan H and Moneer MM: Prognostic value of serum
vascular endothelial growth factor in Egyptian females with
metastatic triple negative breast cancer. Clin Biochem.
42:1420–1426. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Berns EM, Klijn JG, Look MP, et al:
Combined vascular endo-thelial growth factor and TP53 status
predicts poor response to tamoxifen therapy in estrogen
receptor-positive advanced breast cancer. Clin Cancer Res.
9:1253–1258. 2003.PubMed/NCBI
|
|
41
|
Foekens JA, Peters HA, Grebenchtchikov N,
Look MP, Meijer-van Gelder ME, Geurts-Moespot A, van der Kwast TH,
Sweep CG and Klijn JG: High tumor levels of vascular endo-thelial
growth factor predict poor response to systemic therapy in advanced
breast cancer. Cancer Res. 61:5407–5414. 2001.PubMed/NCBI
|
|
42
|
Greenberg S and Rugo HS: Triple-negative
breast cancer: role of antiangiogenic agents. Cancer J. 16:33–38.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Björndahl M, Cao R, Nissen LJ, et al:
Insulin-like growth factors 1 and 2 induce lymphangiogenesis in
vivo. Proc Natl Acad Sci USA. 102:15593–15598. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Moschos SJ and Mantzoros CS: The role of
the IGF system in cancer: from basic to clinical studies and
clinical applications. Oncology. 63:317–332. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Rubin R and Baserga R: Insulin-like growth
factor-I receptor. Its role in cell proliferation, apoptosis, and
tumorigenicity. Lab Invest. 73:311–331. 1995.PubMed/NCBI
|
|
46
|
Dunn SE, Kari FW, French J, Leininger JR,
Travlos G, Wilson R and Barrett JC: Dietary restriction reduces
insulin-like growth factor I levels, which modulates apoptosis,
cell proliferation, and tumor progression in p53-deficient mice.
Cancer Res. 57:4667–4672. 1997.PubMed/NCBI
|
|
47
|
Gooch JL, Van Den Berg CL and Yee D:
Insulin-like growth factor (IGF)-I rescues breast cancer cells from
chemotherapy-induced cell death - proliferative and anti-apoptotic
effects. Breast Cancer Res Treat. 56:1–10. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Decensi A, Veronesi U, Miceli R, Johansson
H, Mariani L, Camerini T, Di Mauro MG, Cavadini E, De Palo G, Costa
A, et al: Relationships between plasma insulin-like growth factor-I
and insulin-like growth factor binding protein-3 and second breast
cancer risk in a prevention trial of fenretinide. Clin Cancer Res.
9:4722–4729. 2003.PubMed/NCBI
|
|
49
|
Gail MH, Costantino JP, Bryant J, Croyle
R, Freedman L, Helzlsouer K and Vogel V: Weighing the risks and
benefits of tamoxifen treatment for preventing breast cancer. J
Natl Cancer Inst. 91:1829–1846. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Yarden RI, Wilson MA and Chrysogelos SA:
Estrogen suppression of EGFR expression in breast cancer cells: a
possible mechanism to modulate growth. J Cell Biochem. 81(Suppl
36): 232–246. 2001. View Article : Google Scholar
|
|
51
|
Haffner MC, Petridou B, Peyrat JP,
Révillion F, Müller-Holzner E, Daxenbichler G, Marth C and Doppler
W: Favorable prognostic value of SOCS2 and IGF-I in breast cancer.
BMC Cancer. 7:1362007. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Shin A, Ren Z, Shu XO, Cai Q, Gao YT and
Zheng W: Expression patterns of insulin-like growth factor 1
(IGF-I) and its receptor in mammary tissues and their associations
with breast cancer survival. Breast Cancer Res Treat. 105:55–61.
2007. View Article : Google Scholar
|
|
53
|
Minami CA, Chung DU and Chang HR:
Management options in triple-negative breast cancer. Breast Cancer
(Auckl). 5:175–199. 2011.
|
|
54
|
Sarfstein R, Maor S, Reizner N,
Abramovitch S and Werner H: Transcriptional regulation of the
insulin-like growth factor-I receptor gene in breast cancer. Mol
Cell Endocrinol. 252:241–246. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Valentinis B, Romano G, Peruzzi F,
Morrione A, Prisco M, Soddu S, Cristofanelli B, Sacchi A and
Baserga R: Growth and differentiation signals by the insulin-like
growth factor 1 receptor in hemopoietic cells are mediated through
different pathways. J Biol Chem. 274:12423–12430. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Munagala R, Aqil F and Gupta RC: Promising
molecular targeted therapies in breast cancer. Indian J Pharmacol.
43:236–245. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Shimizu C, Hasegawa T, Tani Y, Takahashi
F, Takeuchi M, Watanabe T, Ando M, Katsumata N and Fujiwara Y:
Expression of insulin-like growth factor 1 receptor in primary
breast cancer: immunohistochemical analysis. Hum Pathol.
35:1537–1542. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Pizon M, Zimon DS, Pachmann U and Pachmann
K: Insulin-like growth factor receptor I (IGF-IR) and vascular
endothelial growth factor receptor 2 (VEGFR-2) are expressed on the
circulating epithelial tumor cells of breast cancer patients. PLoS
One. 8:e568362013. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
Grau AM, Wen W, Ramroopsingh DS, Gao YT,
Zi J, Cai Q, Shu XO and Zheng W: Circulating transforming growth
factor-beta-1 and breast cancer prognosis: results from the
Shanghai Breast Cancer Study. Breast Cancer Res Treat. 112:335–341.
2008. View Article : Google Scholar
|