Introduction
Cell division cycle-associated protein 4 (CDCA4),
also known as HEPP/SEI-3/TRIP-Br3, was originally identified by
Abdullah et al in 2001, as a novel gene involved in the
intrinsic regulation of hematopoietic stem cell and progenitor cell
lineage commitment and differentiation (1). Gene-based analysis suggested that
genes of the chenodeoxycholic acid (CDCA) family were closely
co-expressed with known cell cycle genes, including CDC2, CDC7 and
cyclins (2). CDCA4 was categorized
as SEI3 of the selected with INK4A (SEI) gene family, based on its
characteristic SEI-1, RBT1 and TARA domain and as a transcriptional
regulator interacting with the PHD bromodomain (TRIP-Br)3 of the
TRIP-Br gene family, according to its unique PHD-bromo-binding
domain (3,4). The SEI gene family consists of four
genes: SEI-1, SEI-2, RBT1 and CDCA4. The close association between
the overexpression of SEI family genes and tumorigenesis has been
confirmed in various studies (5,6).
Previous studies have indicated that SEI-1 and SEI-2 interact with
dimerization partner 1 (DP1) and regulate the transcriptional
activity of E2F-1 (7). SEI genes
also interact with bromodomain-containing transcriptional
cofactors, including lysine acetyltransferase 2B (PCAF), SPT7-like
STAGA complex γ subunit and kinesin-II-associated protein 1
(3). The overexpression of SEI
family genes, including CDCA4, can enhance the transactivation
function of p53, leading to p53-independent growth inhibition in
HeLa and U2OS cells (4). CDCA4 and
SEI-1 can inhibit breast cancer cell apoptosis by interacting with
an X-linked inhibitor of apoptosis protein, a potent apoptosis
inhibitor (8,9). Human CDCA4 mRNA is expressed at
higher levels in actively proliferating tissues, for example the
pancreas, thymus, testis, spleen, liver and placenta. Of note, the
mRNA expression of CDCA4 is only marginal or is undetected in
tissues consisting of permanent cells, including the brain,
skeletal muscle and heart (10).
CDCA4 affects the mRNA expression of the Jun proto-oncogene and
determines cell fate (11). In
addition, CDCA4 has a dual role in cell cycle regulation by
modulating the transcriptional activity of activator E2F
transcription factors and p53 (10). Evidence suggests that the SEI-1
protein can inhibit the MCF-7 cell senescence induced by
doxorubicin (12). SEI-2 is
frequently overexpressed in several types of human tumor.
Clinicopathological evidence and Kaplan-Meier survival analysis
indicates that the overexpression of SEI-2 is directly linked to
poor clinical outcomes in hepatocellular carcinoma (5). MicroRNA-15a downregulates the
expression of CDCA4 through targeting the 3′-untranslated region of
CDCA4, and the downregulation of CDCA4 can inhibit proliferation,
causing cell cycle arrest and reducing the invasiveness of melanoma
cells (13). In addition, The
Cancer Genome Atlas (TCGA) public databases show that CDCA4 has
been found in numerous samples of breast cancer tissue; its mRNA
expression being significantly higher compared with its expression
in adjacent tissues. In addition, GeneChip analysis has suggested
that CDCA4 is a downstream gene of the nuclear factor erthyroid
2-related factor 2 (Nrf2) signaling pathway. Nrf2 has been shown to
regulate the resistance of cancer cells to chemotherapeutic drugs
(14). Based on the aforementioned
information, the present study hypothesized that CDCA4 may be
closely associated with breast cancer cells. The present study
focused on the effect of RNA interference targeting CDCA4 on cell
proliferation, cycle progression and apoptosis in a human breast
cancer cell line. The results suggested that CDCA4 RNA interference
reduced the proliferation of human breast cancer cells to <50%,
and that CDCA4 regulated cell proliferation, at least in part,
through cell cycle progression. In addition, RNA interference of
CDCA4 resulted in a significant increase in the apoptotic rate of
cells.
Materials and methods
GeneChip analysis
Total RNA was extracted from three different batches
of Nrf2 shRNA-transfected MCF-7/ADR cells and are referred to as
knockdown (KD) group samples and three different batches of control
shRNA-transfected MCF-7/ADR cells that are referred to as
nonsilencing control (NC) group samples using TRIzol reagent
(Shanghai Pufei Biotechnology, Co., Ltd., Shanghai, China), and
then quantified using a Thermo Nanodrop 2000 spectrophotometer
(Thermo Fisher Scientific, Inc., Waltham, MA, USA) and Agilent 2100
Bioanalyzer (Agilent Technologies, Inc., Santa Clara, CA, USA). The
samples for microarray hybridization were prepared based on the
manufacturer's protocols. The cDNA was hybridized to the arrays at
2 × g for 18 h at 45°C. The chips were processed in the GeneChip
Fluidics Station 450 (Affymetrix, Inc., Santa Clara, CA, USA).
Microarray images were captured using the Scanner 3000 (Affymetrix,
Inc.), and data were extracted using Affymetrix Power Tools
software v1.8 (Affymetrix; Thermo Fisher Scientific, Inc., Waltham,
MA, USA).
Cell culture
The MCF-7/AD-M human breast cancer cell line was
obtained from the Cell Bank of the Chinese Academy of Sciences
(Shanghai, China). The MCF-7/ADM cells were cultured in
high-glucose Dulbecco's modified Eagle's medium (DMEM; (GE
Healthcare Life Sciences, Logan, UT, USA), supplemented with 10%
fetal bovine serum (FBS, HyClone; GE Healthcare Life Sciences),
penicillin (100 U/ml) and streptomycin (10 µg/ml). They were
incubated at 37°C and 5% CO2 in a humidified chamber
atmosphere.
RNA interference
Different regions of CDCA4 mRNA and nonsilencing
control were designed, chemically synthesized, and ligated into the
GV115 vector according to the manufacturer's protocol (GeneChem
Co., Ltd., Shanghai, China). The target sequence of human CDCA4
with the significant interference effect was
5′-TAGACCTAAGAGTAAATTA-3′. The nonsilencing short hairpin (sh)RNA
was designed as a nonspecific sequence, with no matching to the
human genome, using the Basic Local Alignment Search Tool
(www.ncbi.nlm.nih.gov). For each
interference experiment, equal numbers of MCF-7/ADM cells were
transfected with shRNAs of CDCA4 and control shRNA by
Lentivirus-mediated method. The antibiotic-resistant transfected
cells were selected by applying puromycin (Clontech Laboratories,
Inc., Mountainview, CA, USA) in DMEM, and the stably transfected
MCF-7/ADM cells were checked under phase contrast and fluorescence
microscopes. In a similar manner, MCF-7/ADM cells were transfected
with shRNAs of Nrf2 and control shRNA. The target sequence of Nrf2
was 5′-GGCATTTCACTAAACACAA-3′.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR) analysis
Total RNA was isolated from the cultured cells using
TRIzol reagent (Shanghai Pufei Biotechnology, Co., Ltd., Shanghai,
China). cDNA was synthesized from the DNaseI-treated total RNA (1
µg) using oligo-(dT) primer and reverse transcriptase of Moloney
murine leukemia virus (Promega Corporation, Madison, WI, USA) in a
10-µl reaction mixture. The RT-qPCR analysis was performed using
SYBR Premix Ex Taq (Takara Bio, Inc., Otsu, Japan). In brief, 1 µl
cDNA was diluted with 10 µl SYBR premix ex taq, 0.5 µl sense
primers, 0.5 µl antisense primers and 8 µl RNase-Free
H2O to 20 µl of reaction mixture, and the mixture was
subjected to PCR in an MX3000pq-PCR system (Agilent Technologies,
Inc., Santa Clara, CA, USA). The following primer sets were used:
Forward 5′-ATTTGAAACGCTGGAGACT-3′ and reverse
5′-CCCATCATGCCTGTCAGTA-3′ for human CDCA4 and Forward
5′-TGACTTCAACAGCGACACCCA-3′ and reverse 5′- CAC CCT GTT GCT GTA GCC
AAA-3′ for GAPDH. RT-qPCR was performed as followed: One cycle of
pre denaturation (95°C for 30 sec), 45 cycles of amplification
(95°C for 5 sec and 60°C for 30 sec), and a cooling period. The
expression of GAPDH was used as the internal control for detecting
mRNAs and results were analyzed by the 2−ΔΔCq method
(15). In a similar manner,
silencing effect of MCF-7/ADM cells was examined.
Western blot analysis
The cells were washed in an ice-cold
phosphate-buffered saline buffer and then lysed in
radioimmunoprecipitation assay lysis buffer. Following protein
quantification using a bicinchoninic acid assay, equal quantities
of 20 µg protein sample were separated using 10% sodium dodecyl
sulfate-polyacrylamide gel electrophoresis and transferred onto a
polyvinylidene difluoride membrane. The membranes were blocked with
5% nonfat dry milk and then incubated with primary antibody
(anti-CDCA4, cat. no. QC17060, 1:500 dilution, Sigma-Aldrich; Merck
MGaA, Darmstadt, Germany) for 12 h at 4°C, followed by horseradish
peroxidase-conjugated secondary antibody (Anti-rabbit IgG, cat.no.
sc-2004, 1:2,000 dilution, Santa Cruz Biotechnology, Inc., Dallas,
TX, USA) for 1 h at 26°C. Protein bands were visualized using a
Pierce™ ECL Western Blotting Substrate (Thermo Fisher Scientific,
Inc.) and the levels of expression were quantified by Image J
software v1.46r.
Flow cytometric analysis of cell
cycle
Cells were harvested by trypsinization, centrifuged
at 1,200 × g for 5 min at 26°C, and washed twice with D-Hanks (pH
7.2–7.4) at 4°C. The cell pellets were fixed by slowly adding 70%
ethanol at 20°C and then stored at 4°C. To determine the DNA
content, the cell pellets were centrifuged at 1,200 × g for 5 min
at 26°C and washed with cold D-Hanks and resuspended in a solution
of propidium iodide (Sigma-Aldrich; Merck KGaA) and RNase A
(Fermentas; Thermo Fisher Scientific, Inc.) in D-Hanks for 30–60
min at 37°C. The cell samples were analyzed using a Guava easyCyte
5HT flow cytometer (EMD Millipore, Billerica, MA, USA). The data
were analyzed to determine the proportion of cells in each stage of
the cell cycle. The experiments were performed in triplicate.
3-(4,5-dimethylthia-zol-2-yl)-2,
5-diphenyltetrazolium bromide (MTT) assay
An MTT assay was performed to measure cell growth.
The cells were seeded at 2,000 per well in 96-well plates and
incubated at 37°C for a designed time period. For each test, 20 µl
of MTT solution (5 mg/ml; Genview Tallahasses, FL, USA) was added
to each of the wells, followed by incubation at 37°C for 4 h.
Subsequently, 100 µl of dimethyl sulfoxide solution was added to
resolve the formazan crystals overnight at 37°C. The optical
density at 490 nm was measured the following day to determine the
quantities of formazan formed by cleaving MTT in living cells. For
each treatment, the cells were cultured for 5 days and the samples
were prepared for the quantitative MTT assay once per day following
plating. Three independent assays were performed, and two samples
were measured at each time point.
Colony formation assay
About 400 cells suspended in DMEM containing 10% FBS
were plated on each well of six-well plates. The plates were
incubated at 37°C in a 5% CO2 incubator for 14 days and
colonies of >50 cells were counted. Each dose was examined in
triplicate, and the experiments were performed three times. The
control shRNA (shCtrl)-transfected MCF-7/ADM cells were detected
using the same methods.
Apoptosis assay using flow cytometric
analysis
The cells were cultured for 5 days, washed twice in
D-Hanks (pH 7.2–7.4), and then resuspended in binding buffer.
Annexin V-APC (10 µl) was added to a final concentration of 1 µg/ml
Annexin V-APC. The mixture was incubated for 15 min in the dark at
room temperature and then analyzed using a flow cytometer (Guava
easyCyte HT; EMD Millipore). The reproducibility of the assay was
examined by two independent experiments.
Statistical analysis
Data are presented as the mean ± standard deviation
and were analyzed using SPSS 23. 0 (IBM SPSS, Armonk, NY, USA).
Student's t-test was used to measure statistical significance
between the experimental and control samples. P<0.05 was
considered to indicate a statistically significant difference.
Results
Expression of CDCA4 is significantly
upregulated following Nrf2 knockdown in breast cancer cells
To identify the genes exhibiting change in
expression when Nrf2 was knocked down, GeneChip analysis was
performed. The resulting heat map showed the differences in the
expression of genes between the nonsilencing control group (NC1,
NC2 and NC3) and silencing/knockdown group (KD1, KD2 and KD3)
samples (Fig. 1A). The expression
profiles of the MCF-7/ADM breast cancer cells were analyzed prior
to and following Nrf2 shRNA transfection by GeneChip. A total of
878 significantly different genes were screened. Among the
identified genes, the present study focused on CDCA4, which was
upregulated in the Nrf2 shRNA-transfected cells, compared with the
control shRNA-transfected cells (fold-change=1.6128796). CDCA4 was
found in several samples of breast cancer tissue, with The Cancer
Gene Atlas (TCGA) public databases showing that the mRNA expression
of CDCA4 was significantly higher, compared with its expression in
adjacent tissues (Table I and
Fig. 1B). These findings suggested
that CDCA4 may be closely associated with breast cancer cells.
 | Table I.Comparison of breast cancer tissues
with normal tissues for mRNA levels of CDCA4 in 106 samples from
The Cancer Gene Atlas public databases. |
Table I.
Comparison of breast cancer tissues
with normal tissues for mRNA levels of CDCA4 in 106 samples from
The Cancer Gene Atlas public databases.
| Gene | FC | P-value | Total samples
(n) | Expression unchanged
(n) | Increased expression
(n) | Decreased expression
(n) |
|---|
| CDCA4 | 2.03 | 3.05E-42 | 106 | 51 | 55 | 0 |
Silencing effect of shRNA on the
expression of CDCA4 in the MCF-7/ADM human breast cancer cell
line
To examine the function of CDCA4 in human breast
cancer, the CDCA4 shRNA-mediated suppression of expression in the
MCF-7/ADM human breast cancer cell line was examined using western
blot and RT-qPCR analyses. The expression of CDCA4 was
significantly decreased in the MCF-7/ADM cells (Fig. 2A and B). Therefore, MCF-7/ADM cells
were selected as the model for subsequent experiments.
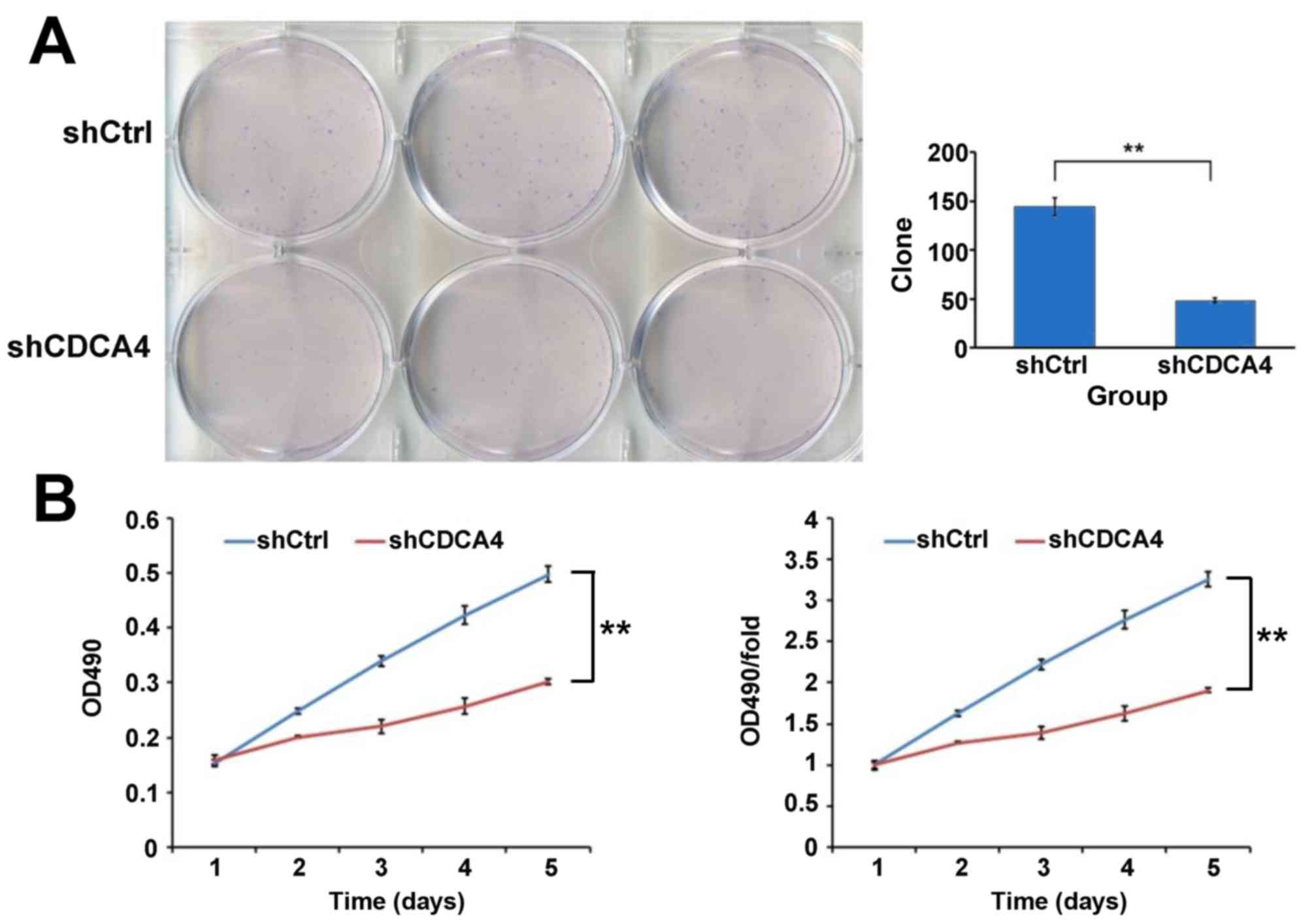
Knockdown of the expression of CDCA4
inhibits the growth of breast cancer cells
A colony formation assay was used to evaluate the
growth of the breast cancer cells in which CDCA4 was silenced. As
shown in Fig. 3A, CDCA4 knockdown
in the MCF-7/ADM cells led to the formation of significantly fewer
colonies on soft agar, compared with the shCtrl group of cells To
further examine the negative effect of CDCA4 knockdown on breast
cancer cell growth, an MTT assay was performed and growth curves
were generated (Fig. 3B). As shown
by the curves, the proliferation of shCDCA4-transfected group cells
was slower, compared with that of the shCtrl-transfected group of
cells during the first 5 days following plating of the cells. The
marked reduction in colony formation and growth of the
CDCA4-silenced cells suggested that the suppression of CDCA4 may
negatively regulate breast cancer cell growth. Together, these data
suggested that CDCA4 enhanced the growth and proliferation of the
human breast cancer cells.
Effect of suppressing the expression
of CDCA4 on cell cycle control in MCF-7/ADM breast cancer
cells
Flow cytometry was used to examine whether the
promoting effect of CDCA4 on MCF-7/ADM cell growth and
proliferation was mediated through cell cycle progression. In the
shCDCA4-transfected group of cells, 40.10% of cells were in the
G0/G1phase, 29.34% were in the S phase and 30.56% were in the G2/M
phase of the cell cycle, which were significantly lower, compared
with the percentages in the shCtrl group of cells (Fig. 4). These data indicated that CDCA4
was significantly associated with the cell cycle of the MCF-7/ADM
cells. Therefore, CDCA4 regulated cell proliferation, at least in
part, through cell cycle progression.
CDCA4 knockdown increases the
apoptotic rate of MCF-7/ADM cells
To investigate whether CDCA4 shRNA-transfected cells
and shCtrl-transfected cells showed differences in apoptotic rate,
an apoptosis assay was performed on the experimental and control
groups of cells using flow cytometry. Apoptosis occurred in
~4.29±0.36% of the control group cells in the 5 days following
plating of the cells. The apoptotic rate of the CDCA4-knockdown
MCF-7/ADM cells increased to 10.1±0.09%. The apoptotic rate of the
shCDCA4 group was significantly higher, compared with that of the
shCtrl group (Fig. 5). These
results indicated that CDCA4 knockdown increased the apoptotic rate
of the MCF-7/ADM cells. Therefore, the present study provided
direct evidence that CDCA4 affected the survival of MCF-7/ADM
cells.
Discussion
Breast cancer is a global health challenge. Patients
with early-stage breast cancer following treatment with surgery
followed by postoperative radiotherapy have a satisfactory survival
rate. However, a substantial number of patients with terminal
breast cancer are unable to undergo surgery due to symptomatic
metastasis. The tolerance of these patients is often too weak to
accept chemotherapy or radiotherapy. Gene-targeted therapy has been
shown to be an effective supplement for conventional therapy for
several types of cancer. The results of the National Surgical
Adjuvant Breast and Bowel Project trial B-31 and the North Central
Cancer Treatment Group trial N9831 suggested that treatment with
trastuzumab following chemotherapy substantially reduced the rate
of recurrence and significantly improved outcomes for patients with
HER2-positive breast cancer (16).
Gene-targeted therapy can be considered as a novel cancer therapy
although it requires further investigation.
In a preliminary study (data not shown), which aimed
to identify potential downstream transcriptional target genes
regulated by Nrf2 in breast cancer cells, GeneChip data were
analyzed, and CDCA4 was shown to be upregulated in Nrf2
shRNA-transfected cells. Considering relevant findings on the SEI
family and analysis of TCGA public databases, it was suggested that
CDCA4 may be closely associated with breast cancer cells.
Few studies (8,11,13)
have demonstrated the importance of CDCA4 in the development of
cancer and cancer cell survival. However, the exact role of CDCA4
in the progression of cancer remains to be fully elucidated, and
the function of CDCA4 in the cell cycle of breast cancer has not
been investigated previously. Whether CDCA4 is a cell fate
regulator important in human breast cancer has not been elucidated.
To address these issues, the present study evaluated the expression
of CDCA4 in terms of its possible direct correlations with the
proliferation and apoptosis of human breast cancer cells.
The present study aimed to examine the effect of RNA
interference targeting CDCA4 on cell proliferation, cell cycle
progression and apoptosis in the MCF-7/ADM human breast cancer cell
line. A couple of studies (10,17)
have revealed that CDCA4 regulates cell proliferation in a variety
of cancer cells. However, the effect of the expression of CDCA4 on
the growth of breast cancer cells remains to be fully elucidated.
The results of the MTT and colony formation assays in the present
study indicated that the suppression of CDCA4 led to the inhibition
of cell proliferation and growth. RNA interference of CDCA4
resulted in a significant increase in the apoptotic rate. These
findings provided direct evidence that the expression of CDCA4 can
affect the survival of human breast cells in vitro, at least
in part. This may be associated with the function of CDCA4 in cell
cycle control. The results of the present study suggested that
CDCA4 is important in regulating the fate of human breast cancer
cells through enhancing proliferation and reducing apoptosis.
In conclusion, the expression of CDCA4 was found to
be associated with the fate of breast cancer cells. Using RNA
interference technology, the results of in vitro experiments
indicated that a downregulation in the expression of CDCA4
inhibited proliferation and induced apoptosis in the MCF-7/ADM
human breast cancer cell line. These data provide a scientific
rationale for potential gene-targeted therapy of breast cancer.
Other potential functions of CDCA4 in breast cancer require further
elucidation through additional relevant investigations.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China (grant no. 81260341).
References
|
1
|
Abdullah JM, Jing X, Spassov DS, Nachtman
RG and Jurecic R: Cloning and characterization of Hepp, a novel
gene expressed preferentially in hematopoietic progenitors and
mature blood cells. Blood Cells Mol Dis. 27:667–676. 2001.
View Article : Google Scholar
|
|
2
|
Walker MG: Drug target discovery by gene
expression analysis: Cell cycle genes. Curr Cancer Drug Targets.
1:73–83. 2001. View Article : Google Scholar
|
|
3
|
Lai IL, Wang SY, Yao YL and Yang WM:
Transcriptional and subcellular regulation of the TRIP-Br family.
Gene. 388:102–109. 2007. View Article : Google Scholar
|
|
4
|
Watanabe-Fukunaga R, Iida S, Shimizu Y,
Nagata S and Fukunaga R: SEI family of nuclear factors regulates
p53-dependent transcriptional activation. Genes Cells. 10:851–860.
2005. View Article : Google Scholar
|
|
5
|
Cheong JK, Gunaratnam L, Zang ZJ, Yang CM,
Sun X, Nasr SL, Sim KG, Peh BK, Rashid SB, Bonventre JV, et al:
TRIP-Br2 promotes oncogenesis in nude mice and is frequently
overexpressed in multiple human tumors. J Transl Med. 7:82009.
View Article : Google Scholar :
|
|
6
|
Tang DJ, Hu L, Xie D, Wu QL, Fang Y, Zeng
Y, Sham JS and Guan XY: Oncogenic transformation by SEI-1 is
associated with chromosomal instability. Cancer Res. 65:6504–6508.
2005. View Article : Google Scholar
|
|
7
|
Hsu SI, Yang CM, Sim KG, Hentschel DM,
O'Leary E and Bonventre JV: TRIP-Br: A novel family of PHD zinc
finger- and bromodomain-interacting proteins that regulate the
transcriptional activity of E2F-1/DP-1. EMBO J. 20:2273–2285. 2001.
View Article : Google Scholar :
|
|
8
|
Li C, Jung S, Lee S, Jeong D, Yang Y, Kim
KI, Lim JS, Cheon CI, Kim C, Kang YS and Lee MS: Nutrient/serum
starvation derived TRIP-Br3 down-regulation accelerates apoptosis
by destabilizing XIAP. Oncotarget. 6:7522–7535. 2015. View Article : Google Scholar :
|
|
9
|
Hong SW, Kim CJ, Park WS, Shin JS, Lee SD,
Ko SG, Jung SI, Park IC, An SK, Lee WK, et al: p34SEI-1 inhibits
apoptosis through the stabilization of the X-linked inhibitor of
apoptosis protein: p34SEI-1 as a novel target for anti-breast
cancer strategies. Cancer Res. 69:741–746. 2009. View Article : Google Scholar
|
|
10
|
Hayashi R, Goto Y, Ikeda R, Yokoyama KK
and Yoshida K: CDCA4 is an E2F transcription factor family-induced
nuclear factor that regulates E2F-dependent transcriptional
activation and cell proliferation. J Biol Chem. 281:35633–35648.
2006. View Article : Google Scholar
|
|
11
|
Tategu M, Nakagawa H, Hayashi R and
Yoshida K: Transcriptional co-factor CDCA4 participates in the
regulation of JUN oncogene expression. Biochimie. 90:1515–1522.
2008. View Article : Google Scholar
|
|
12
|
Lee SL, Hong SW, Shin JS, Kim JS, Ko SG,
Hong NJ, Kim DJ, Lee WJ, Jin DH and Lee MS: p34SEI-1 inhibits
doxorubicin-induced senescence through a pathway mediated by
protein kinase C-delta and c-Jun-NH2-kinase 1 activation in human
breast cancer MCF7 cells. Mol Cancer Res. 7:1845–1853. 2009.
View Article : Google Scholar
|
|
13
|
Alderman C, Sehlaoui A, Xiao Z and Yang Y:
MicroRNA-15a inhibits the growth and invasiveness of malignant
melanoma and directly targets on CDCA4 gene. Tumour Biol.
37:13941–13950. 2016. View Article : Google Scholar
|
|
14
|
Wang XJ, Sun Z, Villeneuve NF, Zhang S,
Zhao F, Li Y, Chen W, Yi X, Zheng W, Wondrak GT, et al: Nrf2
enhances resistance of cancer cells to chemotherapeutic drugs, the
dark side of Nrf2. Carcinogenesis. 29:1235–1243. 2008. View Article : Google Scholar :
|
|
15
|
Livak KJ and Scmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
16
|
Romond EH, Perez EA, Bryant J, Suman VJ,
Geyer CE Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman
PA, et al: Trastuzumab plus adjuvant chemotherapy for operable
HER2-positive breast cancer. N Engl J Med. 353:1673–1684. 2005.
View Article : Google Scholar
|
|
17
|
Wang L, Zhu G, Li Q, Li Y, Xu X, He D and
Zeng C: The spindle function of CDCA4. Cell Motil Cytoskel.
65:581–593. 2008. View
Article : Google Scholar
|