Introduction
Oral cancer is the sixth most common malignant
neoplasm worldwide (1); oral
squamous cell carcinoma (OSCC) is the most common type of oral
cancer and accounts for ~90% of all oral cancer cases, with an
estimated 300,000 new cases diagnosed per year (2). Human papillomavirus infection,
alcohol consumption and smoking were identified as major risk
factors of OSCC (3). Currently,
the main effective treatments for patients are radical operation in
combination with radiotherapy, neoadjuvant chemotherapy and
targeted therapy (4). Despite
remarkable improvements in diagnostic strategies and surgery, the
clinical outcome of patients diagnosed with OSCC remains
unsatisfactory, with a 5-year survival rate of <50% (5). High local recurrence rates and
metastasis are largely responsible for the poor prognosis of
patients with OSCC (6). Therefore,
further elucidation of the mechanisms underlying the pathogenesis
of OSCC may aid the development of tumor-specific biomarkers and
novel therapeutic methods for early diagnosis and therapy of
patients with this malignancy.
microRNAs (miRNAs) are a class of endogenous, highly
conserved, noncoding, short RNAs that serve a role in gene
regulation (7). miRNAs interact
directly with the 3′-untranslated region (UTR) of their target
mRNAs in a base-pairing manner and induce mRNA degradation and/or
inhibit transcription (8). A miRNA
may regulate the expression of various mRNAs simultaneously; it has
been estimated that the expression of ~67% of all human protein
coding genes are modulated by miRNAs (9). Previous studies have reported that
abnormally expressed miRNAs may serve a role in a number of human
disorders, particularly cancers (10). For example, abnormal miRNA
expression was reported in OSCC (11), bladder cancer (12), gastric cancer (13), thyroid cancer (14) and lung cancer (15). Dysregulated miRNAs may serve as
oncogenes or tumor suppressors, and are involved in the regulation
of numerous cellular biological processes, including cell
proliferation, apoptosis, invasion, metastasis, angiogenesis and
epithelial-mesenchymal transition (16–18).
Therefore, miRNAs may serve as novel, effective biomarkers for
cancer diagnosis, therapy and prognosis.
miRNA-655-3p (miR-655), which is located on
chromosome 14q32, was previously reported to be aberrantly
expressed in multiple human cancers, including hepatocellular
carcinoma (19,20), triple-negative breast cancer
(21) and esophageal squamous cell
carcinoma (22). However, the
expression, role and molecular mechanisms of miR-655 in OSCC have
not yet been elucidated. Therefore, the aim of the present study
was to detect miR-655 expression in OSCC tissues and cell lines,
and to investigate its biological roles in OSCC. The mechanisms
underlying the involvement of miR-655 in OSCC were also
investigated. Bioinformatics analyses were performed to determine
potential targets of miR-655, and the results revealed that
metadherin (MTDH) was a candidate target of miR-655. Subsequent
experiments were performed to determine whether MTDH was a direct
target gene of miR-655 in OSCC cells. MTDH has previously been
reported to contribute to the regulation of the phosphatase and
tensin homolog (PTEN)/protein kinase (AKT) signaling pathway
(23,24); therefore, the present study also
investigated whether miR-655 participated in the regulation of the
PTEN/AKT signaling pathway in OSCC cells.
Materials and methods
Collection of OSCC tumoral
tissues
A total of 26 pairs of OSCC tissues and adjacent
non-tumoral oral tissues were collected from patients diagnosed
with OSCC and treated with radical surgery at Yidu Central Hospital
of Weifang (Weifang, China) between November 2014 and February
2017. All patients (15 males and 11 females; aged 49–72 years old)
enrolled in the study received no radiotherapy, chemotherapy,
targeted therapy or other treatments prior to surgery. Patients
treated with radiotherapy, chemotherapy, targeted therapy or other
treatments prior to surgical resection was excluded from the
present study. All tissues were quickly frozen in liquid nitrogen
following surgery and stored in liquid nitrogen until used in
subsequent experiments. The present study was approved by the
Ethics Committee of Yidu Central Hospital of Weifang, and written
informed consent was provided by all patients prior to sample
collection.
Cell culture and transfection
assay
The OSCC cell lines Tca8113, CAL-27 and SCC-9 were
purchased from the American Type Culture Collection (Manassas, VA,
USA) and cultured in Dulbecco's modified Eagle's medium/Ham's F-12
(DMEM/F-12) supplemented with 10% fetal bovine serum (FBS), 100
U/ml penicillin and 100 µg/ml streptomycin (all from Gibco; Thermo
Fisher Scientific, Inc., Waltham, MA, USA). Primary normal human
oral keratinocytes (NHOK) were acquired from ScienCell Research
Laboratories, Inc. (San Diego, CA, USA) and grown in the oral
keratinocyte medium (ScienCell Research Laboratories, Inc.). All
cell lines were maintained at 37°C in a humidified chamber with 5%
CO2.
miR-655 mimics and mimic negative-controls (miR-NC)
were synthesized by Guangzhou Ruibo Biological Technology, Co.,
Ltd. (Guangzhou, China). The miR-655 mimic sequence was
5′-AUAAUACAUGGUUAACCUCUUU-3′ and the miR-NC sequence was
5′-UUCUCCGAACGUGUCACGUTT-3′. MTDH overexpression vector (pCMV-MTDH)
and empty pCMV vector were produced by Shanghai GenePharma Co.,
Ltd. (Shanghai, China). For transfection assays, cells
(5×105 cells/well) were plated onto 6-well plates and
transfected with miR-655 mimics (100 pmol), miR-NC (100 pmol),
pCMV-MTDH (4 µg) or empty pCMV vectors (4 µg) using
Lipofectamine® 2000 (Invitrogen; Thermo Fisher
Scientific, Inc.) at room temperature, according to the
manufacturer's protocol. Following transection for 24 h, CCK-8
assay was performed. Reverse transcription-quantitative polymerase
chain reaction (RT-qPCR) and cell invasion assays were performed a
total of 48 h post-transfection. Western blot analysis was used to
detect protein expression in transfected cells a total of 72 h
post-incubation.
RNA isolation and RT-qPCR
RT-qPCR was performed to determine the expression
levels of miR-655 and MTDH mRNA. Total RNA was extracted from
tissue samples (100 mg) or cells (1×106) using the
TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. To detect miR-655
expression, reverse transcription was carried out using a TaqMan
miRNA Reverse Transcription kit followed by qPCR with a TaqMan
miRNA assay kit (both from Applied Biosystems; Thermo Fisher
Scientific, Inc.). The temperature protocol for RT was as follows:
16°C for 30 min, 42°C for 30 min and 85°C for 5 min. The
thermocycling conditions used for qPCR were as follows: 50°C for 2
min and 95°C for 10 min; followed by 40 cycles of denaturation at
95°C for 15 sec; and subsequently annealing/extension at 60°C for
60 sec. For the quantification of MTDH mRNA levels, a PrimeScript
RT Reagent kit was used to synthesize cDNA, and qPCR was performed
using SYBR Premix Ex Taq II (both from Takara Biotechnology
Co., Ltd., Dalian, China). The temperature protocol for RT was as
follows: 37°C for 15 min and 85°C for 5 sec. The thermocycling
conditions used for qPCR were as follows: 5 min at 95°C; followed
by 40 cycles of 95°C for 30 sec and 65°C for 45 sec. RT-qPCR was
performed in an ABI PRISM 7000 Fluorescent Quantitative PCR system
(Applied Biosystems; Thermo Fisher Scientific, Inc.). U6 small
nuclear RNA and GAPDH were used as the endogenous controls to
normalize the levels of miR-655 and MTDH mRNA, respectively. The
primers used were as follows: miR-655 forward,
5′-TCCGAACATGGTTAA-3′ and reverse, 5′-GTGCAGGGTCCGAGGT-3′; U6
forward, 5′-CTCGCTTCGGCAGCACA-3′ and reverse,
5′-AACGCTTCACGAATTTGCGT-3′; MTDH forward, 5′-TGTTGAAGTGGCTGAGGG-3′
and reverse, 5′-CAGGAAATGATGCGGTTG-3′; and GAPDH forward,
5′-CGGAGTCAACGGATTTGGTCGTAT-3′ and reverse,
5′-AGCCTTCTCCATGGTGGTGAAGAC-3′. Relative gene expression was
calculated using the 2−ΔΔCq method (25).
MTT cell proliferation assay
Following transfection for 24 h, cells (2,000
cells/well) were seeded into 96-well plates, and the extent of
proliferation was detected at room temperature by performing an MTT
assay at 0, 24, 48 and 72 h. Briefly, a total of 20 µl MTT solution
(5 mg/ml; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) was added
to each well. The culture plates were incubated at 37°C for 4 h.
Subsequently, the supernatant containing the MTT solution was
discarded and 150 µl dimethyl-sulfoxide was added into each well to
dissolve the formazan precipitate. The absorbance was read at a
wavelength of 490 nm using an ELISA microplate reader (Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Cell invasion assay
Transfected cells were trypsinized, collected and
washed with FBS-free DMEM/F-12 medium at 48 h post-transfection. A
total of 1×105 cells were suspended in 200 µl FBS-free
DMEM/F-12 medium and seeded in the upper Transwell chambers (8 µm;
Corning Inc., Corning, NY, USA) coated with Matrigel (BD
Biosciences, San Jose, CA, USA). DMEM/F-12 medium (500 µl)
containing 10% FBS was added into the lower chamber. Following 24 h
incubation at 37°C with 5% CO2, the non-invading cells
on the upper membrane were scraped off with cotton swabs. The
invasive cells were fixed with 95% ethanol for 15 min and stained
with 0.5% crystal violet for 15 min, both at room temperature. The
number of invasive cells was counted in at least five randomly
selected fields using an IX51 inverted microscope (magnification,
×200; Olympus Corporation, Tokyo, Japan).
Bioinformatics analysis
TargetScan (version 7.2; http://www.targetscan.org) and PicTar (http://pictar.mdc-berlin.de) were used to predict the
potential targets of miR-655.
Luciferase report assay
Human MTDH 3′-UTRs containing putative wild-type
(WT) or mutated (Mut) binding sites for miR-655 were amplified by
Shanghai GenePharma Co., Ltd., cloned into the psiCHECK-2 reporter
vector (Promega Corporation, Madison, WI, USA), and designated
MTDH-3′-UTR-WT or MTDH-3′-UTR-Mut. Cells were seeded into 24-well
plates (1.5×105 cells/well) 1 day prior to transfection
and incubated at 37°C with 5% CO2. miR-655 mimics (50
pmol) or miR-NC (50 pmol) were co-transfected into cells with
either MTDH-3′-UTR-WT (100 ng) or MTDH-3′-UTR-Mut (100 ng) using
Lipofectamine 2000, and incubated at 37°C with 5% CO2
for 48 h, according to the manufacturer's protocol. Luciferase
activities were evaluated using the Dual Luciferase Assay kit
(Promega Corporation), according to the manufacturer's protocol.
Firefly luciferase activities were normalized to Renilla
luciferase activities.
Western blot analysis
The Total Protein Extraction kit (Nanjing KeyGen
Biotech Co., Ltd., Nanjing, China) was used to extract total
protein from tissue samples (200 mg) or cells (1×106).
Subsequently, the concentration of total protein was quantified
using a Bicinchoninic Acid Assay kit (Nanjing KeyGen Biotech Co.,
Ltd., Nanjing, China). Equivalent amounts of protein (30 µg) were
separated by 10% SDS-PAGE and electroblotted onto polyvinylidene
fluoride membranes (EMD Millipore, Billerica, MA, USA). The
membranes were blocked with 5% non-fat milk in TBS containing 0.1%
Tween-20 (TBST) at room temperature for 1 h and incubated with the
following primary antibodies overnight at 4°C: Mouse anti-human
MTDH antibody (1:1,000; cat. no. sc-517220; Santa Cruz
Biotechnology, Inc., Dallas, TX, USA), mouse anti-human PTEN
antibody (1:1,000; cat. no. ab77161; Abcam, Cambridge, UK), mouse
anti-human RAC-α serine/threonine-AKT antibody (1:1,000; cat. no.
sc-56878), mouse anti-human phosphorylated (p)-AKT antibody
(1:1,000; cat. no. sc-271966; both Santa Cruz Biotechnology, Inc.)
and mouse anti-human GAPDH antibody (1:1,000; cat. no. ab125247;
Abcam). Membranes were washed three times with TBST, followed by
incubation with horseradish peroxidase-conjugated goat anti-mouse
secondary antibody (1:5,000; cat. no. ab205719; Abcam). Following
extensive washes with TBST, protein signals were visualized with an
Enhanced Chemiluminescence Detection System (Pierce; Thermo Fisher
Scientific, Inc.). GAPDH was used as a loading control for
normalization of protein expression levels. Densitometric analysis
was performed using Quantity One software version 4.62 (Bio-Rad
Laboratories, Inc., Hercules, CA, USA).
Statistical analysis
Data are presented as the mean ± standard deviation,
based on the results of at least three repeated experiments.
Analyses were performed with SPSS 19.0 software (IBM Corporation,
Armonk, NY, USA). Differences between two groups were analyzed with
a two-tailed Student's t-test. One-way analysis of variance
followed by Student-Newman-Keuls post hoc test was performed to
investigate the differences between more than two groups.
Spearman's correlation analysis was used to examine the correlation
between miR-655 and MTDH mRNA expression levels in OSCC tissues.
P<0.05 was considered to indicate a statistically significant
difference.
Results
miR-655 expression is downregulated in
OSCC tissues and cell lines
To examine the role of miR-655 in OSCC, miR-655
expression levels were determined in a total of 26 pairs of OSCC
tissues and adjacent non-tumoral tissues using RT-qPCR. Compared
with expression in the adjacent non-tumor tissues, miR-655
expression was significantly lower in OSCC tissues (Fig. 1A; P<0.05). In addition, miR-655
expression levels were examined in three OSCC cell lines (Tca8113,
CAL-27 and SCC-9) and NHOK cells. The data indicated that miR-665
expression was significantly lower in OSCC cell lines compared with
expression in NHOK cells (Fig. 1B;
P<0.05). These results suggested that the expression of miR-655
is significantly reduced in OSCC; this reduced expression may be
related to OSCC progression.
miR-655 inhibits Tca8113 and CAL-27
cell proliferation and invasion
Tca8113 and CAL-27 cells exhibited relatively low
miR-655 expression among the three OSCC cell lines; therefore,
these cells were selected for subsequent experiments. To explore
the effects of miR-655 in OSCC, miR-655 mimics were transfected
into Tca8113 and CAL-27 cells to increase miR-655 expression
levels. RT-qPCR analysis confirmed that miR-655 was significantly
increased in Tca8113 and CAL-27 cells transfected with miR-655
mimics compared expression levels in cells transfected with miR-NC
(Fig. 2A; P<0.05).
MTT and cell invasion assays were performed to
examine the effects of miR-655 overexpression on proliferation and
invasion, respectively, of Tca8113 and CAL-27 cells. The results
indicated that transfection with miR-655 mimics led to a
significant reduction in proliferation and invasion compared with
the respective miR-NC groups (Fig. 2B
and C, respectively; P<0.05). These results suggested that
miR-655 may have tumor suppressive roles in OSCC growth and
metastasis.
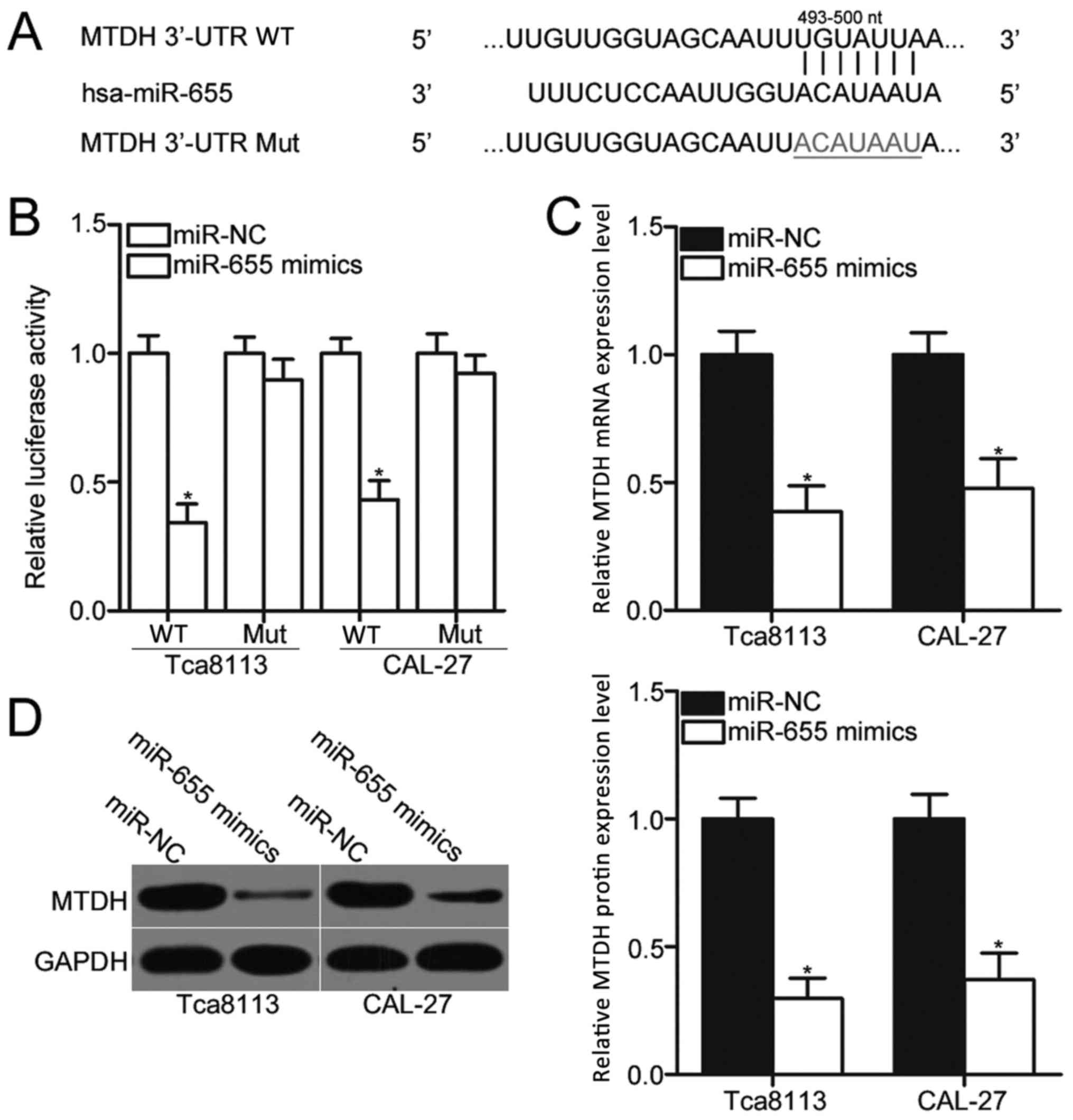
miR-655 directly targets and
downregulates MTDH mRNA expression in OSCC cells
The biological roles of miRNAs in human malignancies
depend on their specific targets; therefore, bioinformatics
analysis was performed to predict the potential targets of miR-655.
A total of 780 conserved sites were revealed, including A
disintegrin metallopeptidase domain-containing protein 10;
pituitary tumor-transforming 1 interacting protein; and membrane
associated guanylate kinase, WW and PDZ domain containing 2. Among
these candidates, MTDH was predicted as a major target of miR-655
(Fig. 3A), which has been
previously reported to be involved in OSCC occurrence and
development (26–29). To confirm this hypothesis,
luciferase reporter assays were performed to determine whether the
3′-UTR of MTDH was directly targeted by miR-655 in OSCC cells.
Luciferase activity was significantly reduced in MTDH-3′-UTR-WT +
miR655 mimics transfected Tca8113 and CAL-27 cells compared with
cells co-transfected with MTDH-3′-UTR-WT + miR-NC (Fig. 3B; P<0.05), whereas no
significant differences in luciferase activities were identified in
cells co-transfected with miR-655 mimics or miR-NC and
MTDH-3′-UTR-Mut. Furthermore, RT-qPCR and western blot assays
demonstrated that overexpression of miR-655 significantly reduced
the expression levels of MTDH mRNA and protein (Fig. 3C and D, respectively; P<0.05) in
Tca8113 and CAL-27 cells compared with miR-NC transfected cells.
These data indicated that MTDH is a direct target of miR-655 in
OSCC cells.
miR-655 expression is inversely
correlated with MTDH mRNA expression levels in OSCC tissues
To further explore the association between miR-655
and MTDH in OSCC, MTDH mRNA expression levels were determined in
the 26 pairs of OSCC tissues and adjacent non-tumoral tissues.
RT-qPCR analysis revealed that MTDH mRNA expression was
significantly higher in OSCC tissues compared with the expression
levels in the adjacent non-tumoral tissues (Fig. 4A; P<0.05). In addition, western
blot analysis was performed to detect MTDH protein levels in OSCC
tissues and adjacent non-tumor tissues, which demonstrated that the
protein expression level of MTDH was notably higher in OSCC tissues
compared to that in adjacent non-tumor tissues (Fig. 4B). Furthermore, the expression of
miR-655 exhibited an inverse correlation with MTDH mRNA expression
in OSCC tissues (Fig. 4C;
r=−0.5949; P<0.05). These results further indicated that MTDH
may be a novel target of miR-655 in OSCC.
Inhibiting MTDH is crucial for the
inhibitory effects of miR-655 expression in OSCC cells
Rescue experiments were performed to further
determine whether the suppressive roles of miR-655 were mediated
through MTDH in OSCC cells. The MTDH overexpression vector
pCMV-MTDH or the pCMV empty vector was transfected into Tca8113 and
CAL-27 cells. RT-qPCR analysis was performed to confirm that MTDH
mRNA expression was significantly increased in Tca8113 and CAL-27
cells that were transfected with pCMV-MTDH compared with pCMV empty
vector-transfected cells (Fig. 5A;
P<0.05). To perform rescue experiments, Tca8113 and CAL-27 cells
were co-transfected with pCMV-MTDH or pCMV empty vector along with
miR-655 mimics. Western blot analysis revealed that the MTDH
protein expression that was decreased by miR-655 mimics was
recovered in cells co-transfected with pCMV-MTDH (Fig. 5B; P<0.05). Subsequent MTT and
cell invasion assays demonstrated that the restoration of MTDH
expression reversed the suppressive effects of miR-655
overexpression on cell proliferation (Fig. 5C and D; P<0.05) and invasion
(Fig. 5E; P<0.05). These
results suggested that the tumor-suppressive roles of miR-655 in
OSCC cells may be mediated, at least part, by the inhibition of
MTDH expression.
miR-655 overexpression inactivates the
PTEN/AKT pathway in OSCC cells
MTDH has previously been reported to contribute to
the regulation of the PTEN/AKT signaling pathway (23,24).
To examine whether miR-655 was able to affect the PTEN/AKT pathway,
the protein expression levels of PTEN, p-AKT and AKT were detected
in Tca8113 and CAL-27 cells co-transfected with miR-655 mimics and
either pCMV-MTDH or pCMV empty vector. In miR-655 mimics + pCMV
empty vector co-transfected cells, there was a notable increase in
PTEN expression and a notable decrease in p-AKT expression compared
with miR-NC transfected cells (Fig.
6); total AKT expression appeared to be unaffected. Conversely,
co-transfection with pCMV-MTDH restored the reduced expression of
PTEN and p-AKT caused by miR-655 overexpression. These results
suggested that miR-655 may inhibit the activation of the PTEN/AKT
signaling pathway in OSCC through the negative regulation of MTDH
expression.
Discussion
Numerous studies have indicated that miRNAs are
important regulators for a variety of biological processes, and
their dysregulation is closely related to cancer formation and
progression (30–32). Therefore, the identification of
aberrantly expressed miRNAs may provide important insight into the
diagnosis and therapy of patients with OSCC. In the present study,
miR-655 expression in OSCC was investigated as well as the roles
and underlying mechanisms associated with miR-655 in the
progression of OSCC. To the best of our knowledge, the present
study is the first to demonstrate that miR-655 may have exhibited
tumor suppressive roles in OSCC by directly targeting MTDH and
regulating the PTEN/AKT signaling pathway, which suggested that
miR-655 may represent an effective therapeutic agent in the
treatment of patients with OSCC.
In the present study, miR-655 expression was
demonstrated to be significantly lower in OSCC tissues and cell
lines compared with expression levels in adjacent non-tumor tissues
and NHOK cells. miR-655 has been previously reported to be
dysregulated in several types of tumors. For example, miR-655
expressed in low levels in hepatocellular carcinoma tissues, and
its expression levels were strongly correlated with tumor size,
portal vein tumor thrombosis status, microvascular invasion,
tumor-node-metastasis stage and lymph node metastasis (19,20).
Patients with hepatocellular carcinoma with low miR-655 expression
levels exhibited lower survival time compared with those patients
with high miR-655 levels, and multivariate analysis identified
miR-655 as an independent risk factor for patients with
hepatocellular carcinoma (19). In
triple-negative breast cancer, the expression of miR-655 was
reduced in tumoral tissues and was significantly associated with
lymph node metastasis (21). In
esophageal squamous cell carcinoma, miR-655 expression was lower in
tumoral tissues compared with expression in adjacent non-tumoral
tissues and was negatively correlated with of lymph node metastases
(22). These findings suggested
that miR-655 expression may be frequently reduced in human cancers
and should be investigated as a potential biomarker for the
diagnosis and prognosis of patients with these specific cancer
types.
The results from the present study further
demonstrated that ectopic expression of miR-655 reduced the
proliferative and invasive abilities of OSCC cells. Dysregulation
of miR-655 expression was implicated in the occurrence and
development of several types of human malignancies. For instance,
overexpression of miR-655 inhibited cell growth and metastasis in
hepatocellular carcinoma (20).
Another study reported that upregulation of miR-655 decreased
migration, invasion and epithelial-to-mesenchymal transition of
triple-negative breast cancer cells (21). miR-655 overexpression was reported
to restrict the migratory capacity of esophageal squamous cell
carcinoma cells (22). It was also
previously reported that miR-655 upregulation prohibited cell
proliferation and motility, and promoted apoptosis in pituitary
tumor cells (33). These findings
suggested that miR-655 may be a novel and promising therapeutic
candidate target for anticancer treatment.
Many direct targets of miR-655 have been validated,
including ADAM metallopeptidase domain 10 in hepatocellular
carcinoma (20), paired-related
homeobox 1 in triple-negative breast cancer (21), pituitary tumor-transforming 1 in
esophageal squamous cell carcinoma (22) and membrane-associated guanylate
kinase, WW and PDZ domain-containing 2 in lung adenocarcinoma
(34). In the present study, MTDH
was identified as a direct target gene of miR-655 in OSCC. MTDH is
located on chromosome 8q22 (35);
it has been reported to be highly expressed in various types of
human cancer, including glioma (36), breast cancer (37), gastric cancer (38), bladder cancer (39) and cervical cancer (40). In OSCC, both mRNA and protein
levels of MTDH were overexpressed in tumor tissues and their
overexpression was positively correlated with differentiation,
clinical stage, T classification and lymph node metastasis. OSCC
patients with high MTDH levels exhibited shorter overall survival
rates relative to those patients with low MTDH expression (26,27).
In addition, MTDH was previously confirmed as an independent
prognostic factor for overall survival rates in OSCC patients
(26,27). Highly expressed MTDH was reported
to serve important roles in the onset and the progression of OSCC
by affecting tumor cell growth, colony formation, migration and
invasion (26,28,29).
MTDH has previously been reported to be involved in the regulation
of the PTEN/AKT signaling pathway (23,24).
It has been well established that activation of the PTEN/AKT
pathway contributes to OSCC development (41,42).
In the present study, it was demonstrated that restoration of MTDH
expression inactivated the PTEN/AKT pathway in OSCC cells via
regulation of MTDH. These findings suggested that MTDH is an
effective candidate for molecular targeted therapy for OSCC.
To the best of our knowledge, the present study is
the first to demonstrate that miR-655 expression was reduced in
OSCC tissues and cell lines. In addition, miR-655 overexpression
suppressed cell proliferation and invasion in OSCC by directly
targeting MTDH and regulating the PTEN/AKT pathway. These results
may improve our understanding of OSCC pathogenesis and may also
provide a theoretical basis for the identification of miR-655 as a
potential tumor suppressor in OSCC.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the present
study are available from the corresponding author on reasonable
request.
Authors' contributions
HJ and QW designed the research; QW, LL and YL
performed functional experiments. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The present study was approved by the Ethics
Committee of Yidu Central Hospital of Weifang (Weifang, China) and
was performed in accordance with The Declaration of Helsinki and
the guidelines of the Ethics Committee of Yidu Central Hospital of
Weifang. Written informed consent was obtained from all patients
for the use of their clinical tissues, prior to enrolment in the
present study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Min A, Zhu C, Peng S, Rajthala S, Costea
DE and Sapkota D: MicroRNAs as important players and biomarkers in
oral carcinogenesis. Biomed Res Int. 2015:1869042015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Warnakulasuriya S: Global epidemiology of
oral and oropharyngeal cancer. Oral Oncol. 45:309–316. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Zaravinos A: An updated overview of
HPV-associated head and neck carcinomas. Oncotarget. 5:3956–3969.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Leemans CR, Braakhuis BJ and Brakenhoff
RH: The molecular biology of head and neck cancer. Nat Rev Cancer.
11:9–22. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
5
|
Perez-Sayans M, Suarez-Penaranda JM,
Padin-Iruegas ME, Gayoso-Diz P, Reis-De Almeida M, Barros-Angueira
F, Gandara-Vila P, Blanco-Carrion A and Garcia-Garcia A: The loss
of p16 expression worsens the prognosis of OSCC. Appl
Immunohistochem Mol Morphol. 23:724–732. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Liang L, Zhang T, Kong Q, Liang J and Liao
G: A meta-analysis on selective versus comprehensive neck
dissection in oral squamous cell carcinoma patients with clinically
node-positive neck. Oral Oncol. 51:1076–1081. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Moreno-Moya JM, Vilella F and Simon C:
MicroRNA: Key gene expression regulators. Fertil Steril.
101:1516–1523. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
John B, Enright AJ, Aravin A, Tuschl T,
Sander C and Marks DS: Human MicroRNA targets. PLoS Biol.
2:e3632004. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vasudevan S, Tong Y and Steitz JA:
Switching from repression to activation: microRNAs can up-regulate
translation. Science. 318:1931–1934. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Calin GA and Croce CM: MicroRNA signatures
in human cancers. Nat Rev Cancer. 6:857–866. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang YJ, Zhang ZF, Fan SH, Zhuang J, Shan
Q, Han XR, Wen X, Li MQ, Hu B, Sun CH, et al: MicroRNA-433 inhibits
oral squamous cell carcinoma cells by targeting FAK. Oncotarget.
8:100227–100241. 2017.PubMed/NCBI
|
|
12
|
Yang D, Du G, Xu A, Xi X and Li D:
Expression of miR-149-3p inhibits proliferation, migration and
invasion of bladder cancer by targeting S100A4. Am J Cancer Res.
7:2209–2219. 2017.PubMed/NCBI
|
|
13
|
Zabaglia LM, Bartolomeu NC, Dos Santos MP,
Peruquetti RL, Chen E, de Arruda Cardoso, Smith M, Payao SLM and
Rasmussen LT: Decreased MicroRNA miR-181c expression associated
with gastric cancer. J Gastrointest Cancer. 49:97–101. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Celano M, Rosignolo F, Maggisano V, Pecce
V, Iannone M, Russo D and Bulotta S: MicroRNAs as biomarkers in
thyroid carcinoma. Int J Genomics. 2017:64965702017. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Kang M, Shi J, Peng N and He S:
MicroRNA-211 promotes non-small-cell lung cancer proliferation and
invasion by targeting MxA. Onco Targets Ther. 10:5667–5675. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Tavazoie SF, Alarcon C, Oskarsson T, Padua
D, Wang Q, Bos PD, Gerald WL and Massague J: Endogenous human
microRNAs that suppress breast cancer metastasis. Nature.
451:147–152. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cui F, Li X, Zhu X, Huang L, Huang Y, Mao
C, Yan Q, Zhu J, Zhao W and Shi H: MiR-125b inhibits tumor growth
and promotes apoptosis of cervical cancer cells by targeting
phosphoinositide 3-kinase catalytic subunit delta. Cell Physiol
Biochem. 30:1310–1318. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Zuo QF, Zhang R, Li BS, Zhao YL, Zhuang Y,
Yu T, Gong L, Li S, Xiao B and Zou QM: MicroRNA-141 inhibits tumor
growth and metastasis in gastric cancer by directly targeting
transcriptional co-activator with PDZ-binding motif, TAZ. Cell
Death Dis. 6:e16232015. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhao XQ, Liang B, Jiang K and Zhang HY:
Down-regulation of miR-655-3p predicts worse clinical outcome in
patients suffering from hepatocellular carcinoma. Eur Rev Med
Pharmacol Sci. 21:748–752. 2017.PubMed/NCBI
|
|
20
|
Wu G, Zheng K, Xia S, Wang Y, Meng X, Qin
X and Cheng Y: MicroRNA-655-3p functions as a tumor suppressor by
regulating ADAM10 and β-catenin pathway in hepatocellular
carcinoma. J Exp Clin Cancer Res. 35:892016. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lv ZD, Kong B, Liu XP, Jin LY, Dong Q, Li
FN and Wang HB: miR-655 suppresses epithelial-to-mesenchymal
transition by targeting Prrx1 in triple-negative breast cancer. J
Cell Mol Med. 20:864–873. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Wang Y, Zang W, Du Y, Ma Y, Li M, Li P,
Chen X, Wang T, Dong Z and Zhao G: Mir-655 up-regulation suppresses
cell invasion by targeting pituitary tumor-transforming gene-1 in
esophageal squamous cell carcinoma. J Transl Med. 11:3012013.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Li J, Li C, Li H, Zhang T, Hao X, Chang J
and Xu Y: MicroRNA30a5p suppresses tumor cell proliferation of
human renal cancer via the MTDH/PTEN/AKT pathway. Int J Mol Med.
41:1021–1029. 2018.PubMed/NCBI
|
|
24
|
Li L and Zhang H: MicroRNA-379 inhibits
cell proliferation and invasion in glioma via targeting metadherin
and regulating PTEN/AKT pathway. Mol Med Rep. 17:4049–4056.
2018.PubMed/NCBI
|
|
25
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Xia X, Du R, Zhao L, Sun W and Wang X:
Expression of AEG-1 and microvessel density correlates with
metastasis and prognosis of oral squamous cell carcinoma. Hum
Pathol. 45:858–865. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Seyedmajidi M, Sohanian S, Abbaszadeh H,
Moslemi D and Bijani A: Astrocyte elevated gene 1 (AEG-1): A
promising candidate for molecular targeted therapy in oral squamous
cell carcinomas. Asian Pac J Cancer Prev. 18:3301–3305.
2017.PubMed/NCBI
|
|
28
|
Wang Y, Wang T, Sun Y, Sun W and Wang X:
Astrocyte elevated gene-1 promotes tumour growth and invasion by
inducing EMT in oral squamous cell carcinoma. Sci Rep. 7:154472017.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wang YP, Liu IJ, Chiang CP and Wu HC:
Astrocyte elevated gene-1 is associated with metastasis in head and
neck squamous cell carcinoma through p65 phosphorylation and
upregulation of MMP1. Mol Cancer. 12:1092013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Garzon R, Calin GA and Croce CM: MicroRNAs
in Cancer. Annu Rev Med. 60:167–179. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Garzon R and Marcucci G: Potential of
microRNAs for cancer diagnostics, prognostication and therapy. Curr
Opin Oncol. 24:655–659. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Schickel R, Boyerinas B, Park SM and Peter
ME: MicroRNAs: Key players in the immune system, differentiation,
tumorigenesis and cell death. Oncogene. 27:5959–5974. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Liang HQ, Wang RJ, Diao CF, Li JW, Su JL
and Zhang S: The PTTG1-targeting miRNAs miR-329, miR-300, miR-381
and miR-655 inhibit pituitary tumor cell tumorigenesis and are
involved in a p53/PTTG1 regulation feedback loop. Oncotarget.
6:29413–29427. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kitamura K, Seike M, Okano T, Matsuda K,
Miyanaga A, Mizutani H, Noro R, Minegishi Y, Kubota K and Gemma A:
MiR-134/487b/655 cluster regulates TGF-beta-induced
epithelial-mesenchymal transition and drug resistance to gefitinib
by targeting MAGI2 in lung adenocarcinoma cells. Mol Cancer Ther.
13:444–453. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Su ZZ, Kang DC, Chen Y, Pekarskaya O, Chao
W, Volsky DJ and Fisher PB: Identification and cloning of human
astrocyte genes displaying elevated expression after infection with
HIV-1 or exposure to HIV-1 envelope glycoprotein by rapid
subtraction hybridization, RaSH. Oncogene. 21:3592–3602. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
He Z, He M, Wang C, Xu B, Tong L, He J,
Sun B, Wei L and Chu M: Prognostic significance of astrocyte
elevated gene-1 in human astrocytomas. Int J Clin Exp Pathol.
7:5038–5044. 2014.PubMed/NCBI
|
|
37
|
Li J, Zhang N, Song LB, Liao WT, Jiang LL,
Gong LY, Wu J, Yuan J, Zhang HZ, Zeng MS and Li M: Astrocyte
elevated gene-1 is a novel prognostic marker for breast cancer
progression and overall patient survival. Clin Cancer Res.
14:3319–3326. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Dong L, Qin S, Li Y, Zhao L, Dong S, Wang
Y, Zhang C and Han S: High expression of astrocyte elevated gene-1
is associated with clinical staging, metastasis and unfavorable
prognosis in gastric carcinoma. Tumour Biol. 36:2169–2178. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Nikpour M, Emadi-Baygi M, Fischer U,
Niegisch G, Schulz WA and Nikpour P: MTDH/AEG-1 contributes to
central features of the neoplastic phenotype in bladder cancer.
Urol Oncol. 32:670–677. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Yu JQ, Zhou Q, Zhu H, Zheng FY and Chen
ZW: Overexpression of astrocyte elevated gene-1 (AEG-1) in cervical
cancer and its correlation with angiogenesis. Asian Pac J Cancer
Prev. 16:2277–2281. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Alyasiri NS, Mehdi SJ, Alam MS, Ali A,
Mandal AK, Gupta S, Singh I and Rizvi MM: PTEN-mediated AKT
activation contributes to the reduced apoptosis among Indian oral
squamous cell carcinoma patients. J Cancer Res Clin Oncol.
138:103–109. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Gan YH and Zhang S: PTEN/AKT pathway
involved in histone deacetylases inhibitor induced cell growth
inhibition and apoptosis of oral squamous cell carcinoma cells.
Oral Oncol. 45:e150–e154. 2009. View Article : Google Scholar : PubMed/NCBI
|