Introduction
Ovarian cancer is a highly metastatic malignancy.
The survival rate of ovarian cancer remains low and has only
slightly increased during the last several decades, as many
patients are still diagnosed at an advanced stage. In the United
States, ovarian cancer is the fifth leading cause of cancer-related
death in postmenopausal women (1).
Recently, the mortality rate of ovarian cancer patients
demonstrated an increasing trend in China from 2000 to 2011
(2). The main reason for this high
mortality rate is not only the fact that many patients are
diagnosed at an advanced stage, but also the development of
drug-resistance such as cisplatin-resistance despite improvements
in chemotherapies. Therefore, in addition to the discovery of novel
targets for early detection, new therapeutic strategies are
necessary to improve the outcomes of patients with ovarian
cancer.
Emodin is a natural anthraquinone isolated from
several plants including Rhubarb and Polygonum cuspidatum
that are widely applied in traditional Chinese medicine (3). It has been demonstrated that emodin
exhibits an antitumor effect in vitro through several
mechanisms. Inhibition of angiogenesis by suppressing
angiogenesis-associated microRNA (miR)-155, miR-210 and miR-20b was
observed in pancreatic cancer (4).
Gastric cancer cell proliferation was suppressed by emodin via
downregulation of c-myc expression (5). Additionally, studies have revealed
that emodin induces cell cycle arrest and apoptosis in colon cancer
cells by increasing the expression of caspase-6 (6) and p53 (7). Recent studies have demonstrated that
emodin also plays an inhibitory role in transforming growth factor
(TGF)-β-induced epithelial-mesenchymal transition (EMT) (8,9). A low
concentration of emodin was shown to enhance paclitaxel-induced
apoptosis (10). Combined use of
emodin and cisplatin reduced the growth of human ovarian carcinoma
cells by downregulating multidrug resistance-related protein 1
expression (11).
Although studies have investigated the mechanisms
underlying the effects of emodin on ovarian cancer since the last
decade, the conclusions are inconsistent. A deeper insight into the
in vitro role of emodin in inhibiting ovarian cancer cell
growth is warranted. In the present study, we demonstrated that
emodin inhibited the expression of TGF-β2 by regulating miR-199a
and forkhead box D3 (FOXD3) in ovarian cancer cells.
Materials and methods
Cell culture
A2780 human epithelial ovarian cancer cell line was
purchased from the Cell Bank of the China Academy of Sciences
(Shanghai, China). Cells were cultured in RPMI-1640 medium modified
(HyClone Laboratories; GE Healthcare, Chicago, IL, USA),
supplemented with 10% fetal bovine serum (FBS), 100 U/ml penicillin
and 100 µg/ml streptomycin in humidified air at 37°C with 5%
CO2. For treatment, 20 µM emodin (cat. no. 30269, purity
≥97.0%; Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) or dimethyl
sulfoxide (DMSO) was added to the cell medium for 0, 6, 12 or 24 h.
Trypsin (0.25%) was used to detach the cells from the plates.
RNA extraction and cDNA synthesis
Total RNA was extracted using TRIzol reagent
(Invitrogen; Thermo Fisher Scientific, Inc., Waltham, MA, USA)
following the manufacturer's protocol. RNA purity was assessed by
Thermo NanoDrop 2000 (Thermo Fisher Scientific, Inc.) by standard
absorbance ratios as A260/A280 ≥1.8 and A260/A230 ≥1.5.
Complementary DNAs were synthesized from 1 µg of total RNA using
TaqMan Reverse Transcription reagents (Life Technologies; Thermo
Fisher Scientific, Inc.).
Microarray analysis
A2780 cells were pre-treated with 20 µM emodin or
DMSO for 24 h. To quantify miRNAs, TaqMan® MicroRNA
Reverse Transcription kit (Invitrogen; Thermo Fisher Scientific,
Inc.) was used. For the microarray assay, RNA was amplified with
the GeneChip 3′IVT Express kit (Affymetrix Inc.; Thermo Fisher
Scientific, Inc.). Quality of the samples was verified by the
NanoDrop results. The amplified transcripts were hybridized to
Affymetrix Human Genome U133 Plus 2.0 Array (Affymetrix; Thermo
Fisher Scientific, Inc.) according to the manufacturer's protocol.
The results were analyzed using GeneSpring 12.6 (Agilent
Technologies, Inc., Santa Clara, CA, USA).
Computational miRNA target prediction
and quantitative real-time PCR
TargetScan 6.0 (http://www.targetscan.org/) was used to predict
potential miRNAs binding to TGF-β2. The predicted miRNAs were
tested using quantitative real-time (qRT)-PCR. For miRNA analysis,
qRT-PCR was performed using TaqMan microRNA Assay (Applied
Biosystems; Thermo Fisher Scientific, Inc.) according to the
manufacturer's instructions with specific TaqMan probes (Applied
Biosystems; Thermo Fisher Scientific, Inc.). For quantitation of
mRNA, a Bio-Rad CFX96 Real-Time PCR system (Bio-Rad Laboratories,
Hercules, CA, USA) was employed according to the manufacturer's
instructions. All mRNA and miRNA quantification data were
normalized to GAPDH and U6, respectively. hTGF-β2 sense,
5′-TGGTGAAAGCAGAGTTCAGAG-3′ and antisense,
5′-CACAACTTTGCTGTCGATGTAG-3′; GAPDH sense,
5′-AGCCTCCCGCTTCGCTCTCT-3′ and antisense,
5′-GCGCCCAATACGACCAAATCCGT-3′; U6 sense,
5′-GCTTCGGCAGCACATATACTAAAAT-3′ and antisense,
5′-CGCTTCACGAATTTGCGTGTCAT-3′; FOXD3 sense,
5′-GTCCGCTGGGAATAACTTTCCGTA-3′ and antisense,
5′-ATGTACAAAGAATGTCCCTCCCACCC-3′ were used
Luciferase assay
The three mutations in the TGF-β2 3′ untranslated
region (UTR) and pGL3-Report pri-miR-199a-1 constructs were
generated using QuikChange Site-Directed Mutagenesis kits (Agilent
Technologies, Inc., Santa Clara, CA, USA). Cells were transfected
using Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific,
Inc.). Cells (5×103) were plated into 96-well plates and
incubated overnight before transfection. Luciferase activities were
measured by a Dual-Luciferase reporter assay system (Promega,
Madison, WI, USA) according to the manufacturer's instructions.
Renilla luciferase vector (Promega) was applied as an
internal control.
Western blotting
Protein extract was obtained using RIPA Lysis Buffer
(Beyotime Institute of Biotechnology, Haimen, China) containing 1%
phenylmethylsulfonyl fluoride (PMSF; Sigma-Aldrich; Merck KGaA).
Equal amounts of samples were loaded and separated by 10% SDS-PAGE
and transferred onto a nitrocellulose (NC) membrane (Sigma-Aldrich;
Merck KGaA). Membranes were blocked with 5% skim milk and then
incubated with anti-TGF-β2 produced in rabbit (1:400; cat. no.
SAB4502956; Sigma-Aldrich; Merck KGaA) and GAPDH produced in rabbit
(1:200; cat. no. G9546; Sigma-Aldrich; Merck KGaA) diluted in
Tris-buffered saline containing 0.1% (v/v) Tween-20 (PBST)
overnight at 4°C. After washing with PBST, the membranes were
incubated with horseradish peroxidase-conjugated goat anti-rabbit
or anti-mouse IgG (cat. no. 65–6120; Thermo Fisher Scientific,
Inc.). The results were visualized by ECL kit (Thermo Fisher
Scientific, Inc.) and exposed to X-ray film (Thermo Fisher
Scientific, Inc.).
Cell viability
Cell viability was measured by Cell Counting Kit-8
(CCK-8) assay purchased from BestBio (Shanghai, China). Briefly,
cells were seeded at a density of 3,500 cells/well in a 96-well
plate. Emodin (20 µM) was added into each well after an overnight
incubation. After 48 h of transfection, 10 µl of diluted CCK-8
solution was added to each well and incubated for 2 h at 37°C. The
absorbance values were read at wavelengths of 450 nm using a
microplate reader (Bio-Rad Laboratories).
Colony formation
For the colony formation assay, ovarian cancer cells
were pre-treated with 20 µM emodin for 24 h. Then, the cells were
seeded in 6-well plates at a density of 300 cells/well. After 2
weeks of incubation, the cells were washed twice with
phosphate-buffered saline (PBS) and fixed in 4%
paraformaldehyde-methanol for 10 min. The colonies were stained
using 0.1% crystal violet (Sigma-Aldrich; Merck KGaA) and the
number of colonies were counted. The colonies were calculated as
the mean number of cells in 10 randomly selected fields using
phase-contrast microscopy (Olympus Corp., Tokyo, Japan)
Human ovarian cancer specimens
Human ovarian cancer specimens were derived from the
patients in our hospital between May 2016 and March 2017. Written
consent has been obtained from each participant. The present study
was approved by the ethics committee in our hospital.
Statistical analysis
All data are shown as the mean ± SD from three
independent experiments. Two-tailed Student's t-test was used to
analyze the differences between samples. Spearman's correlation
coefficient was employed for analysis of the correlation between
the expression levels of miR-199a and TGF-β2 in human ovarian
cancer specimens. Statistical significance was considered as
P<0.05.
Results
Downregulation of TGF-β2 by emodin in
A2780 cells
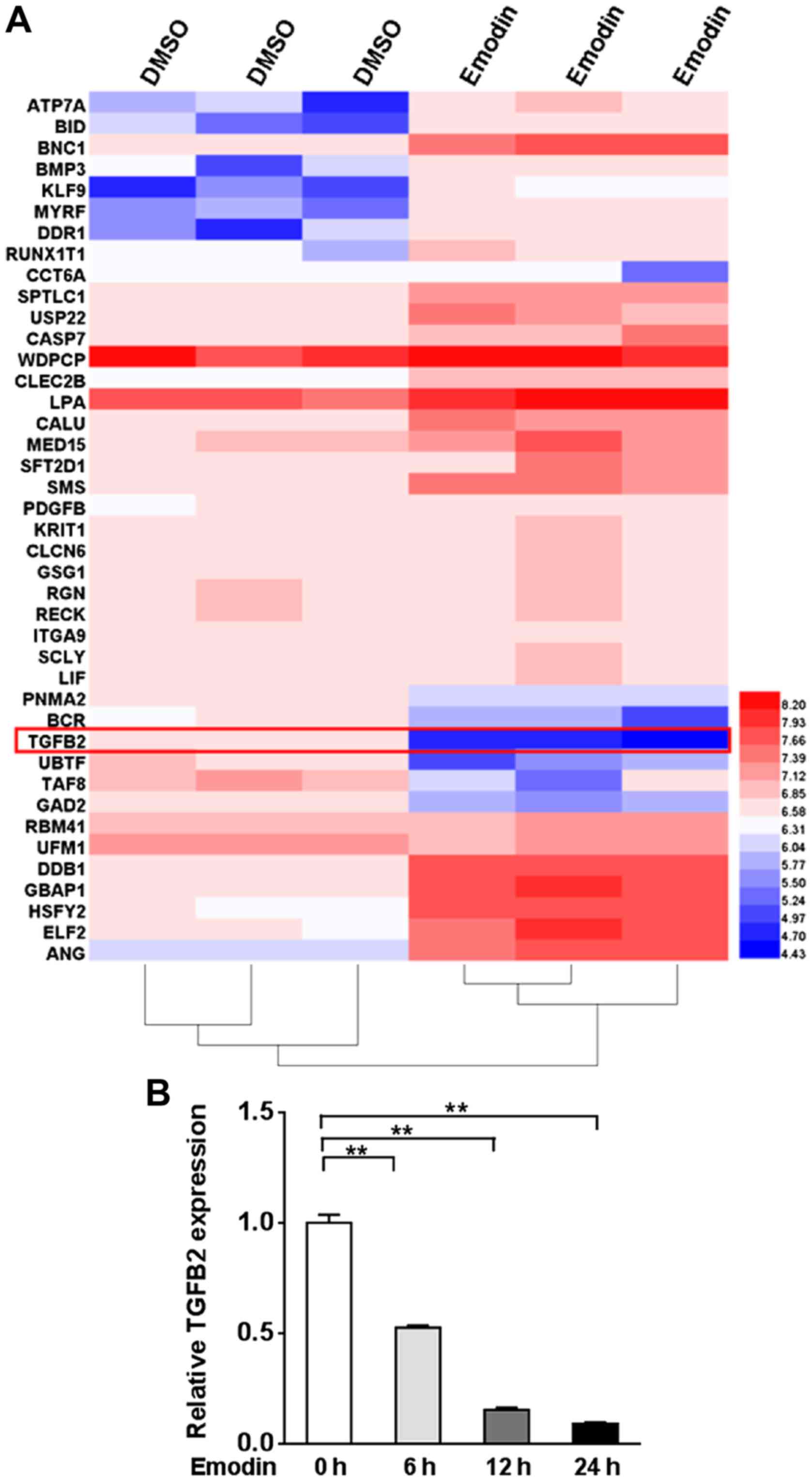
To assess the genes affected by emodin, we performed
microarray analysis in A2780 cells treated with emodin compared to
cells treated with DMSO. Among all of the gene transcripts, TGF-β2
was markedly reduced ~1.5-fold in the emodin-treated A2780 cells
(Fig. 1A). We further examined the
TGF-β2 expression level by qRT-PCR in A2780 cells treated with
emodin for 0, 6, 12 and 24 h. TGF-β2 expression was significantly
lower than that noted in the untreated cells from 6 h, and further
reductions were observed after 12 and 24 h (P<0.01, Fig. 1B).
Elevated expression of miR-199a is
induced by emodin
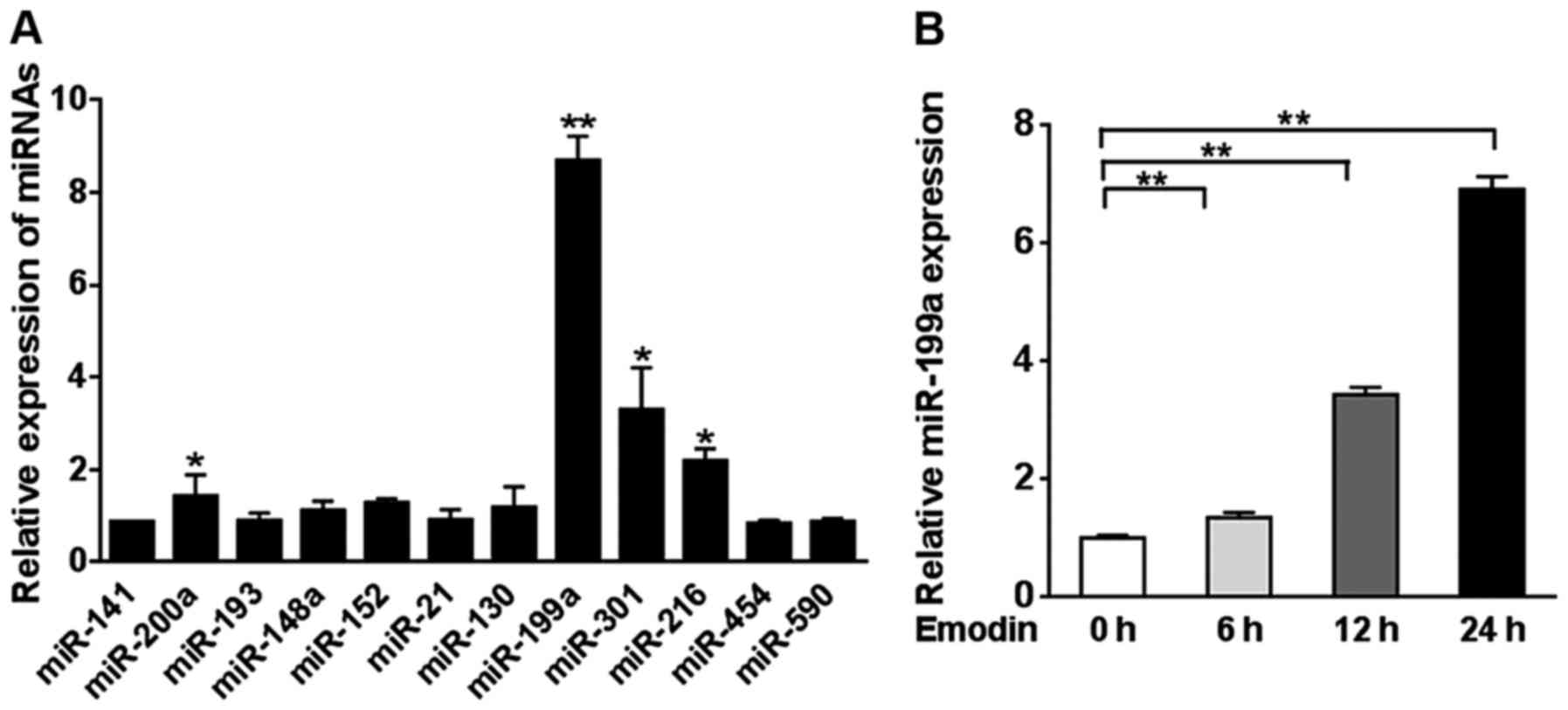
We identified 12 potential miRNAs that may bind to
TGF-β2 using TargetScan 6.0. The expression levels of the 12 miRNAs
were measured in A2780 cells treated with 20 µM emodin or DMSO for
24 h. miR-199a demonstrated a significantly higher expression level
compared to miR-141, miR-200a, miR-193, miR-148a, miR-152, miR-21,
miR-130, miR-301, miR-216, miR-454 and miR-590 (Fig. 2A). Then, we assessed the miR-199a
expression level in A2780 cells treated with emodin for 0, 6, 12
and 24 h. A significant increase in the miR-199a level was observed
from 6 h of emodin treatment (P<0.01, Fig. 2B).
miR-199a directly targets the TGF-β
signaling pathway
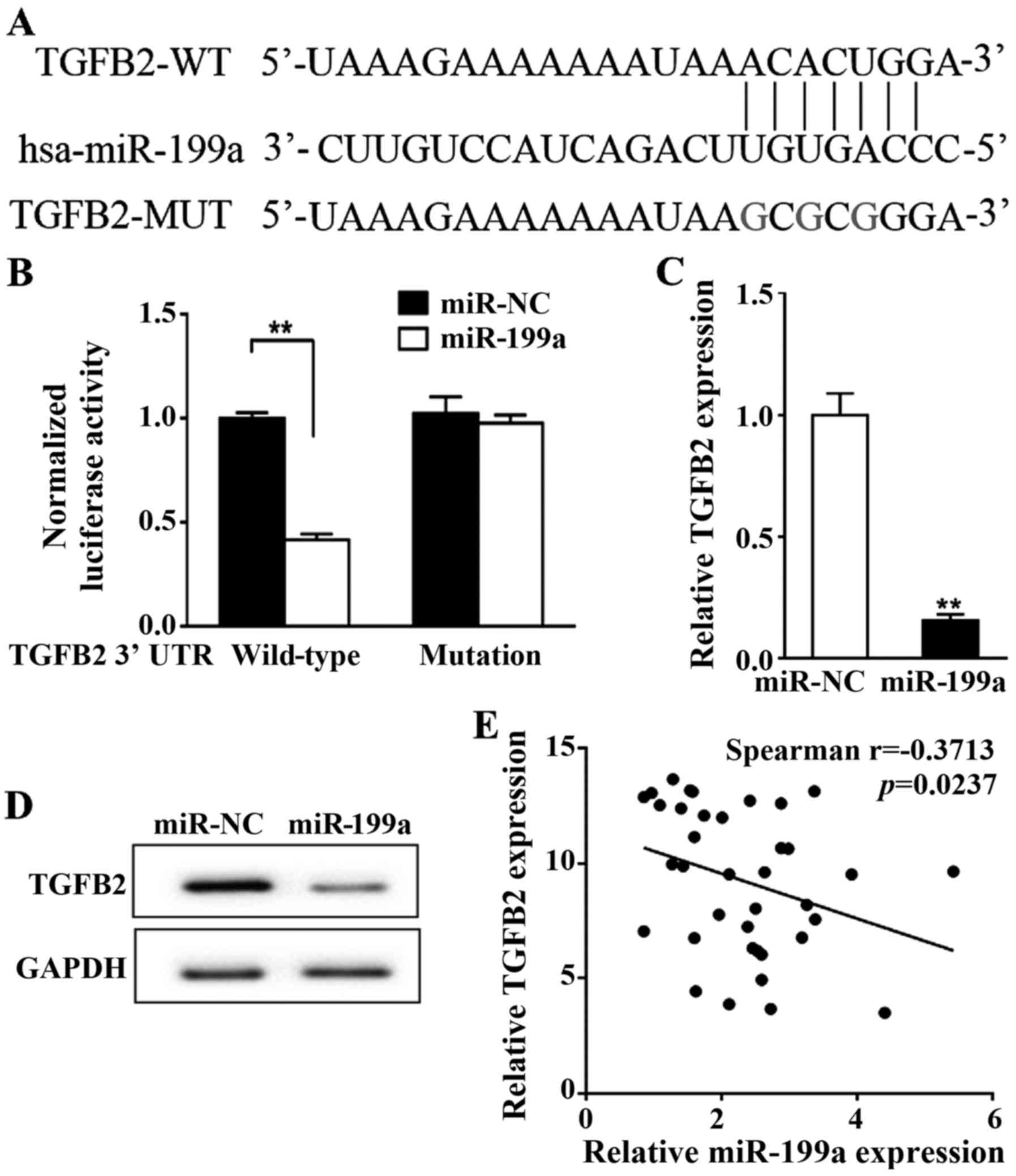
To confirm whether miR-199a directly binds to
TGF-β2, we mutated three nucleotides within the predicted miR-199a
binding site of TGF-β2 3′-UTR (Fig.
3A) and performed a luciferase assay. No change in luciferase
activity was observed in the cells transfected with mutated TGF-β2
compared with the negative control (miR-NC) whereas miR-199a
wild-type significantly reduced the TGF-β2 transcriptional activity
compared to miR-NC (P<0.01, Fig.
3B). The mRNA and protein levels of TGF-β2 were compared
between A2780 cells transfected with miR-199a and cells with
control miR-NC. Both transcriptional and translational levels of
TGF-β2 were significantly inhibited by miR-199a (P<0.01,
Fig. 3C and D). Spearman's
correlation analysis revealed a significant inverse correlation
between the expression of miR-199a and TGF-β2 in human ovarian
cancer specimens (n=37, P<0.05, Fig.
3E).
miR-199a mediates emodin-induced
TGF-β2 downregulation
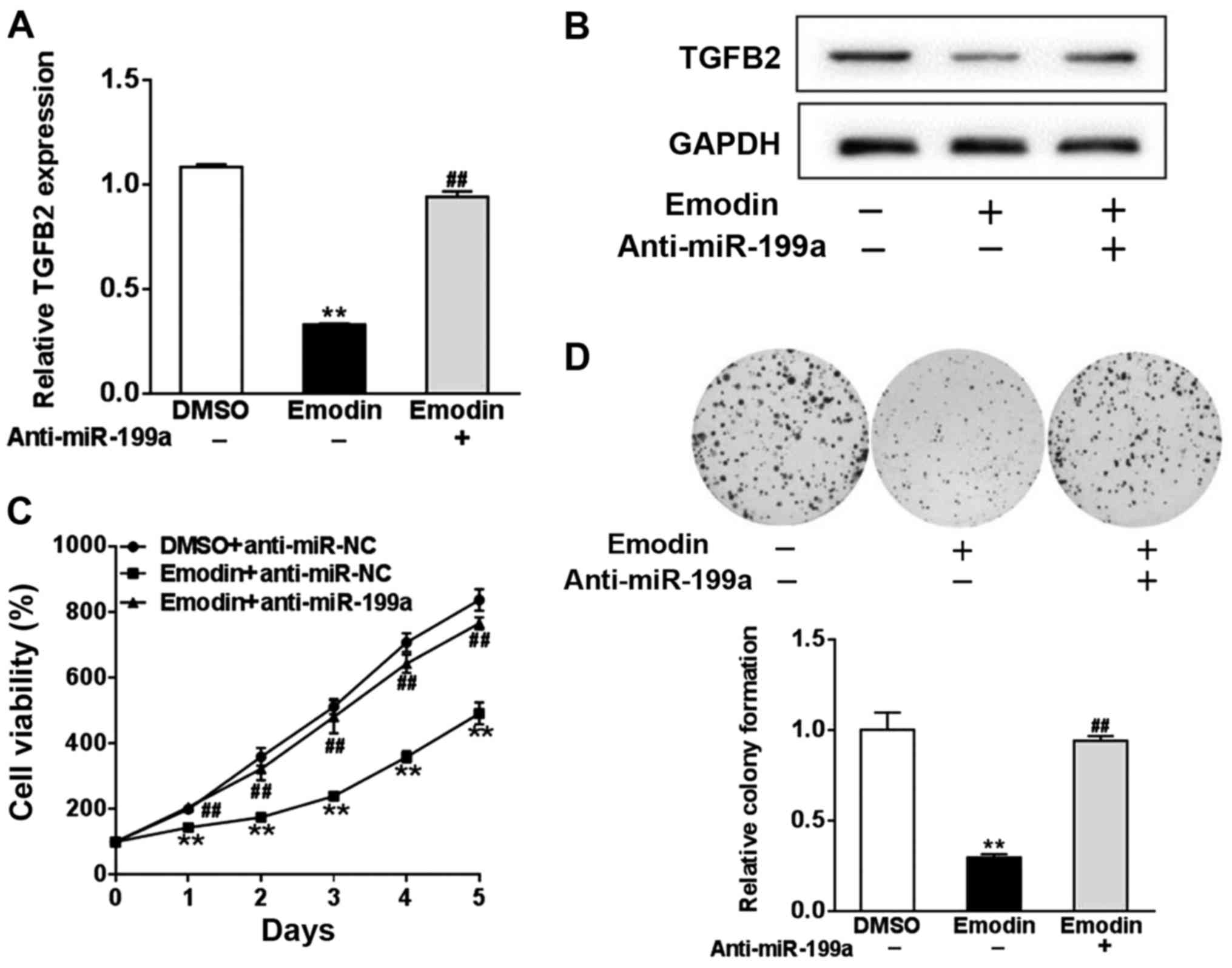
To investigate whether miR-199a was affected by
emodin to regulate TGF-β2, we transfected A2780 cells with miR-199a
inhibitor (anti-miR-199a) or control antisense RNA (anti-miR-NC),
and treated the cells with 20 µM emodin or DMSO. Protein and mRNA
expression of TGF-β2 were significantly reduced by emodin treatment
in comparison with DMSO, whereas anti-miR-199a inhibited the effect
of emodin on TGF-β2 (P<0.01, Fig. 4A
and B). Given the role of TGF-β in cell proliferation, we next
assessed whether miR-199a mediated the effect of TGF-β2 on cell
viability. In comparison with the untreated cells, emodin treatment
significantly reduced the growth of A2780 cells, which was blocked
by anti-miR-199a (P<0.01, Fig.
4C). Colony formation assay revealed that A2780 cells
transfected with anti-miR-199a significantly reversed the reduction
in colony formation induced by emodin (P<0.01, Fig. 4D).
Emodin inhibits TGF-β2 through the
FOXD3/miR-199a signaling axis
Both primary mature miR-199a-1 and miR-199a-2 are
precursors of mature miR-199a. We analyzed their transcripts in
A2780 cells treated with 20 µM emodin or DMSO for 24 h, and only
pri-miR-199a-1 was regulated by emodin (Fig. 5A). Additionally, we revealed that
there is a potential FOXD3 binding site within miR-199a-1 (Fig. 5B). FOXD3 is an important factor in
the suppression of tumorigenesis. Emodin treatment of A2780 cells
significantly increased the mRNA level of FOXD3 compared to that
noted in the untreated cells (P<0.01, Fig. 5C). FOXD3 markedly promoted the
expression level of miR-199a compared with the control, while
emodin further enhanced this promotion (P<0.01, Fig. 5D). As expected, targeted knockdown
of FOXD3 led to a significant decrease in the emodin-induced
increase of miR-199a expression (P<0.01, Fig. 5E). Consistent with these results,
luciferase activity of miR-199a-1 was significantly upregulated by
FOXD3 compared with of a mutated miR-199a-1 reporter (mut
PGL3-miR-199a-1, P<0.01, Fig.
5F).
Discussion
TGF-β functions as an inhibitor of normal cell
proliferation, but the role of TGF-β in cancer progression has been
widely suggested. It is an important growth factor involved in the
regulation of the proliferation of ovarian surface epithelial cells
where 80% of ovarian cancers originate (12) and in the promotion of ovarian cancer
cell proliferation by elevating the expression level of insulin
growth factor 1 receptor (13).
TGF-β enhances the secretion of matrix metalloproteinases that
participate in gynecological cancer metastasis (14,15).
TGF-β contains three isoforms, TGF-β1, TGF-β2 and TGF-β3. An early
clinical study reported that 44, 66 and 66% of malignant ovarian
cancer patients demonstrated overexpression of TGF-β1, TGF-β2 and
TGF-β3, respectively (16). The
dual or triple co-expression of TGF-β isoforms suggests a similar
role for them in ovarian cancer (16). The poor prognosis of patients with
ovarian cancer was reported to be associated with TGF-β activation
(17) suggesting the potential
functions of TGF-β in ovarian cancer development. Our results
demonstrated a reduction in TGF-β2 induced by emodin, providing
supportive evidence of the antitumor property of emodin in
vitro.
In recent years, studies have begun to concentrate
on the correlation between miRNAs and cancer. miRNAs are ~22-nt
RNAs that bind to the 3′-UTR of target mRNAs to negatively regulate
the expression of target genes (18). Thus, overexpression of miRNAs may
function as either oncogenes or tumor-suppressing genes (19). By comparing the expression profiles
of miRNAs between ovarian cancer tissues and normal ovaries, Iorio
et al (20) first discovered
that several miRNAs, including miR-141, miR-200a, miR-200b and
miR-200c, were upregulated, while miR-125b1, miR-140, miR-145 and
miR-199a were downregulated in ovarian cancer. As an important
factor in tumorigenesis, TGF-β signaling has also been shown to be
linked to several miRNAs. miR-181a was identified to promote
TGF-β-induced EMT by inhibiting Smad7, a negative regulator of
TGF-β (17). Additionally, miR-200
was reported to inhibit TGF-β signaling to suppress cell invasion
and upregulate epithelial gene expression (21). The present study demonstrated that
TGF-β2 expression was significantly reduced in A2780 cells treated
with emodin. To identify whether miRNAs have a role in regulating
TGF-β2, we used TargetScan to predict the miRNAs with conserved
binding sites to TGF-β2, and found that miR-199a was a potential
regulator of TGF-β2. This was further evidenced by the influence of
emodin at both the transcriptional and translational levels of
TGF-β2. Thus, regarding the low expression level of miR-199a in
ovarian cancer shown in previous studies, it is possible that
emodin exerts it functions via upregulating miR-199a to repress
TGF-β2 signaling in ovarian cancer cell lines in vitro.
To date, few miRNAs have been identified to be
modulated by emodin including angiogenesis-associated miR-155,
miR-210 and miR-20b (4). The
combination of emodin and curcumin synergistically increased
miR-34a expression conferring an antitumor effect against breast
cancer (22). However, there are
several miRNAs reported in previous studies that are involved in
controlling the TGF-β2 signaling pathway. miR-153 was demonstrated
to inhibit proliferation and invasion of osteosarcoma cells by
repressing TGF-β2 (23). Evidence
showed that TGF-β2 also plays a crucial role in glioma cell
proliferation and metastasis, which can be repressed by miR-141
(24). miR-200a at a high level in
ovarian cancer cells was found to directly target TGF-β2 to inhibit
renal carcinoma cell proliferation, migration and invasion
(25). We also observed statistical
significance in the correlation between miR-200a and TGF-β2, but
miR-191a showed a much higher significant relevance to TGF-β2. This
may be caused by the distinction between different types of tumors.
Furthermore, we revealed that miR-199a was involved in
emodin-induced antitumor effect in vitro, and both
transcriptional and translational levels of TGF-β2 were increased
by miR-199a. In particular, only pri-miR-199a-1 was influenced by
emodin. A potential binding sites of FOXD3 was found within
miR-199a.
We observed that the alteration of miR-199a induced
by emodin was mediated by FOXD3, known as an important factor in
the development of chemoresistance via enhancing EMT (26). Previous studies have demonstrated
the involvement of FOXD3 in tumorigenesis in several types of
cancers, such as human hepatocellular carcinoma (27) colorectal cancer (28) and gastric carcinogenesis (29). No studies have characterized the
function of FOXD3 in ovarian cancer, but its role in the regulation
of miR-137 has been studied. FOXD3 directly controls the
transcriptional activity of miR-137 to suppress the growth and
metastasis of hepatocellular carcinoma (27). Of note, miR-137 expression was
reported to be reduced in ovarian cancer specimens, and its
function as a tumor suppressor was evidenced (30) suggesting that FOXD3 may be also be
dysregulated in ovarian cancer. This needs to be evidenced in
further study. Our results indicated that FOXD3 expression was
significantly increased by emodin treatment in ovarian cancer cells
in vitro. Thus, it is possible that emodin directly enhanced
FOXD3 expression and sequentially activated miR-199a, which
inhibited the expression of TGF-β2 in ovarian cancer cells in
vitro.
Together, the present study demonstrated a possible
signaling pathway activated by emodin in a cisplatin-resistant
ovarian cancer cell line. We identified that emodin exerts
antitumor effect via activating FOXD3 and miR-199a to suppress the
TGF-β2 expression which is at high level in ovarian cancer tumors.
Nevertheless, experiments performed in the present study were
exclusively in vitro, which limited the further clinical
significance of our results. Further studies are needed to expand
our understanding of the antitumor function of emodin, in order to
potentiate its clinical applications.
Acknowledgements
Not applicable.
References
|
1
|
Siegel RL, Miller KD and Jemal A: Cancer
statistics, 2016. CA Cancer J Clin. 66:7–30. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Dong X, Fu J, Yin X, Cao S, Li X, Lin L
and Ni J: Huyiligeqi: Emodin: A review of its pharmacology,
toxicity and pharmacokinetics. Phytother Res. 30:1207–1218. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lin SZ, Xu JB, Ji X, Chen H, Xu HT, Hu P,
Chen L, Guo JQ, Chen MY, Lu D, et al: Emodin inhibits angiogenesis
in pancreatic cancer by regulating the transforming growth
factor-β/drosophila mothers against decapentaplegic pathway and
angiogenesis-associated microRNAs. Mol Med Rep. 12:5865–5871. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Guo J, Xiao B, Liu Q, Gong Z and Le Y:
Suppression of C-myc expression associates with anti-proliferation
of aloe-emodin on gastric cancer cells. Cancer Invest. 26:369–374.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Suboj P, Babykutty S, Srinivas P and
Gopala S: Aloe emodin induces G2/M cell cycle arrest and apoptosis
via activation of caspase-6 in human colon cancer cells.
Pharmacology. 89:91–98. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Xie MJ, Ma YH, Miao L, Wang Y, Wang HZ,
Xing YY, Xi T and Lu YY: Emodin-provoked oxidative stress induces
apoptosis in human colon cancer HCT116 cells through a
p53-mitochondrial apoptotic pathway. Asian Pac J Cancer Prev.
15:5201–5205. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Gao R, Chen R, Cao Y, Wang Y, Song K,
Zhang Y and Yang J: Emodin suppresses TGF-β1-induced
epithelial-mesenchymal transition in alveolar epithelial cells
through Notch signaling pathway. Toxicol Appl Pharmacol. 318:1–7.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hsu HC, Liu LC, Wang HY, Hung CM, Lin YC,
Ho CT and Way TD: Stromal fibroblasts from the interface zone of
triple negative breast carcinomas induced epithelial-mesenchymal
transition and its inhibition by emodin. PLoS One. 12:e01646612017.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Li J, Liu P, Mao H, Wanga A and Zhang X:
Emodin sensitizes paclitaxel-resistant human ovarian cancer cells
to paclitaxel-induced apoptosis in vitro. Oncol Rep.
21:1605–1610. 2009.PubMed/NCBI
|
|
11
|
Ma J, Yang J, Wang C, Zhang N, Dong Y,
Wang C, Wang Y and Lin X: Emodin augments cisplatin cytotoxicity in
platinum-resistant ovarian cancer cells via ROS-dependent MRP1
downregulation. BioMed Res Int. 2014:1076712014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Auersperg N, Wong AS, Choi KC, Kang SK and
Leung PC: Ovarian surface epithelium: Biology, endocrinology, and
pathology. Endocr Rev. 22:255–288. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Alsina-Sanchis E, Figueras A, Lahiguera Á,
Vidal A, Casanovas O, Graupera M, Villanueva A and Viñals F: The
TGFβ pathway stimulates ovarian cancer cell proliferation by
increasing IGF1R levels. Int J Cancer. 139:1894–1903. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lin SW, Lee MT, Ke FC, Lee PP, Huang CJ,
Ip MM, Chen L and Hwang JJ: TGFbeta1 stimulates the secretion of
matrix metalloproteinase 2 (MMP2) and the invasive behavior in
human ovarian cancer cells, which is suppressed by MMP inhibitor
BB3103. Clin Exp Metastasis. 18:493–499. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Peng H, Liu L and Zhao X: Prognostic
significance of matrix metalloproteinase-2 in gynecological cancer:
A systemic review of the literature and meta-analysis. J BUON.
18:202–210. 2013.PubMed/NCBI
|
|
16
|
Bartlett JM, Langdon SP, Scott WN, Love
SB, Miller EP, Katsaros D, Smyth JF and Miller WR: Transforming
growth factor-beta isoform expression in human ovarian tumours. Eur
J Cancer. 33:2397–2403. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Parikh A, Lee C, Joseph P, Marchini S,
Baccarini A, Kolev V, Romualdi C, Fruscio R, Shah H, Wang F, et al:
microRNA-181a has a critical role in ovarian cancer progression
through the regulation of the epithelial-mesenchymal transition.
Nat Commun. 5:29772014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Moss EG: MicroRNAs: Hidden in the genome.
Curr Biol. 12:R138–R140. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lewis BP, Shih IH, Jones-Rhoades MW,
Bartel DP and Burge CB: Prediction of mammalian microRNA targets.
Cell. 115:787–798. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Iorio MV, Visone R, Di Leva G, Donati V,
Petrocca F, Casalini P, Taccioli C, Volinia S, Liu CG, Alder H, et
al: MicroRNA signatures in human ovarian cancer. Cancer Res.
67:8699–8707. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Perdigão-Henriques R, Petrocca F,
Altschuler G, Thomas MP, Le MT, Tan SM, Hide W and Lieberman J:
miR-200 promotes the mesenchymal to epithelial transition by
suppressing multiple members of the Zeb2 and Snail1 transcriptional
repressor complexes. Oncogene. 35:158–172. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Guo J, Li W, Shi H, Xie X, Li L, Tang H,
Wu M, Kong Y, Yang L, Gao J, et al: Synergistic effects of curcumin
with emodin against the proliferation and invasion of breast cancer
cells through upregulation of miR-34a. Mol Cell Biochem.
382:103–111. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Niu G, Li B, Sun L and An C: MicroRNA-153
inhibits osteosarcoma cells proliferation and invasion by targeting
TGF-β2. PLoS One. 10:e01192252015. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Peng T, Zhang S, Li W, Fu S, Luan Y and
Zuo L: MicroRNA-141 inhibits glioma cells growth and metastasis by
targeting TGF-β2. Am J Transl Res. 8:3513–3521. 2016.PubMed/NCBI
|
|
25
|
Lu R, Ji Z, Li X, Qin J, Cui G, Chen J,
Zhai Q, Zhao C, Zhang W and Yu Z: Tumor suppressive microRNA-200a
inhibits renal cell carcinoma development by directly targeting
TGFB2. Tumour Biol. 36:6691–6700. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Zhou Z, Zhang L, Xie B, Wang X, Yang X,
Ding N, Zhang J, Liu Q, Tan G, Feng D, et al: FOXC2 promotes
chemoresistance in nasopharyngeal carcinomas via induction of
epithelial mesenchymal transition. Cancer Lett. 363:137–145. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liu LL, Lu SX, Li M, Li LZ, Fu J, Hu W,
Yang YZ, Luo RZ, Zhang CZ and Yun JP: FoxD3-regulated microRNA-137
suppresses tumour growth and metastasis in human hepatocellular
carcinoma by targeting AKT2. Oncotarget. 5:5113–5124. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
He GY, Hu JL, Zhou L, Zhu XH, Xin SN,
Zhang D, Lu GF, Liao WT, Ding YQ and Liang L: The
FOXD3/miR-214/MED19 axis suppresses tumour growth and metastasis in
human colorectal cancer. Br J Cancer. 115:1367–1378. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Cheng AS, Li MS, Kang W, Cheng VY, Chou
JL, Lau SS, Go MY, Lee CC, Ling TK, Ng EK, et al: Helicobacter
pylori causes epigenetic dysregulation of FOXD3 to
promote gastric carcinogenesis. Gastroenterology. 144:122–133.e9.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Zhang L, Li Z, Gai F and Wang Y:
MicroRNA-137 suppresses tumor growth in epithelial ovarian cancer
in vitro and in vivo. Mol Med Rep. 12:3107–3114.
2015. View Article : Google Scholar : PubMed/NCBI
|