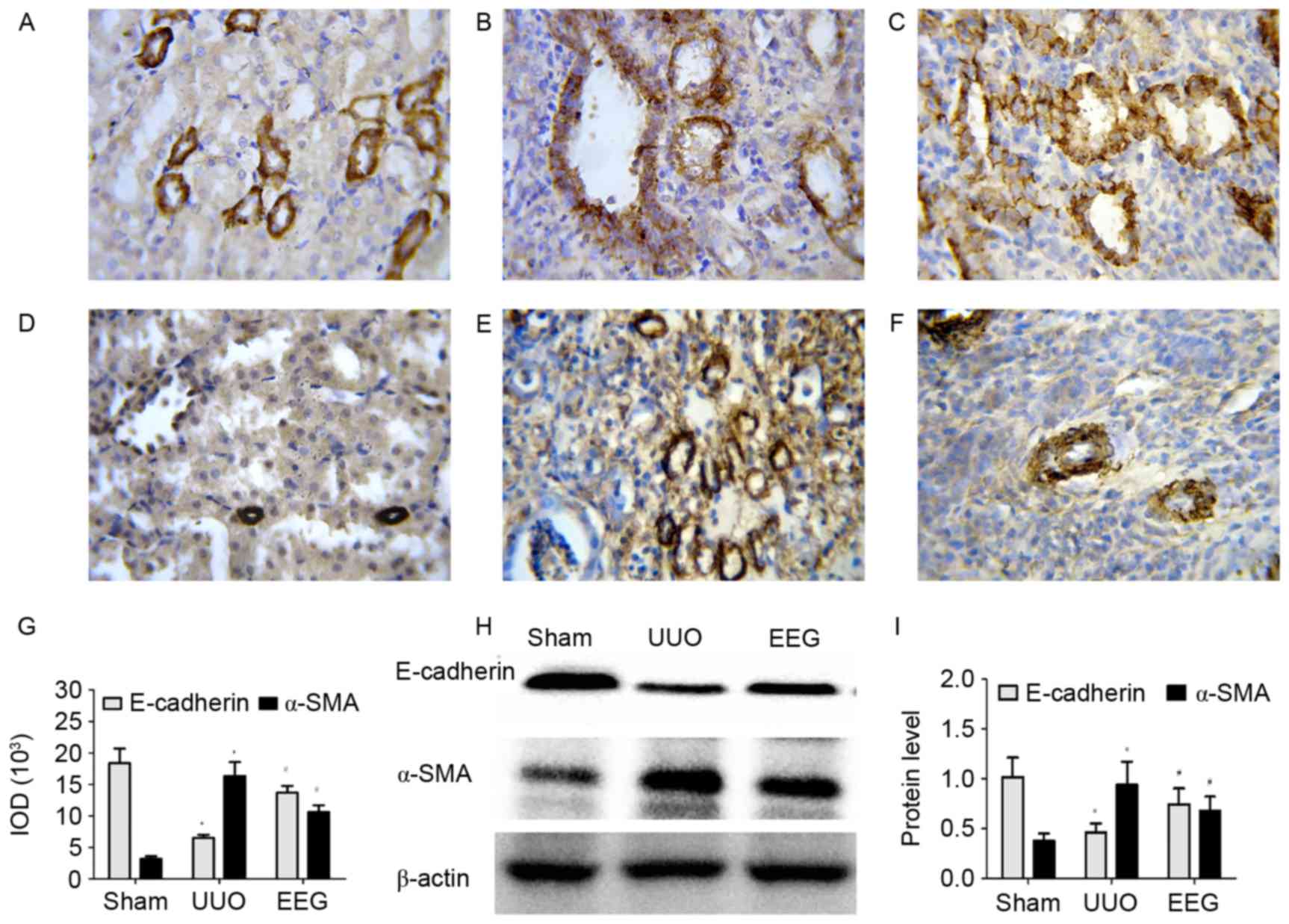
|
1
|
Jha V, Garcia-Garcia G, Iseki K, Li Z,
Naicker S, Plattner B, Saran R, Wang AY and Yang CW: Chronic kidney
disease: Global dimension and perspectives. Lancet. 382:260–272.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Lozano R, Naghavi M, Foreman K, Lim S,
Shibuya K, Aboyans V, Abraham J, Adair T, Aggarwal R, Ahn SY, et
al: Global and regional mortality from 235 causes of death for 20
age groups in 1990 and 2010: A systematic analysis for the global
burden of disease study 2010. Lancet. 380:2095–2128. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Sun YB, Qu X, Caruana G and Li J: The
origin of renal fibroblasts/myofibroblasts and the signals that
trigger fibrosis. Differentiation. 92:102–107. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Duffield JS, Lupher M, Thannickal VJ and
Wynn TA: Host responses in tissue repair and fibrosis. Annu Rev
Pathol. 8:241–276. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Bertinat R, Silva P, Mann E, Li X, Nualart
F and Yáñez AJ: In vivo sodium tungstate treatment prevents
E-cadherin loss induced by diabetic serum in HK-2 cell line. J Cell
Physiol. 230:2437–2446. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Wang JY, Gao YB, Zhang N, Zou DW, Wang P,
Zhu ZY, Li JY, Zhou SN, Wang SC, Wang YY and Yang JK: miR-21
overexpression enhances TGF-β1-induced epithelial-to-mesenchymal
transition by target smad7 and aggravates renal damage in diabetic
nephropathy. Mol Cell Endocrinol. 392:163–172. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Farris AB and Colvin RB: Renal
interstitial fibrosis: Mechanisms and evaluation. Curr Opin Nephrol
Hypertens. 21:289–300. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
López-Hernández FJ and López-Novoa JM:
Role of TGF-β in chronic kidney disease: An integration of tubular,
glomerular and vascular effects. Cell Tissue Res. 347:141–154.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Phanish MK, Winn SK and Dockrell ME:
Connective tissue growth factor-(CTGF, CCN2)-a marker, mediator and
therapeutic target for renal fibrosis. Nephron Exp Nephrol.
114:e83–e92. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cammarata PR, Neelam S and Brooks MM:
Inhibition of hypoxia inducible factor-1 α downregulates the
expression of epithelial to mesenchymal transition early marker
proteins without undermining cell survival in hypoxic lens
epithelial cells. Mol Vis. 21:1024–1035. 2015.PubMed/NCBI
|
|
11
|
Sun Y, Wang H, Liu M, Lin F and Hua J:
Resveratrol abrogates the effects of hypoxia on cell proliferation,
invasion and EMT in osteosarcoma cells through downregulation of
the HIF-1α protein. Mol Med Rep. 11:1975–1981. 2015.PubMed/NCBI
|
|
12
|
Hakroush S, Moeller MJ, Theilig F,
Kaissling B, Sijmonsma TP, Jugold M, Akeson AL, Traykova-Brauch M,
Hosser H, Hähnel B, et al: Effects of increased renal tubular
vascular endothelial growth factor (VEGF) on fibrosis, cyst
formation, and glomerular disease. Am J Pathol. 175:1883–1895.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sis B, Husain S, Chang J, Halloran P and
Osasan S: Decreased renal VEGF-A signaling as a mechanism for
kidney transplant fibrosis and failure. Am J Transplant. 13 Suppl
5:S1102013.
|
|
14
|
Wang YR, Chen Y, Deng L, Cai S, Liu J, Li
W, Du L, Cui G, Xu X, Lu T, et al: Systematic separation and
purification of iridoid glycosides and crocetin derivatives from
Gardenia jasminoides Ellis by high-speed counter-current
chromatography. Phytochem Anal. 26:202–208. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hong YJ and Yang KS: Anti-inflammatory
activities of crocetin derivatives from processed Gardenia
jasminoides. Arch Pharm Res. 36:933–940. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fu YH, Liu B, Liu J, Liu Z, Liang D, Li F,
Li D, Cao Y, Zhang X, Zhang N and Yang Z: Geniposide, from Gardenia
jasminoides Ellis, inhibits the inflammatory response in the
primary mouse macrophages and mouse models. Int Immunopharmacol.
14:792–798. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Koo HJ, Lim KH, Jung HJ and Park EH:
Anti-inflammatory evaluation of gardenia extract, geniposide and
genipin. J Ethnopharmacol. 103:496–500. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chang KH, Chen WL, Wu YR, Lin TH, Wu YC,
Chao CY, Lin JY, Lee LC, Chen YC, Lee-Chen GJ and Chen CM: Aqueous
extract of Gardenia jasminoides targeting oxidative stress to
reduce polyQ aggregation in cell models of spinocerebellar ataxia
3. Neuropharmacology. 81:166–175. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Chen YH, Lan T, Li J, Qiu CH, Wu T, Gou HJ
and Lu MQ: Gardenia jasminoides attenuates hepatocellular injury
and fibrosis in bile duct-ligated rats and human hepatic stellate
cells. World J Gastroenterol. 18:7158–7165. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mizuno S, Matsumoto K and Nakamura T:
Hepatocyte growth factor suppresses interstitial fibrosis in a
mouse model of obstructive nephropathy. Kidney Int. 59:1304–1314.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Mizuguchi Y, Miyajima A, Kosaka T, Asano
T, Asano T and Hayakawa M: Atorvastatin ameliorates renal tissue
damage in unilateral ureteral obstruction. J Urol. 172:2456–2459.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou C, Bai W, Chen Q, Xu Z, Zhu X, Wen A
and Yang X: Protective effect of crocetin against burn-induced
intestinal injury. J Surg Res. 198:99–107. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Song Y, Zhu L and Li M: Antifibrotic
effects of crocetin in scleroderma fibroblasts and in
bleomycin-induced sclerotic mice. Clinics (Sao Paulo).
68:1350–1357. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cai J, Yi FF, Bian ZY, Shen DF, Yang L,
Yan L, Tang QZ, Yang XC and Li H: Crocetin protects against cardiac
hypertrophy by blocking MEK-ERK1/2 signalling pathway. J Cell Mol
Med. 13:909–925. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Park JH, Yoon J, Lee KY and Park B:
Effects of geniposide on hepatocytes undergoing
epithelial-mesenchymal transition in hepatic fibrosis by targeting
TGFβ/Smad and ERK-MAPK signaling pathways. Biochimie. 113:26–34.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Chen P, Chen Y, Wang Y, Cai S, Deng L, Liu
J and Zhang H: Comparative evaluation of hepatoprotective
activities of geniposide, crocins and crocetin by CCl4-induced
liver injury in mice. Biomol Ther (Seoul). 24:156–162. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma T, Huang C, Zong G, Zha D, Meng X, Li J
and Tang W: Hepatoprotective effects of geniposide in a rat model
of nonalcoholic steatohepatitis. J Pharm Pharmacol. 63:587–593.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sanz-Rosa D, de las Heras N, Ortega MR, et
al: Role of CTGF on renal fibrosis in spontaneously hypertensive
rats. Effect of treatment with candesartan. J Hypertens. 23:327.
2005.
|
|
29
|
Haase VH: Pathophysiological consequences
of HIF activation: HIF as a modulator of fibrosis. Ann N Y Acad
Sci. 1177:57–65. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Nørregaard R, Bødker T, Jensen BL,
Stødkilde L, Nielsen S and Frøkiaer J: Increased renal
adrenomedullin expression in rats with ureteral obstruction. Am J
Physiol Regul Integr Comp Physiol. 296:R185–R192. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Harris AL: Hypoxia-a key regulatory factor
in tumour growth. Nat Rev Cancer. 2:38–47. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Higgins DF, Kimura K, Bernhardt WM,
Shrimanker N, Akai Y, Hohenstein B, Saito Y, Johnson RS, Kretzler
M, Cohen CD, et al: Hypoxia promotes fibrogenesis in vivo via HIF-1
stimulation of epithelial-to-mesenchymal transition. J Clin Invest.
117:3810–3820. 2007.PubMed/NCBI
|
|
33
|
Hellwig-Bürgel T, Stiehl DP, Katschinski
DM, Marxsen J, Kreft B and Jelkmann W: VEGF production by primary
human renal proximal tubular cells: Requirement of HIF-1,
PI3-kinase and MAPKK-1 signaling. Cell Physiol Biochem. 15:99–108.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Taoka R, Jinesh GG, Xue W, Safe S and
Kamat AM: CF3DODA-Me induces apoptosis, degrades Sp1, and blocks
the transformation phase of the blebbishield emergency program.
Apoptosis. 22:719–729. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Milanini-Mongiat J, Pouysségur J and Pagès
G: Identification of two Sp1 phosphorylation sites for p42/p44
mitogen-activated protein kinases: Their implication in vascular
endothelial growth factor gene transcription. J Biol Chem.
277:20631–20639. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Wahab NA and Mason RM: A critical look at
growth factors and epithelial-to-mesenchymal transition in the
adult kidney. Interrelationships between growth factors that
regulate EMT in the adult kidney. Nephron Exp Nephrol. 104:129–134.
2006. View Article : Google Scholar
|
|
37
|
Song YR, You SJ, Lee YM, Chin HJ, Chae DW,
Oh YK, Joo KW, Han JS and Na KY: Activation of hypoxia-inducible
factor attenuates renal injury in rat remnant kidney. Nephrol Dial
Transplant. 25:77–85. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Burt LE, Forbes MS, Thornhill BA, Kiley SC
and Chevalier RL: Renal vascular endothelial growth factor in
neonatal obstructive nephropathy. I. Endogenous VEGF. Am J Physiol
Renal Physiol. 292:F158–F167. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Lian YG, Zhou QG, Zhang YJ and Zheng FL:
VEGF ameliorates tubulointerstitial fibrosis in unilateral ureteral
obstruction mice via inhibition of epithelial-mesenchymal
transition. Acta Pharmacol Sin. 32:1513–1521. 2011. View Article : Google Scholar : PubMed/NCBI
|