Introduction
Low-intensity focused ultrasound penetrates the
topical tissues or organs as a low-dose mechanical energy, and is
widely used in the treatment of musculoskeletal injury (1) and uterine involution. While the role of
low-intensity focused ultrasound in postpartum uterine involution
has attracted increasing attention and recognition, its mechanism
in promoting uterine smooth muscle contraction remains to be
elucidated. A randomized controlled study was performed to examine
the clinical application of low-intensity focused ultrasound in
uterine involution by observing its effect on the levels of
endothelin-1 (ET-1), nitrogen monoxide (NO) and oxytocin receptor
(OXTR) in uterine tissues of Sprague-Dawley (SD) rats following
abortion.
Materials and methods
Animals
A total of 60 healthy SD rats (55 females and 5
males), aged ≤3 months (nulliparous females, 180–250 g; males,
250–350 g) were maintained in individually ventilated cages in the
Animal Center of Chongqing Medical University (Chongqing, China)
(room temperature, 18–25°C; humidity, 40–60%; noise, ≤60 dB). The
animal research protocol was approved by the Animal Care and Use
Committee of Chongqing Medical University.
Drugs and reagents
Mifepristone tablets configured in a liquid with a
concentration of 1.66 mg/ml and 0.01 g/ml (approval numbers: Zhunzi
H20033551 and H20073696, respectively) were purchased from Gedian
Humanwell Hubei Pharmaceutical Co., Ltd. (Hubei, China). The ET-1
enzyme-linked immunosorbent assay (ELISA) detection kit was from
Nanjing Jiancheng Technology Co., Ltd. (Nanjing, China). The rabbit
anti-mouse OXTR antibody (ab101617) was purchased from Abcam
(Cambridge, MA, USA) and the streptavidin-horseradish peroxidase
rabbit secondary antibody kit was from KangWei Century Technology
Co., Ltd. (Beijing, China).
Instruments
The ultrasound therapeutic device for involution of
the uterus (CKC100) was purchased from Chongqing Haifu Medical
Technology Co., Ltd., (Chongqing, China), the microplate reader
(352) was from LabSystems Multiskan MS (Helsinki, Finland), the
washer (ACB) was purchased from Thermo LabSystems (Helsinki,
Finland), the micro-speed centrifuge (TG16W) was from Changsha
Pingfan Instrument and Meter Co., Ltd. (Changsha, China), the
water-incubator (GNP-9080) was from Shanghai Wujiu Automation
Equipment, Co., Ltd. (Shanghai, China), the microwave
[G70F23CN2P-BM1 (SO)] used was from Glanze (Guangdong, China), the
incubator (ZDP-A2160) was purchased from Hangzhou, Seoul Equipment
Co., Ltd. (Hangzhou, China) and the microscope (IX73) was from
Olympus Corp. (Tokyo, Japan).
Modeling
A total of 55 female and 5 male SD rats were
randomly selected. The ratio of mated male to female rats at night
was 2:1. Sperm were observed in the vaginal smear under the
microscope and a vaginal plug was observed after 12 h (next day),
which was considered to be the first day of pregnancy (2). Otherwise, after 7 days of separation
between the male and female rats, the same process was repeated
until they became pregnant. After 7 days of pregnancy, 16.6 mg/kg
of mifepristone was administered at 8:00 a.m. and 0.1 mg/kg of
misoprostol was administered at 6:00 p.m. causing the sac to
discharge and vaginal bleeding. The rats were dissected 6 days
post-treatment and no pregnancy tissue was identified in the
uterus. Pathological examination showed a complete abortion. Thus,
a rat model of complete termination (abortion) of early pregnancy
was established (3).
Randomization
The rats undergoing abortion were numbered from 1 to
30 using a computerized random number table, and divided into the
ultrasound irradiation or the sham irradiation group.
Treatment
From the first day of abortion, low-intensity
focused ultrasound irradiation (sound intensity, 2
W/cm2; frequency, 0.8 MHz) was applied to the abdominal
area around the uterine tissue for 30 min daily for 5 consecutive
days in the ultrasound irradiation group. Subsequent to shaving the
abdomen, the anesthetized rats were placed in the supine position
on the surgical table. The abdomen, bladder and the treatment head
were coated with ultrasound coupling agent. Appropriate pressure
was used to regulate the thickness of the bladder. The distance
between the therapeutic transducer and abdominal skin was 1.5–2.5
cm (to ensure that the uterine tissue was within the ultrasonic
focal region). The therapeutic transducer was rotated slowly for 30
min.
The sham irradiation group was similarly treated
without any power output in the therapeutic transducer (sound
intensity, 0 W/cm2; frequency, 0 MHz).
Detection indicators and methods
The measurement of ET-1 and NO was as follows: The
proportion of tissue weight (g): Normal saline (ml) = 1:9. The
tissues were centrifuged at 2,500 r/min for 10 min and the
supernatants were used to measure ET-1 and NO levels by ELISA and
chemical testing.
The OXTR measurement was as follows: Uterine tissue
samples were fixed in 4% paraformaldehyde, embedded in paraffin wax
and sliced at of 4–40 µm. The slices were mounted on slides,
dehydrated using alcohol washes of increasing concentrations (such
as 50, 75, 90, 95 and 100%), and cleared using xylene. OXTR was
detected by immunohistochemistry.
Image analysis
Cells with yellow particles were considered to
exhibit a positive expression of OXTR. A total of 5 high-power
fields were randomly selected in each slice. The number of positive
cells <10% was recorded as 0; 10–30% as 1; 30–50% as 2; 50–70%
as 3; and >70% as 4. Color intensity was also used for scoring
as follows: Pale yellow was recorded as 1, yellow as 2 and brown as
3. The two scores (0–7) were added to calculate the following:
Negative (0, −), weakly positive (1–2, +), moderately positive
(3–4, ++) and strongly positive (5–7, +++).
Statistical analysis
The data were processed using SPSS for Windows
Version 20.0 (IBM Corp., Armonk, NY, USA). Measurement data are
described as mean ± standard deviation, and categorical data as
ratios or rates. When the measurement data showed normality and
homogeneity of variance between the two groups, t-tests were used.
Otherwise, the Wilcoxon rank-sum test was used. Categorical data
were analyzed by Pearson's χ2 test. P<0.05 was
considered to indicate a statistically significant difference.
Results
Pathology
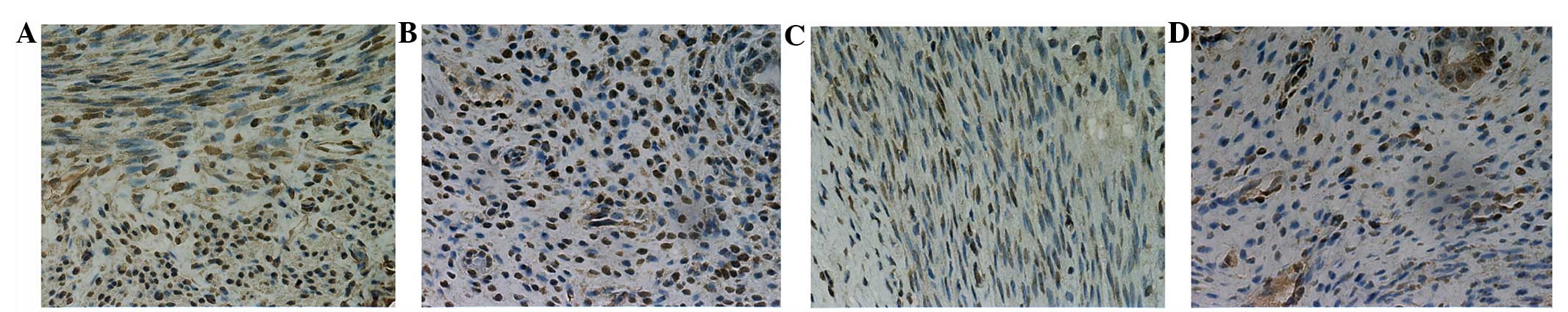
In the ultrasound irradiation group, the uterine
muscle fibers were slender, the inner muscles were arranged in a
ring and the outer muscles were longitudinal (Fig. 1A and B). In the sham irradiation group,
the uterine muscle fibers showed mild hypertrophy and were
haphazardly arranged. Additionally, the myometrium was thicker and
the uterine cavity was larger compared to the ultrasound
irradiation group (Fig. 1C and D).
ET-1 level in the uterine tissue
The ET-1 level in the uterine tissues was
significantly higher in the ultrasound irradiation group compared
to the sham irradiation group (P<0.05) (Table I).
 | Table I.Effect of low-intensity ultrasound on
ET-1 and NO levels of uterine tissue. |
Table I.
Effect of low-intensity ultrasound on
ET-1 and NO levels of uterine tissue.
| Groups | No. | ET-1 (ng/l) | NO (µmol/g prot) |
|---|
| Ultrasound
radiation | 15 |
98.83±27.72 |
1.614±0.849 |
| Sham radiation | 15 |
69.47±24.92 |
2.231±1.735 |
| t/Z |
| t=3.051 | Z=−0.933 |
| P-values |
| P=0.005 | P=0.367 |
NO level in the uterine tissue
The NO level in the uterine tissues was lower in the
ultrasound irradiation group compared to the sham irradiation group
(P>0.05) (Table I).
OXTR expression in the myometrium
Positive OXTR expression was observed in the
myometrium of the two groups. However, 80% uterine tissues in the
ultrasound irradiation group showed strong positive expression, in
contrast to only 40% in the sham irradiation group. The difference
was statistically significant (P<0.05) (Table II) (Fig. 2A
and C).
 | Table II.OXTR expression in the myometrium in
the two groups. |
Table II.
OXTR expression in the myometrium in
the two groups.
| Group | No. | (−) | (+) | (++) | (+++) | (>++%) |
|---|
| Ultrasound
radiation | 15 | 0 | 0 | 3 | 12 | 80.00 |
| Sham radiation | 15 | 0 | 2 | 7 | 6 | 40.00 |
| Total | 30 | 0 | 2 | 10 | 18 |
|
OXTR expression in the
endometrium
Positive OXTR expression was observed in the
endometrium of the two groups. However, 86.7% uterine tissues in
the ultrasound irradiation group showed strong positive expression,
in contrast to only 40% in the sham irradiation group. The
difference was statistically significant (P<0.05) (Table III) (Fig. 2B
and D).
 | Table III.OXTR expression in the endometrium of
the uterus in the two groups. |
Table III.
OXTR expression in the endometrium of
the uterus in the two groups.
| Group | No. | (−) | (+) | (++) | (+++) | (>++%) |
|---|
| Ultrasound
radiation | 15 | 0 | 0 | 2 | 13 | 86.70 |
| Sham radiation | 15 | 0 | 2 | 7 | 6 | 40.00 |
| Total | 30 | 0 | 2 | 9 | 18 |
|
Discussion
Incomplete uterine involution frequently occurs
postpartum, which seriously affects the physical and mental health
of women. Its incidence has increased with the increase in cesarean
section deliveries (4). Slow decline
of uterine fundus prevents the uterus from returning to a normal
size in puerperium, causes uterine displacement, increased lochia
amount, duration, and even hemorrhage or infections in puerperium.
Uterine smooth muscle contraction is necessary for uterine
involution and 70–80% of postpartum hemorrhages are caused by
uterine inertia (5).
ET-1 and NO are involved in the regulation of
uterine contraction and relaxation (6). ET is the strongest and most persistent
vasoconstrictor peptide known, and has four isoenzyme isomers:
ET-1, ET-2, ET-3 and vasoactive intestinal peptide contraction.
Uterine tissue mainly generates ET-1 (7), which promotes muscle contraction by
Gq-phospholipase C-inositol 1,4,5-trisphosphate signal transduction
pathways, protein kinase Cθ (PKCθ) and phospholipase A2 to activate
the synthesis of arachidonic acid, PKC and sphingosine kinase,
which in turn activate RhoA/Rho kinase (7–9). NO is an
endothelium-derived relaxing factor, which activates the guanylyl
cyclase (GC) to inhibit spontaneous uterine smooth muscle
contraction (10,11). GC is widely present in smooth muscle
cells, and its activation increases the cyclic guanosine
monophosphate (cGMP) level, which facilitates smooth muscle
contraction and relaxation.
Oxytocin (OT) is a neuropeptide that is mainly
produced in the supraoptic nuclei and paraventricular nuclei of the
hypothalamus (12), and is released
into the central nervous system and peripheral blood (13). It can stimulate uterine muscle
contraction, promote milk secretion, and has a role in the
functions of the breast, ovary, brain and uterus. OT exerts its
biological effects by binding to the OXTR (14), which enhances uterine muscle
contraction in rats, humans, rabbits and cows (15). Prolonged OT stimulation will
desensitize OXTR in the uterine smooth muscle (16,17).
Therefore, increasing the expression of OXTR in postpartum uterine
smooth muscle cells may be effective in managing incomplete uterine
involution.
In 1978, Ter Haar et al (18) discovered that ultrasound (3 MHz, 2
W/cm2) can promote the contraction of uterine smooth
muscle in pregnant mice, and enhance the frequency and amplitude of
spontaneous uterine contraction. Zhang et al and Rui-Hong
et al (19–21) reported that ultrasonic irradiation can
promote the contraction of uterine smooth muscle in rats and
humans, which were confirmed by in vivo studies. After
ultrasonic (frequency, 0.8 MHz; sound intensity, 2
W/cm2) irradiation for 10 min, the frequency, amplitude,
tension and contraction of the isolated uterus in rats increased
significantly. These parameters were considered to be the most
appropriate radiation intensity.
The present study used low-intensity focused
ultrasound (frequency=0.8 MHz, sound intensity=2 W/cm2)
to irradiate the uterus following medical abortion in rats. In the
ultrasound irradiation group, uterine muscle fibers were slender,
inner muscles were arranged in a ring and the outer muscles were
longitudinal. In the sham irradiation group, uterine muscle fibers
showed mild hypertrophy and were haphazardly arranged. In addition,
the myometrium was thicker and the uterine cavity was larger
compared to the ultrasound irradiation group. These results
suggested that low-intensity ultrasound can promote contraction of
uterine muscle, thereby promoting uterine involution. Low-intensity
ultrasound affected the levels of NO and ET-1 in aborted uterine
tissue of rats. ET-1 levels in the uterine tissues were
significantly higher in the ultrasound irradiation group compared
to the sham irradiation group (P<0.05). However, NO levels in
the uterine tissues were slightly less in the ultrasound compared
to the sham irradiation group, but there was no statistical
significance (P>0.05). Under normal circumstances, ET and NO
exhibit negative feedback regulation and promote their synthesis by
endothelin receptors, while NO uses cGMP to inhibit the production
of ET. The two are synthesized and released in dynamic equilibrium,
thereby maintaining a relatively stable ratio. Any imbalance
results in a pathological state. These results suggest that
low-intensity ultrasound altered the balance of ET-1 and NO to
reach a new homeostasis, which was more suitable for the
contraction of the uterus.
Zhang et al (21) showed that ultrasound can significantly
increase OT-induced contraction of uterine smooth muscle. The
frequency and amplitude of contraction, and uterine activity were
significantly enhanced. Therefore, whether ultrasound promotes
uterine involution was investigated by increasing the expression of
OXTR in the uterine tissue. OT combined with OXTR, which is
expressed on myometrium cells, causes direct contraction of uterine
smooth muscle. OXTR can stimulate epithelial cells to release
prostaglandins, and promote contraction by a paracrine mechanism
(22). Therefore, the present study
analyzed OXTR expression in the endometrium and myometrium of the 2
groups, and identified that the strong positive expression of OXTR
was significantly higher in the ultrasound irradiation group
compared to the sham irradiation group (P<0.05) thereby
suggesting that increased expression of OXTR is an important
mechanism by which low-intensity ultrasound promotes uterine
involution.
Low-intensity focused ultrasound (sound intensity, 2
W/cm2; frequency, 0.8 MHz) could promote uterine
involution in rats that had undergone complete abortion, and thus
promote the recovery of tissue morphology. The underlying
mechanisms include an increase in ET-1 levels, modifying the
balance of ET-1 and NO, and the expression of OXTR in the uterine
myometrium and endometrium.
Acknowledgements
The present study was supported by the National
Natural Science Fund by the Chinese National Science Foundation
(grant nos. 81127901 and 31000435), the National Basic Research
Program of China (grant nos. 2011CB707900 and 2012CB722402), the
National Twelfth Five-year Science and Technology Support Program
(grant no. 2011BAI141301) and Science and Technology Project
affiliated to the Education Department of Chongqing Municipality
(grant nos. KJ110325 and KJ130329). The authors thank Mrs. Chongyan
Li, Mr. Jie Xu, Mrs. Chengdan Ran and Mrs. Cuiping Wang for their
contribution to this study.
References
|
1
|
Menetrey J, Kasemkijwattana C, Fu FH,
Moreland MS and Huard J: Suturing versus immobilization of a muscle
laceration. A morphological and functional study in a mouse model.
Am J Sports Med. 27:222–229. 1999.PubMed/NCBI
|
|
2
|
Li Y, Shao L, Li S, Deng Z and Zhu C:
Experimental observation on the Pessaries for the identification of
pregnant rat model. Chinese Journal of Laboratory Animal Science.
1:62002.
|
|
3
|
You Z, Zeng J, You H, Zha Y, Deng H and Li
D: Effect of gynecology hemostasis acesodyne granule on the
expression of estrogen-progestin and its receptor in rats model of
medical abortion. Chinese Journal of Information on Traditional
Chinese Medicine. 17:19–21. 2010.
|
|
4
|
Wu Z, Zeng Y and Zhu L: Analysis of 238
cases postpartum subinvolution of uterus. The Modern Medicine.
11:41–42. 2011.
|
|
5
|
Heinemann U: Basic mechanisms of partial
epilepsies. Curr Opin Neurol. 17:155–159. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang J, Yang T and Cui X: Effect of Sheng
Hua Tang to the level of NO and ET-1 in abortion rat model. Journal
of Liaoning Traditional Chinese Medicine. 11:2002–2003. 2009.
|
|
7
|
Dallot E, Pouchelet M, Gouhier N, Cabrol
D, Ferré F and Breuiller-Fouché M: Contraction of cultured human
uterine smooth muscle cells after stimulation with endothelin-1.
Biol Reprod. 68:937–942. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Di Liberto G, Dallot E, Eude-Le Parco I,
Cabrol D, Ferré F and Breuiller-Fouché M: A critical role for PKC
zeta in endothelin-1-induced uterine contractions at the end of
pregnancy. Am J Physiol Cell Physiol. 285:C599–C607. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tanfin Z, Leiber D, Robin P, Oyeniran C
and Breuiller-Fouché M: Endothelin-1: Physiological and
pathological roles in myometrium. Int J Biochem Cell Biol.
43:299–302. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Buhimschi I, Yallampalli C, Dong Y-L and
Garfield RE: Involvement of a nitric oxide-cyclic guanosine
monophosphate pathway in control of human uterine contractility
during pregnancy. Am J Obstet Gynecol. 172:1577–1584. 1995.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Anggård EE: Endogenous and exogenous
nitrates. Acta Anaesthesiol Scand Suppl. 97:7–10. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Robinson KJ, Hazon N, Lonergan M and
Pomeroy PP: Validation of an enzyme-linked immunoassay (ELISA) for
plasma oxytocin in a novel mammal species reveals potential errors
induced by sampling procedure. J Neurosci Methods. 226:73–79. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Neumann ID and Landgraf R: Balance of
brain oxytocin and vasopressin: Implications for anxiety,
depression, and social behaviors. Trends Neurosci. 35:649–659.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Liu Q, Qian DM, Liu QL and Gao H: Research
progress of oxytocin and its analogues. Chin J Pharm Anal.
31:609–613. 2011.
|
|
15
|
Murata T, Narita K and Ichimaru T: Rat
uterine oxytocin receptor and estrogen receptor α and β mRNA levels
are regulated by estrogen through multiple estrogen receptors. J
Reprod Dev. 60:55–61. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Jia R and Liu X: Effects of oxytocin and
prostaglandins on oxytocin receptor expression of human myometrial
smooth muscle cell. Chin J Biochem Pharmaceuties. 30:379–382.
2009.
|
|
17
|
Magalhaes JK, Carvalho JC, Parkes RK,
Kingdom J, Li Y and Balki M: Oxytocin pretreatment decreases
oxytocin-induced myometrial contractions in pregnant rats in a
concentration-dependent but not time-dependent manner. Reprod Sci.
16:501–508. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Haar Ter G, Dyson M and Talbert D:
Ultrasonically induced contractions in mouse uterine smooth muscle
in vivo. Ultrasonics. 16:275–276. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Zhang Y, Sun JC, Chang SF, Wang ZB, Wang
GZ and Liu YM: Dose effect of low intensity ultrasound on uterine
smooth muscle in rats. Acta Academiae Medicinae Militaris Tertiae.
16:1566–1568. 2007.
|
|
20
|
Rui-Hong L, Jiangchuan S and Shufang C:
The effect of low intensity ultrasound on the contraction of
uterine smooth muscle in rats in vivo. Journal of Chongqing Medical
University. 6:162008.
|
|
21
|
Zhang Y, Sun JC, Chang SF and Wang ZB: A
comparative study: Low intensity ultrasound and oxytocin on the
contraction of uterine smooth muscle in rats. Chinese Journal of
Medical Imaging Technology. 2:22008.
|
|
22
|
Romero R, Sibai BM, Sanchez-Ramos L,
Valenzuela GJ, Veille JC, Tabor B, Perry KG, Varner M, Goodwin TM,
Lane R, et al: An oxytocin receptor antagonist (atosiban) in the
treatment of preterm labor: A randomized, double-blind,
placebo-controlled trial with tocolytic rescue. Am J Obstet
Gynecol. 182:1173–1183. 2000. View Article : Google Scholar : PubMed/NCBI
|