Introduction
Hepatic encephalopathy (HE) is a severe complication
among patients with cirrhosis, as it is associated with poor
survival outcomes and a reduced quality of life (1). The diagnosis of overt HE (OHE) is
relatively simple, as patients exhibit reduced consciousness and
neurological deficits, and treatment can typically be started at
the onset of the symptoms. However, minimal HE (MHE) is much more
difficult to diagnose, as patients with MHE do not exhibit overt
neurological deficits, and sensitive psychometric tests are
required to diagnose these patients. The recent guidelines
regarding the clinical management of MHE encourage the use of these
tests (2), although they require large
amounts of time and specialized equipment. For example, the
diagnosis of MHE in Japan is typically performed using a personal
computer with a specific touch panel (3). Other studies have reported that magnetic
resonance spectroscopy (4),
neuroelectrical latency (5) and
critical flicker frequency (6) can be
used to diagnose MHE, although these techniques are also limited by
their requirement for specialized equipment. Serum levels of
nitrotyrosine (7) and inflammatory
cytokines (8) may also be useful for
diagnosing MHE, although these techniques are not commercially
available in Japan. However, it is critical to diagnose MHE in
patients with cirrhosis, as MHE is a risk factor for OHE (9), reduces the 5-year survival rate for
cirrhosis (10), impairs the ability
of an individual to drive a vehicle (11), increases the incidence of motor vehicle
accidents (12,13) and increases the risk of falls (14). Furthermore, MHE is a highly prevalent
cognitive disorder that severely affects the health-related quality
of life for individuals (15).
Additionally, MHE was diagnosed in 30.1% of cirrhotic patients in
Japan (16). It shows that MHE is not
a rare condition in liver cirrhosis. Therefore, a simple and
effective screening method is urgently needed to identify patients
with MHE.
Our previous study reported that sleep disturbances
in patients with cirrhosis are mainly caused by restless legs
syndrome (RLS) (17), and that
Child-Pugh scores (CPS) are associated with cirrhosis-related
symptom scores (CSS), which are calculated using questionnaire
items that were developed in our previous study (18). Furthermore, sleep disturbances and RLS
could be accurately diagnosed using this questionnaire and the
sleep disturbances in patients without OHE improved following
consuming a branched-chain amino acid (BCAA)-enriched snack
(18). Moreover, our previous study
reported that patients who underwent living donor liver
transplantation also experienced sleep disturbances, which were
caused by RLS, sleep apnea syndrome and MHE (19). Among these patients, the Japanese
36-item short-form health survey (SF-36) scores were associated
with MHE (20), which was diagnosed
using a two-dimensional operation system (3). Therefore, the present study aimed to
evaluate the associations between MHE, clinical parameters and
questionnaire item scores for sleep disturbances, RLS and CSS.
Patients and methods
Patients
The present study evaluated 91 patients (41 women
and 50 men) with cirrhosis who were being evaluated for liver
transplantation at Nagasaki University Hospital (Nagasaki, Japan)
between July 2011 and May 2014. All the patients had liver
cirrhosis, which was diagnosed using laboratory data and imaging
findings. None of the patients had OHE at their initial evaluation,
and any OHE was subsequently diagnosed using clinical findings.
Diagnosis of MHE
The neuropsychological test (NPT) system is designed
to assess psychomotor, attention, memory and special functions in
order to diagnose MHE. This system consists of eight tests: Number
connection tests A and B, a figure position test, a digit symbol
test, a block design test and reaction time tests A-C. The system
was simplified to accommodate two-dimensional manipulation using a
computer and all tests can be completed in ~20 min, which includes
the time that is required for practice and reading the operation
guide. The software for this system was developed by Otsuka
Pharmaceutical Co., Ltd. (Tokyo, Japan), Kokuyo Co., Ltd. (Osaka,
Japan) and ISB Co., Ltd. (Tokyo, Japan).
Clinical and laboratory
parameters
All the patients underwent anthropometric
measurements to calculate body mass index (kg/m2).
Laboratory testing was also performed to obtain data regarding the
following parameters: White blood cells, red blood cells, platelets
(PLT), prothrombin time, blood urea nitrogen, creatinine, aspartate
aminotransferase, alanine aminotransferase, γ-glutamyl
transpeptidase, alkaline phosphatase, total bilirubin, total
protein, albumin (ALB), high-density lipoprotein, low-density
lipoprotein, cholinesterase (ChE), triglycerides, fasting blood
glucose and ammonia. Each patient was questioned regarding whether
they had a history of OHE, variceal bleeding, ascites or oral
medication (including BCAA supplements). In addition, the Model for
End-stage Liver Disease (MELD) score and CPS were calculated at
entry for each patient.
Questionnaires
The CSS questionnaire contained items regarding
cirrhotic symptoms, which included hand tremors, appetite loss,
foot muscle cramps, fatigue, decreased strength, anxiety, abdominal
fullness, abdominal pain, a feeling of low energy, difficulty
falling asleep, sleeping poorly and being sleepy during the
daytime. An ‘impact factor’ for each item was calculated, which was
defined as the product of the frequency of the item and the mean
importance that the patients attributed to the item. The impact
factor for each item ranged from 0 to 3, and the CSS was calculated
as the sum of the impact factors (18). The Epworth Sleepiness Scale (ESS)
(21) was used to evaluate daytime
hypersomnolence; ESS scores range from 0 to 24, and a score of ≥10
indicates significant daytime hypersomnolence. Sleep quality was
evaluated using the Japanese version of the Pittsburgh Sleep
Quality Index (PSQI) (22,23). Responses to the PSQI questionnaire were
used to generate seven components, which are scored from 0 (normal)
to 3 (extremely poor). Health-related quality of life was evaluated
using the Japanese SF-36 [version 2; Medical Outcomes Trust
(Hanover, NH, USA), Health Lab (Hanover, NH, USA), QualityMetric
(Lincoln, RI, USA), and Shunichi Fukuhara (iHope International;
Kyoto, Japan)]. This tool contains 1 item that evaluates the
perceived change in health status, and the remaining 35 items are
used to generate eight subscales of 0–100 that evaluate physical
functioning, role limitations due to poor physical health, bodily
pain, general health perception, vitality, social functioning, role
limitations due to poor emotional health, and role limitations due
to poor mental health. All the patients were evaluated for the
presence of RLS using a written survey that was developed by the
International Restless Legs Syndrome Research Group in 2003.
Patients were diagnosed with RLS if they fulfilled all four
criteria and exhibited symptoms of RLS that occurred at least twice
per week.
Statistical analysis
All the data were analyzed using Stat Flex software
(version 6.0; Artech Co., Ltd., Osaka, Japan) and P<0.05 was
considered to indicate a statistically significant difference.
Differences in the laboratory data were analyzed using the t-test
or χ2 test, as appropriate. A multivariate analysis was
performed using binary logistic regression analysis to calculate
the odds ratios for development of MHE. Receiver operating
characteristic analysis was used to evaluate the association
between MHE and CSS with a history of OHE.
Results
Patients and the NPT
All 91 patients completed the eight tests in the
NPT, and abnormal scores were observed in the number connection
test A (21 patients), number connection test B (34 patients),
figure position test (79 patients), digit symbol test (60
patients), block design test (29 patients), reaction time test A
(65 patients), reaction time test B (62 patients) and reaction time
test C (66 patients). In the present study, MHE was defined based
on ≥2 abnormal scores in number connection test A, number
connection test B, digit symbol test and block design, which
identified MHE in 42 of the 91 patients. The clinical
characteristics of the normal and MHE groups are shown in Table I. Compared to the normal group, the MHE
group exhibited significantly higher values for MELD, CSS, PSQI,
RLS, CPS, OHE history, BCAA supplementation, ammonia and
prothrombin time-international normalized ratio. Furthermore,
compared to the normal group, the MHE group exhibited significantly
lower levels of ALB, total protein, ChE, PLT, high-density
lipoprotein and low-density lipoprotein. Among the SF-36 items, the
MHE group exhibited significantly lower bodily pain, emotional
health and mental health scores when compared to the normal group.
The results of the univariate analyses of the associations between
MHE and the characteristics of the patient and scores are listed in
Table II. Significant associations
were observed between MHE and gender (male), CSS, PSQI, RLS, bodily
pain, emotional health, mental health, OHE history, ALB levels, PLT
counts and high-density lipoprotein levels. However, only a history
of OHE was independently associated with MHE in the multivariate
analysis.
 | Table I.Clinical characteristics of MHE in
patients with cirrhosis. |
Table I.
Clinical characteristics of MHE in
patients with cirrhosis.
| Characteristics | Normal (n=49) | MHE (n=42) | P-value |
|---|
| Disease, B:C:N | 7:16:26 | 5:12:25 | NS |
| Age, years | 60±14 | 59±9 | NS |
| Gender, f:m | 27:22 | 14:28 | 0.056 |
| BMI,
kg/m2 | 23.9±4.0 | 24.7±4.9 | NS |
| CSS | 7.2±4.7 | 10.0±4.7 | 0.007 |
| ESS | 5.8±3.9 |
5.6±3.5 | NS |
| PSQI | 5.9±3.7 |
7.8±4.1 | 0.03 |
| RLS, no:yes | 43:6 | 25:13 | 0.01 |
| PFN | 36.6±15.9 |
33.2±15.1 | NS |
| RPN | 77.1±27.0 |
28.7±16.2 | NS |
| BPN | 50.4±11.4 |
44.4±11.1 | 0.01 |
| GHN | 40.3±8.4 | 38.6±8.6 | NS |
| VTN | 44.9±11.8 |
41.1±13.3 | NS |
| SFN | 44.3±12.7 |
39.6±15.4 | NS |
| REN | 41.0±13.9 |
34.2±15.3 | 0.03 |
| MHN | 47.7±10.7 | 42.0±8.7 | 0.007 |
| PCS | 39.0±13.3 |
36.6±12.9 | NS |
| MCS | 50.3±9.6 | 48.9±9.5 | NS |
| RCS | 53.5±72.6 |
35.5±14.8 | NS |
| MELD | 10.0±4.3 | 13.2±4.3 | 0.0007 |
| CPS | 6.2±2.6 |
8.5±2.8 | 0.0001 |
| OHE, no:yes | 47:2 | 25:17 | 0.0001 |
| BCAA, no:yes | 30:19 | 15:27 | 0.02 |
| ALB | 3.7±0.7 |
3.1±0.6 | 0.0001 |
| TP | 7.26±0.77 |
6.89±0.87 | 0.03 |
| ALT | 44.3±43.7 | 45.8±38 | NS |
| ChE | 215.8±104.3 | 130.3±93 | 0.0001 |
| Cr | 0.78±0.28 | 1.15±1.4 | 0.08 |
| PLT | 13.4±7.7 |
8.6±6.4 | 0.002 |
| PT-INR | 1.21±0.36 |
1.37±0.35 | 0.03 |
| TB | 1.8±2.5 |
2.5±2.3 | NS |
| HDL | 47.4±21.6 |
36.1±14.9 | 0.006 |
| LDL | 82.9±32.2 |
66.5±39.3 | 0.04 |
| NH3 | 46.5±34.4 |
82.8±45.2 | 0.0001 |
 | Table II.Factors associated with minimal
hepatic encephalopathy among patients with cirrhosis. |
Table II.
Factors associated with minimal
hepatic encephalopathy among patients with cirrhosis.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Factors | P-value | OR | 95% CI | P-value | OR | 95% CI |
|---|
| Gender | 0.03 | 2.46 | 1.045–5.763 | NS |
|
|
| CSS | 0.01 | 1.14 | 1.029–1.251 | NS |
|
|
| PSQI | 0.03 | 1.13 | 1.133–1.006 | NS |
|
|
| RLS | 0.01 | 3.73 |
1.258–11.038 | NS |
|
|
| BPN | 0.01 |
0.954 | 0.918–0.991 | NS |
|
|
| REN | 0.03 |
0.969 | 0.941–0.988 | NS |
|
|
| MHN | 0.01 |
0.942 | 0.900–0.986 | NS |
|
|
| OHE | 0.0004 | 15.98 |
3.414–74.805 | 0.04 | 9.014 | 1.078–75.378 |
| ALB | 0.0002 | 0.278 | 0.141–0.548 | 0.06 | 0.339 | 0.107–1.071 |
| PLT | 0.004 | 0.93 | 0.841–0.969 | NS |
|
|
| HDL | 0.009 | 0.967 | 0.944–0.992 | NS |
|
|
Associations for patients without a
history of OHE
The 69 patients without a history of OHE (Table III) were also examined and 22
patients were identified who exhibited MHE. Among the patients
without a history of OHE, patients with MHE exhibited significantly
higher values for CSS, MELD, CPS, prothrombin time-international
normalized ratio and ammonia levels when compared to the normal
patients. Furthermore, the patients with MHE and no history of OHE
exhibited significantly lower levels of ALB, ChE and PLT compared
to the normal patients with no history of OHE. However, the
multivariate analyses revealed that only CSS was independently
associated with MHE among the patients without a history of OHE
(Table IV). When the associations
between MHE and the CSS items were examined, patients with MHE
exhibited significantly higher scores for hand tremors, appetite
loss and decreased strength, as well as non-significant increases
in the scores for fatigue and anxiety (verses the normal patients)
(Table V). However, no significant
differences were observed when we compared the scores for muscle
cramping, abdominal fullness, abdominal pain, a feeling of low
energy and the three sleep-related items.
 | Table III.Clinical characteristics of MHE among
patients with cirrhosis and no history of overt hepatic
encephalopathy. |
Table III.
Clinical characteristics of MHE among
patients with cirrhosis and no history of overt hepatic
encephalopathy.
|
Characteristics | Normal (n=47) | MHE (n=22) | P-value |
|---|
| CSS |
6.8±4.4 |
10.0±4.7 | 0.007 |
| MELD |
9.6±3.3 |
12.2±3.7 | 0.002 |
| CPS |
6.0±2.5 |
7.3±2.5 | 0.03 |
| ALB |
3.7±0.7 |
3.2±0.6 | 0.003 |
| ChE |
220.1±103.2 |
149.5±105.8 | 0.007 |
| PLT |
13.6±7.8 |
9.2±7.4 | 0.02 |
| PTI-NR |
1.12±0.17 |
1.35±0.43 | 0.01 |
| NH3 |
45.2±34.6 |
75.1±45.5 | 0.004 |
 | Table IV.Factors associated with minimal
hepatic encephalopathy among patients with cirrhosis and no history
of overt hepatic encephalopathy. |
Table IV.
Factors associated with minimal
hepatic encephalopathy among patients with cirrhosis and no history
of overt hepatic encephalopathy.
|
| Univariate
analysis | Multivariate
analysis |
|---|
|
|
|
|
|---|
| Factors | P-value | OR | 95% CI | P-value | OR | 95% CI |
|---|
| CSS | 0.01 |
1.17 | 1.034–1.313 | 0.02 | 1.187 | 1.023–1.379 |
| ALB | 0.005 |
0.35 | 0.167–0.782 | NS |
| ChE | 0.01 |
0.993 | 0.993–0.998 | NS |
| NH3 | 0.007 |
1.019 | 1.005–1.033 | NS |
| PLT | 0.03 |
0.918 | 0.849–0.991 | NS |
| PT-INR | 0.02 | 27.19 | 1.531–482.865 | NS |
 | Table V.Comparing the cirrhosis-related
symptom scores among patients with and without MHE. |
Table V.
Comparing the cirrhosis-related
symptom scores among patients with and without MHE.
| Questionnaire
items | MHE | Normal | P-value |
|---|
| Hand tremors | 0.524 | 0.196 | 0.01 |
| Appetite loss | 0.957 | 0.413 |
0.004 |
| Muscle
cramping | 1.174 | 0.957 | NS |
| Fatigue | 1.478 | 1.064 | 0.09 |
| Decreased
strength | 1.957 | 1.468 | 0.02 |
| Anxiety | 1.174 | 0.787 | 0.06 |
| Abdominal
fullness | 0.957 | 0.638 | NS |
| Abdominal pain | 0.391 | 0.340 | NS |
| Feeling of low
energy | 1.391 | 1.109 | NS |
| Difficulty falling
asleep | 1.391 | 1.128 | NS |
| Poor sleep
quality | 1.478 | 1.289 | NS |
| Sleepy during the
daytime | 1.318 | 1.340 | NS |
Predicting MHE
Based on these results, the factors that may predict
MHE were evaluated. A history of OHE was the best marker for
predicting MHE (odds ratio: 9.014), and CSS was the best marker for
predicting MHE in patients without a history of OHE (odds
ratio=1.187). Therefore, a prediction score was developed that
combined a history of OHE (no: 0 points, yes: 10 points) and CSS
(range, 0–36 points), and evaluated its ability to predict MHE
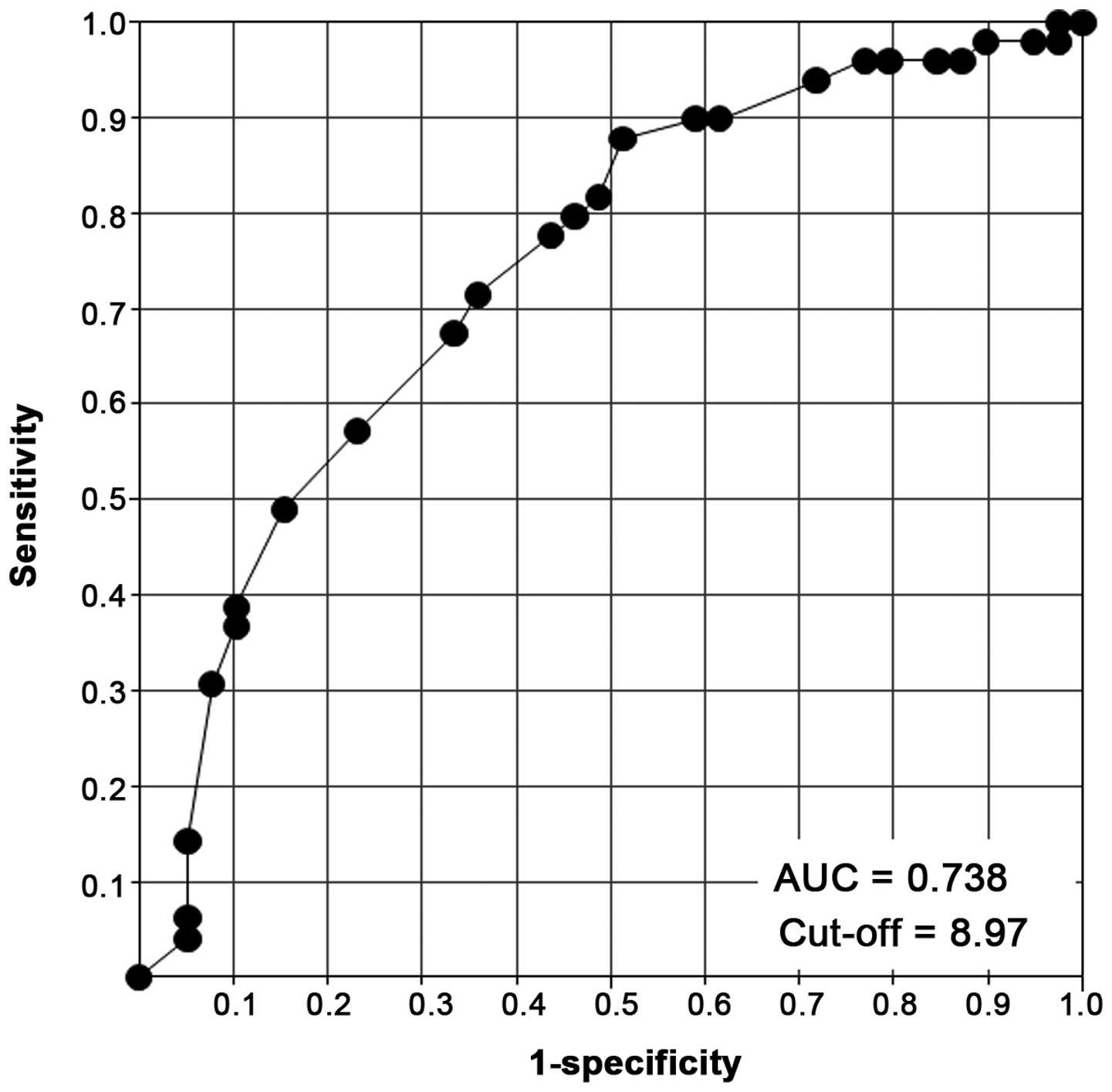
using receiver operating characteristic curve analysis (Fig. 1). The area under the curve was 0.738
and the cut-off value was 8.97. Based on a cut-off score of 9
points, the prediction score provided a sensitivity of 0.671 and a
specificity of 0.333 for MHE.
Discussion
In the present study, patients were evaluated for
signs of MHE, which was indicated by severe liver damage, MELD
score, CPS and a history of OHE. In addition, the CSS, PSQI scores
and SF-36 items were associated with MHE. The multivariate analyses
revealed that a history of OHE was the best predictor of MHE.
However, among patients without a history of OHE, MHE was
associated with indicators of severe liver damage, such as CPS,
MELD score and platelet counts, and CSS was the best predictor of
MHE among those patients. Furthermore, a novel prediction score was
developed for MHE using a history of OHE and CSS, which provided an
area under the curve of 0.738 and a sensitivity of 0.671.
Similar to these findings, it has been reported that
patients with MHE exhibit severe liver damage (as indicated by a
high MELD score) (10,24,25) and high
CPS (24). However, the previous
studies did not include patients with a history of OHE (23,24).
Moreover, OHE and MHE are not fully treated via liver
transplantation (19,26), and patients with a history of OHE may
experience impaired cognitive function, even subsequent to
resolving the OHE (27). Therefore,
patients with resolved OHE may be at risk for developing MHE,
regardless of the extent of any liver damage. Furthermore, MHE is a
risk factor for OHE (9), and
resolution of OHE is a risk factor for MHE, which highlights the
importance of accurately determining whether patients with
cirrhosis have a history of OHE. The present findings confirm this
concept, as a history of OHE was the best predictor of MHE among
all the variables that were evaluated.
It is well known that BCAA affects MHE and OHE
(16), there were a few patients with
a history of OHE who had already treated with BCAA in this study.
Therefore, the MHE group had a significantly higher number of
patients who were treated with BCAA supplements compared to the
normal group. Thus, there was a possibility of the effects that
BCAA supplements improve OHE to MHE.
Patients without a history of OHE were also
evaluated, and as speculated, their sleep disturbances may be
associated with MHE. Notably, low PSQI scores and frequent RLS were
associated with MHE among patients with a history of OHE, although
these associations were not observed among patients without a
history of OHE. Furthermore, CSS was the best predictor of MHE
among the patients without a history of OHE. In this context, CSS
is calculated using items from a cirrhosis symptom questionnaire
that was developed in our previous study, and three of the 12 items
in this tool are associated with sleep disturbances. Moreover, our
previous study reported that CSS, excluding the scores for the
three sleep disturbance-related items, is associated with liver
damage (18). In the present study,
three CSS items (hand tremors, appetite loss and decreased
strength) were significantly associated with MHE, and these
associations validate the previously reported associations between
MHE and the following responses: ‘I do not maintain balance’, ‘I
act irritable or impatient with myself’, ‘I am not doing any of my
usual physical recreations or activity’ and ‘I am eating much less
than usual’ (25). Another study has
also reported that worry was strongly associated with MHE in a
questionnaire regarding chronic liver disease (24). Although the majority of the patients
with MHE experience symptoms that contribute to cognitive
impairment, their other limitations (such as hand tremors, appetite
loss and decreased strength) may also reflect restrictions in their
activities of daily living. By contrast, MHE was not associated
with difficulty in falling asleep, sleeping poorly and being sleepy
during the daytime, which would indicate that patients with MHE,
but not OHE, do not experience sleep disturbances.
In conclusion, the present findings indicate that a
history of OHE was the best predictor of MHE, and CSS was the best
predictor of MHE among patients without a history of OHE.
Therefore, a prediction score was developed using a history of OHE
and CSS, and this score appears to be effective in screening for
MHE among patients with cirrhosis. However, this score is not
highly specific for MHE, and additional parameters are required to
develop a more sensitive screening tool. As patients with cirrhosis
have multiple symptoms, which may be associated with cognitive
impairment (28), the incorporation of
cognitive impairment symptoms into the present prediction score may
increase its sensitivity for MHE.
Glossary
Abbreviations
Abbreviations:
|
HE
|
hepatic encephalopathy
|
|
OHE
|
overt hepatic encephalopathy
|
|
NPT
|
neuropsychological test
|
|
MHE
|
minimal hepatic encephalopathy
|
|
PLT
|
platelets
|
|
ALB
|
albumin
|
|
ChE
|
cholinesterase
|
|
MELD
|
Model for End-stage Liver Disease
|
|
CPS
|
Child-Pugh score
|
|
CSS
|
cirrhosis-related symptom score
|
|
ESS
|
Epworth Sleepiness Scale
|
|
PSQI
|
Pittsburgh Sleep Quality Index
|
|
RLS
|
restless legs syndrome
|
|
BCAA
|
branched-chain amino acid
|
References
|
1
|
Romero-Gómez M, Montagnese S and Jalan R:
Hepatic encephalopathy in patients with acute decompensation of
cirrhosis and acute-on-chronic liver failure. J Hepatol.
62:437–447. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Ferenci P, Lockwood A, Mullen K, Tarter R,
Weissenborn K and Blei AT: Hepatic encephalopathy - definition,
nomenclature, diagnosis, and quantification: Final report of the
working party at the 11th World Congresses of Gastroenterology,
Vienna, 1998. Hepatology. 35:716–721. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kato A, Watanabe Y, Sawara K and Suzuki K:
Diagnosis of sub-clinical hepatic encephalopathy by
Neuropsychological Tests (NP-tests). Hepatol Res. 38(Suppl 1):
S122–S127. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Qi R, Zhang LJ, Luo S, Ke J, Kong X, Xu Q,
Liu C, Lu H and Lu GM: Default mode network functional
connectivity: A promising biomarker for diagnosing minimal hepatic
encephalopathy: CONSORT-compliant article. Medicine (Baltimore).
93:e2272014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Cona G, Montagnese S, Bisiacchi PS, Gatta
A, Cillo U, Angeli P, Amodio P and Schiff S: Early markers of
neural dysfunction and compensation: A model from minimal hepatic
encephalopathy. Clin Neurophysiol. 125:1138–1144. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kircheis G, Hilger N and Häussinger D:
Value of critical flicker frequency and psychometric hepatic
encephalopathy score in diagnosis of low-grade hepatic
encephalopathy. Gastroenterology. 146:961–969. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Montoliu C, Cauli O, Urios A, ElMlili N,
Serra MA, Giner-Duran R, González-Lopez O, Del Olmo JA, Wassel A,
Rodrigo JM, et al: 3-nitro-tyrosine as a peripheral biomarker of
minimal hepatic encephalopathy in patients with liver cirrhosis. Am
J Gastroenterol. 106:1629–1637. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Montoliu C, Piedrafita B, Serra MA, del
Olmo JA, Urios A, Rodrigo JM and Felipo V: IL-6 and IL-18 in blood
may discriminate cirrhotic patients with and without minimal
hepatic encephalopathy. J Clin Gastroenterol. 43:272–279. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Riggio O, Amodio P, Farcomeni A, Merli M,
Nardelli S, Pasquale C, Pentassuglio I, Gioia S, Onori E, Piazza N,
et al: A Model for Predicting Development of Overt Hepatic
Encephalopathy in Patients With Cirrhosis. Clin Gastroenterol
Hepatol. 13:1346–1352. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Ampuero J, Simón M, Montoliú C, Jover R,
Serra MÁ, Córdoba J and Romero-Gómez M: Minimal hepatic
encephalopathy and critical flicker frequency are associated with
survival of patients with cirrhosis. Gastroenterology.
149:1483–1489. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Bajaj JS, Saeian K, Schubert CM,
Hafeezullah M, Franco J, Varma RR, Gibson DP, Hoffmann RG, Stravitz
RT, Heuman DM, et al: Minimal hepatic encephalopathy is associated
with motor vehicle crashes: The reality beyond the driving test.
Hepatology. 50:1175–1183. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bajaj JS, Pinkerton SD, Sanyal AJ and
Heuman DM: Diagnosis and treatment of minimal hepatic
encephalopathy to prevent motor vehicle accidents: A
cost-effectiveness analysis. Hepatology. 55:1164–1171. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Kawaguchi T, Taniguchi E and Sata M: Motor
vehicle accidents: How should cirrhotic patients be managed? World
J Gastroenterol. 18:2597–2599. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Román E, Córdoba J, Torrens M, Torras X,
Villanueva C, Vargas V, Guarner C and Soriano G: Minimal hepatic
encephalopathy is associated with falls. Am J Gastroenterol.
106:476–482. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bianchi G, Giovagnoli M, Sasdelli AS and
Marchesini G: Hepatic encephalopathy and health-related quality of
life. Clin Liver Dis. 16:159–170. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kato A, Tanaka H, Kawaguchi T, Kanazawa H,
Iwasa M, Sakaida I, Moriwaki H, Murawaki Y, Suzuki K and Okita K:
Nutritional management contributes to improvement in minimal
hepatic encephalopathy and quality of life in patients with liver
cirrhosis: A preliminary, prospective, open-label study. Hepatol
Res. 43:452–458. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Matsuzaki T, Ichikawa T, Kondo H, Taura N,
Miyaaki H, Isomoto H, Takeshima F and Nakao K: Prevalence of
restless legs syndrome in Japanese patients with chronic liver
disease. Hepatol Res. 42:1221–1226. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ichikawa T, Naota T, Miyaaki H, Miuma S,
Isomoto H, Takeshima F and Nakao K: Effect of an oral branched
chain amino acid-enriched snack in cirrhotic patients with sleep
disturbance. Hepatol Res. 40:971–978. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Akahoshi M, Ichikawa T, Taura N, Miyaaki
H, Yamaguchi T, Yoshimura E, Takahara I, Soyama A, Takatsuki M,
Kondo H, et al: Sleep disturbances and quality of life in patients
after living donor liver transplantation. Transplant Proc.
46:3515–3522. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Fukuhara S and Suzukamo Y: Manual of the
SF-36v2 Japanese Version. Institute for Health Outcomes and Process
Evaluation Research. Kyoto, Japan: 2004.
|
|
21
|
Johns MW: A new method for measuring
daytime sleepiness: The Epworth sleepiness scale. Sleep.
14:540–545. 1991.PubMed/NCBI
|
|
22
|
Doi Y, Minowa M, Uchiyama M, Okawa M, Kim
K, Shibui K and Kamei Y: Psychometric assessment of subjective
sleep quality using the Japanese version of the Pittsburgh Sleep
Quality Index (PSQI-J) in psychiatric disordered and control
subjects. Psychiatry Res. 97:165–172. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Buysse DJ, Reynolds CF III, Monk TH,
Berman SR and Kupfer DJ: The Pittsburgh Sleep Quality Index: A new
instrument for psychiatric practice and research. Psychiatry Res.
28:193–213. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Hirano H, Saito M, Yano Y, Momose K,
Yoshida M, Tanaka A and Azuma T: Chronic liver disease
questionnaire would be a primary screening tool of neuropsychiatric
test detecting minimal hepatic encephalopathy of cirrhotic
patients. Hepatol Res. 45:994–1003. 2015. View Article : Google Scholar
|
|
25
|
Nabi E, Thacker LR, Wade JB, Sterling RK,
Stravitz RT, Fuchs M, Heuman DM, Bouneva I, Sanyal AJ, Siddiqui MS,
et al: Diagnosis of covert hepatic encephalopathy without
specialized tests. Clin Gastroenterol Hepatol. 12:1384–1389.e2.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Lin WC, Chou KH, Chen CL, Chen HL, Lu CH,
Li SH, Huang CC, Lin CP and Cheng YF: Longitudinal brain white
matter alterations in minimal hepatic encephalopathy before and
after liver transplantation. PLoS One. 9:e1058872014. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Umapathy S, Dhiman RK, Grover S, Duseja A
and Chawla YK: Persistence of cognitive impairment after resolution
of overt hepatic encephalopathy. Am J Gastroenterol. 109:1011–1019.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Córdoba J: New assessment of hepatic
encephalopathy. J Hepatol. 54:1030–1040. 2011. View Article : Google Scholar : PubMed/NCBI
|