Introduction
Chronic hepatitis B (CHB), which is defined as
persistence of hepatitis B surface antigen for six months or more,
is a major public health problem. According to the World Health
Organisation, there are an estimated 240 million chronically
infected individuals worldwide, particularly in low- and
middle-income countries (1). The major
complications of CHB are cirrhosis and hepatocellular carcinoma
(HCC). Between 20 and 30% of those who become chronically infected
will develop these complications, and an estimated 650,000
individuals will succumb due to CHB annually (1). The majority of individuals are unaware of
their hepatitis B virus (HBV) infection, and, therefore, often
present at an advanced stage of the disease (1). China has been identified as a region with
a high prevalence of HBV; based on the National Disease Supervision
Information Management System of China, the mean reported incidence
of HBV was 84.3 per 100,000 in China between 2005 and 2010
(2). Despite the reduction in the
incidence of the HBV infection, there are ~93 million individuals
chronically infected with HBV and ~20 million cases of symptomatic
CHB in China currently (3). The high
prevalence of CHB poses heavy social and financial burdens for
China.
The dynamic natural history of CHB infection
involves a complex interaction between the host immune system and
the virus. Broadly, there are four phases, of varying duration,
which are defined as follows: Immune-tolerant, immune-elimination
(or immune-clearance), inactive and reactivation (4,5). The
immune-tolerant phase is characterized by a high level of HBV
replication, HBeAg positivity, and a normal or minimally elevated
alanine aminotransferase (ALT) level (6). Low ALT level CHB-infected patients (ALT
<80 U/l) may be in the HBV immune-tolerant phase (7). According to local therapeutic guidelines
of prevention and treatment of CHB, patients exhibiting mild ALT
elevation (40–80 IU/l) should receive antiviral therapy subsequent
to confirmation of hepatic fibrosis status via biopsy or
non-invasive examinations (8–10). For patients whose ALT level fluctuates,
but never normalizes, the degree of liver inflammation and fibrosis
were more serious, and the occurrence rate of cirrhosis and
hepatocellular carcinoma may be higher (11). In clinical practice, antiviral
treatment cannot be administered to CHB patients with low ALT
levels due to lack of timely histological diagnosis; this may cause
silent progression of the disease and result in severe
sequelae.
Bicyclol is a liver-protective substance
administered to patients with various types of liver disease. Its
mechanism of action may be closely associated with free
radical-scavenging properties, protection against lipid
peroxidation, protection of hepatic cell membranes and
mitochondrial function (12), and
inhibition of inflammatory cytokines (13). It was demonstrated in experimental and
clinical studies that Bicyclol decreased serum transaminase levels
and resulted in improvements as observed by pathology (14–16).
Although various studies have examined the clinical efficacy of
Bicyclol in virus hepatitis patients, none, to the best of our
knowledge, have investigated the effectiveness and safety of
Bicyclol in a clinical setting.
The current study selected hospitalization data of
CHB patients exhibiting mild ALT elevation from a hospital
information system (HIS) database of 18 third-grade class A
hospitals in China. The ALT normalization rate served as the
evidence of successful treatment, and the curative effect of
Bicyclol tablets on patients with HBV accompanied by mild ALT
elevation was discussed in order to propose practical clinical
applications.
Patients and methods
Data sources and standardization
Hospitalization data of patients included in the
present study was obtained from a HIS database (compiled by the
Institute of Basic Research in Clinical Medicine, China Academy of
Chinese Medical Science, Beijing, China) using data from 18
third-grade class A hospitals in China. Informed consent was not
considered necessary due to the retrospective nature of the study.
Research was performed subsequent to discussion and approval by the
Ethics Committee of the Institute of Basic Research in Clinical
Medicine, China academy of Chinese Medical Sciences. The HIS
database was standardized prior to data extraction and data
analysis, with the standardization process involving exclusion of
redundant data in patients' general information, discordant data
and advice from doctors that was deemed irrelevant. Dosages were
standardized, as were physiochemical indexes. Following data
integration, data meeting the following criteria were included in
the current study.
Treatment analysis data inclusion
criteria
Exposed group: Patients with HBV that were taking
Bicyclol tablets were selected from the viral hepatitis database;
the ALT examination was conducted 7 days prior to Bicyclol
administration; the result revealed a mild abnormality in ALT level
(40–80 U/l); ALT examination was conducted within 7 days of drug
withdrawal; and medication consumption had been for >15 and
<30 days. Non-exposed group: Patients with HBV that were not
taking Bicyclol tablets were selected from the viral hepatitis
database; duration of hospitalization was >15 days and <30
days; ≥2 ALT examinations had been conducted, with the first result
revealing that there was mild abnormality (40–80 U/l).
Safety data inclusion criteria
Exposed group: Patients taking Bicyclol tablets were
selected from the viral hepatitis database;
creatinine/hemoglobin/leucocyte level measurements had been
conducted 7 days prior to Bicyclol administration and within 7 days
of Bicyclol withdrawal; and patients with an initially abnormal
result were excluded. Non-exposed group: Patients not taking
Bicyclol tablets were selected from the viral hepatitis database;
duration of hospitalization was <30 days; ≥2
creatinine/hemoglobin/leucocyte level examinations had been
conducted; and patients with an initially abnormal result were
excluded. A 1:1 match of every index was made between the members
of the exposed and non-exposed groups according to gender, age and
state of illness on admission.
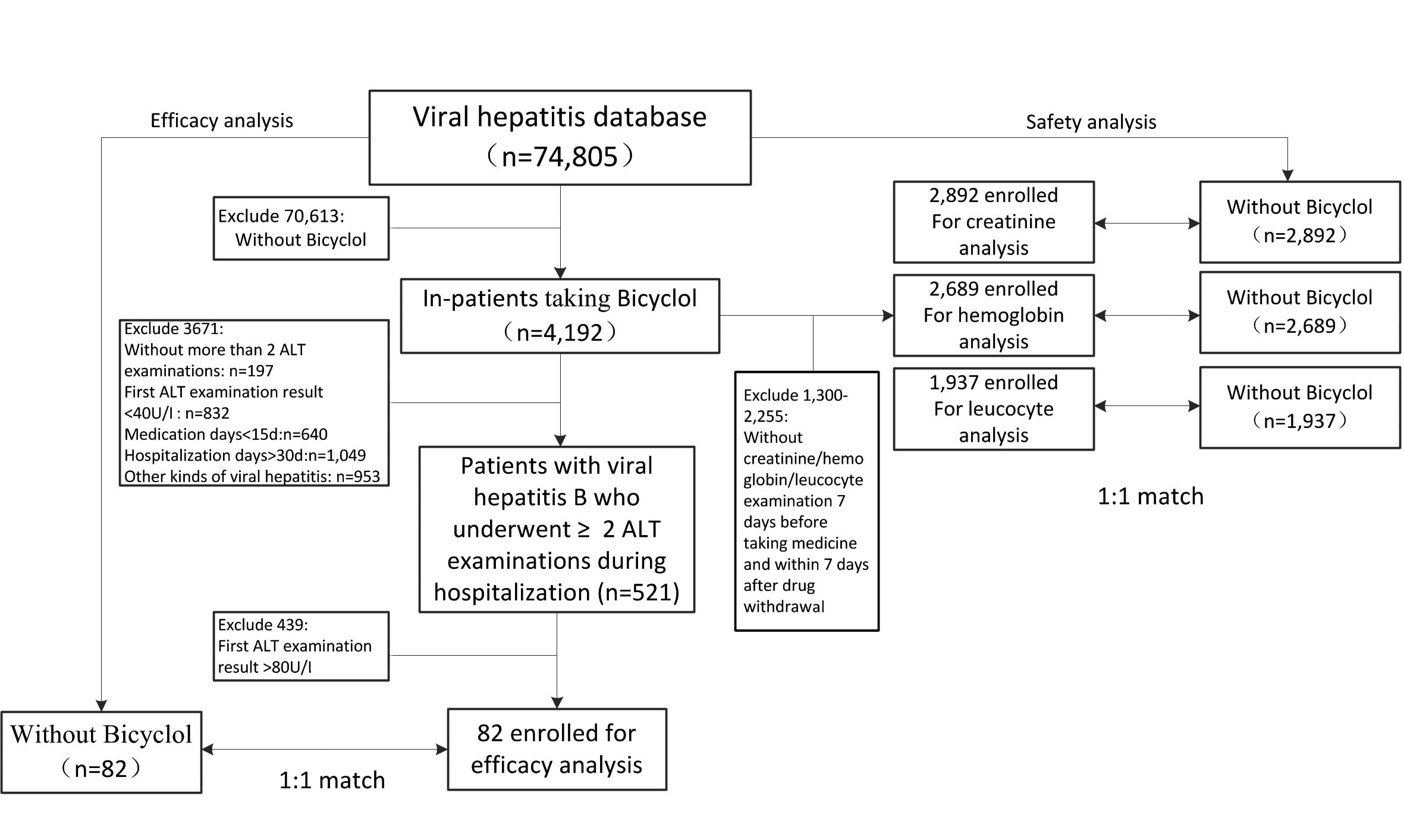
Data extraction and data analysis
All standardized data was extracted from the HIS
database in strict accordance with the inclusion criteria; the data
extraction and analysis process is presented in Fig. 1.
Definition of a treatment effect
The criteria for judging the curative effect indexes
were as follows: The ALT examination result for patients of the
exposed group was measured as close to the time of Bicyclol
withdrawal as possible and no later than 7 days subsequently. For
the patients in the non-exposed group, the last ALT detection value
during hospitalization was used. When the final ALT detection value
was ≤40 U/l, it was defined as normalization, when the final ALT
detection value was >40 U/l, it was defined as not
normalized.
Statistical analysis
Between-group differences with continuous variables
were analyzed using the independent-samples T test or
non-parametric test. Between-group differences in categorical data
and single factor analysis of between-group ALT normalization rates
were analyzed using χ2-test (Pearson's method). In
addition, multifactorial analysis of between-group ALT
normalization rates was conducted using unconditional logistic
regression. Variables such as the age of patients, gender, baseline
ALT levels, initial illness severity, hospitalization conditions,
hospitalization duration, drug combination (with/without reduced
glutathione; GSH) and treatment regimen were converted to
dichotomous variables, and modeled for Wald analysis. Subgroup
analysis was performed using a single factor logistic regression
model, and the ALT normalization rate was taken as a dependent
variable and the groups as independent variables; the differences
in ALT normalization rates were compared between the two groups.
Statistical tests were two-sided and P<0.05 was considered to
indicate a statistically significant difference. Statistical
analysis was performed using SQL Sever 2008 (Microsoft, Inc.,
Redmond, WA, USA) and SPSS 17.0 (SPSS, Inc., Chicago, IL, USA).
Results
Assessment of curative effect
General information of enrolled patients
There were 82 CHB patients taking Bicyclol tablets
in the exposed group, which met the inclusion criteria, and 82 CHB
patients not taking Bicyclol tablets in the non-exposed group. The
patients in the two groups were selected from the viral hepatitis
database according to gender, age and state of illness on admission
with a 1:1 match between groups. Differences between general
information, such as age, gender, and baseline ALT levels of
patients in the two groups were not identified as being
statistically significant (P>0.05), therefore the experimental
results of the two groups are comparable (Table I).
 | Table I.Comparison of patient information
between the two groups. |
Table I.
Comparison of patient information
between the two groups.
| Variables | Exposed group
(n=82) | Non-exposed group
(n=82) | P-value |
|---|
| Age (years, means ±
SD) | 47.38±16.79 | 47.82±16.85 | 0.690 |
| Gender |
|
|
|
| Male | 63 | 63 | 1.000 |
|
Female | 19 | 19 |
|
| Baseline ALT (U/l,
means ± SD) | 58.80±11.06 | 55.60±10.30 | 0.062 |
| State of illness on
admission |
|
|
|
|
Common | 62 | 62 | 1.000 |
|
Urgent | 20 | 20 |
|
| Pattern of
hospitalization |
|
|
|
|
Outpatient | 72 | 67 | 0.277 |
| Emergency
patient | 10 | 15 |
|
| Hospitalization days
(means ± SD) | 20.93±4.47 | 21.32±4.01 | 0.399 |
| Drug combination |
|
|
|
|
Combination with GSH | 30 | 37 | 0.266 |
|
Non-combination with GSH | 52 | 45 |
|
Single factor analysis of ALT normalization
rates
No serious deterioration of liver function occurred
in either of the two groups, and the ALT normalization rates of the
exposed and non-exposed groups were 65.85 and 50.00%, respectively.
Between-group differences were identified as being statistically
significant (χ2=4.228; P=0.040; Table II).
 | Table II.Comparison of ALT normalization rates
between the two groups. |
Table II.
Comparison of ALT normalization rates
between the two groups.
|
| Cases of
outcome/total cases (%) |
|
|
|---|
|
|
|
|
|
|---|
| Variables | Exposed group | Non-exposed
group | Pearson's
χ2 | P-value |
|---|
| Without ALT
normalization | 28/82 (34.15) | 41/82 (50.00) | 4.228 | 0.040 |
| With ALT
normalization | 54/82 (65.85) | 41/82 (50.00) |
|
|
Multifactorial analysis of ALT normalization
rates
After revising patient age, gender, baseline ALT
levels, illness severity on admission, patterns of hospitalization,
duration of hospitalization and drug combination (with/without GSH)
using a multifactorial model, the results revealed that the
differing group type was the independent influencing factor
(P=0.025) that influenced ALT normalization. The patients in the
exposed group possessed higher ALT normalization rates [odds ratio
(OR)=2.156; 95% confidence interval (CI), 1.103–4.215] compared
with the non-exposed group (Table
III).
 | Table III.Multfactorial analysis of ALT
normalization rates. |
Table III.
Multfactorial analysis of ALT
normalization rates.
| Subgroup | Wald
χ2 | P-value | Adjusted OR (95%
CI) |
|---|
| Group |
|
|
|
| (exposed
vs. non-exposed) | 5.049 | 0.025 | 2.156
(1.103–4.215) |
| Age |
|
|
|
| (≥45 vs.
<45) | 0.320 | 0.571 | 1.226
(0.606–2.479) |
| Gender |
|
|
|
| (male vs.
female) | 0.195 | 0.659 | 0.837
(0.380–1.842) |
| Baseline ALT
levels |
|
|
|
| (≥60 U/l
vs. <60 U/l) | 0.664 | 0.415 | 0.756
(0.385–1.482) |
| State of illness on
admission |
|
|
|
| (common
vs. urgent) | 0.035 | 0.852 | 0.924
(0.402–2.124) |
| Pattern of
hospitalization |
|
|
|
|
(outpatients vs. emergency
patients) | 1.604 | 0.205 | 0.524
(0.193–1.424) |
| Hospitalization
days |
|
|
|
| (≥20 vs.
<20) | 2.983 | 0.084 | 1.804
(0.923–3.526) |
| Combination with
GSH |
|
|
|
| (yes vs.
no) | 2.377 | 0.123 | 0.593
(0.305–1.152) |
Subgroup analysis of ALT normalization rates
Subgroup analysis was performed for two levels of
each of the above-mentioned variables (Table IV). For male patients or those whose
baseline ALT levels were <60 U/l, consumption of Bicyclol
tablets resulted in increased ALT normalization rates when compared
with non-exposed group, and the difference was statistically
significant (P<0.01). The ALT normalization rates of the two
groups were not identified to be statistically significant when
compared with the rest of the subgroup analysis.
 | Table IV.Comparison of ALT normalization rates
in subgroup analysis. |
Table IV.
Comparison of ALT normalization rates
in subgroup analysis.
|
| Cases of ALT
normalization/total (%) |
|
|
|
|---|
|
|
|
|
|
|
|---|
| Subgroup | Exposed group | Non-exposed
group | Pearson's
χ2 | P-value | OR (95% CI) |
|---|
| Age (years) |
|
|
|
|
|
|
≥45 | 38/57 (66.66) | 29/56 (51.79) | 2.569 | 0.109 | 1.862
(0.871–3.983) |
|
<45 | 16/25 (64.00) | 12/26 (46.15) | 1.621 | 0.203 | 2.074
(0.675–6.377) |
| Gender |
|
|
|
|
|
|
Male | 43/63 (68.25) | 28/63 (44.44) | 7.106 | 0.008 | 2.687
(1.299–5.559) |
|
Female | 11/19 (57.89) | 13/19 (68.42) | 0.450 | 0.502 | 0.635
(0.168–2.396) |
| Baseline ALT levels
(U/l) |
|
|
|
|
|
|
≥60 | 22/41 (53.66) | 15/27 (55.55) | 0.024 | 0.878 | 0.926
(0.349–2.945) |
|
<60 | 32/41 (78.05) | 26/55 (47.27) | 8.816 | 0.003 | 3.966
(1.597–9.847) |
| State of illness on
admission |
|
|
|
|
|
|
Common | 39/62 (62.90) | 30/62 (48.39) | 2.627 | 0.105 | 1.809
(0.883–3.703) |
|
Urgent | 15/20 (75.00) | 11/20 (55.00) | 1.720 | 0.190 | 2.455
(0.642–9.391) |
| Pattern of
hospitalization |
|
|
|
|
|
|
Outpatients | 45/72 (62.50) | 33/67 (49.25) | 2.457 | 0.117 | 1.717
(0.873–3.376) |
|
Emergency patients | 9/10 (90.00) | 8/15 (53.33) | 3.088 | 0.079 | 7.875
(0.788–78.671) |
| Hospitalization
days |
|
|
|
|
|
|
≥20 | 32/46 (69.57) | 29/50 (58.00) | 1.374 | 0.241 | 1.655
(0.713–3.844) |
|
<20 | 22/36 (61.11) | 12/32 (37.50) | 3.705 | 0.054 | 2.619
(0.983–6.981) |
| Combination with
GSH |
|
|
|
|
|
|
Yes | 18/30 (60.00) | 16/37 (43.24) | 1.843 | 0.175 | 1.969
(0.740–5.235) |
| No | 36/52 (69.23) | 25/45 (55.56) | 1.916 | 0.166 | 1.800
(0.783–4.137) |
Safety analysis
As shown in Table V,
the safety data sets reveal that the occurrence rates of abnormal
creatinine, hemoglobin and leucocyte levels in the exposed group
were lower than those in the non-exposed group, and the difference
was statistically significant (P<0.05). The findings indicate
that Bicyclol tablets possess a good safety profile.
 | Table V.Occurrence rates (%) of
creatinine/hemoglobin/leucocyte level abnormalities between the two
groups. |
Table V.
Occurrence rates (%) of
creatinine/hemoglobin/leucocyte level abnormalities between the two
groups.
| Observation
indexes | Exposed group %
(n/N) | Non-exposed group %
(n/N) | Pearson's
χ2 | P-value |
|---|
| Creatinine | 5.74
(166/2892) | 7.92
(229/2892) | 10.7846 | 0.0010 |
| Hemoglobin | 24.62
(662/2689) | 28.93
(778/2689) | 12.7614 | 0.0004 |
| Leucocyte | 34.9
(674/1931) | 38.22
(738/1931) | 4.5727 | 0.0325 |
Discussion
CHB is a global health problem affecting millions of
people worldwide. The clinical spectrum is wide, ranging from a
subclinical inactive carrier state, to progressive chronic
hepatitis, cirrhosis, decompensation and HCC (4). According to Chinese Guidelines for
Prevention and Treatment of Chronic Hepatitis B (8), when ALT levels are between 1 × upper
limit of normal (ULN) and 2 × ULN, liver biopsy or non-invasive
examinations (such as transient elastography) should be performed
in order to evaluate the state of liver fibrosis and establish
whether antiviral treatment is required. In China, numerous
patients were unable to undergo relevant examinations due to
financial constraints, low prevalence of noninvasive equipment or
resistance to invasive liver biopsy. A study conducted in Taiwan
revealed that the cumulative occurrence rate of HCC at 3, 5 and 10
years (2.3, 5.3 and 13.4%, respectively) in patients with a
continuously low (15–44 U/l) ALT level were lower than those of
patients with ALT abnormalities (3.7, 8.5 and 21.0%, respectively)
(17). Thus, CHB patients with mild
ALT elevation should be observed in clinical practice and the
elevated ALT levels should be controlled to within a normal
range.
Health care administrative policy and financial
support greatly accelerated the adoption of electronic health
records (EHRs) in China. According to a 2012 national survey in
China, ~50% of 1,004 responding hospitals had adopted at least the
basic forms of EHR and practice management systems often referred
to as HIS (18). Retrospective studies
based on HIS data guarantee objectivity and veracity, identify
potential patterns from within large quantities of data, and
provide a reference point and ideas for prospective studies and
clinical practice. All data in the current study was extracted from
the HIS database, which contains diagnostic and treatment
information for ~3 million patients from 39 large third-grade class
A hospitals capable of more advanced levels of diagnosis and
treatment. Detailed information, including disease diagnosis,
laboratory exams, treatment regimens, and disease outcome
information is also compiled. There is an obvious advantage in
veracity of data, large sample sizes, multidimensional analysis,
multiple assessment indices and from obtaining information
regarding diagnosis and treatment.
The single factor analysis result revealed that the
ALT normalization rate of the exposed group was significantly
higher than that of the non-exposed group (P<0.05). After
adjusting for individual patient variables, the ALT normalization
OR (95% CI) of the exposed group was 2.156 (1.103–4.215) compared
with that of the non-exposed group. Subgroup analysis indicated
that when gender or baseline ALT levels were taken as
stratification factors, differences in ALT normalization rates
between the two groups were statistically significant (P<0.05).
This indicates that males or patients with baseline ALT levels that
are <60 U/l are the dominant population, and the administration
of Bicyclol results in improved curative effects. Furthermore,
safety data analysis revealed that kidney and the occurrence rates
of blood abnormalities in patients in the exposed group were lower
than those of the non-exposed group (P<0.05), which demonstrates
that Bicyclol possesses a good safety profile.
Bicyclol may improve injured liver cell function and
alleviate pathological injuries of the liver. Pharmacological
studies show that Bicyclol may inhibit the expression and activity
of key inflammatory regulatory factors, such as nuclear
transcription factor-κB, tumor necrosis factor-α, interleukin-1β,
transforming growth factor-β1 and inducible nitric oxide (NO)
synthase. In addition, Bicyclol may inhibit inflammatory injury,
oxidative damage and abnormal apoptosis of hepatocytes caused by
generation of reactive oxygen species and NO generation (19–22), thereby
improving the structure and function of the hepatocyte and
mitochondrial membranes (23,24). Previous clinical studies indicate that
orally administered Bicyclol possesses a good safety profile and is
advantageous in the setting of CHB-induced ALT elevation (14,25).
In conclusion, the present study demonstrated that
Bicyclol tablets improved the ALT normalization rate of viral CHB
patients exhibiting mild ALT elevation. However, there were certain
limitations of the present study due to its inherent retrospective
nature, and the number of cases and observation indices were,
therefore, comparatively small. Large sample, randomized,
controlled studies are required to provide more powerful,
evidence-based treatment strategies for use in clinical practice
concerning Bicyclol administration in the setting of HBV.
Acknowledgements
The present study was supported by the National
Science and Technology Major Project of China (grant no.
2015ZX09501004-001-002), the Independent Project of China Academy
of Traditional Chinese Medical Sciences (grant no. PY1303), the
Independent Project of Fundamental Research of China Academy of
Traditional Chinese Medical Sciences (grant no. Z0406) and the
Special Research Project of TCMs by the State Administration of
Traditional Chinese Medicine (grant no. 201507003).
References
|
1
|
World Health Organization, . Guidelines
for the prevention, care and treatment of persons with chronic
hepatitis B infection. March;2015.
|
|
2
|
Yan YP, Su HX, Ji ZH, Shao ZJ and Pu ZS:
Epidemiology of hepatitis B virus infection in China: Current
status and challenges. J Clin Transl Hepatol. 2:15–22.
2014.PubMed/NCBI
|
|
3
|
Zheng X, Wang J and Yang D: Antiviral
therapy for chronic hepatitis B in China. Med Microbiol Immunol
(Berl). 204:115–120. 2015. View Article : Google Scholar
|
|
4
|
Tang CM, Yau TO and Yu J: Management of
chronic hepatitis B infection: current treatment guidelines,
challenges, and new developments. World J Gastroenterol.
20:6262–6278. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Manzoor S, Saalim M, Imran M, Resham S and
Ashraf J: Hepatitis B virus therapy: What's the future holding for
us? World J Gastroenterol. 21:12558–12575. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nebbia G, Peppa D and Maini MK: Hepatitis
B infection: Current concepts and future challenges. QJM.
105:109–113. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Expert Committee on Antiviral Therapy for
Special Patients with Chronic Hepatitis B, . Expert Consensus on
Antiviral Therapy for Special Patients with Chronic Hepatitis B: An
update in 2015. J Clin Hepatol. 31:1185–1192. 2015.
|
|
8
|
Chinese Society of Hepatology: CMA and
Society of Infectious Diseases, CMA, . Guidelines for Prevention
and Treatment of Chronic Hepatitis B (2015 Edition). Chin J Exp
Clin Infect Dis. 9:570–589. 2015.
|
|
9
|
European Association For The Study Of The
Liver: EASL clinical practice guidelines, . Management of chronic
hepatitis B virus infection. J Hepatol. 57:167–185. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Sarin SK, Kumar M, Lau GK, Abbas Z, Chan
HLY, Chen CJ, Chen DS, Chen HL, Chen PJ, Chien RN, et al:
Asian-Pacific clinical practice guidelines on the management of
hepatitis B: A 2015 update. Hepatol Int. 10:1–98. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Li XN, Wu XP and Xiang TX: Characteristics
of liver biopsy in chronic hepatitis B virus-infected patients with
normal and minimally raised ALT. J Nanchang Univ. 51:73–76.
2011.
|
|
12
|
Liu GT, Li Y, Wei HL, Zhang H, Xu JY and
Yu LH: Mechanism of protective action of bicyclol against
CCl-induced liver injury in mice. Liver Int. 25:872–879. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang H and Li Y: Protective effect of
bicyclol on acute hepatic failure induced by lipopolysaccharide and
D-galactosamine in mice. Eur J Pharmacol. 534:194–201. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Pan SY, Dong H, Yu ZL, Zhao XY, Xiang CJ,
Wang H, Fong WF and Ko KM: Bicyclol, a synthetic
dibenzocyclooctadiene derivative, decreases hepatic lipids but
increases serum triglyceride level in normal and
hypercholesterolaemic mice. J Pharm Pharmacol. 59:1657–1662. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Xie W, Shi G, Zhang H, Zhao G, Yu Z, Lang
Z, Zhao H, Yan J and Cheng J: A randomized, multi-central,
controlled study of patients with hepatitis B e antigen-positive
chronic hepatitis B treated by adefovir dipivoxil or adefovir
dipivoxil plus bicyclol. Hepatol Int. 6:441–448. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Han Y, Shi JP, Ma AL, Xu Y, Ding X-D and
Fan J-G: Randomized, vitamin E-controlled trial of bicyclol plus
metformin in non-alcoholic fatty liver disease patients with
impaired fasting glucose. Clin Drug Investig. 34:1–7. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Lin CL and Kao JH: Risk stratification for
hepatitis B virus related hepatocellular carcinoma. J Gastroenterol
Hepatol. 28:10–17. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Lei J, Sockolow P, Guan P, Meng Q and
Zhang J: A comparison of electronic health records at two major
Peking University Hospitals in China to United States meaningful
use objectives. BMC Med Inform Decis Mak. 13:962013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Yu YN, Chen H and Li Y: Effect of bicyclol
on cisplatin-induced hepatotoxicity in the hepatocarcinoma 22
tumour-bearing mice. Basic Clin Pharmacol Toxicol. 104:300–305.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yao XM, Chen H and Li Y: Protective effect
of bicyclol on liver injury induced by hepatic warm
ischemia/reperfusion in rats. Hepatol Res. 39:833–842. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Lou XE, Xu N, Yao HP and Chen Z: Bicyclol
attenuates pro-inflammatory cytokine and chemokine productions in
CpG-DNA-stimulated L02 hepatocytes by inhibiting p65-NF-κB and
p38-MAPK activation. Pharmazie. 65:206–212. 2010.PubMed/NCBI
|
|
22
|
Bao XQ and Liu GT: Involvement of HSP70 in
the protection of bicyclol on apoptosis of HepG2 cells intoxicated
by d-galactosamine. J Asian Nat Prod Res. 12:313–323. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Yu HY, Wang BL, Zhao J, Yao XM, Gu Y and
Li Y: Protective effect of bicyclol on tetracycline-induced fatty
liver in mice. Toxicology. 261:112–118. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Liu GT: Bicyclol: A novel drug for
treating chronic viral hepatitis B and C. Med Chem. 5:29–43. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wang H, Qiang L and Pei L: Assessment of
Bicyclol tablets' Effect on Viral CHB. Chin J Exp Clin Virol.
21:165–167. 2007.
|