Introduction
Thymidylate synthase (TS), as the de novo
source of thymidylate synthesis, is an essential enzyme involved in
DNA replication and cell growth (1). It catalyzes the conversion of
deoxyuridylate (dUMP) to deoxythymidylate (dTMP), which is critical
for DNA synthesis and repair. The substrate for TS is a central
metabolite in folate metabolism. Due to its important role in the
folate biosynthesis pathway, it has become one of the major targets
of antitumor agents, such as 5-fluorouracil (5-Fu) and pemetrexed
(PEM).
PEM is a multitarget antifolate agent that has
produced excellent clinical outcomes in first-line, second-line and
maintenance treatment in advanced non-small cell lung cancer
(NSCLC) (2–4). Notably, PEM-based treatment provided
more favorable clinical outcomes in patients with adenocarcinoma
(ADC) compared with those with squamous cell carcinoma (SCC). A
post hoc analysis of three randomized global trials confirmed the
superiority of PEM in non-squamous non-small cell lung cancer
(NSNSCLC) (5). A plausible
explanation for this superiority may involve the expression of TS
in different histological types. A Japanese study (6) collected 2621 NSCLC patients and
examined TS expression in postoperative tissue samples obtained
from this population. TS expression was categorized according to
TS/β-actin values. In univariate analysis, TS gene expression in
formalin-fixed and paraffin-embedded (FFPE) tumor samples was
higher for SCC (mean TS/β-actin 4.3), compared with ADC (mean
TS/β-actin 2.3) (p<0.01). Another study also confirmed that the
mean scoring of TS was significantly higher in the non-ADC than in
the ADC subgroup (2.79±0.61 vs. 1.98±0.88, p<0.0001) (7). Notably, the above analysis revealed
that patients with positive expression of TS had a lower 5-year
progression-free survival (PFS) rate than those presenting negative
expression of TS (48.6 vs. 79.1%, p<0.0001). The 5-year overall
survival (OS) rate was also significantly lower in the positive
group (67.5 vs. 86.1%, p=0.0002). Among patients with ADC, the
5-year PFS rate was 30.5% in the TS expression positive group and
83.1% in the negative group (p<0.0001). The 5-year OS rates were
61.1 and 90.1%, respectively (p<0.0001). Sun et al
(8) also demonstrated an
association between a higher response rate for PEM-based
chemotherapy and TS-negativity (33.7 vs. 14.1%, p=0.002). PFS for
PEM-based treatment was significantly longer in the ADC population
(2.9 vs. 1.4 months, p=0.001) and TS-negative subgroup (4.1 vs. 2.0
months, p=0.001). Multivariate analysis revealed that TS-negativity
was associated with longer PFS [hazard ratio (HR), 0.70; 95%
confidence interval (CI), 0.51–0.97].
The functional polymorphisms in the TS gene have
been suggested to be a regulator of downstream protein expression
and mRNA level (9,10). The most closely studied
polymorphisms have focused on the variable number of tandem repeats
(VNTR) of a 28-bp sequence (2R/3R) in the 5′-untranslated region
(UTR), a single nucleotide polymorphism (SNP) in the second repeat
of 3R allele (G>C) and a 6-bp deletion or insertion in 3′-UTR of
the TS gene. Preclinical study revealed that the triple repeat
occurring in 5′-UTR plus the 6-bp insertion conferred a higher
transcriptional efficiency and greater mRNA stability than the
double repeat plus 6-bp deletion in vitro (10,11).
The association between these polymorphisms and efficacy of
5-Fu-based chemotherapy has been demonstrated in certain solid
tumors, such as gastric, colorectal and breast cancer (12–14).
A previous study suggested that VNTR and SNP in the 5′-UTR of the
TS gene in combination with a C667T polymorphism of
methylenetetrahydrofolate reductase (MTHFR) were associated with
prognosis of NSCLC (15). However,
there was no difference in prognosis between different genotypes
when TS and MTHFR groups were considered separately.
Based on the data mentioned above, the present study
was conducted to further investigate the association between
polymorphisms of the TS gene and efficacy of PEM-based treatment in
advanced NSCLC.
Materials and methods
Patients
This study was approved by the ethics and research
committee of our hospital. Data were obtained from 90 patients
presenting with advanced NSCLC receiving PEM-based treatment
between March 2009 and September 2011 at the Department of Medical
Oncology in Shanghai Pulmonary Hospital. Diagnoses were confirmed
by cytological or histological examinations. The extent of disease
was determined according to the 7th edition of the American Joint
Committee on Cancer/International Union Against Cancer (AJCC/UICC)
classification (16,17). Non-smokers were defined as having
smoked fewer than 100 cigarettes in their lifetime and without use
of other tobacco products.
DNA extraction and genotyping
Peripheral blood samples were drawn into ethylene
diamine tetra-acetic acid (EDTA)-containing tubes and preserved at
−20°C in a refrigerator for final test. Genomic DNA was extracted
from 100 μl of blood using the Takara genomic DNA extraction kit
(Takara Biotechnology, China). Polymorphisms were detected by
Taqman probes designed and synthesized by Invitrogen. The sequences
of probes and primers are listed in Table I. PCR analysis was performed as
previously reported (9). PCR
products were confirmed by capillary electrophoresis) and then
detected using a 3730XL DNA detection device (Applied Biosystems,
USA). Polymerase chain reaction (PCR) for TS 3′-UTR genotyping was
carried out in a volume of 20 μl solution containing 1.0 μl genomic
DNA, 9.7 μl ultrapure water, 2 μl 10X PCR buffer, 1.2 μl 50 mmol/l
MgCl2, 0.5 μl 10 mmol/l (10 mM) dNTPs, 4 μl 5X GC
buffer, 0.5 μl of each primer (10 μM), 0.2 μl of probe-FAM (10 μM),
0.2 μl of probe-VIC (10 μM) and 0.2 μl 5 U/μl Taq DNA polymerase
(Invitrogen). Cycling parameters were as follows: pre-PCR at 50°C
for 1 min; held at 95°C for 10 min; 40 cycles of 95°C for 15 sec,
60°C for 1 min; and post-PCR at 60°C for 1 min. PCR was performed
using QPCR 7500 fast instrument supplied by Applied Biosystems
(USA). PCR products were analyzed using a real-time fluorescent PCR
allele discrimination system for genotyping.
 | Table ISequences of the primers and
probes. |
Table I
Sequences of the primers and
probes.
| Primer/probes | Sequences
(5′-3′) |
|---|
| Primers | |
| rs2853542 | |
| Forward |
GTGGCTCCTGCGTTTCCCCC |
| Reverse |
GCGGAGGATGTGTTGGATCT |
| rs16430 | |
| Forward |
CGTGGACGAATGCAGAACACT |
| Reverse |
TTCACAAGCTATTCCCTCAAATCT |
| Probes | |
| rs16430-del |
FAM-ACAACTATAAAGTTCATAACCA-MGB |
| rs16430-CTTTAA |
VIC-ATAACTTTAAAGTTCATAACC-MGB |
Treatment
Each patient received at least one cycle of
PEM-based treatment. PEM was administered at 500
mg/m2/cycle every 3 weeks with platinum regimens as
first-line treatment or a single-agent treatment in second-line or
further. All patients received premedication with folic acid (0.4
mg/day) and vitamin B12 (1 mg intramuscularly every 9 weeks). Oral
corticosteroids (fluorodexamethasone 4 mg, twice/day) and
ranitidine (0.15 g, twice/day) were administered on the day before,
the day of and the day after chemotherapy. The number of cycles was
documented.
Evaluation of efficacy
The Response Evaluation Criteria in Solid Tumors
(RECIST) version 1.1 was used to assess the efficacy independently
(18). Complete response (CR) was
defined as the disappearance of all target lesions as well as any
pathological lymph nodes (whether target or non-target) reduced to
<10 mm in short axis. Partial response (PR) was defined as a
decrease of no less than 30% in the sum of diameters of target
lesions compared with the baseline sum diameters. Progressive
disease (PD) was defined as an increase of no less than 20% in the
sum of diameters of target lesions compared with the smallest sum.
In addition to the relative increase of 20%, the sum must also
demonstrate an absolute increase of at least 5 mm. The appearance
of one or more new lesions was also considered progression
according to the new criteria. As for non-target lesions, the
assessment was also performed complying with RECIST version
1.1.
Statistical analyses
The effect of each TS polymorphism was assessed
independently and in combination. 5′ polymorphisms were categorized
as 2R if the genotype contained a functional 2R allele (2R/2R,
2R/3R) and 3R if the genotype contained a 3R allele (3R/3R, 3R/4R)
(9). 3′ polymorphisms were
categorized as ins6 if the genotype was ins6/ins6 and del6 if
genotypes were ins6/del6 and del6/del6 (19). The combined effects of TS
polymorphisms were analyzed by grouping the 5′ and 3′ genotypes,
yielding three categories, 2R&del6, 2R&ins6 and
3R&del6. The statistical analyses were performed using SPSS
version 16. The interval between dates of first administration of
PEM and first recorded disease progression, unacceptable toxicity
or last visit was defined as progression-free survival (PFS). The
date of the last visit was December 30, 2011. Chi-square test and
Pearson’s test were used to carry out univariate analyses. Survival
curves were generated by the Kaplan-Meier method and statistical
differenceswere evaluated using the log-rank test. The significance
level of p-value was set at p<0.05.
Results
Patients and polymorphisms
A total of 90 patients were studied and their
baseline features are listed in Table
II. The majority of the patients were under 70 years of age
(79/87.8%). Each gender represented half of the study population.
There was a dominant proportion (86/95.6%) of stage IV NSNSCLC. The
number of non-smokers was greater than that of smokers, accounting
for 62.2% of the entire group. The majority of patients (56/62.2%)
received PEM in second-line or further. The median cycle of PEM
administration was 4. At the final cut-off date (December 30,
2011), more than 80% of the patients had experienced progressive
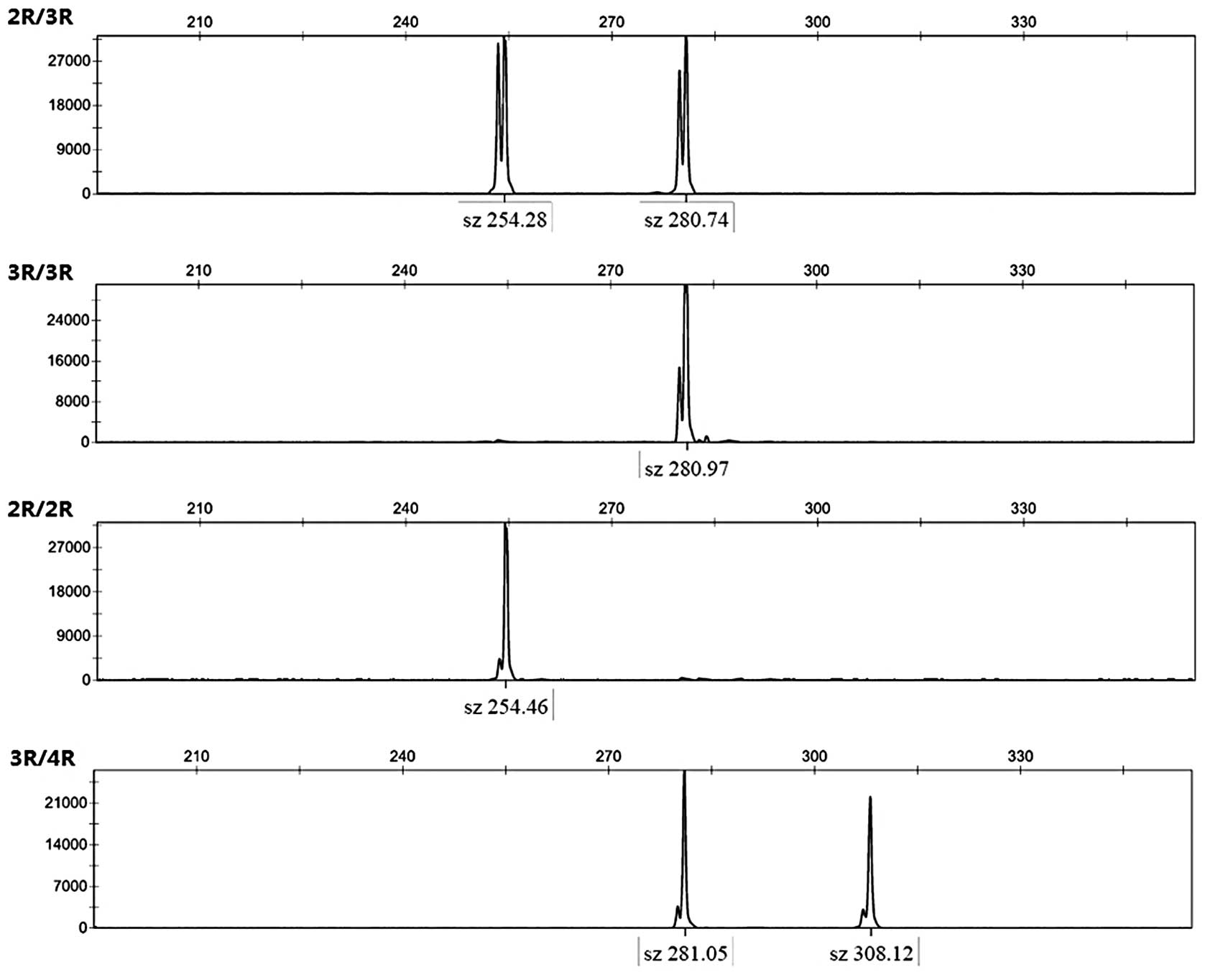
disease. Four types of VNTR in the 5′-UTR region of the TS gene
were detected (Fig. 1). The
discrimination system detected three polymorphisms located in the
3′-UTR region (Fig. 2). Among the
study population, 2 patients exhibited 2R/2R (2.2%), 54 exhibited
3R/3R (60%), 33 exhibited 2R/3R (36.7%) and one exhibited 3R/4R
(1.1%). Distribution of the 3′-UTR polymorphism was: 40 (44.4%)
cases of del6/del6, 43 (47.8%) of ins6/del6 and 7 (7.8%) of
ins6/ins6. No differences were observed in the distribution of
various genotypes according to age, gender, smoking status, line of
treatment and cycles (Table
III).
 | Table IIDemographic and baseline
characteristics of the study population. |
Table II
Demographic and baseline
characteristics of the study population.
| Characteristics | No. of patients
(%) |
|---|
| Overall | 90 (100) |
| Age (years) | median 58; range,
34–78 |
| ≤70 | 79 (87.8) |
| >70 | 11 (12.2) |
| Performance
status | |
| 0/1 | 90 (100) |
| Gender | |
| Male | 45 (50) |
| Female | 45 (50) |
| Clinical stage | |
| IIIB | 4 (4.4) |
| IV | 86 (95.6) |
| Smoking status | |
| Non-smoker | 56 (62.2) |
| Smoker | 34 (37.8) |
| Line of
treatment | |
| First-line | 34 (37.8) |
| Second-line or
further | 56 (62.2) |
| Cycle | |
| ≤4 | 64 (71.1) |
| >4 | 26 (28.9) |
| Histological
type | |
| Adenocarcinoma | 83 (92.2) |
|
Non-adenocarcinoma | 7 (7.8) |
| PD events | |
| PD | 75 (83.3) |
| Non-PD | 15 (16.7) |
 | Table IIIAssociation between polymorphisms and
patient clinical features. |
Table III
Association between polymorphisms and
patient clinical features.
| A, 5′-UTR | | | |
|
| Variable | 2R/2R+2R/3R n
(%) | 3R/3R+3R/4R n
(%) | P-value
|
| Age (years) | | | |
| ≤70 | 28 (35.4) | 51 (64.6) | |
| >70 | 7 (63.6) | 4 (36.4) | 0.072 |
| Gender | | | |
| Male | 21 (46.7) | 24 (53.3) | |
| Female | 14 (31.1) | 31 (68.9) | 0.130 |
| Smoking status | | | |
| Non-smoker | 22 (39.3) | 34 (60.7) | |
| Smoker | 13 (38.2) | 21 (61.8) | 0.921 |
| Line of
treatment | | | |
| First-line | 12 (35.3) | 22 (64.7) | |
| Second-line or
further | 23 (41.1) | 33 (58.9) | 0.586 |
| Cycle | | | |
| ≤4 | 27 (42.2) | 37 (57.8) | |
| >4 | 8 (30.8) | 18 (69.2) | 0.314 |
|
| B, 3′-UTR | | | |
|
| Variable | Ins6/del6+del6/del6
n (%) | Ins6/ins6 n
(%) | P-value |
|
| Age (years) | | | |
| ≤70 | 74 (93.7) | 5 (6.3) | |
| >70 | 9 (81.8) | 2 (18.2) | 0.064 |
| Gender | | | |
| Male | 42 (93.3) | 3 (6.7) | |
| Female | 41 (91.1) | 4 (8.9) | 0.987 |
| Smoking status | | | |
| Non-smoker | 51 (91.1) | 5 (8.9) | |
| Smoker | 32 (94.1) | 2 (5.9) | 0.601 |
| Line of
treatment | | | |
| 1st-line | 32 (94.1) | 2 (5.9) | 0.601 |
| 2nd-line or
further | 51 (91.1) | 5 (8.9) | |
| Cycle | | | |
| ≤4 | 59 (92.2) | 5 (7.8) | |
| >4 | 24 (92.3) | 2 (7.7) | 0.985 |
Association between polymorphisms and
clinical outcomes
All patients were available for evaluation of
response rate and PFS. For the entire study population, the
objective response rate (ORR) and the disease control rate (DCR)
was 11.1 and 65.5%, respectively. Median PFS was 112 days. In the
5′-UTR group, patients harboring 2R alleles had similar clinical
outcomes compared with those harboring 3R alleles (PFS, p=0.518;
DCR, p=0.631; ORR, p=0.541), as shown in Table IV and Fig. 3A. In 3′-UTR group, no difference
was observed between patients carrying 6- allele (del6/del6 and
ins6/del6) and those carrying 6+ allele (ins6/ins6), as shown in
Table IV and Fig. 3B. Median PFS were 112 and 147 days,
respectively (p=0.776). Combination of genotypes did not yield
significant differences among different groups. In 2R&del6,
2R&ins6 and 3R&del6 subset, DCR was 74.1, 57.1 and 62.5%,
respectively (p=0.517). Accordingly, ORR was 11.1, 0 and 12.5%
(p=0.611); median PFS was 86, 147 and 126 days (p=0.938) (Table IV and Fig. 3C).
 | Table IVAssociation between polymorphisms and
the efficacy of pemetrexed-based treatment in advanced NSCLC. |
Table IV
Association between polymorphisms and
the efficacy of pemetrexed-based treatment in advanced NSCLC.
| Genotype | Frequency n
(%) | DCR n (%) | ORR n (%) |
|---|
| 2R/2R+2R/3R | 35 (38.9) | 24 (68.6) | 3 (8.6) |
| 3R/3R+3R/4R | 55 (61.1) | 35 (63.6) | 7 (12.7) |
| P-value | 0.631 | 0.541 | |
|
Ins6/del6+del6/del6 | 83 (92.2) | 55 (66.3) | 10 (12) |
| Ins6/ins6 | 7 (7.8) | 4 (57.1) | 0 (0) |
| P-value | 0.626 | 0.330 | |
| 2R/2R&6-/6+;
2R/3R&6-/6-; 2R/3R&6-/6+ | 28 (31.1) | 20 (74.1) | 3 (11.1) |
| 2R/2R&6+/6+;
2R/3R&6+/6+ | 7 (7.8) | 4 (57.1) | 0 (0) |
| 3R/4R&6+/6-;
3R/3R&6+/6-; 3R/3R&6-/6- | 55 (61.1) | 34 (62.5) | 7 (12.5) |
| P-value | | 0.517 | 0.611 |
Discussion
In this study, we evaluated the correlation between
polymorphisms in the TS gene and clinical outcomes of patients with
advanced NSCLC following PEM-based chemotherapy. To our knowledge,
this is the first study investigating the role of polymorphisms of
the TS gene in PEM-treated advanced NSCLC patients. We demonstrated
the lack of association between the polymorphisms and the efficacy
of PEM-based treatment in advanced NSCLC. No significant
differences were observed in ORR, DCR and PFS according to various
genotypes. To our knowledge, this is the first study conducted in
advanced NSCLC.
We noted an imbalance of stage and histological type
in our study population. Due to the introduction of a new staging
classification (16,17), some patients with malignant pleural
effusion who were previously categorized as stage IIIb were
re-categorized as stage IV. PEM is recommended for adenocarcinomas
in first- and second-line treatment (20), and more than 90% of the study
population presented with ADC. Due to the large disparity existing
in these two subsets, we did not assess the association between
polymorphisms and different stages or histological types.
Frequencies of TS polymorphisms reported by Takehara
et al comprised 20 cases (6.8%) of 2R/2R, 75 (25.4%) of
2R/3R, 199 of 3R/3R and 1 of 3R/5R (67.8%) (15). Furthermore, TS VNTR did not show
any significant association with clinicopathological factors in
this Japanese study. The distribution of 3′-UTR polymorphism in a
Chinese study was documented as follows: 7.6% ins6/ins6, 44.3%
ins6/del6 and 48.1% del6/del6 (19). Based on the same ethnicity, our
study also observed a similar distribution of the 5′-UTR
polymorphism, with 2.2% of patients harboring 2R/2R, 60% harboring
3R/3R, 36.7% harboring 2R/3R and one with 3R/4R. The incidence of
3′-UTR polymorphisms was also consistent with those described in a
previous publication (10). Our
study has confirmed the lack of association between TS
polymorphisms and clinical features in advanced NSCLC. However,
when the imbalance of stage and histological type are taken into
account, there may be deviations in the results, as the analyses
were performed in a highly selected population. Therefore, our
results may not serve as robust evidence.
Although TS VNTR and polymorphisms in the 3′-UTR
region were demonstrated to be associated with efficacy of
5-Fu-based treatment, the majority of existing studies were
performed in gastrointestinal cancers. Huang et al (21) collected 116 gastric cancer cases
who received 5-Fu-based adjuvant chemotherapy. The study found a
significant shorter overall survival (OS) in patients with TS
ins6/ins6 genotype (20.7 months) compared with those with del6/del6
(29.8 months) (p=0.017) and ins6/del6 (41.0 months) (p=0.022)
genotypes. Median relapse-free survival (RFS) was 11.5 months for
the ins6/ins6 group, 20.8 for the ins6/del6 group and 36.9 for the
del6/del6 subset. Another study on stomach cancer also found a
significant correlation between polymorphisms in 3′-UTR of the TS
gene and the sensitivity of gastric cancer to 5-Fu-based
chemotherapy (19). The response
rates of the del6/del6 and del6/ins6 groups were significantly
higher than that of the ins6/ins6 group (p=0.045).
Notably, TS expression was demonstrated to be
different in various histological types (8,9). The
regulation of TS expression may be influenced by numerous uncertain
factors (22). Due to the
complicated communication between molecules occurring in the
signaling pathways, it is assumed that the complex modulation of
signaling pathways may also lead to a different expression level of
TS in patients with tumors located in different organs and further
result in different clinical outcomes. Gorlick et al
reported a higher TS mRNA expression in pulmonary metastases (mean
TS/β-actin ratio 19.7) when compared with hepatic lesions (mean
TS/β-actin ratio 4.7) of colorectal cancer (23). Therefore, the TS expression in lung
adenocarcinoma may differ from gastrointestinal cancers, which may
also be a reasonable explanation for the converse results of our
analysis and previous studies. However, we did not obtain data
regarding TS expression. This was a limitation of our analysis in
addition to the small scale of the study population.
In conclusion, our investigation demonstrated that
the polymorphisms of the TS gene were unable to predict response to
PEM-based treatment. However, this study was performed in a highly
selected population. Therefore, further investigation should be
conducted to yield a deeper insight into the overall value of these
markers.
References
|
1
|
Rahman L, Voeller D, Rahman M, et al:
Thymidylate synthase as an oncogene: a novel role for an essential
DNA synthesis enzyme. Cancer Cell. 5:341–351. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Scagliotti GV, Parikh P, von Pawel J, et
al: Phase III study comparing cisplatin plus gemcitabine with
cisplatin plus pemetrexed in chemotherapy-naive patients with
advanced-stage non-small-cell lung cancer. J Clin Oncol.
26:3543–3551. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Hanna N, Shepherd FA, Fossella FV, et al:
Randomized phase III trial of pemetrexed versus docetaxel in
patients with non-small-cell lung cancer previously treated with
chemotherapy. J Clin Oncol. 22:1589–1597. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Ciuleanu T, Brodowicz T, Zielinski C, et
al: Maintenance pemetrexed plus best supportive care versus placebo
plus best supportive care for non-small-cell lung cancer: a
randomised, double-blind, phase III study. Lancet. 374:1432–1440.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Scagliotti G, Brodowicz T, Shepherd FA, et
al: Treatment-byhistology interaction analyses in three phase III
trials show superiority of pemetrexed in nonsquamous non-small cell
lung cancer. J Thorac Oncol. 6:64–70. 2011.PubMed/NCBI
|
|
6
|
Tanaka F, Wada H, Fukui Y and Fukushima M:
Thymidylate synthase gene expression in primary lung cancer
patients: a large-scale study in Japanese population. Ann Oncol.
22:1791–1797. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kaira K, Ohde Y, Nakagawa K, Okumura T, et
al: Thymidylate synthase expression is closely associated with
outcomes in patients with pulmonary adenocarcinoma. Med Oncol.
29:1663–1672. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Sun JM, Han J, Ahn JS, Park K and Ahn MJ:
Significance of thymidylate synthase and thyroid transcription
factor 1 expression in patients with nonsquamous non-small cell
lung cancer treated with pemetrexed-based treatment. J Thorac
Oncol. 6:1392–1399. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
De Bock CE, Garg MB, Scott N, Sakoff JA,
Scorgie FE, Ackland SP and Lincz LF: Association of thymidylate
synthase enhancer region polymorphisms with thymidylate synthase
activity in vivo. Pharmacogenomics J. 11:307–314. 2011.PubMed/NCBI
|
|
10
|
Mandola MV, Stoehlmacher J, Zhang W, et
al: A 6 bp polymorphism in the thymidylate synthase gene causes
message instability and is associated with decreased intratumoral
TS mRNA levels. Pharmacogenetics. 14:319–327. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mandola MV, Stoehlmacher J, Muller-Weeks
S, Cesarone G, Yu MC, Lenz HJ and Ladner RD: Ladner, a novel single
nucleotide polymorphism within the 5′ tandem repeat polymorphism of
the thymidylate synthase gene abolishes USF-1 binding and alters
transcriptional activity. Cancer Res. 63:2898–2904. 2003.
|
|
12
|
Lurje G, Manegold PC, Ning Y, Pohl A,
Zhang W and Lenz HJ: Thymidylate synthase gene variations:
predictive and prognostic markers. Mol Cancer Ther. 8:1000–1007.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Shitara K, Muro K, Ito S, et al: Folate
intake along with genetic polymorphisms in
methylenetetrahydrofolate reductase and thymidylate synthase in
patients with advanced gastric cancer. Cancer Epidemiol Biomarkers
Prev. 19:1311–1319. 2010. View Article : Google Scholar
|
|
14
|
Kumar K, Vamsy M and Jamil K: Thymidylate
synthase gene polymorphisms effecting 5-FU response in breast
cancer patients. Cancer Biomark. 6:83–93. 2010.PubMed/NCBI
|
|
15
|
Takehara A, Kawakami K, Ohta N, Oyama K,
Ota Y, Oda M and Watanabe G: Prognostic significance of the
polymorphisms in thymidylate synthase and methylenetetrahydrofolate
reductase gene in lung cancer. Anticancer Res. 25:4455–4461.
2005.
|
|
16
|
Sobin LH, Gospodarowicz MK and Wittekind
CH: International Union Against Cancer (UICC) TNM Classification of
Malignant Tumours. 7th edition. Wiley-Blackwell; Oxford: pp. 1–256.
2009
|
|
17
|
Edge SB, Byrd DR, Carducci MA, et al:
American Joint Committee on Cancer (AJCC) Cancer Staging Handbook.
7th edition. Springer-Verlag; New York: pp. 1–195. 2009
|
|
18
|
Eisenhauer EA, Therasse P, Bogaerts J, et
al: New response evaluation criteria in solid tumours: revised
RECIST guideline (version 1.1). Eur J Cancer. 45:228–247. 2009.
View Article : Google Scholar
|
|
19
|
Lu JW, Gao CM, Wu JZ, Cao HX, Tajima K and
Feng JF: Polymorphism in the 3′-untranslated region of the
thymidylate synthase gene and sensitivity of stomach cancer to
fluoropyrimidine-based chemotherapy. J Hum Genet. 51:155–160.
2006.
|
|
20
|
Ettinger DS, Akerley W, Borghaei H, et al:
NCCN Clinical Practice Guidelines in Oncology. version 2.2012.
National Comprehensive Cancer Network; Fort Washington, PA: pp.
1–112. 2011, Available at https://www.nccn.com.
|
|
21
|
Huang ZH, Hua D and Li LH: The
polymorphisms of TS and MTHFR predict survival of gastric cancer
patients treated with fluorouracil-based adjuvant chemotherapy in
Chinese population. Cancer Chemother Pharmacol. 63:911–918. 2009.
View Article : Google Scholar
|
|
22
|
Giovannetti E, Backus HH, Wouters D, et
al: Changes in the status of p53 affect drug sensitivity to
thymidylate synthase (TS) inhibitors by altering TS levels. Br J
Cancer. 96:769–775. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Gorlick R, Metzger R, Danenberg KD, et al:
Higher levels of thymidylate synthase gene expression are in
pulmonary as compared with hepatic metastases of colorectal
adenocarcinoma. J Clin Oncol. 16:1465–1469. 1998.PubMed/NCBI
|