Introduction
Superficial siderosis of the central nervous system
(SSCNS) is a rare disease of the CNS resulting from hemosiderin
deposits in neuronal tissues close to the cerebrospinal fluid.
Since this disease was first reported by Hamill in 1908 (1), there have been only ~300 reported
cases of SSCNS in the literature (2). The clinical syndrome is characterized
by sensorineural deafness, cerebellar ataxia, dementia and signs of
pyramidal tract dysfunction (3).
SSCNS is mainly caused by subarachnoid hemorrhage due to idiopathic
bleeding, which accounts for 35% of all cases (4). Other known causes of subarachnoid
hemorrhage leading to SSCNS include brain tumors, trauma and
vascular abnormalities (4). To
date, no effective treatment is available for SSCNS. In the past,
SSCNS was primarily diagnosed by postmortem examination or surgical
biopsy (3). However, with advances
in radiological imaging techniques, cranial magnetic resonance
imaging (MRI) has become the primary method for the diagnosis of
SSCNS (5). MRI can detect the
presence of hemosiderin on the surface of the brain and spinal cord
with considerable sensitivity.
The present study reports the case of a 48-year-old
male patient who was diagnosed with SSCNS using MRI. The patient
presented with dizziness, ataxia and nystagmus, but without the
typical SSCNS symptom of hearing loss.
Case report
A 48-year-old male patient was admitted to the First
Hospital of Jilin University (Changchun, China) on October
11th 2013 after presenting with dizziness, ataxia and
slurred speech, with symptoms aggravating over five months. The
patient’s speech had begun to slur five months previously with no
evident cause; however, the patient did not suffer from any
communication difficulties. The patient also exhibited ataxia and
had lost the ability to walk independently; however, the lower
extremities of the patient moved normally while in the supine
position. These symptoms worsened progressively, developing into
blurred vision and dizziness. The patient exhibited no hearing loss
in either ear, and no headache, nausea or vomiting. The patient had
suffered from a pontine hemorrhage two years previously, with a
sequelae of mild slurred speech and ataxic gait, but was able to
walk independently at that time. In addition, the patient had
experienced hypertension for two years, but had no history of
diabetes or venereal disease exposure. Written informed consent was
obtained from the patient prior to their inclusion in the present
study.
On examination, the patient was conscious and
exhibited dysarthria. Both eyes exhibited horizontal and rotary
nystagmus, and the pupils were round and equal in diameter (3.0 mm)
with a normal pupillary light reflex. The patient showed normal
symmetrical nasolabial folds, a normal midline tongue protrusion,
bilateral powerful lift of the soft palate and a reduced gag
reflex. Muscle strength was 5/5 in the extremities, with normal
muscle tone and tendon reflexes. The Chaddock sign was positive,
and the deep and shallow sensations were normal. Responses to the
finger-nose pointing and heel-knee-shin tests were unsteady and
inaccurate. The patient exhibited a positive Romberg’s sign, while
meningeal irritation and the Kernig sign were negative.
Laboratory tests were normal, consisting of routine
blood and urine analyses, blood coagulation, liver and kidney
functions and blood ion concentrations. In addition, the patient’s
antinuclear antibody, anti-O antibody, erythrocyte sedimentation
rate, thyroid function and tumor markers were all normal. A lumbar
puncture revealed clear and colorless cerebrospinal fluid with a
pressure of 160 mmH2O. The white blood cell count was
zero, the protein level was 0.31 g/l, the glucose level was 2.9
mmol/l and the chloride content was 126 mmol/l in the cerebrospinal
fluid. Pandy’s test, as well as tests for Mycobacterium
tuberculosis, Cryptococcus neoformans, Treponema
pallidum and HIV were all negative.
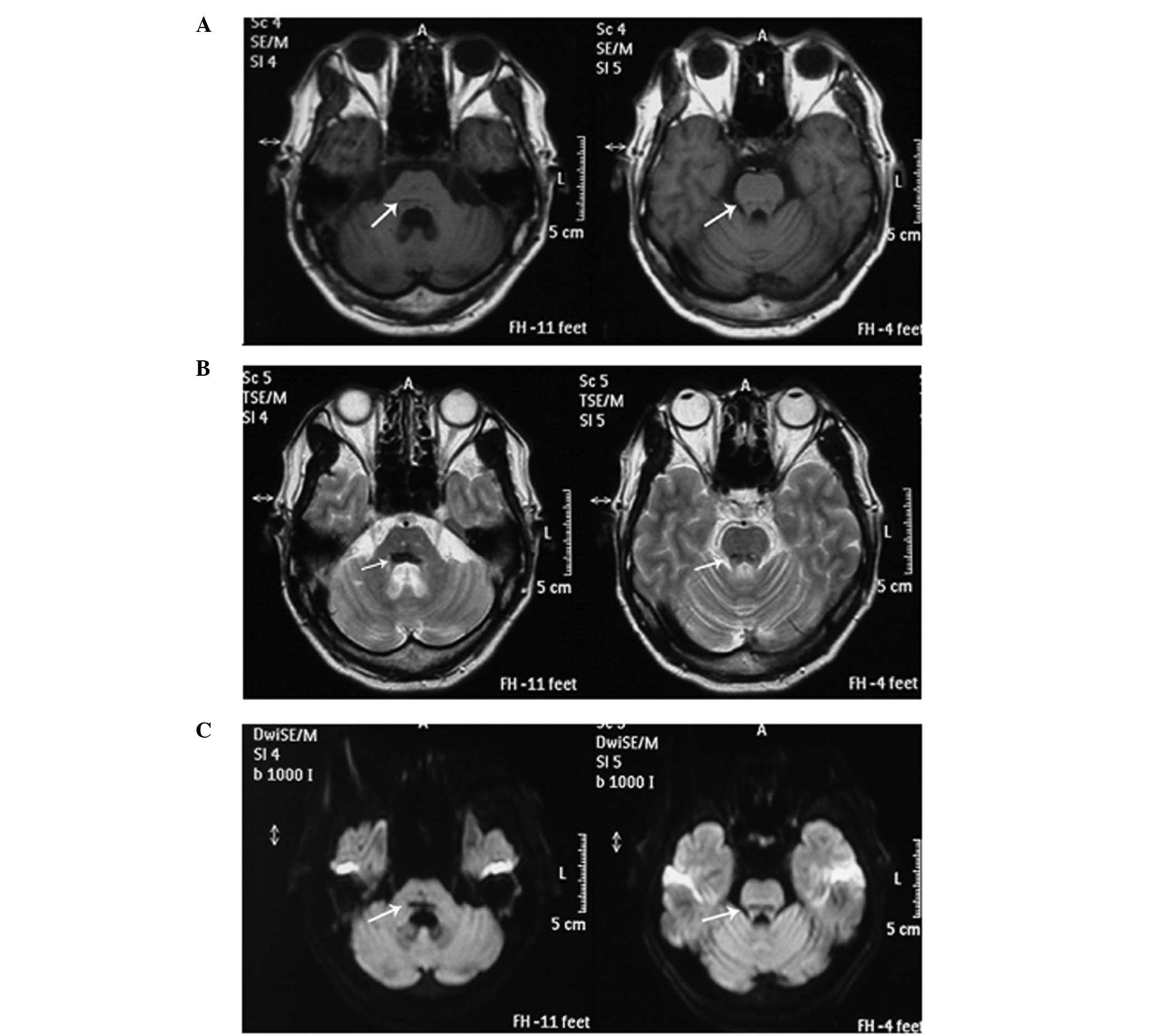
Cross-sectional MRI scans of the cranium were
captured using T1-weighted imaging (T1WI), T2-weighted imaging
(T2WI) and diffusion-weighted imaging (DWI), which revealed areas
of linear hypointensity on the surface of the pons (Fig. 1). The size of hypointensity due to
hemosiderin was larger in the T2WI scans than in the T1WI and DWI
scans. The images revealed widened and deepened sulci and gyri, an
enlarged ambient cistern and mild cerebellar atrophy. The patient
received oral administration of vitamins B1 (20 mg, 3 times/day)
and B12 (0.05 mg, 3 times/day) for 2 months. The patient was
followed up at 2 months following discharge. Audiometric tests
showed normal hearing during the hospital stay and at the two-month
follow-up examination.
Discussion
SSCNS is a rare disease caused by chronic repeated
subarachnoid hemorrhage (3).
Clinical symptoms can occur as late as 8–37 years after the initial
onset of the subarachnoid hemorrhage (6). In the present study, the patient
showed a slow disease progression, presenting with clinical
symptoms of ataxia, dizziness and slurred speech two years after
the initial pontine hemorrhage.
Idiopathic bleeding is the major source of
subarachnoid bleeding, accounting for 35% of all cases, which is
followed by CNS tumors (15%), brain trauma (13%) and arteriovenous
malformation (9%) (4). Other rare
sources of subarachnoid bleeding include subdural surgery, brachial
plexus injury and nerve root avulsion. The pathology of SSCNS
primarily results from hemosiderin deposits and subsequent
impairment of the surface of the cerebrum, cerebellar vermis,
cranial nerve VIII and corpora quadrigemina, as well as the
longitudinal and lateral fissures and sulci of the brain (7) and pia mater of the spinal cord
(8). Macroscopically, a brownish
discoloration is observed in the leptomeninges and the adjacent
surface of the brain parenchyma and ventricular walls. Adhesion in
the subarachnoid space is often observed. Microscopically, there is
hemosiderin deposition, leptomeningeal fibrosis, neuronal loss and
increased macrophage infiltration, as well as reactive gliosis,
axonal demyelination and intracellular ovoid bodies (9).
Prior to the development of MRI, the diagnosis of
SSCNS depended primarily on postmortem examination or surgical
biopsy (3). MRI has become the
primary method for diagnosis of SSCNS since first being used for
this purpose in 1985 (5). The
appearance of SSCNS in MRI scans is due to the presence of ferric
ions on the surface of CNS tissues (5). Compared with T2WI, head
susceptibility-weighted imaging is more sensitive to hemosiderin
and can more accurately detect hypointense signals in the brain
(3), which provides more valuable
imaging information for the diagnosis of SSCNS. Although fast
fluid-attenuated inversion-recovery MRI is sensitive to acute
subarachnoid hemorrhage (10), it
is limited in the detection of hemosiderin deposits.
T2WI MRI reveals a characteristic linear
hypointensity due to hemosiderin deposition that is in contrast to
the hyperintense signals of the cerebrospinal fluid (11). Therefore, since the characteristic
hypointensity is easily detected by T2WI MRI, this technique is
often used to diagnose SSCNS. In addition, cerebellar atrophy is
commonly identified by cranial MRI (12). In the present case, a
characteristic linear hypointensity on the surface of the pons with
mild cerebellar atrophy was observed in the T2WI MRI scans,
supporting the diagnosis of SSCNS.
The characteristic clinical features of SSCNS are
bilateral sensorineural deafness, progressive cerebellar ataxia and
pyramidal tract signs (3). Ali
et al (13) reported that
sensorineural deafness occurred in 95% of SSCNS cases, ataxia in
88% and signs of pyramidal tract dysfunction in 76%. In cases of
ataxia due to injury of the cerebellar vermis, gait ataxia occurs
more commonly than limb ataxia in SSCNS patients (8). Other clinical symptoms of SSCNS
include dementia, loss of smell, bladder dysfunction, somatic
sensory dysfunction, unequally sized pupils, headache and back pain
(14). In addition, certain SSCNS
patients experience the sudden onset of headaches and
meningitis-like symptoms (15). In
rare cases, extraocular muscle paralysis, dysarthria, sciatica and
motor neuron damage in the lower extremities also occur. In the
early stages of the disease, symmetrical or asymmetrical
high-frequency hearing loss often occurs, and dizziness can occur
when the brain lesions involve cranial nerve VIII (3).
In the present case, the 48-year-old patient
presented with clinical symptoms and signs of ataxia that consisted
of dizziness, nystagmus, dysarthria and abnormal finger-nose
pointing and heel-knee-shin tests two years after suffering from a
pontine hemorrhage. In addition, the patient exhibited a positive
Chaddock sign, suggesting that the pyramidal tract was impaired.
These clinical symptoms were consistent with the diagnosis of
SSCNS. The characteristic areas of linear hypointensity on MRI
further confirmed the diagnosis of SSCNS.
In the present case, SSCNS was likely to have been
caused by the pontine hemorrhage. The onset of clinical symptoms
varies greatly among SSCNS patients. A number of SSCNS patients
present no clinical symptoms during their lifetime and are only
diagnosed by postmortem autopsy. In rare cases, SSCNS patients may
present clinical symptoms in the early stages of the disease;
however, the majority of patients exhibit no clinical symptoms
(16). SSCNS may be found
incidentally by cranial MRI, showing the presence of hemosiderin
deposits. The onset and duration of clinical symptoms may be
determined by the source and extent of the bleeding.
One of the most common clinical symptoms of SSCNS is
sensorineural deafness. However, in the present case, the patient
did not present with any signs of hearing loss. The development of
sensorineural deafness is a chronic process and the progressive
deterioration of hearing commonly lasts for several years until
hearing is completely lost (17).
Although the current patient had normal hearing at the two-month
follow-up examination, a long-term follow-up is required to monitor
the hearing of the patient.
Currently, no effective treatment is available for
SSCNS. Surgery is feasible for the removal of blood from the site
of hemorrhage, and it is possible to surgically correct the cause
of the subarachnoid hemorrhage, such as nerve root avulsion,
arteriovenous malformation and dural lesions. However, surgery has
been effective in very few patients, with a number of patients
experiencing aggravated symptoms following surgery (18). Ion chelation, large doses of
vitamin C and E and hormones (alone or in combination with
immunosuppressive therapy) have been clinically used, and are able
to effectively halt disease progression in certain patients
(19). Han et al (20) found that penicillamine was
effective in the treatment of SSCNS. In the present case, hormone
therapy with dexamethasone (started at a 15-mg dose and gradually
tapered, once daily for one month) was found to reduce dizziness,
with the patient able to walk with the aid of a walker after
receiving the treatment.
In summary, SSCNS is a rare disease that is easily
overlooked by clinicians. The present study described the case of
an SSCNS patient who presented with dizziness, ataxia and
nystagmus, without loss of hearing. MRI is important for the
diagnosis of SSCNS, and improving the understanding of SSCNS is
important for clinicians to identify SSCNS patients who do not
exhibit typical clinical symptoms.
References
|
1
|
Hamill RC: Report of a case of melanosis
of the brain, cord, and meninges. J Nerv Men Dis. 35:5941908.
View Article : Google Scholar
|
|
2
|
Posti JP, Juvela S, Parkkola R and Roine
S: Three cases of superficial siderosis of the central nervous
system and review of the literature. Acta Neurochir (Wien).
153:2067–2073. 2011. View Article : Google Scholar
|
|
3
|
Fearnley JM, Stevens JM and Rudge P:
Superficial siderosis of the central nervous system. Brain.
118:1051–1066. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Sahin S, Agilkaya S and Karsidag S:
Superficial siderosis of the central nervous system: An unusual
cause for headache and hearing loss. Neurol Asia. 11:145–149.
2006.
|
|
5
|
Koeppen AH, Michael SC, Li D, et al: The
pathology of superficial siderosis of the central nervous system.
Acta Neuropathol. 116:371–382. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Anderson NE, Sheffield S and Hope JK:
Superficial siderosis of the central nervous system: a late
complication of cerebellar tumors. Neurology. 52:163–169. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Fearnley J and Rudge P: Treatment of
superficial siderosis of the central nervous system. Mov Disord.
10:6851995. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kumar N, Cohen-Gadol AA, Wright RA, et al:
Superficial siderosis. Neurology. 66:1144–1152. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Koeppen AH, Dickson AC, Chu RC and Thach
RE: The pathogenesis of superficial siderosis of the central
nervous system. Ann Neurol. 34:646–653. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Noguchi K, Seto H, Kamisaki Y, Tomizawa G,
Toyoshima S and Watanabe N: Comparison of fluid-attenuated
inversion-recovery MR imaging with CT in a simulated model of acute
subarachnoid hemorrhage. AJNR Am J Neuroradiol. 21:923–927.
2000.PubMed/NCBI
|
|
11
|
Kumar N: Superficial siderosis:
associations and therapeutic implications. Arch Neurol. 64:491–496.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Bracchi M, Savoiardo M, Triulzi F, et al:
Superficial siderosis of the CNS: MR diagnosis and clinical
findings. AJNR Am J Neuroradiol. 14:227–236. 1993.PubMed/NCBI
|
|
13
|
Ali S, Moriarty J, Mullatti N and David A:
Psychiatric comorbidity in adult patients with hypothalamic
hamartoma. Epilepsy Behav. 9:111–118. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kumar N, Miller GM, Piepgras DG and Mokri
B: A unifying hypothesis for a patient with superficial siderosis,
low-pressure headache, intraspinal cyst, back pain, and prominent
vascularity. J Neurosurg. 113:97–101. 2010. View Article : Google Scholar
|
|
15
|
Koeppen AH and Dentinger MP: Brain
hemosiderin and superficial siderosis of the central nervous
system. J Neuropathol Exp Neurol. 47:249–270. 1988. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Miliaras G, Bostantjopoulou S,
Argyropoulou M, Kyritsis A and Polyzoidis K: Superficial siderosis
of the CNS: report of three cases and review of the literature.
Clin Neurol Neurosurg. 108:499–502. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tyler GK, Martin TP and Baguley DM:
Systematic review of outcome of cochlear implantation in
superficial siderosis. Otol Neurotol. 33:976–982. 2012.PubMed/NCBI
|
|
18
|
Payer M, Sottas C and Bonvin C:
Superficial siderosis of the central nervous system: secondary
progression despite successful surgical treatment, mimicking
amyotrophic lateral sclerosis. Case report and review. Acta
Neurochir (Wien). 152:1411–1416. 2010. View Article : Google Scholar
|
|
19
|
Leussink VI, Flachenecker P,
Brechtelsbauer D, et al: Superficial siderosis of the central
nervous system: pathogenetic heterogeneity and therapeutic
approaches. Acta Neurol Scand. 107:54–61. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Han X, Tao HY and Wu JJ: A case of
superficial siderosis of the central nervous system and review of
the literature. Zhongguo Lin Chuang Shen Jing Ke Xue. 20:537–542.
2012.(In Chinese).
|