Introduction
Adenocarcinoma of the esophagogastric junction (AEG)
is a lethal malignancy originating from the distal esophagus and
the esophagogastric junction (1,2). The
incidence of AEG has increased rapidly worldwide during the past
two decades (3–6). The prognosis of AEG is poor due to
distant metastasis at the time of diagnosis and the limited
treatment options (7,8). Trastuzumab, a monoclonal antibody
targeting human epidermal growth factor receptor 2 (HER2), has
emerged as an effective therapeutic option for AEG when combined
with chemotherapy (9).
In a phase III, open-label, international,
randomized controlled trial, Bang et al (9) observed that patients treated with
trastuzumab plus chemotherapy had longer median follow-up and
median overall survival times compared with patients treated with
chemotherapy alone. The authors suggested that trastuzumab in
combination with chemotherapy may be a new standard option for the
first-line treatment of HER2-positive advanced gastric or
gastro-esophageal junction cancer (9). International National Comprehensive
Cancer Network Guidelines recommend the detection of HER2 in
patients with AEG in order to guide the selection of further
clinical treatment options (10).
However, trastuzumab treatment is invalid for nearly half of
HER2-positive patients (9), and the
mechanisms of drug resistance are currently unknown. Therefore, the
identification of sensitive predictive biomarkers for HER2-positive
patients in whom trastuzumab treatment is invalid is important for
the prognosis of advanced AEG.
Sex determining region Y-box 9 (SOX9) is a nuclear
transcription factor that belongs to the group E subgroup of the
SOX protein family, and its activity has been associated with
several types of cancer (11–13).
Chakravarty et al (14) and
Riemenschnitter et al (15)
suggested SOX9 to be a prognostic marker in adjuvant oncological
settings. It was reported that breast cancer patients with a high
SOX9 level following chemotherapy contributes to a negative
prognosis and shortened overall survival (15). More importantly, the predictive
efficacy of SOX9 is higher than that of other clinico-pathological
parameters, such as HER2-status or pathological stage (15). However, to the best of our knowledge,
the specific biological function of SOX9 in AEG has not yet been
reported.
In the present study, in vitro experiments
were performed on ESO26 cells to investigate the effect of SOX9 on
cell proliferation and apoptosis in the presence or absence of
trastuzumab. Furthermore, whether SOX9 affects the activation of
the phosphatidylinositol-3-kinase (PI3K)/AKT (also known as protein
kinase B) signaling pathway, an intracellular signaling pathway
that is involved in the resistance of gastric cancer to trastuzumab
(16,17), was explored. The results of this
study may elucidate the mechanism underlying trastuzumab resistance
and improve the efficacy of trastuzumab treatment.
Materials and methods
Cell culture and trastuzumab
treatment
The ESO26 cell line was purchased from the European
Collection of Cell Cultures (Salisbury, UK). The cells were
cultured in RPMI-1640 medium (Invitrogen; Thermo Fisher Scientific,
Inc., Waltham, MA, USA) supplemented with 2 mM glutamine
(Invitrogen; Thermo Fisher Scientific, Inc.) and 10% fetal bovine
serum (FBS; Invitrogen; Thermo Fisher Scientific, Inc.), and
maintained at 37°C in a humidified atmosphere of 5% CO2.
Trastuzumab was provided by Roche Pharmaceuticals Ltd. (Shanghai,
China). The cells at a density of 3×105 cells/well were
treated with 0, 0.5, 1, 2 and 4 mg/ml trastuzumab for 24 h in
RPMI-1640 medium.
Cell transfection
Small interfering RNA (Si)-control and si-SOX9 were
obtained from Zhonghong Boyuan Biological Technology Co., Ltd.
(Shenzhen, China), and were transfected into ESO26 cells using
Lipofectamine 2000 (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's instructions. Briefly, cells were
seeded into 6-well plates at a density of 3×105
cells/well and incubated at 3°C overnight. At 4 h prior to
transfection, the medium was changed to Opti-MEM (Invitrogen;
Thermo Fisher Scientific, Inc.). Si-control/si-SOX9 and
Lipofectamine 2000 were each diluted in 250 µl Opti-MEM, and then
mixed and incubated at 37°C for 20 min. The mixture was added to
each well and the cells were incubated at 37°C for 6 h. After that,
the medium was replaced with complete RPMI-1640 medium. Following
transfection, the cells were incubated with 4 mg/ml trastuzumab for
24 h in RPMI-1640 medium supplemented with 2 mM glutamine and 10%
FBS at 37°C.
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted from the ESO26 cells using
TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.)
according to the manufacturer's protocol. cDNA was then synthesized
from 1 µg total RNA using a SuperScript® VILO™ cDNA
Synthesis kit (Thermo Fisher Scientific Inc.). The temperature for
the reverse transcription reaction was at 42°C. Primers used for
PCR were: SOX9 forward, 5′-ttgagccttaaaacggtgct-3′ and reverse,
5′-tggtgttctgagaggcacag-3′; β-actin forward,
5′-ggacttcgagcaagagatgg-3′ and reverse, 5′-agcactgtgttggcgtacag-3′.
The qPCR was performed using the LightCycler 480 Instrument II
(Roche Diagnostics, Basel, Switzerland) using LightCycler 480
SYBR-Green I Master (Roche Diagnostics) in accordance with the
manufacturer's instructions. Thermocycler conditions were: 95°C for
10 min, followed by 40 cycles of 95°C for 15 sec, 60°C for 30 sec,
and 72°C for 1 min. All reactions were performed in triplicate. The
expression of SOX9 was normalized against the β-actin reference
gene using the 2−ΔΔCq method (18).
Western blot analysis
The ESO26 cells were harvested and lysed with
radioimmunoprecipitation assay lysis buffer (Beyotime Institute of
Biotechnology, Shanghai, China) at room temperature for 5 sec, and
then were centrifuged at 13,000 × g for 5 min at room temperature.
Protein concentration was quantified using a Bradford Protein Assay
kit (Beyotime Institute of Biotechnology). A total of 50 µg protein
per lane was separated by 10% SDS-PAGE, and then transferred onto
polyvinylidene fluoride membranes (Bio-Rad Laboratories, Inc.,
Hercules, CA, USA). The membranes were blocked by incubation with
5% non-fat milk at 4°C overnight, and washed with Tris-buffered
saline with Tween-20 (TBST) three times. Subsequently, the
membranes were incubated with the following primary antibodies:
SOX9 rabbit polyclonal antibody (ab26414; 1:400 dilution; Abcam,
Cambridge, MA, USA), phospho-AKT (Ser473) rabbit polyclonal
antibody (cat. no. 9271; 1:800 dilution; Cell Signaling Technology,
Inc., Beverly, MA, USA), AKT mouse monoclonal antibody (cat. no.
2967; 1:1,000 dilution; Cell Signaling Technology, Inc.) and GAPDH
mouse monoclonal antibody (ab8245; 1:1,000 dilution; Abcam) at 4°C
overnight. After washing with TBST, the membranes were incubated
with horseradish peroxidase (HRP)-labeled rabbit anti-mouse IgG
(ab6728; 1:2,000 dilution; Abcam) or HRP-labeled goat anti-mouse
IgG (ab6789; 1:2,000 dilution; Abcam) at 37°C for 2 h. The protein
bands were visualized using an ECL Western Blotting Detection kit
(Pierce; Thermo Fisher Scientific, Inc.). Protein levels were
analyzed using densitometric analysis using ImageJ software version
1.46 (National Institutes of Health; Bethesda, MD, USA). The
experiment was performed in triplicate.
MTT assay
MTT assay was used to investigate the proliferation
of the transfected ESO26 cells with and without trastuzumab
treatment. The cells were incubated in 96-well plates at a density
of 3×103 cells per well, and were exposed to 4 mg/ml
trastuzumab for 24, 48, 72 and 96 h. MTT solution (Sigma-Aldrich;
Merck KGaA, Darmstadt, Germany) was then added to each well, and
the cells were incubated at 37°C for 4 h. Medium with no cells was
used as the blank control. Subsequently, 150 µl dimethyl sulfoxide
(Sigma-Aldrich; Merck KGaA) was added to each well to dissolve the
formazan crystals. The optical density was measured at a wavelength
of 490 nm using a microplate reader (ELx800; BioTek Instruments,
Inc., Winooski, VT, USA).
Flow cytometry (FCM)
Cell apoptosis of the transfected ESO26 cells with
and without trastuzumab treatment was determined using an Annexin
V-fluorescein isothiocyanate (FITC) Apoptosis Detection kit
(BioVision Inc., Milpitas, CA, USA). The cells were collected and
washed with PBS. They were then resuspended in Binding Buffer, and
incubated with 5 µl Annexin V-FITC and 10 µl propidium iodide (PI)
at room temperature for 15 min in the dark. Subsequently, 400 µl
Binding Buffer were added. The stained cells were detected and cell
apoptosis rates were determined using a flow cytometer
(FACSCalibur; BD Biosciences, San Jose, CA, USA) with CellQuest Pro
software (BD Biosciences).
Statistical analysis
Statistical analysis was performed using SPSS 19.0
statistical analysis software (IBM Corp., Armonk, NY, USA). Data
are presented as the mean ± or + standard deviation. Statistical
significance for experiments was determined by one-way analysis of
variance followed by the least significant difference test.
P<0.05 was to indicate a statistically significant
difference.
Results
Effect of trastuzumab on SOX9
expression in ESO26 cells
To investigate the effect of trastuzumab on SOX9
expression, ESO26 cells were treated with various concentrations of
trastuzumab for 24 h, and then SOX9 expression was determined using
RT-qPCR and western blotting. The mRNA and protein levels of SOX9
were significantly upregulated in ESO26 cells treated with 1, 2 or
4 mg/ml trastuzumab compared with those in the untreated ESO26
cells (Fig. 1).
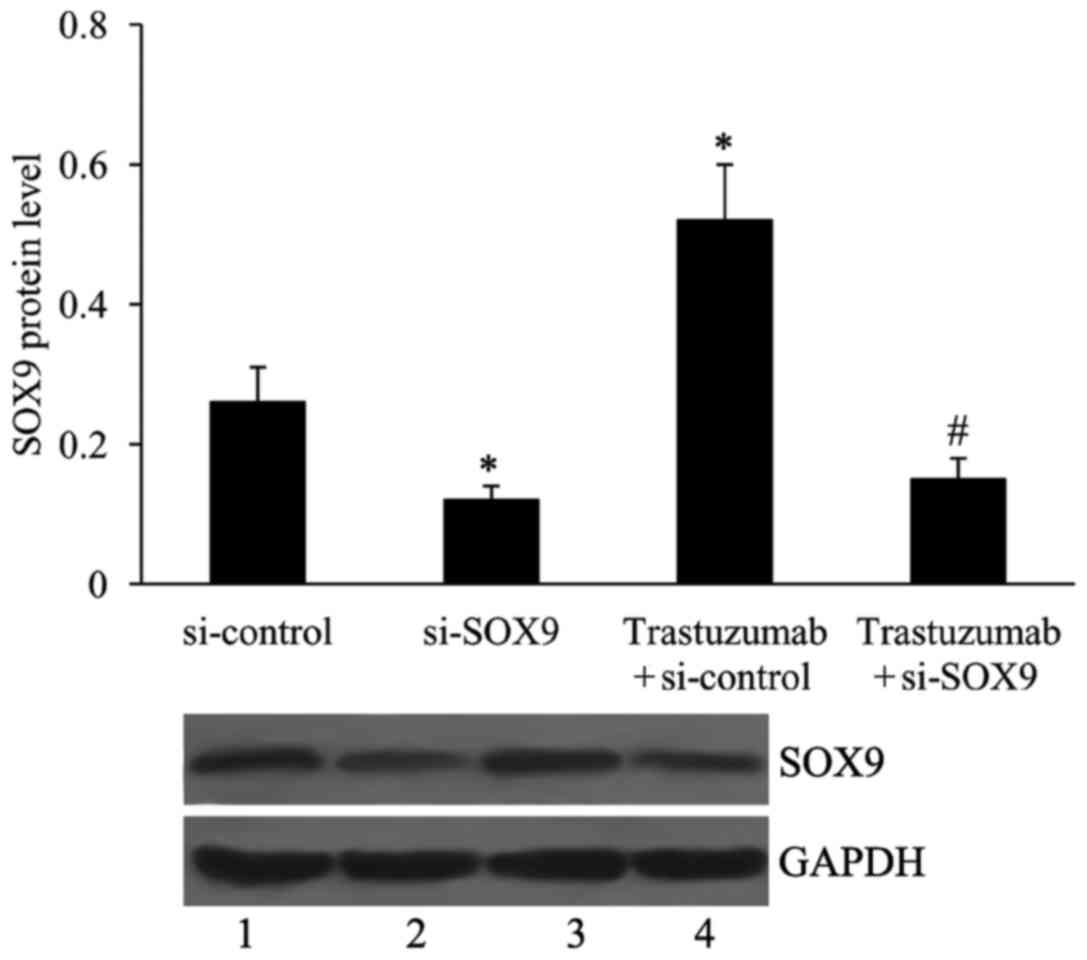
Effect of si-SOX9 on SOX9 expression
in ESO26 cells
ESO26 cells were transfected with si-SOX9, and then
incubated with 4 mg/ml trastuzumab for 24 h. Subsequently, SOX9
expression was examined using western blotting. The SOX9 protein
level was significantly downregulated in the si-SOX9-transfected
cells compared with the si-control cells in the presence or absence
of trastuzumab (Fig. 2).
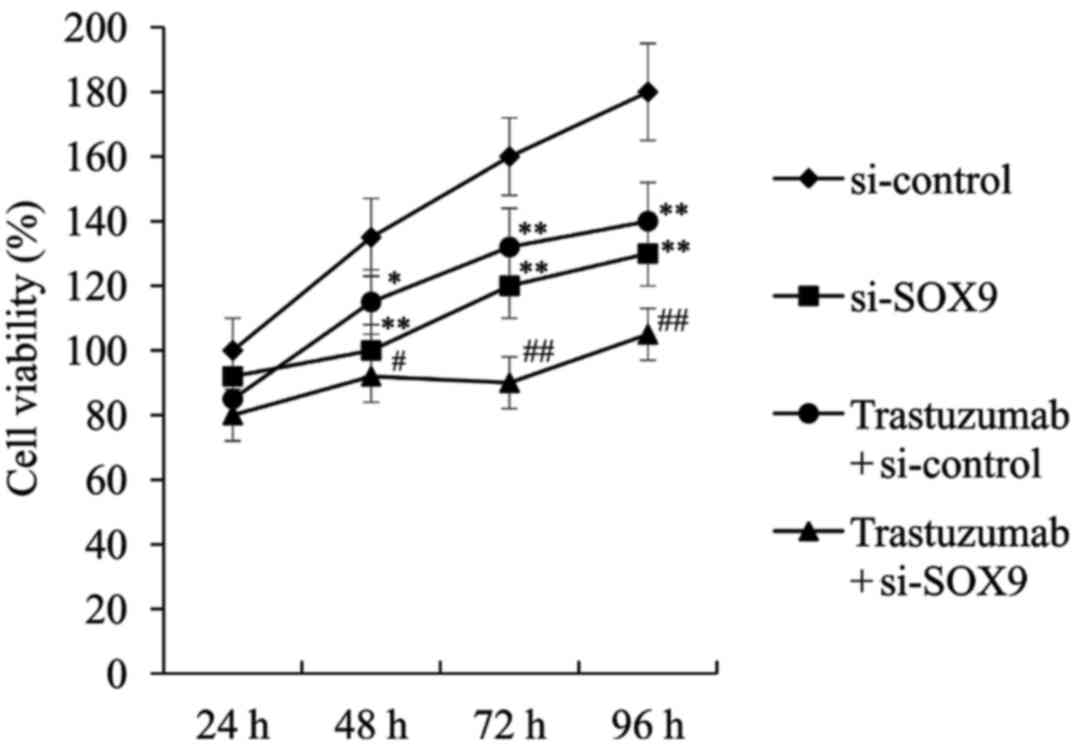
Effect of si-SOX9 on ESO26 cell
proliferation
Cell proliferation following transfection with
si-control or si-SOX9 was determined using an MTT assay. As shown
in Fig. 3 the proliferation of the
cells transfected with the si-control was significantly suppressed
by trastuzumab treatment. In the SOX9 knockdown cells,
proliferation was also significantly inhibited by trastuzumab.
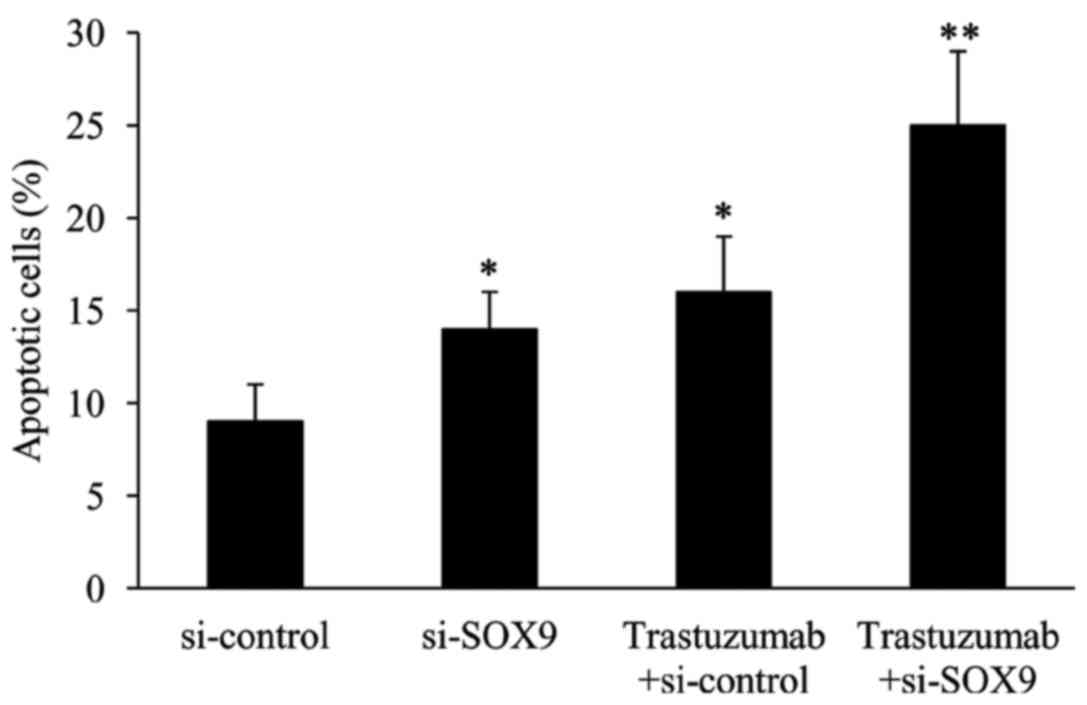
Effect of si-SOX9 on ESO26 cell
apoptosis
The effect of si-SOX9 on ESO26 cell apoptosis was
examined using FCM analysis. The results revealed that compared
with the cells in the si-control group, the number of apoptotic
cells was significantly increased by transfection with si-SOX9 and
by treatment with trastuzumab. In the presence of trastuzumab, the
number of apoptotic cells was greater in the SOX9 knockdown cells
compared with the si-control cells (Fig.
4).
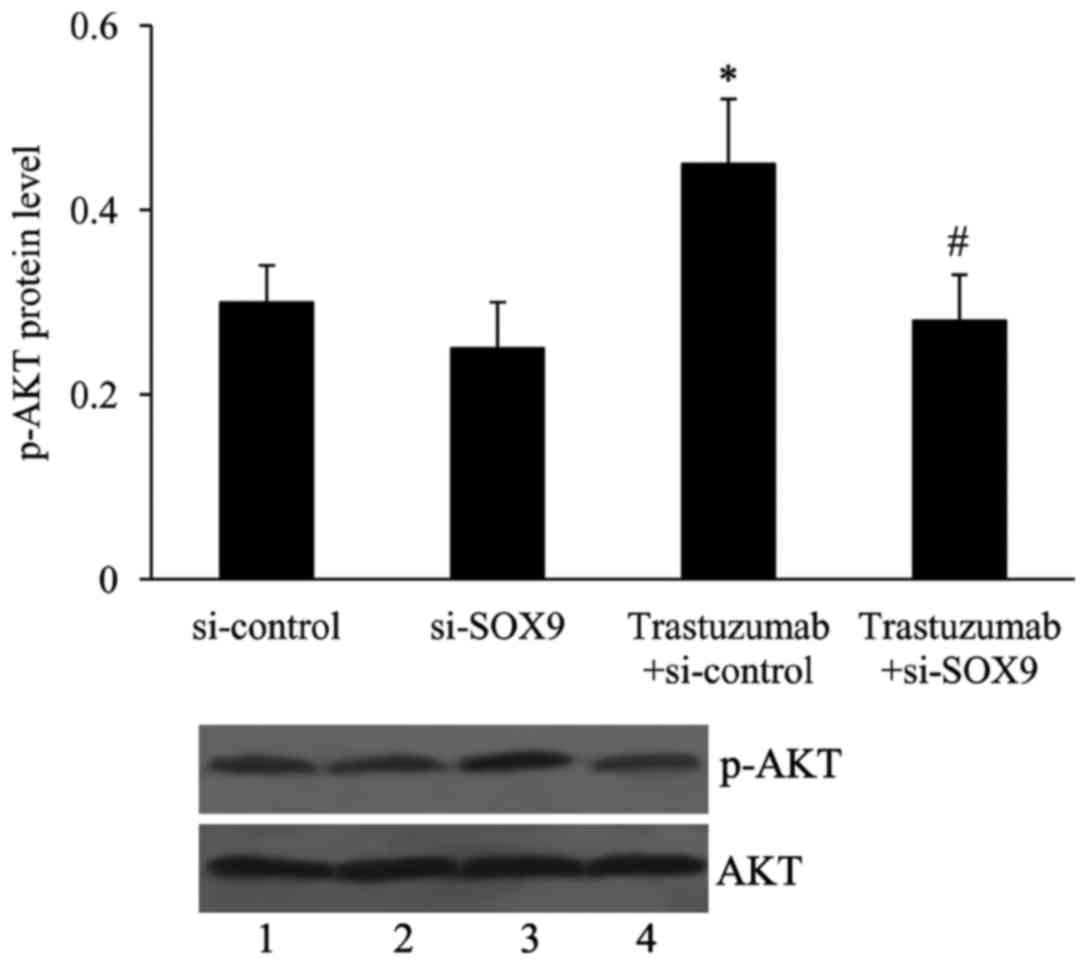
Effect of si-SOX9 on the PI3K/AKT
pathway in ESO26 cells
To investigate the effect of si-SOX9 on PI3K/AKT
activation, the expression of phosphorylated AKT was measured. As
shown in Fig. 5, trastuzumab
treatment activated the PI3K/AKT signaling pathway, as demonstrated
by an elevated level of phosphorylated AKT in the
trastuzumab-treated cells compared with the respective untreated
cells. Si-SOX9 transfection did not exhibit any effects on PI3K/AKT
signaling activation in untreated ESO26 cells; however, the level
of phosphorylated AKT was significantly decreased by si-SOX9 in
trastuzumab-treated cells.
Discussion
In the present study, it was demonstrated that
trastuzumab induced SOX9 expression in a concentration-dependent
manner. SOX9 knockdown sensitized ESO26 cells to trastuzumab by
inhibiting cell proliferation and increasing cell apoptosis.
Furthermore, the trastuzumab-induced activation of PI3K/AKT
signaling was suppressed by SOX9 knockdown.
SOX9, plays vital roles in embryonic development
(19). The importance of SOX9 in
tumorigenesis has been identified, and the dysregulation of SOX9
has been implicated in several cancers, including prostate cancer,
lung adenocarcinoma and gastric carcinoma (20–22).
Overexpression of SOX9 has been demonstrated to increase tumor
growth, invasion and angiogenesis (14,23). In
the present study, it was revealed that SOX9 knockdown led to
suppressed cell proliferation and increased cell apoptosis,
indicating that SOX9 is a tumor promoter in AEG. This finding is
consistent with the role of SOX9 in other types of tumor (20–22). It
has previously been reported that SOX9 mediates the effect of
certain antitumor drugs; SOX9 expression was markedly inhibited in
mice tumors by combination treatment with ABT-263 and 5-FU
(24). In renal cell carcinoma, SOX9
was demonstrated to be involved in resistance to tyrosine kinase
inhibitors (TKIs) and indicated to be a promising biomarker
predicting the response to TKI treatment; patients who were SOX9
negative exhibited a better response to TKI treatment than did
those who were SOX9 positive (25).
The present study firstly demonstrated that SOX9 is involved in the
resistance of AEG to trastuzumab. SOX9 expression was shown to be
induced by trastuzumab in a concentration-dependent manner in ESO26
cells. In addition, the trastuzumab treatment suppressed cell
proliferation and promoted the apoptosis of ESO26 cells, and these
effects were further increased by transfection with si-SOX9. These
findings suggested that SOX9 promotes the resistance of AEG cells
to trastuzumab.
The PI3K/AKT signaling pathway is an intracellular
signaling pathway important in regulating cell proliferation, cell
cycle, apoptosis and differentiation (26–28).
PI3K activation leads to phosphorylation and activation of the
serine/threonine kinase AKT (29);
therefore, AKT phosphorylation may be used to assess the activity
of the PI3K/AKT signaling pathway. Aberrant PI3K/AKT signaling has
been detected in several cancer cell lines, and it has been
reported that activation of the PI3K/AKT signaling pathway may
predict the resistance to chemotherapy and molecularly targeted
therapy (30–32). Recently, Liu et al (16) and Zuo et al (17) reported that activation of the
PI3K/AKT signaling pathway leads to the resistance of gastric
cancer to trastuzumab. To investigate whether the activity of the
PI3K/AKT signaling pathway was affected by SOX9, si-SOX9 was used
to transfect ESO26 cells in the present study. It was found that
the trastuzumab-induced phosphorylation of AKT was suppressed by
SOX9 knockdown. These results demonstrated that SOX9 mediates the
effect of trastuzumab on PI3K/AKT signaling activation.
A limitation of the present study is that only one
cell line, ESO26, was used to investigate the role of SOX9 in
trastuzumab resistance in AEG. Further studies using additional
cell lines are required to support the results.
In conclusion, the present study demonstrated that
SOX9 participated in trastuzumab resistance by affecting cell
proliferation and apoptosis. In addition, SOX9 affected
trastuzumab-induced PI3K/AKT signaling activation in AEG. The
present study provided in vitro evidence of the role of SOX9
in the resistance of AEG to trastuzumab. These issues require
further elucidation in clinical studies.
Acknowledgements
The present study was supported by the Shenzhen
Science and Technology Project (grant no.
JCYJ20150403101028177).
References
|
1
|
Pohl H and Welch HG: The role of
overdiagnosis and reclassification in the marked increase of
esophageal adenocarcinoma incidence. J Natl Cancer Inst.
97:142–146. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rüdiger Siewert J, Feith M, Werner M and
Stein HJ: Adenocarcinoma of the esophagogastric junction: Results
of surgical therapy based on anatomical/topographic classification
in 1,002 consecutive patients. Ann Surg. 232:353–361. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Botterweck AA, Schouten LJ, Volovics A,
Dorant E and van Den Brandt PA: Trends in incidence of
adenocarcinoma of the oesophagus and gastric cardia in ten European
countries. Int J Epidemiol. 29:645–654. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Edgren G, Adami HO, Weiderpass E and Nyrén
O: A global assessment of the oesophageal adenocarcinoma epidemic.
Gut. 62:1406–1414. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dikken JL, Lemmens VE, Wouters MW,
Wijnhoven BP, Siersema PD, Nieuwenhuijzen GA, van Sandick JW, Cats
A, Verheij M, Coebergh JW and van de Velde CJ: Increased incidence
and survival for oesophageal cancer but not for gastric cardia
cancer in the Netherlands. Eur J Cancer. 48:1624–1632. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Brown LM, Devesa SS and Chow WH: Incidence
of adenocarcinoma of the esophagus among white Americans by sex,
stage, and age. J Natl Cancer Inst. 100:1184–1187. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
de Jonge PJ, van Blankenstein M, Grady WM
and Kuipers EJ: Barrett's oesophagus: Epidemiology, cancer risk and
implications for management. Gut. 63:191–202. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Siewert JR, Stein HJ and Feith M:
Adenocarcinoma of the esophago-gastric junction. Scand J Surg.
95:260–269. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Bang YJ, Van Cutsem E, Feyereislova A,
Chung HC, Shen L, Sawaki A, Lordick F, Ohtsu A, Omuro Y, Satoh T,
et al: Trastuzumab in combination with chemotherapy versus
chemotherapy alone for treatment of HER2-positive advanced gastric
or gastro-oesophageal junction cancer (ToGA): A phase 3,
open-label, randomised controlled trial. Lancet. 376:687–697. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Strong VE, D'Amico TA, Kleinberg L and
Ajani J: Impact of the 7th Edition AJCC staging classification on
the NCCN clinical practice guidelines in oncology for gastric and
esophageal cancers. J Natl Compr Canc Netw. 11:60–66. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ma F, Ye H, He HH, Gerrin SJ, Chen S,
Tanenbaum BA, Cai C, Sowalsky AG, He L, Wang H, et al: SOX9 drives
WNT pathway activation in prostate cancer. J Clin Invest.
126:1745–1758. 2016. View
Article : Google Scholar : PubMed/NCBI
|
|
12
|
Marcker Espersen ML, Linnemann D,
Christensen IJ, Alamili M, Troelsen JT and Høgdall E: SOX9
expression predicts relapse of stage II colon cancer patients. Hum
Pathol. 52:38–46. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Wang X, Ju Y, Zhou MI, Liu X and Zhou C:
Upregulation of SOX9 promotes cell proliferation, migration and
invasion in lung adenocarcinoma. Oncol Lett. 10:990–994. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Chakravarty G, Moroz K, Makridakis NM,
Lloyd SA, Galvez SE, Canavello PR, Lacey MR, Agrawal K and Mondal
D: Prognostic significance of cytoplasmic SOX9 in invasive ductal
carcinoma and metastatic breast cancer. Exp Biol Med (Maywood).
236:145–155. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Riemenschnitter C, Teleki I, Tischler V,
Guo W and Varga Z: Stability and prognostic value of Slug, Sox9 and
Sox10 expression in breast cancers treated with neoadjuvant
chemotherapy. Springerplus. 2:6952013. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Liu J, Pan C, Guo L, Wu M, Guo J, Peng S,
Wu Q and Zuo Q: A new mechanism of trastuzumab resistance in
gastric cancer: MACC1 promotes the Warburg effect via activation of
the PI3K/AKT signaling pathway. J Hematol Oncol. 9:762016.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zuo Q, Liu J, Zhang J, Wu M, Guo L and
Liao W: Development of trastuzumab-resistant human gastric
carcinoma cell lines and mechanisms of drug resistance. Sci Rep.
5:116342015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Jo A, Denduluri S, Zhang B, Wang Z, Yin L,
Yan Z, Kang R, Shi LL, Mok J, Lee MJ and Haydon R: The versatile
functions of Sox9 in development, stem cells, and human diseases.
Genes Dis. 1:149–161. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Wang H, McKnight NC, Zhang T, Lu ML, Balk
SP and Yuan X: SOX9 is expressed in normal prostate basal cells and
regulates androgen receptor expression in prostate cancer cells.
Cancer Res. 67:528–536. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jiang SS, Fang WT, Hou YH, Huang SF, Yen
BL, Chang JL, Li SM, Liu HP, Liu YL, Huang CT, et al: Upregulation
of SOX9 in lung adenocarcinoma and its involvement in the
regulation of cell growth and tumorigenicity. Clin Cancer Res.
16:4363–4373. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhou CJ, Guo JQ, Zhu KX, Zhang QH, Pan CR,
Xu WH, Wang HJ and Liu B: Elevated expression of SOX9 is related
with the progression of gastric carcinoma. Diagn Cytopathol.
39:105–109. 2011. View
Article : Google Scholar : PubMed/NCBI
|
|
23
|
Wang H, Leav I, Ibaragi S, Wegner M, Hu
GF, Lu ML, Balk SP and Yuan X: SOX9 is expressed in human fetal
prostate epithelium and enhances prostate cancer invasion. Cancer
Res. 68:1625–1630. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Chen Q, Song S, Wei S, Liu B, Honjo S,
Scott A, Jin J, Ma L, Zhu H, Skinner HD, et al: ABT-263 induces
apoptosis and synergizes with chemotherapy by targeting stemness
pathways in esophageal cancer. Oncotarget. 6:25883–25896.
2015.PubMed/NCBI
|
|
25
|
Li XL, Chen XQ, Zhang MN, Chen N, Nie L,
Xu M, Gong J, Shen PF, Su ZZ, Weng X, et al: SOX9 was involved in
TKIs resistance in renal cell carcinoma via Raf/MEK/ERK signaling
pathway. Int J Clin Exp Pathol. 8:3871–3881. 2015.PubMed/NCBI
|
|
26
|
Yu JS and Cui W: Proliferation, survival
and metabolism: The role of PI3K/AKT/mTOR signalling in
pluripotency and cell fate determination. Development.
143:3050–3060. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Ma Y, Qin H and Cui Y: MiR-34a targets
GAS1 to promote cell proliferation and inhibit apoptosis in
papillary thyroid carcinoma via PI3K/Akt/Bad pathway. Biochem
Biophys Res Commun. 441:958–963. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Wang Y, Wang WL, Xie WL, Li LZ, Sun J, Sun
WJ and Gong HY: Puerarin stimulates proliferation and
differentiation and protects against cell death in human
osteoblastic MG-63 cells via ER-dependent MEK/ERK and PI3K/Akt
activation. Phytomedicine. 20:787–796. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Franke TF, Yang SI, Chan TO, Datta K,
Kazlauskas A, Morrison DK, Kaplan DR and Tsichlis PN: The protein
kinase encoded by the Akt proto-oncogene is a target of the
PDGF-activated phosphatidylinositol 3-kinase. Cell. 81:727–736.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mandell JW: Phosphorylation state-specific
antibodies: Applications in investigative and diagnostic pathology.
Am J Pathol. 163:1687–1698. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Mandell JW: Immunohistochemical assessment
of protein phosphorylation state: The dream and the reality.
Histochem Cell Biol. 130:465–471. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Slomovitz BM and Coleman RL: The
PI3K/AKT/mTOR pathway as a therapeutic target in endometrial
cancer. Clin Cancer Res. 18:5856–5864. 2012. View Article : Google Scholar : PubMed/NCBI
|