Introduction
During bacterial infection, the innate immune system
triggers a rapid and non-specific inflammatory response mechanism
to prevent the spread of bacteria. As one of the first defense
mechanisms used, the phagocytic macrophages serve an important role
in clearing foreign particles, including bacteria and cell debris,
by phagocytosis and by restoring cell homeo-stasis to prevent
tissue damage (1,2). Following microbial or cell debris
recognition, the acute inflammatory response is initiated and
triggers the rapid release of proinflamma-tory mediators such as
interleukin (IL)-1β, IL-6, and tumor necrosis factor-α (TNF-α),
which will subsequently induce the adaptive immune system (3,4).
In addition to their phagocytic activity, macrophages serve an
essential role in the maintenance of tissue homeostasis.
Furthermore, they exhibit an uncommon plasticity and are able to
polarize into subsets of proinflammatory or anti-inflammatory
populations depending on their surrounding environment and their
own phagocytic activity (5). The
mitogen-activated protein kinase (MAPK) signaling pathways, which
include the most studied members ERK, p38 MAPK (p38) and JNK, are
instrumental in all innate immune receptor signaling pathways. The
MAPKs act by modulating the expression of inflammatory cytokines
and chemokines (6,7).
Staphylococcus aureus, a gram-positive human
bacterium, is a commensal opportunistic pathogen. It is a leading
cause of skin and soft tissue infections, osteomyelitis, septic
arthritis, pneumonia and endovascular infections (8). Unfortunately, treatments are
complicated by the prevalence of multi-drug resistance strains,
which result in high mortality and morbidity rates (9). Originally considered an
extracellular pathogen, it has been demonstrated that S.
aureus is also able to invade, survive and replicate within
mammalian phagocytic and non-phagocytic cells using various
survival strategies (10). A
pathogenic characteristic of S. aureus is its ability to
generate intense local and systemic inflammatory responses, evident
in the release of inflammatory cytokines. Once released within the
infected tissue environment, the cytokines act on the surrounding
epithelial, stromal, and circulating cells, triggering secondary
waves of cytokine release, consequently amplifying the host defense
response. Uncontrolled levels of cytokines are detrimental to the
host, resulting in massive tissue destruction.
Sulforaphane (SFN), a degradation product of
gluco-sinolates, is an isothiocyanate derivative naturally present
in cruciferous vegetables such as broccoli sprouts. SFN is a potent
enhancer of phase II detoxification enzymes with chemopreventive,
antitumor, anti-inflammatory and antimicrobial activities against a
variety of bacterial and fungal species (11-13). While several in vitro
studies have described SFN antimicrobial activity, SFN treatment in
Helicobacter pylori infection was the most studied. This
pathogenic bacteria is involved in gastric epithelium infection and
is associated with gastric cancer, and at present, the molecular
mechanism involved in the bactericidal effect of SFN against H.
pylori remains elusive (11,14). Recently, treatment with SFN was
demonstrated to inhibit HIV infection in THP-1-derived macrophages
and primary macrophages, but not primary T cells (15). In this case, the inhibitory effect
of SFN on microbial persistence is exerted through a nuclear factor
erythroid 2-related factor 2 (Nrf2)-dependent mechanism. SFN is a
well-known activator of the transcription factor Nrf2, a key
regulator of genes coding for phase II detoxifying enzymes,
including NAD(P)H quinone dehydrogenase 1 (NQO1) and heme
oxygenase-1 (HO-1), cytoprotective, antioxidant and
anti-inflammatory responses (16,17). Nrf2 is an essential factor in the
attenuation of inflammation since Nrf2-deficient mice exhibit
increased inflammation (18). In
addition, Nrf2 has been suggested to negatively regulate the
transcription of IL-1β and IL-6 proinflammatory cytokine genes in
lipo-polysaccharide (LPS)-treated mouse bone marrow-derived
macrophages (19).
Post-transcriptional regulation by microRNAs
(miRNAs) has emerged as a major regulatory mechanism to control the
expression level of genes involved in a number of fundamental
cellular processes such as inflammation, proliferation, apoptosis
and macrophage polarization upon pathogenic infection (20). miRNAs are molecules measuring
18-24 nucleotides in length and belong to the short non-coding RNA
family. By binding to the 3′ untranslated region of their target
mRNA, each mature miRNA is able to target up to hundreds of mRNAs
and inhibit their expression (21). SFN and other glucosinolate
derivatives have been demonstrated to modulate epigenetic
alterations identified during carcinogenesis, including DNA
methylation, histone modifications and miRNAs (22).
The present study investigated the underlying
cellular mechanisms initiated by the pretreatment of human
THP-1-derived macrophages, primary human peripheral blood
mononuclear cells (PBMC)-derived macrophages, and primary mouse
bone marrow-derived macrophages (BMDMs) with SFN and subsequent
challenge with S. aureus. The effect of SFN pretreatment on
the intracellular survival of S. aureus, macrophage
apoptosis and macrophage inflammatory response was evaluated.
Furthermore, Nrf2−/− BMDMs were used to assess whether
the Nrf2 signaling was involved. Finally, the cellular signaling
pathways, regulated by SFN and involved in the decrease in
intracellular S. aureus survival, were examined using
specific MAPK inhibitors. The results provided novel insights into
the molecular mechanisms underlying the anti-inflammatory and
anti-apoptotic activities mediated by SFN to disrupt S.
aureus-favored environment in macrophages.
Materials and methods
Antibodies and reagents
SFN, phorbol 12-myristate 13-acetate (PMA),
phalloidin-ATTO 594 (cat. no. 51927) were purchased from
Sigma-Aldrich; Merck KGaA. The RPMI-1640 medium, heat-inactivated
fetal bovine serum (FBS), FAM-FLICA caspases-3/7 assay kit
(ImmunoChemistry Technologies, LLC) and human recombinant
granulocyte-macrophage colony-stimulating factor (GM-CSF) were
obtained from Eurobio Scientific. The Tryptic soy broth and Tryptic
soy agar (Condalab) were obtained from Dominique Dutscher SAS. The
Annexin V-fluorescein isothiocyanate apoptosis reagent, and the
inhibitors SB203580 and SP600125 were obtained from Abcam. Rabbit
anti-Nrf2 antibody (cat. no. 16396-1AP) was purchased from
ProteinTech Group, Inc. Gentamycin sulfate, mouse anti-GAPDH (cat.
no. CB1001), rabbit anti-phos-phorylated (p)-ERK (cat. no. 04-797),
rabbit anti-ERK (cat. no. ABS44) and mouse anti-p-p38 (cat. no.
MABS64) antibodies were obtained from Merck KGaA. Mouse anti-p-JNK
(cat. no. 612540), mouse anti-JNK (cat. no. 610627), mouse anti-p38
(cat. no. 612168) antibodies were purchased from BD Biosciences.
The iTaq SYBR-Green Supermix and DC protein assay kit were
purchased from Bio-Rad Laboratories, Inc. Goat anti-rabbit IRDye
680RD (cat. no. 926-68071) and goat anti-mouse IRDye 800CW (cat.
no. 926-32210) antibodies were obtained from LI-COR Biosciences.
Specific miRCURY LNA miRNA PCR primer sets (hsa-miR-142-5p,
has-miR-146a-5p and UniSp6), and the miRCURY LNA RT and miRCURY LNA
SYBR PCR kits were purchased from Qiagen. Dulbecco's modified Eagle
medium (DMEM), Maxima First Strand cDNA synthesis kit, CellRox
Green flow cytometry assay kit and Ficoll-Paque™ (GE Healthcare)
were obtained from Thermo Fisher Scientific, Inc.
Animals
Breeding of the C57BL/6J Nrf2 knockout
(Nrf2−/−) mouse strain, provided by Dr Yamamoto (Tohuki
University) and purchased from Riken BRC (17), was performed at the University of
Versailles-Saint-Quentin-en-Yvelines (license no.
APAFIS8785-201610191723731v3). The protocols were approved by the
Animal Ethics Committee CEEA47 PELVIPHARM of the University of
Versailles-Saint-Quentin-en-Yvelines and the Ministry of Higher
Education, Research and Innovation. Nrf2 heterozygote
(Nrf2+/−) littermates were used as the control
group.
Cell culture, cell differentiation and
treatments
The human THP-1 monocytic cell line [American Type
Culture Collection (ATCC®) TIB-202™] was maintained in
RPMI-1640 supplemented with 10% heat-inactivated FBS, 1 mM sodium
pyruvate, 10 mM HEPES and 0.05 mM β-mercaptoethanol in a humidified
atmosphere at 37°C and 5% CO2. Terminal differentiation
of THP-1 to macrophages was obtained by rinsing the cells twice
with sterile PBS prior to incubation at 37°C with 50 nM PMA for 48
h.
To obtain the PBMC-derived macrophages, human
primary monocytes were isolated from heparinized peripheral blood
purchased from the Etablissement Français du Sang (EFS) using a
Ficoll-Paque™ gradient. Blood samples were diluted with an equal
volume of PBS and centrifuged at room temperature for 30 min at 400
x g. The EFS authorized the Inserm U1179 research unit (agreement
number 15/EFS/012) to use anonymized blood samples for
non-therapeutic purposes. PBMCs were cultured in the abovementioned
culture medium without β-mercaptoethanol with the addition of 20
ng/ml GM-CSF. A total of 7 days after isolation, non-adherent cells
were removed and adherent PBMC-derived macrophages were ready for
treatment.
For the BMDMs isolated from Nrf2−/− and
Nrf2+/− littermate mice, bone marrow was extracted from
mouse femurs and tibias, washed with PBS and cultured in DMEM
supplemented with 20% filtered L929 fibroblast cell
line-conditioned media and 10% heat-inactivated FBS in non-treated
culture dishes. Conditioned medium was harvested from L929
fibroblasts, grown in DMEM supplemented with 10% heat-inactivated
FBS, and 3 days following confluency it was filtered through a
0.22-µm filter and stored at -20°C. A total of 7 days after
harvest, all non-adherent cells were removed, and the remaining
macrophages were detached by resuspending cells in cold PBS, and by
scraping cells after 10 min incubation on ice. BMDMs were stained 1
min at room temperature by diluting the cell suspension with an
equal volume of 0.4% trypan blue, counted using the Countess
automated cell counter (Thermo Fisher Scientific, Inc.), and seeded
in 24-well culture plates at a density of 5×105/well for
use the following day.
When required, macrophages were incubated at 37°C
with p38 or JNK inhibitors (25 µM SB203580 and 25 µM
SP600125, respectively) 1 h prior to addition of 10 µM SFN
(23). A total of 3 h after SFN
treatment, S. aureus was added at a multiplicity of
infection (MOI) of 10, and the cells were incubated at 37°C for an
additional 3 h prior to cell lysis and RNA isolation. For 24 h
post-infection assays, 20 µg/ml gentamycin was added to cell
medium 1 h after S. aureus infection, to eliminate
extracellular bacteria.
Total RNA isolation and reverse
transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA was isolated 3 h after S. aureus
infection of macrophages with TRIzol® (Invitrogen;
Thermo Fisher Scientific, Inc.) and chloroform extraction technique
following the manufacturer's protocol. Total RNA was quantified
using the GE NanoVue spectrophotometer (GE Healthcare). To
determine mRNA expression levels of target genes HO-1, NQO1, IL-1β,
IL-6, TNF-α, C-C motif chemokine receptor 7 (CCR7), IL-23, iNOS,
and 8S ribosomal RNA, 1 µg total RNA was reverse transcribed
to cDNA using Maxima First strand cDNA synthesis kit following the
manufacturer's protocol. cDNAs were then analyzed by RT-qPCR using
iTaq SYBR-Green qPCR mix. The thermo-cycling conditions were as
follows: 95°C for 30 sec, followed by 40 cycles of a 5 sec
denaturation step at 95°C and a 20 sec annealing/extension step at
60°C. The specific oligonucleotides for human and mouse HO-1, NQO1,
IL-1β, IL-6, TNF-α, 18S ribosomal RNA, CCR7, IL-23 and iNOS, listed
in Table I, were synthetized by
Eurogentec. Gene expression levels were normalized to that of the
reference gene 18S ribosomal RNA. To analyze miRNA expression
levels of target genes miR-142-5p and miR-146a-5p, miRNA was
reverse transcribed using the miRCURY LNA RT kit. cDNAs were then
analyzed by qPCR using miRCURY LNA SYBR PCR kit. miRNA expression
levels of target genes were normalized to the internal control U6
snRNA level. RT-qPCR was performed using the CFX96 and CFX384 Touch
thermocyclers (Bio-Rad Laboratories, Inc.), with each sample
performed in triplicate. Data were analyzed on BioRad CFX manager
3.1 using the 2−ΔΔCq method (24). Each RT-qPCR assay was performed
independently and in triplicate.
 | Table IPrimers used for reverse
transcription quantitative polymerase chain reaction analyses. |
Table I
Primers used for reverse
transcription quantitative polymerase chain reaction analyses.
| Target gene | Primer sequence
(5′-3′) |
|---|
| Human CCR7 | F
GATTACATCGGAGACAACACCA |
| R
AGTACATGATAGGGAGGAACCAG |
| Human IL-1β | F
AATGATGGCTTATTACAGTGGCA |
| R
GTCGGAGATTCGTAGCTGGA |
| Human IL-23 | F
GTTCCCCATATCCAGTGTGG |
| R
GAGGCTTGGAATCTGCTGAG |
| Human IL-6 | F
GTAGCCGCCCCACACAGA |
| R
CATGTCTCCTTTCTCAGGGCTG |
| Human TNF-α | F
GGAGAAGGGTGACCGACTC |
| R
TGGGAAGGTTGGATGTTCGT |
| Human 18S rRNA | F
GATAGCTCTTTCTCGATTCCG |
| R
CTAGTTAGCATGCCAGAGTC |
| Mouse HO-1 | F
CACGCATATACCCGCTACCT |
| R
CCAGAGTGTTCATTCGAGCA |
| Mouse IL-1β | F
GCAACTGTTCCTGAACTCAACT |
| R
ATCTTTTGGGGTCCGTCAACT |
| Mouse IL-6 | F
GTTCTCTGGGAAATCGTGGA |
| R
CCAGTTTGGTAGCATCCATC |
| Mouse iNOS | F
TCCTGGAGGAAGTGGGCCGAAG |
| R
CCTCCACGGGCCCGGTACTC |
| Mouse NQO1 | F
TTCTCTGGCCGATTCAGAGT |
| R
GGCTGCTTGGAGCAAAATAG |
| Mouse TNF-α | F
TGTCTACTCCCAGGTTCTCTT |
| R
GCAGAGAGGAGGTTGACTTTC |
| Mouse 18S rRNA | F
CTGAGAAACGGCTACCACATC |
| R
CGCTCCCAAGATCCAACT |
Bacterial strain, growth culture, and
fluorescence labeling of S. aureus
The ATCC 25923TM strain was grown aerobically in
Tryptic soy broth to an optical density of 1 at 37°C under
agitation. Bacterial glycerol stocks were maintained at -20°C. When
required, frozen stocks were thawed and bacteria were diluted to
the appropriate inoculum in sterile PBS. For the intracellular
bacterial survival assay, bacteria were grown on Tryptic soy broth
solidified with 1.5% agar. For the S. aureus internalization
assay, fresh fluorescent S. aureus were generated by
incubating S. aureus with SYTO9 for 15 min (Thermo Fisher
Scientific, Inc.) at room temperature in the dark under gentle
shaking, then washing 3 times in sterile PBS, and resus-pending in
PBS prior to cell infection.
S. aureus internalization assay and
immunofluorescence labeling
THP-1 cells were seeded in 6-well plates at
1×106 cells/well and differentiated with 50 nM PMA for
48 h. THP-1-derived macrophages were infected with SYTO9-labeled
S. aureus at an MOI of 10. After 1 h infection, cells were
washed twice with cold PBS to prevent additional bacterial
internalization and to remove extracellular bacteria. Cells were
then trypsinized at 37°C for 10 min, fixed in 4% paraformaldehyde
(PFA) for 15 min at room temperature, and finally suspended in 500
µl 0.02% EDTA. The quantification of cells infected with
SYTO9-labeled S. aureus were performed using BD LSRFortessa
flow cytometer and analyzed using FACSDiva software 7.0 (BD
Biosciences). A forward and side scatter gate was set to exclude
dead and aggregated cells. A total of 100,000 events per condition
was collected.
For the immunofluorescence labeling, THP-1-derived
macrophages seeded at 5×105 cells/coverslip were
infected with SYTO9-stained S. aureus for 1 h then cells
were rinsed with PBS and fixed for 10 min at room temperature with
4% PFA. Cells were visualized by labeling with phalloidin-ATTO 594.
Images were captured using a laser scanning confocal fluorescence
microscope (Leica SP8; Leica Microsystems, Inc.) and analyzed using
ImageJ v.1.47 software (National Institutes of Health).
Intracellular survival assay of S.
aureus
For the intracellular bacterial survival assay,
macrophages seeded in 24-well plates at 2.5×105
cells/well were infected with S. aureus at an MOI of 10.
After 1 h infection, cells were washed with PBS and extracellular
bacteria were eliminated by the addition of 20 µg/ml
gentamycin in cell culture medium. After 24 h incubation at 37°C,
cells were rinsed with PBS and lysed in 1 ml ice-cold sterile water
for 20 min at 4°C. Intracellular bacteria were diluted in 5-fold
serial dilutions and then plated on Tryptic soy agar plates. Colony
forming units (CFU) counts were determined 24 h after incubation at
37°C.
Quantification of reactive oxygen species
(ROS) production
The CellRox green reagent assay kit (Thermo Fisher
Scientific, Inc.) was used to determine ROS levels. Briefly,
THP-1-derived macrophages were seeded at a density of
1×105 cells/well on coverslip-covered wells in 24-well
plates. Following treatment, macrophages were stained with 5
µM CellRox green reagent for 30 min at 37°C. The cells were
then washed with PBS, fixed in 4% PFA for 15 min at room
temperature and the nuclei were stained with 10 µg/ml
4',6-diamidino-2-phe-nylindole dihydrochloride for 10 min at room
temperature. Analysis of the images captured with the Leica SP8
confocal microscope (Leica Microsystems, Inc.) were performed using
ImageJ v.1.47 software (National Institutes of Health).
Cell viability assay
THP-1-derived macrophages were seeded at
5×104 cells/well in a 96-well plate. Cytotoxicity to SFN
or S. aureus infection was assessed by an MTT cell viability
assay kit (Biotium, Inc.) to measure cellular metabolic activity
following the manufacturer's protocol. Absorbance changes were
measured at 550 nm, and the background absorbance at 600 nm, using
the FLUOstar Omega microplate reader (BMG Labtech).
Caspases-3/7 activity assays
When indicated, BMDMs seeded in 6-well plates at
1×106 cells/well were pretreated for 3 h with 10
µM SFN or DMSO prior to infection with S. aureus (MOI
of 10). Gentamycin 20 µg/ml was added to the culture medium
1 h after infection to eliminate extracellular S. aureus.
Staurosporine (1 µM) was used as an apoptosis positive
control. Following cell treatment, caspases-3/7 activities were
determined using the Green FAM-FLICA Caspases-3/7 assay kit
according to the manufacturer's protocol. Briefly, the cells were
trypsinized and incubated at 37°C in the dark for 1 h with the
FLICA probe, made of the irreversible caspase inhibitor
DEVD-fluoromethyl ketone fused to a carboxyfluorescein
(FAM-DEVD-FMK), which binds specifically and covalently to
activated caspase-3/7 enzymes. Following 2 washes, cells were fixed
using 4% PFA for 10 min at room temperature and resuspended in
0.02% EDTA and the number of FLICA-positive cells was counted using
the BD LSRFortessa™ flow cytometer and analyzed using FACSDiva 7.0
software (BD Biosciences).
Protein extraction and western blot
analysis
For total protein extraction, THP-1-derived
macrophages were rinsed with cold PBS and then lysed with cold RIPA
buffer [150 mM NaCl, 1% Triton X-100, 0.5% sodium deoxycholate,
0.1% SDS and 50 mM Tris-HCl (pH 7.5)], supplemented with protease
and phosphatase inhibitor cocktail mixtures. Protein concentrations
were determined using the DC protein assay kit. Total proteins (20
µg/lane) were resolved by 4-20% SDS-PAGE and transferred to
Immobilon-FL polyvinylidene difluoride membranes (Merck KGaA).
Western blot analysis was then performed at room temperature for ~3
h using the iBind Flex Western system (Invitrogen; Thermo Fisher
Scientific, Inc.) following the manufacturer's protocol. Briefly,
primary antibodies against Nrf2 (1:1,000), GAPDH (1:8,000),
totaland p-ERK (1:2,000 and 1:1,000, respectively), total and p-p38
(1:2,000 and 1:1,000, respectively), total and p-JNK (both
1:1,000), and IRDye680RD- or IRDye800RD-conjugated secondary
antibodies (1:4,000 and 1:3,000, respectively) were diluted in
iBind Flex FD Solution. Fluorescent blot imaging was performed
using Odyssey CLx Imaging system (LI-COR Biosciences).
Densitometric analysis was performed using Image Studio Lite
software v4.0 (LI-COR Biosciences).
Statistical analysis
The data are presented as the mean ± standard error
of the mean (SEM) from at least 3 independent experiments. Imaging
flow cytometry data are presented as the mean ± SEM of at least 3
independent experiments analyzed from 100,000 events each.
Statistical comparisons were performed using two-tailed unpaired
t-tests or ANOVA followed by Tukey's post hoc test. P<0.05 was
considered to indicate a statistically significant difference.
Results
SFN improves intracellular S. aureus
clearance in macro- phages
In order to gain a better understanding of the
effect of SFN on the intracellular fate of S. aureus in
macrophages, THP-1-derived macrophages were utilized. To visualize
the internalization of S. aureus, THP-1 monocytes were first
differentiated into macrophages with PMA and infected with
SYTO9-stained S. aureus for 1 h. Following PFA fixation, the
macrophages, stained with fluorescent phalloidin and intracellular
S. aureus, were visualized by confocal microscopy. Analysis
of the images of infected macrophages demonstrated the
internalization of SYTO9-labeled S. aureus in THP-1-derived
macrophages, including S. aureus enclosed in actin-dependent
structures reminding of early phagocy-tosis (Fig. 1A).
To evaluate the intracellular survival of S.
aureus, THP-1-derived macrophages were pretreated for 3 h with
10 µM SFN or the vehicle DMSO prior to S. aureus
infection. A total of 1 h after infection, gentamycin was added to
the culture medium to eliminate extracellular bacteria and infected
macrophages were incubated for an additional 24 h post-infection.
CFU enumeration indicated that intracellular bacteria counts were
significantly decreased in SFN pretreated THP-1-derived macrophages
compared with the DMSO pretreated cells (Fig. 1B). The inhibitory effect of SFN
observed in the THP-1-derived macrophages was confirmed by
infecting macrophages derived from human peripheral blood
mononuclear cells (PBMCs). PBMCs issued from blood donors and
isolated using a Ficoll-Paque™ gradient were first differentiated
to macrophages for 7 days with GM-CSF prior to pretreatment with
SFN or DMSO and infection with S. aureus. SFN pretreated
PBMC-derived macrophages exhibited a significant decrease in
intracellular bacterial survival 24 h after infection compared to
S. aureus counts issued from DMSO treated macrophages
(Fig. 1C).
Furthermore, to determine whether cytotoxicity
contributed to the apparent decrease in bacterial persistence, cell
viability was assessed in THP-1-derived macrophages incubated with
SFN or infected for 1 h with S. aureus prior to the addition
of gentamycin and 24 h incubation. The MTT assay revealed that
neither SFN nor S. aureus affected THP-1-derived macrophage
viability as compared with that observed in the DMSO-treated cells
(Fig. 1D). Additionally,
quantification by flow cytometry of internalized SYTO9-stained
S. aureus in THP-1-derived macrophages 1 h after infection
indicated that SFN pretreatment of macrophages had no effect on
S. aureus internalization when compared with the
DMSO-treated macrophages infected with S. aureus (Fig. 1E). The modulation of the oxidative
stress by S. aureus in THP-1-derived macrophages was also
assessed. ROS levels were measured in macrophages 24 h after
bacterial challenge in presence of the CellRox fluorogenic probe.
Confocal images and fluorescent intensities were analyzed by ImageJ
software, and the results indicated that there was no significant
modification to ROS production in THP-1-derived macrophages
pretreated with SFN and/or infected with S. aureus compared
with the DMSO-treated macrophages (Fig. S1), indicating that the
SFN-mediated inhibition of intracellular S. aureus survival
in THP-1-derived macrophages was not due to an effect of SFN or
S. aureus on cell viability or ROS production.
SFN affects intracellular S. aureus
survival and cell apoptosis in an Nrf2-independent manner
As SFN is a well-known activator of transcription
factor Nrf2, a key regulator of inducible intracellular defenses in
the innate immune system, the present study aimed to determine
whether SFN activated Nrf2 signaling to impair intracellular
survival of S. aureus in macrophages. Primary bone marrow
stem cells were isolated from Nrf2−/− mice and their
control Nrf2+/− littermates, and differentiated into
BMDMs using L929-conditioned media. Nrf2+/− and
Nrf2−/− BMDMs pretreated with DMSO or SFN were infected
with S. aureus for 1 h prior to the addition of gentamycin
to the culture medium. Total proteins were extracted 24 h after
infection and examined by western blot analysis. SFN significantly
increased the Nrf2 protein levels in both non-infected and infected
Nrf2+/− BMDMs compared with the DMSO-treated
macrophages, whereas no detectable Nrf2 was observed in the
non-infected and infected macrophages pretreated with DMSO
(Fig. 2A). Knockout of Nrf2 in
the Nrf2−/− BMDMs was validated by western blot analysis
and qPCR analysis of 2 target genes of Nrf2 coding for HO-1 and
NQO1. qPCR analysis demonstrated the lack of transcription
upregulation of HO-1 and NQO1 in DMSO-treated or S.
aureus-infected Nrf2+/− BMDM, and in
Nrf2−/− BMDMs pretreated with SFN (Fig. 2A-C).
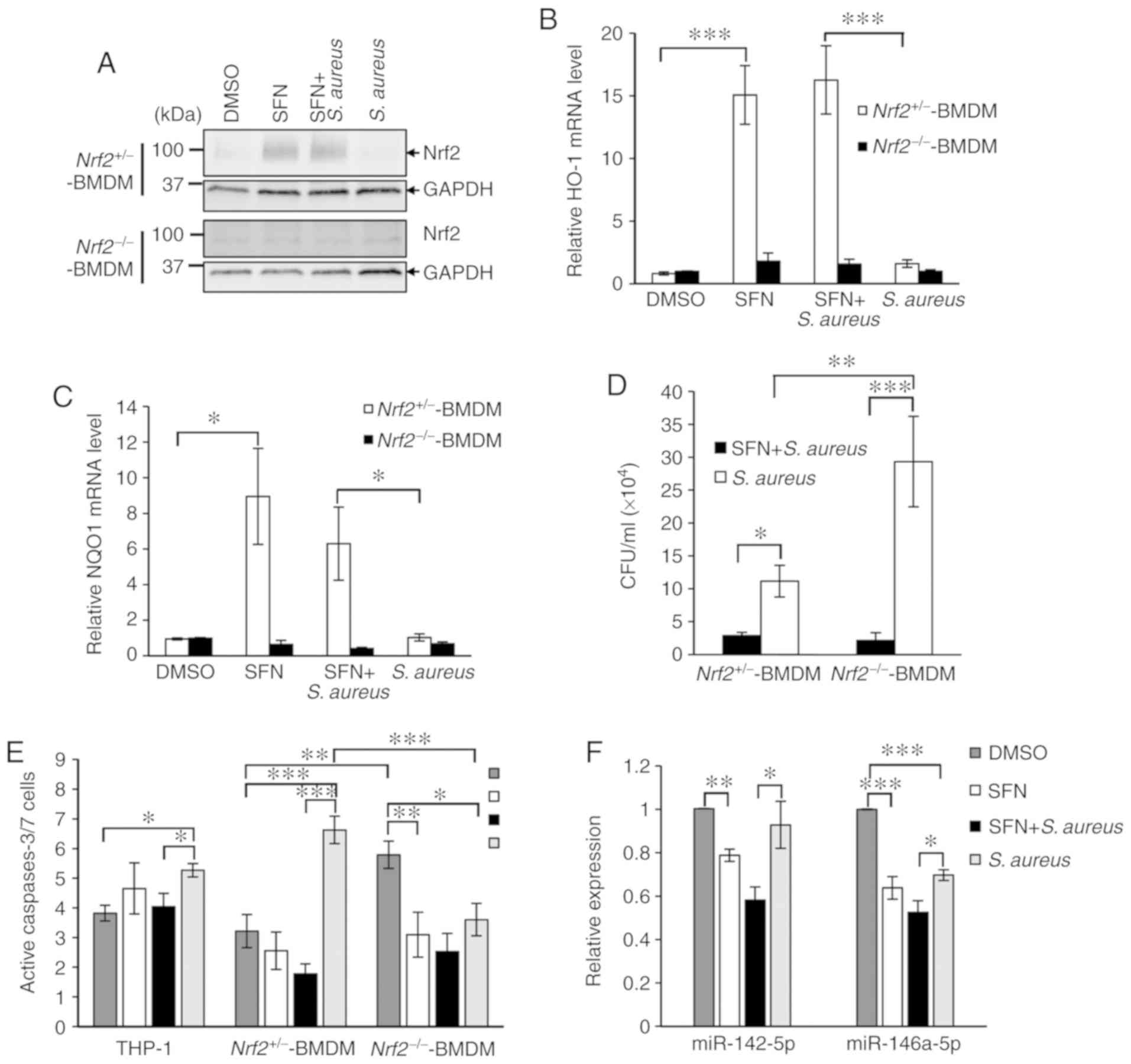 | Figure 2Nrf2-independent modulation of S.
aureus survival and S. aureus-mediated cell apoptosis by
SFN. (A) Primary Nrf2+/− and Nrf2−/− BMDMs
differentiated for 7 days were treated with 10 µM SFN or
DMSO for 24 h. Total protein extracts were analyzed by western blot
analysis using specific antibodies for Nrf2 and GAPDH. The blots
presented are representative of n=3 independent experiments from 3
Nrf2+/− mice and 3 Nrf2−/− mice. Non-specific
signals were detected in Nrf2−/− BMDMs extracts.
Nrf2+/− and Nrf2−/− BMDMs were pretreated
with 10 µM SFN for 3 h prior to S. aureus infection.
RNA extraction was performed 3 h post-infection. (B) HO-1 and (C)
NQO1 mRNA expression levels were quantified by RT-qPCR (n=7
Nrf2+/− mice; n=6 Nrf2−/− mice). (D)
Intracellular survival of S. aureus was determined in
Nrf2+/− and Nrf2−/− BMDMs pretreated with 10
µM SFN or DMSO prior to infection with S. aureus and
addition of gentamycin. After 24 h, CFUs were determined from
Nrf2+/− BMDMs (n=9) and Nrf2−/− BMDMs (n=7).
(E) Caspases 3/7-dependent cell apoptosis was determined 24 h after
S. aureus infection of THP-1-derived macrophages (n=4),
Nrf2+/− BMDMs (n=6) and Nrf2−/− BMDMs (n=5)
pretreated with SFN or DMSO. (F) THP-1-derived macrophages were
pretreated with 10 µM SFN for 3 h prior to infection with
S. aureus. RNA extraction was performed 6 h after
pretreatment (n=5). Expression levels of miR-142-5p and miR-146a-5p
were quantified by RT-qPCR. Statistical analyses were performed
using two-way ANOVA followed by Tukey's test. *P<0.05,
**P<0.01, ***P<0.001. Nrf2, nuclear
factor erythroid 2-related factor 2; S. aureus,
Staphylococcus aureus; SFN, sulforaphane; BMDMs, bone marrow
derived-macrophages; HO-1, heme oxygenase-1; NQO1, NAD(P)H quinone
dehydrogenase 1; RT-qPCR, reverse transcription-quantitative
polymerase chain reaction; CFUs, colony forming units; DMSO,
dimethyl sulfoxide. |
The effect of Nrf2 and Nrf2-knockout on the
intracellular survival of S. aureus in Nrf2−/−
and Nrf2+/− BMDMs pretreated with SFN or DMSO was next
investigated. Similar to THP-1-derived macrophages and primary
PBMC-derived macrophages (Fig. 1B and
C), Nrf2+/− BMDMs exhibited a significant decrease
in intracellular bacterial survival when pretreated with SFN
compared with Nrf2+/− BMDMs pretreated with DMSO
(Fig. 2D). Noticeably, S.
aureus survival levels were significantly decreased following
SFN pretreatment in Nrf2−/− BMDMs, suggesting that the
effect of SFN on bacterial survival was independent of Nrf2.
Furthermore, bacterial survival was increased in the S.
aureus-infected Nrf2−/− BMDMs compared with the
S. aureus-infected Nrf2+/− BMDM, which suggested
a role of Nrf2 signaling in moderating intracellular S.
aureus survival in macrophages, independently of SFN.
Following S. aureus infection, immune cells
can undergo caspase-dependent cell death to contribute to the
control of infection. The resulting apoptotic bodies, comprised of
endo-cytosed pathogens and antigen presenting cell debris,
facilitate T-cell response and clearance by neighboring immune
cells (25). To verify the effect
of SFN on S. aureus mediated caspase-3/7 activity, the
FAM-FLICA caspase-3/7 assay kit was used on THP-1-derived
macrophages or BMDMs pretreated with SFN or DMSO and infected with
S. aureus. Gentamycin was added to the medium 1 h after
infection and cells were incubated for 24 h post-infection.
Apoptosis analysis by flow cytometry indicated that S.
aureus significantly activated caspases-3/7 in THP-1-derived
macrophages and Nrf2+/− BMDMs, while the lack of Nrf2 in
S. aureus-challenged Nrf2−/− BMDMs resulted in a
significant decrease in S. aureus-mediated
caspase-3/7-dependent cell apoptosis as compared with the
DMSO-treated Nrf2−/− BMDMs and S. aureus-infected
Nrf2+/− BMDMs (Fig.
2E). In addition, an increase in caspases-3/7 activity was
detected in the DMSO-treated Nrf2−/− BMDMs as compared
with that measured in the DMSO-treated Nrf2+/− BMDM,
which was inhibited by SFN pretreatment. SFN pretreatment of the
THP-1-derived macrophages and Nrf2+/− BMDMs decreased
the level of S. aureus-triggered caspases-3/7-dependent cell
apoptosis. Notably, this negative effect of SFN was maintained in
the S. aureus-infected Nrf2−/− BMDM. The
expression levels of miR-142-5p and miR-146a-5p have been
associated with cell apoptosis (26,27). Therefore, the effects of SFN on
the levels of these miRNAs were examined. RT-qPCR analyses
demonstrated that SFN pretreatment of the THP-1-derived macrophages
significantly attenuated the S. aureus-mediated increased
expression levels of miR-142-5p and miR-146a-5p (Fig. 2F). Altogether, these results
suggested a negative correlation between decreased S.
aureus-mediated cell apoptosis in Nrf2−/− BMDMs and
increased intracellular bacterial survival. Although Nrf2 clearly
participates in the control of S. aureus intracellular
persistence, the inhibitory effects of SFN on macrophage
bactericidal activity and caspase-dependent cell apoptosis appears
to be Nrf2-independent.
SFN inhibits S. aureus-induced
transcriptional expressions of proinflammatory genes
The inflammatory response triggered by S.
aureus infection in macrophages and the effect of SFN on S.
aureus-induced inflammation were then examined. THP-1-derived
macrophages were pretreated with SFN or DMSO and infected with
S. aureus. Total RNAs were extracted 3 h after infection and
the transcription levels of the genes coding for proinflammatory
cytokines IL-1β, IL-6 and TNF-α were measured by RT-qPCR. The
levels of IL-1β, IL-6 and TNF-α were significantly increased in the
S. aureus-infected THP-1-derived macrophages compared with
the non-infected macrophages (Fig.
3A-C). Pretreatment with SFN prior to bacterial challenge
prevented S. aureus-mediated increase in IL-1β, IL-6 and
TNF-α mRNA expression levels. Similar inhibitory effect of SFN was
observed in primary PBMC-derived macrophages infected with S.
aureus (Fig. 3D-F).
 | Figure 3Inhibitory effect of SFN on the S.
aureus-induced transcription of proinflammatory genes in
macrophages. mRNA expression levels of IL-1β, IL-6 and TNF-α were
determined by reverse transcription-quantitative polymerase chain
reaction at 6 h post-infection in (A-C) THP-1-derived macrophages
(n=7), (D-F) primary PBMC-derived macrophages (n=3), (G-I) primary
Nrf2+/− BMDMs (n=7) and Nrf2−/− BMDMs (n=6).
Fold inductions are presented relative to the expression observed
in the DMSO-treated macrophages. Statistical analyses were
performed using one-way ANOVA followed by Tukey's post hoc test for
A-F and two-way ANOVA followed by Tukey's test for G-I.
*P<0.05 and **P<0.01. SFN,
sulforaphane; S. aureus, Staphylococcus aureus; IL,
interleukin; TNF-α, tumor necrosis factor-α; PBMC, peripheral blood
mononuclear cells; BMDMs, bone marrow derived-macrophages; DMSO,
dimethyl sulfoxide. |
As Nrf2 has been recently demonstrated to directly
affect the transcriptional induction of proinflammatory cytokine
genes inflammation in macrophages, the participation of Nrf2 in the
regulation of SFN-mediated increase was investigated (19). S. aureus significantly
increased the transcriptional expression levels of IL-1β, IL-6 and
TNF-α genes in Nrf2+/− and Nrf2−/− BMDM, and
pretreatment of BMDMs with SFN prevented S. aureus-mediated
transcription of the proinflammatory cytokine genes in both
macrophages expressing Nrf2 and lacking Nrf2 (Fig. 3G-I), suggesting that SFN inhibited
S. aureus-induced inflammation independently of Nrf2.
SFN prevents the S. aureus-induced
expression of M1 marker genes in infected macrophages
Upon bacterial infection, activated macrophages
produce M1-like proinflammatory marker genes including IL-1β, IL-6
and TNF-α (28). The upregulation
of M1 marker genes CCR7 and IL-23 genes in the THP-1-derived
macrophages and the iNOS gene in the Nrf2+/− BMDMs
following S. aureus infection was then confirmed by qPCR
(Fig. 4A-C). SFN pretreatment of
macrophages attenuated the levels of S. aureus-induced M1
marker genes to those observed in non-infected macrophages. These
results suggest that pretreatment with SFN thwarts the classical
activation of macrophages by S. aureus.
SFN modulates the MAPK signaling
pathway
To investigate the molecular mechanisms employed by
SFN to markedly decrease S. aureus-induced inflammation, the
present study focused on the MAPK signaling pathways, as MAPK
cascades are significantly involved in the macrophage inflammatory
responses. ERK, p38 and JNK are the 3 most studied members of the
MAPK family. To determine whether S. aureus activated ERK,
p38 and JNK, THP-1-derived macrophages were pretreated with DMSO or
SFN followed by S. aureus infection. Protein extracts were
examined by western blot analysis, and the phosphorylation states
of ERK, p38 and JNK were determined. Densitometry analysis of total
ERK and phosphorylated ERK protein signals demonstrated a
significant increase in phosphorylated ERK in SFN-treated
macrophages compared with the DMSO-treated macrophages (Fig. 5A and B). No modification in the
activation of the ERK pathway was observed in the S.
aureus-challenged macrophages, while phosphorylation of p38 and
JNK was significantly augmented (Fig.
5C-F). SFN pretreatment of THP-1-derived macrophages notably
prevented activation of p38 and JNK in S. aureus infected
and non-infected macrophages. These results suggested a strong
inhibitory effect of SFN on the p38 and JNK signaling pathways.
MAPK-dependent inhibition of IL-1β, IL-6
and TNF-α mRNA expression levels by SFN
To further investigate whether the anti-inflammatory
effect of SFN on the proin-flammatory cytokine genes IL-1β, IL-6
and TNF-α resulted from the inhibition of p38 or JNK
phosphorylation by SFN, specific p38 and JNK inhibitors were used.
THP-1-derived macrophages were pretreated with the p38 inhibitor
SB203580 or the JNK inhibitor SP600125 prior to SFN treatment
and/or S. aureus infection. qPCR analysis indicated that
SB203580 effectively repressed the S. aureus-mediated
increase in IL-1β, IL-6 and TNF-α gene expression levels when
compared with the DMSO-pretreated macrophages infected with S.
aureus (Fig. 6A-C). In
addition, the decrease in IL-1β and IL-6 gene expression levels by
SB203580 was comparable with that observed in the SFN-pretreated
macrophages challenged with S. aureus. SP600125
significantly decreased the expression levels of IL-6 in the
THP-1-derived macrophages infected with S. aureus compared
with the macrophages pretreated with DMSO and infected with S.
aureus (Fig. 7B). Data
obtained from the Nrf2+/− BMDMs validated the inhibitory
effect of SB203580 on IL-1β and IL-6 gene transcription (Fig. 6D and E), while SP600125
specifically inhibited the transcription of IL-6 and TNF-α genes
(Fig. 6E and F).
 | Figure 6SFN inhibitory effect on p38 and JNK
impacts on S. aureus-mediated expression of proinflammatory
genes and S. aureus survival in macrophages. (A-C)
THP-1-derived macrophages were pretreated with p38 inhibitor SB or
JNK inhibitor SP for 1 h prior to 10 µM SFN or DMSO
treatment. After additional 3 h incubation, cells were infected
with S. aureus. Total RNA was extracted 3 h post-infection
and (A) human IL-1β, (B) IL-6 and (C) TNF-α mRNA expression levels
were determined by RT-qPCR (n=4). (D-F) Nrf2+/− BMDMs
were pretreated with SB203580 and SP600125 prior to 10 µM
SFN or DMSO treatment and S. aureus infection. (D) Mouse
IL-1β, (E) IL-6 and (F) TNF-α mRNA expression levels were
quantified by RT-qPCR (n=5 mice). (G) Intracellular survival of
S. aureus was assessed in THP-1-derived macrophages
pretreated with SB and SP prior to 10 µM SFN or DMSO
treatment and S. aureus infection. CFU counts were
determined 24 h after infection (n=4). *P<0.05 and
***P<0.001 vs. DMSO treatment according to one-way
ANOVA followed by Tukey's post hot test. SFN, sulforaphane; p38,
p38 mitogen-activated protein kinase; JNK, c-Jun N-terminal kinase;
SB, SB203580; SP, SP600125; DMSO, dimethyl sulfoxide; S.
aureus, Staphylococcus aureus; IL, interleukin; TNF-α,
tumor necrosis factor-α; RT-qPCR, reverse
transcription-quantitative polymerase chain reaction; CFU, colony
forming units. |
 | Figure 7Schematic diagram depicting a
hypothetical molecular signaling pathways modulated by SFN in S.
aureus-challenged macrophages. S. aureus activates p38
and JNK MAPK signaling pathways, probably through binding to PRRs
toll-like receptor 2 and/or nucleotide-binding oligomerization
domain-like receptor 2 and upregulation of transcription of genes
coding for proinflammatory cytokines IL-1β, IL-6 and TNF-α. SFN
decreases inflammation and intracellular bacterial survival in
macrophages. SFN decreases cytokine-dependent growth of S.
aureus, cell inflammation and cell apoptosis in S.
aureus-infected macrophages by; i) downregulating transcription
of genes coding for proinflammatory cytokines by inhibiting
phosphorylation of p38 and JNK, and potentially interfering with
S. aureus recognition by PRR; and ii) downregulating
miR-142-5p and miR-146a-5p. SFN, sulforaphane; S. aureus,
Staphylococcus aureus; PRR, pattern-recognition receptors;
p38, p38 mitogen-activated protein kinase; JNK, c-Jun N-terminal
kinase; IL, interleukin; TNF-α, tumor necrosis factor-α; miR,
microRNA. |
As SFN suppressed p38 and JNK phosphorylation it was
of interest to examine whether pretreatment of macrophages with
SB203580 or SP600125 resulted in an attenuation of S. aureus
load in macrophages. The THP-1-derived macrophages were pretreated
with SFN, SB203580 or SP600125 and challenged with S. aureus
for 1 h prior to the addition of gentamycin. CFU counts were
determined after 24 h, and a significant decrease in intracellular
bacterial survival in macrophages treated with SB203580 or SP600125
was observed, at a level comparable with that obtained in SFN
pretreated macrophages (Fig. 6G).
Overall, the results suggested that SFN activation of macrophage
bactericidal activity is based on the inhibition of the macrophage
proinflammatory responses by inhibiting p38 and JNK MAPK
signaling.
Fig. 7 summarizes
the hypothesis regarding the role of SFN in inhibiting S.
aureus recognition, but not its internalization, thereby
preventing p38 and JNK MAPK signaling, and inhibiting
MAPK-dependent upregulation of IL-1β, IL-6 and TNF-α
proinflammatory cytokine genes, as well as potentially averting
macrophage M1 polarization.
Discussion
SFN, a naturally-occurring isothiocyanate present in
abundance in cruciferous vegetables such as broccoli sprouts, has
been demonstrated to possess pleiotropic protective effects against
oxidative stress, various types of cancer, inflammatory diseases
and microbial infections (11-13). In the present study, the molecular
mechanism activated by SFN to improve bacterial clearance in
macrophages challenged with S. aureus was identified. It was
demonstrated that SFN decreases intracellular bacterial survival by
preventing the activation of p38 and JNK signaling pathways
activated by S. aureus, 2 major enzymes implicated in the
production of inflammatory cyto-kines. Macrophages treated with SFN
exhibited a significant decrease in the levels of S.
aureus-induced inflammation, and while the anti-inflammatory
activity of SFN has been previously described, the underlying
cellular mechanisms involved in this process remains unclear. The
use of Nrf2−/− BMDMs also demonstrated that the
inhibitory effects of SFN on intracellular bacterial survival and
caspase-3/7-dependent apoptosis were Nrf2-independent, but
associated with the downregulation of miR-142-5p and
miR-146a-5p.
S. aureus is considered a facultative
intracellular pathogen, as it has been demonstrated to proliferate
extracellularly and intracellularly within various cell types
(29). In chronic and recurrent
infections, S. aureus may persist several days within the
infected macrophages prior to proliferation (30). The present study first validated
that the strain of S. aureus used was able to invade and
survive within the THP-1-derived macrophages. Upon bacterial
recognition by the host innate immune receptors, cascades of highly
dynamic signal transduction systems are sequentially activated and
amplified, resulting in the proinflammatory response required in
the antimicrobial defense mechanism. Therefore, it stands to reason
that bacteria have developed, among their numerous immune evasion
strategies, elaborative mechanisms to exploit the host innate
immune signal transduction pathways (31). The increase in inflammatory
cytokines produced by the inflammatory response in response to
bacterial infection has been associated with an increase in the
rates of bacterial nosocomial infections (32). Bacteria such as S. aureus,
Pseudomonas aeruginosa and Acinetobacter spp.,
incubated with IL-1β, IL-6 and TNF-α, demonstrated cytokine
concentration-dependent growth patterns (32). In patients with atopic dermatitis,
S. aureus recognition mediated an increase in the production
of inflammatory cytokines IL-1β or IFN-γ and promoted the
cytokine-dependent growth enhancement of S. aureus,
increasing the severity of the skin lesion (33). A recent study suggested that S.
aureus-alpha toxin may activate the inflammasome through
activation of the acid sphingomye-linase, which resulted in the
rapid release of cathepsins and the production of IL-1β and TNF-α
in BMDMs (34). The present study
demonstrated that S. aureus infection of macrophages
triggered a robust increase in the transcriptional expression
levels of proinflammatory genes, which was associated with
increased expression levels of M1 marker genes. The intracellular
survival rates of S. aureus were observed to benefit from
this inflammatory environment.
MAPK signaling pathways are central to the
inflammatory response of the innate immune system. In human
bronchial epithelial cells, concomitant infection of influenza
virus and S. aureus synergistically promoted enhanced
phosphorylation of p38, ERK and JNK. Treatment of these cells with
specific inhibitors of p38 and ERK demonstrated that these
signaling pathways were associated with the regulation of IL-6
production (35). Herein, the
THP-1-derived macrophages and Nrf2+/− BMDMs infected
with S. aureus exhibited increased phosphorylation of p38
and JNK MAPK. The rapid inflammatory response was reflected in the
marked increase in IL-1β, IL-6, and TNF-α mRNA expression levels in
the S. aureus-infected macrophages. IL-1β, IL-6 and TNF-α
are proinflammatory cytokines known to initiate and regulate the
immune response and inflammation. The use of specific MAPK
inhibitors for p38 and JNK signaling allowed a better undestanding
of the role served by each member of the MAPK family. Based on the
results, it was determined that the transcriptional expression
levels of the IL-1β, IL-6 and TNF-α genes were p38- and
JNK-dependent.
In previous years, a number of compounds have been
developed to inhibit protein kinases, with several drugs approved
for the treatment of cancer. For treatments of neuroinflammatory
disorders, several anti-inflammatory compounds specifically
targeting the classical MAPKs, such as p38α MAPK inhibitors, were
assessed in clinical trials but were rapidly discontinued as a
result of poor safety profiles or low long-term efficacy (36). Several natural or chemical
compounds have also been previously used to modulate the S.
aureus-mediated inflammatory response by acting on
intracellular signal transduction pathways. In a mouse model of
S. aureus-induced mastitis, mice treated with the natural
compound brazilin, present in Caesalpinia sappan heartwood,
exhibited a decrease in levels of the S. aureus-induced
inflammatory cytokines IL-1β, IL-6 and TNF-α, leading to a decrease
in inflammatory-mediated tissue injury. In addition, an inhibition
of the S. aureus-induced phosphorylation of p38, JNK and ERK
in the brazilin-treated mice was observed (37). Similar regulatory mechanisms of
NF-κB and MAPK signaling pathways were observed in S.
aureus-infected RAW 264.7-derived macrophages treated with
selenium derivatives. The downregulation of NF-κB and MAPK
signaling pathways by selenium was suggested to be associated with
the decrease in TNF-α, IL-1β and IL-6 transcriptional expression
levels and their cytokine release (38). In addition, treatment of an S.
aureus-induced peritonitis mouse model with an ephedrine
derivative increased the survival rate of the infected mice by
decreasing inflammation through the modulation of the PI3K/AKT and
p38 signaling pathways (39).
Based on these data, we hypothesized that SFN, known for its
anti-inflammatory and antioxidant properties, was able to affect
the S. aureus-mediated increase in the inflammatory response
and decrease bacterial survival in S. aureus-infected
macrophages. THP-1-derived macrophages, primary PBMC-derived
macrophages or primary BMDM, pretreated with SFN and challenged
with S. aureus, exhibited a signifi-cant decrease in
bacterial survival 24 h after internalization. In addition, SFN
significantly inhibited the S. aureus-mediated activation of
p38 and JNK MAPK in macrophages. In concordance with these results,
inhibition of p38 and JNK signaling with SB203580 and SP600125
inhibitors resulted in similar beneficial effects on macrophages
compared with those observed in the SFN-treated macrophages, such
as the significant repression of S. aureus-mediated
upregulation of the proinflammatory genes IL-1β, IL-6 and TNF-α,
and the subsequent decrease in intracellular survival rate of S.
aureus.
As the pathogen-associated molecular patterns of
S. aureus are recognized by pattern recognition receptors
(PRRs), there is a strong possibility that the anti-inflammatory
effect of SFN relies on the disruption of the receptors preventing
pathogen recognition. Toll-like receptors (TLR) are PRRs that
recognize pathogen patterns or cell debris, and activate downstream
TLR signaling. S. aureus binds to membrane protein TLR2,
thereby activating a cascade of proinflammatory responses through
NF-κB and MAPK signaling pathways. Watanabe et al (40) suggested that the TLR2-mediated
activation of JNK decreased the level of superoxide in S.
aureus-infected macrophages, resulting in the prolonged
intracellular survival of S. aureus in phagosomes. In
addition, using LPS-stimulated RAW264.7 macrophages, Youn et
al (41) demonstrated that
SFN suppressed the TLR4-mediated increase in inflammation by
hindering TLR4 oligomerization in a thiol-dependent manner.
However, alternative pathogen receptors also participated in S.
aureus recognition, as TLR2-deficient mice were still able to
produce decreased but significant levels of cytokines in response
to S. aureus infection (42). Cytosolic receptors such as
nucleotide-binding oligomerization domain (NOD)-like receptors, in
particular NOD2, participate in intracellular S. aureus
recognition (43). Similar to
TLR2, NOD2 mediates cytokine responses through activation of the
NF-κB and MAPK signaling pathways. Experiments performed on primary
epithelial cells isolated from NOD2-deficient mice and
TLR2-deficient mice indicated that staphylococcal peptidoglycan
co-localized with NOD2 and TLR2. In both mutants, the levels of
IL-1β and IL-6 were decreased by one-half compared with the
wild-type cells, suggesting the additive effects of each receptor
(44). In addition, it is
unlikely that SFN disruption of TLR2 oligomerization affects the
phagocytosis process of macrophages, as TLR2-deficient macrophages
maintain their phagocytic activity (45), which concurs with the similarities
in internalized S. aureus levels between DMSO- and
SFN-pretreated macrophages observed in the present study. Based on
these data, we hypothesize that SFN decreases the recognition of
S. aureus with cell wall components by impeding TLR2
heterodimerization, thereby preventing the downstream activation of
NF-κB and MAPK signaling pathways, and inhibiting the production of
proinflammatory cytokines. Future experiments are required to
determine whether SFN interferes with bacterial invasion,
intracellular survival, and/or replication, whether the
anti-inflammatory and anti-apoptotic molecular mechanisms activated
by SFN are effective against methicillin-resistant S.
aureus, and whether SFN affects the phagosomal maturation
process in bacteria-infected macrophages.
Using BMDMs isolated from the bone marrow of
Nrf2+/− and Nrf2−/− mice, the present study
demonstrated that Nrf2 was necessary in promoting S.
aureus-mediated caspases-3/7-dependent cell death. Previously,
the anti-inflammatory contribution of Nrf2 was hypothesized to be
an indirect effect of the upregulation of genes coding for
antioxidant enzymes, with the removal of ROS resulting in a
decreased inflammation (46).
Herein, as the ROS levels remained unaffected by either S.
aureus challenge or SFN stimulation in THP-1-derived
macrophages, the contribution of ROS in the regulation of
inflammation by SFN was excluded. Despite the marked activation of
ERK MAPK and Nrf2 signaling pathways by SFN, the results of the
present study clearly established that SFN affects intracellular
bacterial survival independently of Nrf2. In contrast with the
observations of the present study, several studies have
demonstrated that the inhibition of microbial survival in
macrophages treated with SFN involved the Nrf2 signaling pathway.
In HIV infection, SFN did not interfere with viral entry, but has
been demonstrated to inhibit HIV infection in macrophages through
Nrf2 activation by preventing viral insertion into host chromosomes
(15). In addition, we have
previously reported that intracellular survival of Mycobacterium
abscessus was decreased through SFN-induced Nrf2-mediated cell
apoptosis (23).
Recent studies have associated the expression
levels of several miRNAs with fundamental cellular processes such
as cell apoptosis, phagocytosis, inflammation and macrophage
polarization upon bacterial infection (20,47). The present study demonstrated that
expression levels of miR-142-5p and miR-146a-5p are negatively
regulated by SFN, suggesting their involvement in SFN-mediated
decrease of cell apoptosis. miR-146a-5p expression levels are
induced by inflammatory stimuli including TNF-α and IL-1β, and
exert an anti-inflammatory effect by inhibiting pro-inflammatory
genes such as IL-1 receptor associated kinase 1 and TNF receptor
associated factor 6 in human adipocytes (48,49). We hypothesize that the lower than
expected expression levels of miR-142-5p and miR-146a-5p observed
in THP-1-derived macrophages infected by S. aureus may be
due to the short incubation time (3 h after infection) in the
experiment in the present study. Additional experiments, including
increasing the infection time period, or using primary cells such
as BMDMs or human PBMCs, will improve the understanding of the
effect of SFN-mediated negative regulation of miR-142-5p,
miR-146a-5p and other miRNAs on cellular processes such as
inflammation, phago-cytosis and macrophage polarization, as well as
identify their target mRNAs.
In conclusion, this study proposed a more
comprehensive view of the molecular mechanism activated by SFN to
promote bacteria clearance. Unlike pharmacological compounds that
specifically target protein kinases, the present study demonstrated
that due to the pleiotropic effects of SFN, SFN may possibly affect
a wide range of molecular targets in S. aureus-activated
macrophages. The present results suggested that using SFN to
regulate the MAPK-inflammatory response pathway and interfere with
S. aureus-induced apoptosis may be a promising therapeutic
approach to decrease S. aureus intracellular survival.
Supplementary Data
Acknowledgements
Not applicable.
Funding
This study was supported by the Legs Poix funding
from the Chancellerie des Universités de Paris (MB), the National
Institute of Health and Medical Research funding (MB and TD), and
the University of Versailles-Saint-Quentin-en-Yvel ines funding (MB
and TD).
Availability of data and materials
The datasets used and/or analyzed during the
current study are available from the corresponding author on
reasonable request.
Authors' contributions
TBD conceived, designed and performed the
experiments, analyzed and interpreted data, and wrote the
manuscript. MA performed the experiments. SV interpreted data and
reviewed the manuscript. MB conceived the study, interpreted data
and reviewed the manuscript. All authors read and approved the
final manuscript.
Ethics approval and consent to
participate
The protocols were approved by the Institutional
Animal Ethics Committee CEEA47 PELVIPHARM of the University of Vers
ailles-Saint-Quentin-en-Yvelines (Montigny-le-Bretonneux, France),
and the Ministry of Higher Education, Research and Innovation.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Arango Duque G and Descoteaux A:
Macrophage cytokines: Involvement in immunity and infectious
diseases. Front Immunol. 5:4912014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Arthur JS and Ley SC: Mitogen-activated
protein kinases in innate immunity. Nat Rev Immunol. 13:679–692.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Chen GY and Nunez G: Sterile inflammation:
Sensing and reacting to damage. Nat Rev Immunol. 10:826–837. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Iwasaki A and Medzhitov R: Regulation of
adaptive immunity by the innate immune system. Science.
327:291–295. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Murray PJ and Wynn TA: Protective and
pathogenic functions of macrophage subsets. Nat Rev Immunol.
11:723–737. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Gomez MI, Lee A, Reddy B, Muir A, Soong G,
Pitt A, Cheung A and Prince A: Staphylococcus aureus protein A
induces airway epithelial inflammatory responses by activating
TNFR1. Nat Med. 10:842–848. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kujime K, Hashimoto S, Gon Y, Shimizu K
and Horie T: p38 mitogen-activated protein kinase and
c-jun-NH2-terminal kinase regulate RANTES production by influenza
virus-infected human bronchial epithelial cells. J Immunol.
164:3222–3228. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
DeLeo FR, Otto M, Kreiswirth BN and
Chambers HF: Community-associated meticillin-resistant
Staphylococcus aureus. Lancet. 375:1557–1568. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Parker D, Ahn D, Cohen T and Prince A:
Innate immune signaling activated by MDR bacteria in the airway.
Physiol Rev. 96:19–53. 2016. View Article : Google Scholar :
|
|
10
|
Fraunholz M and Sinha B: Intracellular
Staphylococcus aureus: Live-in and let die. Front Cell Infect
Microbiol. 2:432012. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fahey JW, Haristoy X, Dolan PM, Kensler
TW, Scholtus I, Stephenson KK, Talalay P and Lozniewski A:
Sulforaphane inhibits extracellular, intracellular, and
antibiotic-resistant strains of Helicobacter pylori and prevents
benzo[a] pyrene-induced stomach tumors. Proc Natl Acad Sci USA.
99:7610–7615. 2002. View Article : Google Scholar
|
|
12
|
Romeo L, Iori R, Rollin P, Bramanti P and
Mazzon E: Isothiocyanates: An overview of their antimicrobial
activity against human infections. Molecules. 23:E6242018.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sita G, Hrelia P, Graziosi A and Morroni
F: Sulforaphane from cruciferous vegetables: Recent advances to
improve glioblastoma treatment. Nutrients. 10:E17552018. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Johansson NL, Pavia CS and Chiao JW:
Growth inhibition of a spectrum of bacterial and fungal pathogens
by sulforaphane, an isothiocyanate product found in broccoli and
other cruciferous vegetables. Planta Med. 74:747–750. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Furuya AK, Sharifi HJ, Jellinger RM,
Cristofano P, Shi B and de Noronha CM: Sulforaphane Inhibits HIV
Infection of Macrophages through Nrf2. PLoS Pathog.
12:e10055812016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Itoh K, Chiba T, Takahashi S, Ishii T,
Igarashi K, Katoh Y, Oyake T, Hayashi N, Satoh K, Hatayama I, et
al: An Nrf2/small Maf heterodimer mediates the induction of phase
II detoxifying enzyme genes through antioxidant response elements.
Biochem Biophys Res Commun. 236:313–322. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Itoh K, Mochizuki M, Ishii Y, Ishii T,
Shibata T, Kawamoto Y, Kelly V, Sekizawa K, Uchida K and Yamamoto
M: Transcription factor Nrf2 regulates inflammation by mediating
the effect of 15-deoxy-Delta(12,14)-prostaglandin j(2). Mol Cell
Biol. 24:36–45. 2004. View Article : Google Scholar :
|
|
18
|
Ishii Y, Itoh K, Morishima Y, Kimura T,
Kiwamoto T, Iizuka T, Hegab AE, Hosoya T, Nomura A, Sakamoto T, et
al: Transcription factor Nrf2 plays a pivotal role in protection
against elastase-induced pulmonary inflammation and emphysema. J
Immunol. 175:6968–6975. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Kobayashi EH, Suzuki T, Funayama R,
Nagashima T, Hayashi M, Sekine H, Tanaka N, Moriguchi T, Motohashi
H, Nakayama K and Yamamoto M: Nrf2 suppresses macrophage
inflammatory response by blocking proinflammatory cytokine
transcription. Nat Commun. 7:116242016. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Zhou X, Li X and Wu M: miRNAs reshape
immunity and inflammatory responses in bacterial infection. Signal
Transduct Target Ther. 3:142018. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Selbach M, Schwanhausser B, Thierfelder N,
Fang Z, Khanin R and Rajewsky N: Widespread changes in protein
synthesis induced by microRNAs. Nature. 455:58–63. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Fuentes F, Paredes-Gonzalez X and Kong AN:
Dietary glucosinolates sulforaphane, phenethyl isothiocyanate,
indole-3-carbinol/3,3′-diin-dolylmethane: Anti-oxidative
stress/inflammation, Nrf2, epigenetics/epigenomics and in vivo
cancer chemopreventive efficacy. Curr Pharmacol Rep. 1:179–196.
2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Bonay M, Roux AL, Floquet J, Retory Y,
Herrmann JL, Lofaso F and Deramaudt TB: Caspase-independent
apoptosis in infected macrophages triggered by sulforaphane via
Nrf2/p38 signaling pathways. Cell death Discov. 1:150222015.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
25
|
Jorgensen I, Rayamajhi M and Miao EA:
Programmed cell death as a defence against infection. Nat Rev
Immunol. 17:151–164. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Cheng D, Li J, Zhang L and Hu L:
miR-142-5p suppresses proliferation and promotes apoptosis of human
osteosarcoma cell line, HOS, by targeting PLA2G16 through the
ERK1/2 signaling pathway. Oncol Lett. 17:1363–1371. 2019.PubMed/NCBI
|
|
27
|
Shu L, Zhang W, Huang G, Huang C, Zhu X,
Su G and Xu J: Troxerutin attenuates myocardial cell apoptosis
following myocardial ischemia-reperfusion injury through inhibition
of miR-146a-5p expression. J Cell Physiol. 234:9274–9282. 2019.
View Article : Google Scholar
|
|
28
|
Atri C, Guerfali FZ and Laouini D: Role of
human macrophage polarization in inflammation during infectious
diseases. Int J Mol Sci. 19:E18012018. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Sendi P and Proctor RA: Staphylococcus
aureus as an intracellular pathogen: The role of small colony
variants. Trends Microbiol. 17:54–58. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Hamza T and Li B: Differential responses
of osteoblasts and macrophages upon Staphylococcus aureus
infection. BMC Microbiol. 14:2072014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Reddick LE and Alto NM: Bacteria fighting
back: How pathogens target and subvert the host innate immune
system. Mol Cell. 54:321–328. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Meduri GU, Kanangat S, Stefan J, Tolley E
and Schaberg D: Cytokines IL-1beta, IL-6, and TNF-alpha enhance in
vitro growth of bacteria. Am J Respir Crit Care Med. 160:961–967.
1999. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Di Domenico EG, Cavallo I, Bordignon V,
Prignano G, Sperduti I, Gurtner A, Trento E, Toma L, Pimpinelli F,
Capitanio B and Ensoli F: Inflammatory cytokines and biofilm
production sustain Staphylococcus aureus outgrowth and persistence:
A pivotal interplay in the pathogenesis of atopic dermatitis. Sci
Rep. 8:95732018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Ma J, Gulbins E, Edwards MJ, Caldwell CC,
Fraunholz M and Becker KA: Staphylococcus aureus α-toxin induces
inflammatory cytokines via lysosomal acid sphingomyelinase and
ceramides. Cell Physiol Biochem. 43:2170–2184. 2017. View Article : Google Scholar
|
|
35
|
Klemm C, Bruchhagen C, van Kruchten A,
Niemann S, Löffler B, Peters G, Ludwig S and Ehrhardt C:
Mitogen-activated protein kinases (MAPKs) regulate IL-6
over-production during concomitant influenza virus and
Staphylococcus aureus infection. Sci Rep. 7:424732017. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Cohen P: Targeting protein kinases for the
development of anti-inflammatory drugs. Curr Opin Cell Biol.
21:317–324. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Gao XJ, Wang TC, Zhang ZC, Cao YG, Zhang
NS and Guo MY: Brazilin plays an anti-inflammatory role with
regulating Toll-like receptor 2 and TLR 2 downstream pathways in
Staphylococcus aureus-induced mastitis in mice. Int
Immunopharmacol. 27:130–137. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Bi CL, Wang H, Wang YJ, Sun J, Dong JS,
Meng X and Li JJ: Selenium inhibits Staphylococcus aureus-induced
inflammation by suppressing the activation of the NF-κB and MAPK
signalling pathways in RAW264.7 macrophages. Eur J Pharmacol.
780:159–165. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
He W, Ma J, Chen Y, Jiang X, Wang Y, Shi
T, Zhang Q, Yang Y, Jiang X, Yin S, et al: Ephedrine hydrochloride
protects mice from Staphylococcus aureus-induced peritonitis. Am J
Transl Res. 10:670–683. 2018.
|
|
40
|
Watanabe I, Ichiki M, Shiratsuchi A and
Nakanishi Y: TLR2-mediated survival of Staphylococcus aureus in
macrophages: A novel bacterial strategy against host innate
immunity. J Immunol. 178:4917–4925. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Youn HS, Kim YS, Park ZY, Kim SY, Choi NY,
Joung SM, Seo JA, Lim KM, Kwak MK, Hwang DH and Lee JY:
Sulforaphane suppresses oligomerization of TLR4 in a
thiol-dependent manner. J Immunol. 184:411–419. 2010. View Article : Google Scholar
|
|
42
|
Takeuchi O, Hoshino K and Akira S: Cutting
edge: TLR2-deficient and MyD88-deficient mice are highly
susceptible to Staphylococcus aureus infection. J Immunol.
165:5392–5396. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hruz P, Zinkernagel AS, Jenikova G, Botwin
GJ, Hugot JP, Karin M, Nizet V and Eckmann L: NOD2 contributes to
cutaneous defense against Staphylococcus aureus through
alpha-toxin-dependent innate immune activation. Proc Natl Acad Sci
USA. 106:12873–12878. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Muller-Anstett MA, Muller P, Albrecht T,
Nega M, Wagener J, Gao Q, Kaesler S, Schaller M, Biedermann T and
Götz F: Staphylococcal peptidoglycan co-localizes with Nod2 and
TLR2 and activates innate immune response via both receptors in
primary murine keratinocytes. PLoS One. 5:e131532010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Villamon E, Gozalbo D, Roig P, O'Connor
JE, Fradelizi D and Gil ML: Toll-like receptor-2 is essential in
murine defenses against Candida albicans infections. Microbes
infect. 6:1–7. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Kong X, Thimmulappa R, Kombairaju P and
Biswal S: NADPH oxidase-dependent reactive oxygen species mediate
amplified TLR4 signaling and sepsis-induced mortality in
Nrf2-deficient mice. J Immunol. 185:569–577. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Tanaka K, Kim SE, Yano H, Matsumoto G,
Ohuchida R, Ishikura Y, Araki M, Araki K, Park S, Komatsu T, et al:
MiR-142 is required for staphylococcus aureus clearance at skin
wound sites via small GTPase-mediated regulation of the neutrophil
actin cytoskeleton. J Invest Dermatol. 137:931–940. 2017.
View Article : Google Scholar
|
|
48
|
Lambert KA, Roff AN, Panganiban RP,
Douglas S and Ishmael FT: MicroRNA-146a is induced by inflammatory
stimuli in airway epithelial cells and augments the
anti-inflammatory effects of glucocorticoids. PLoS One.
13:e02054342018. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Roos J, Enlund E, Funcke JB, Tews D,
Holzmann K, Debatin KM, Wabitsch M and Fischer-Posovszky P:
miR-146a-mediated suppression of the inflammatory response in human
adipocytes. Sci Rep. 6:383392016. View Article : Google Scholar : PubMed/NCBI
|





















