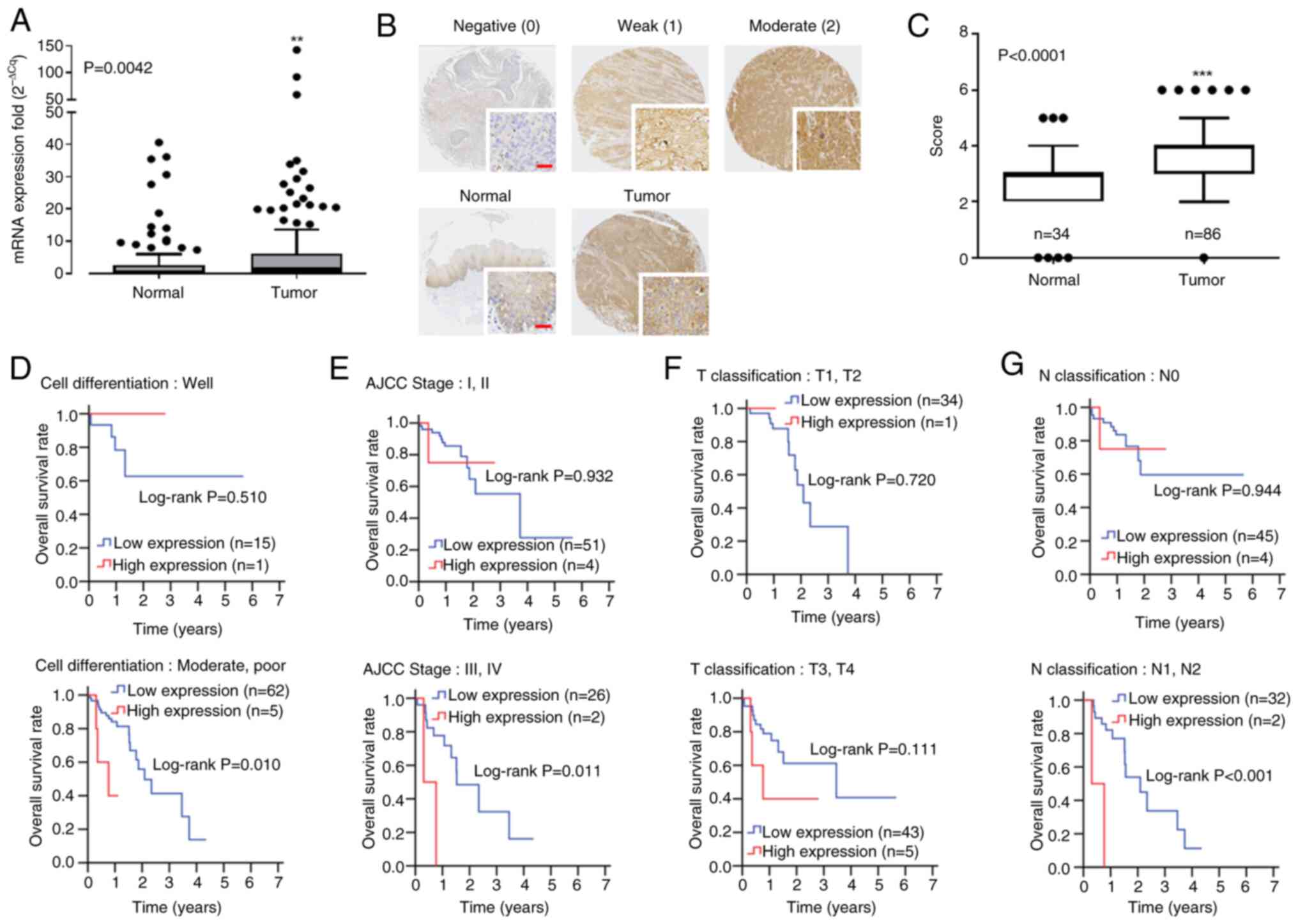
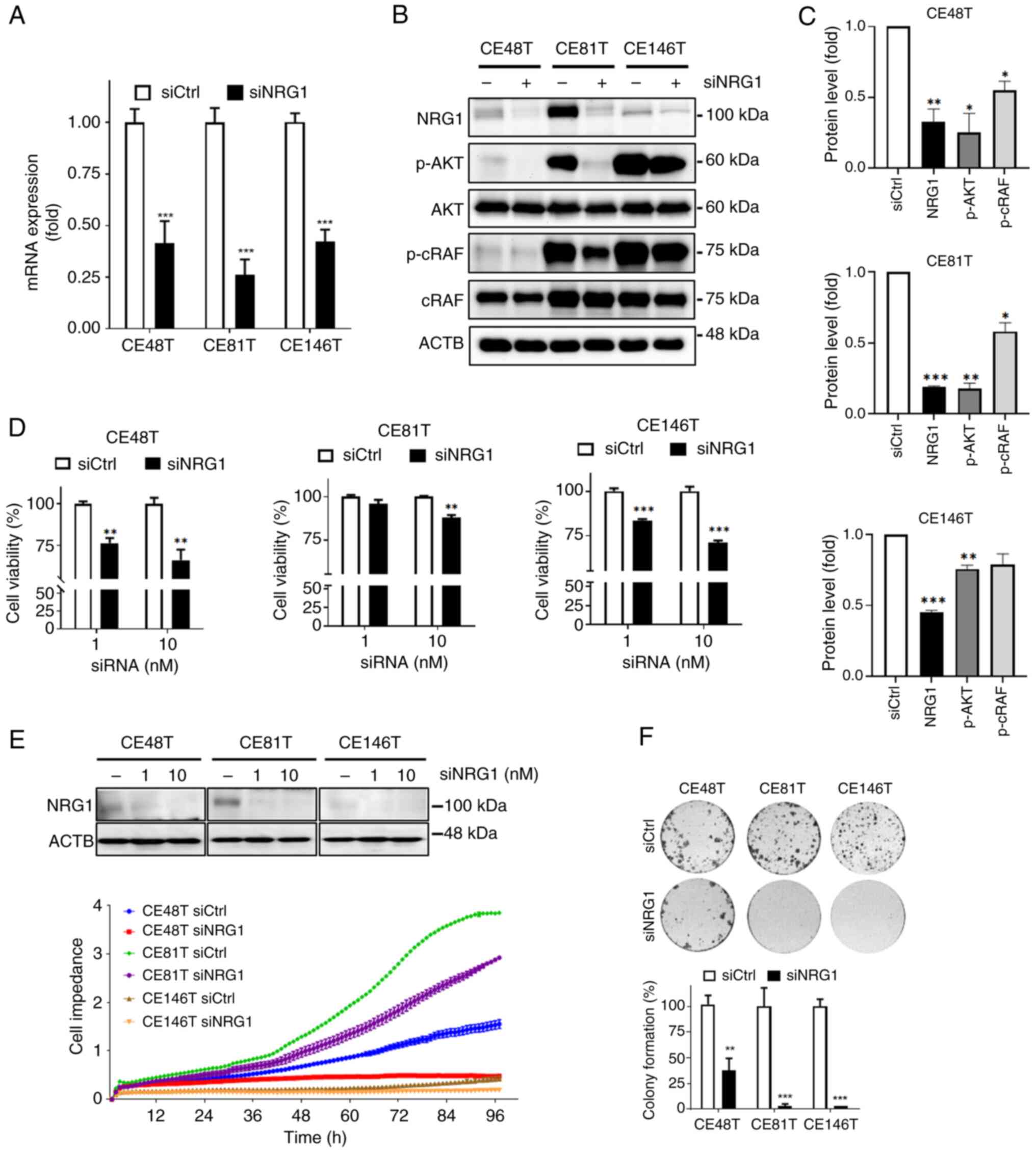
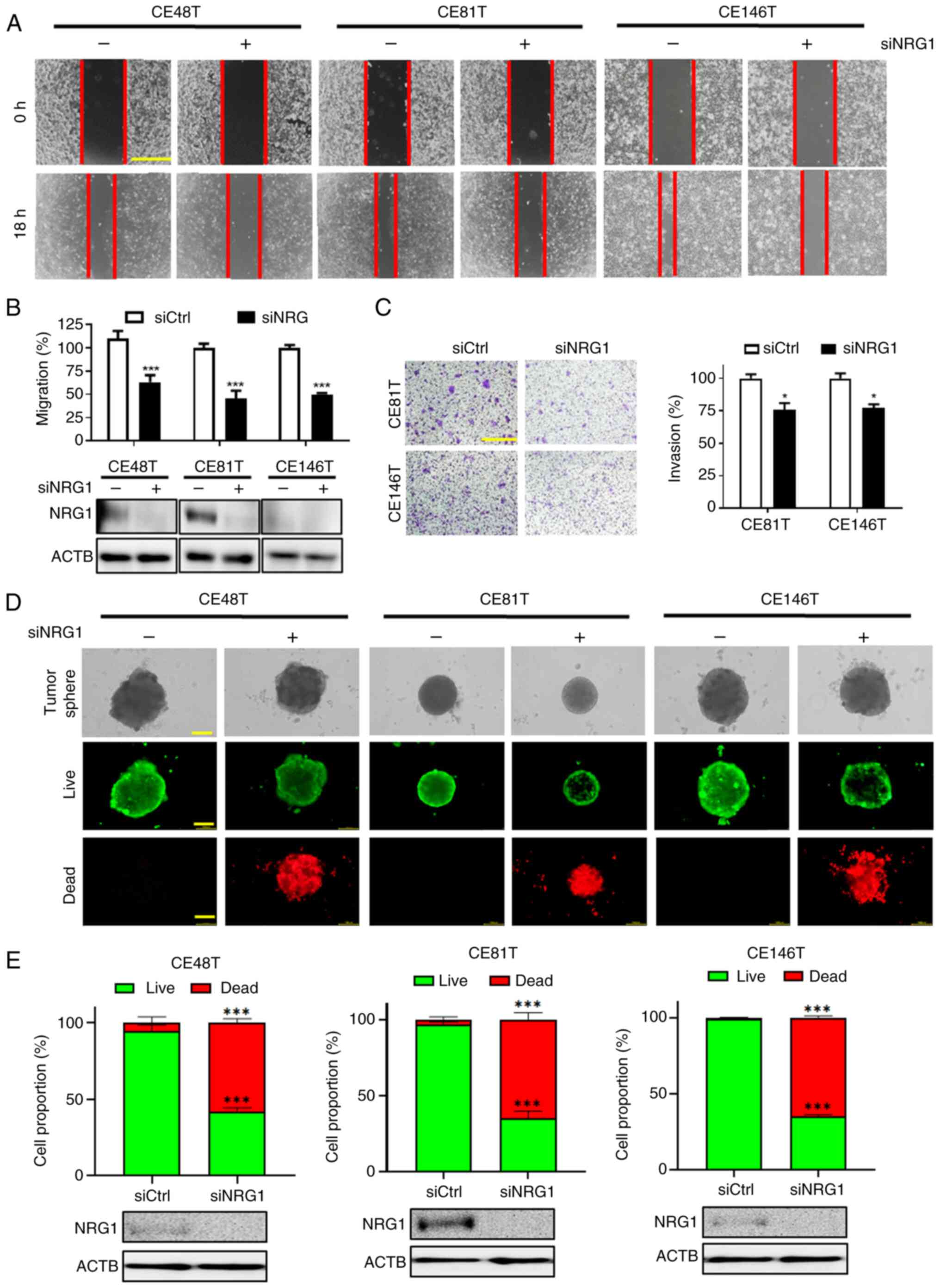
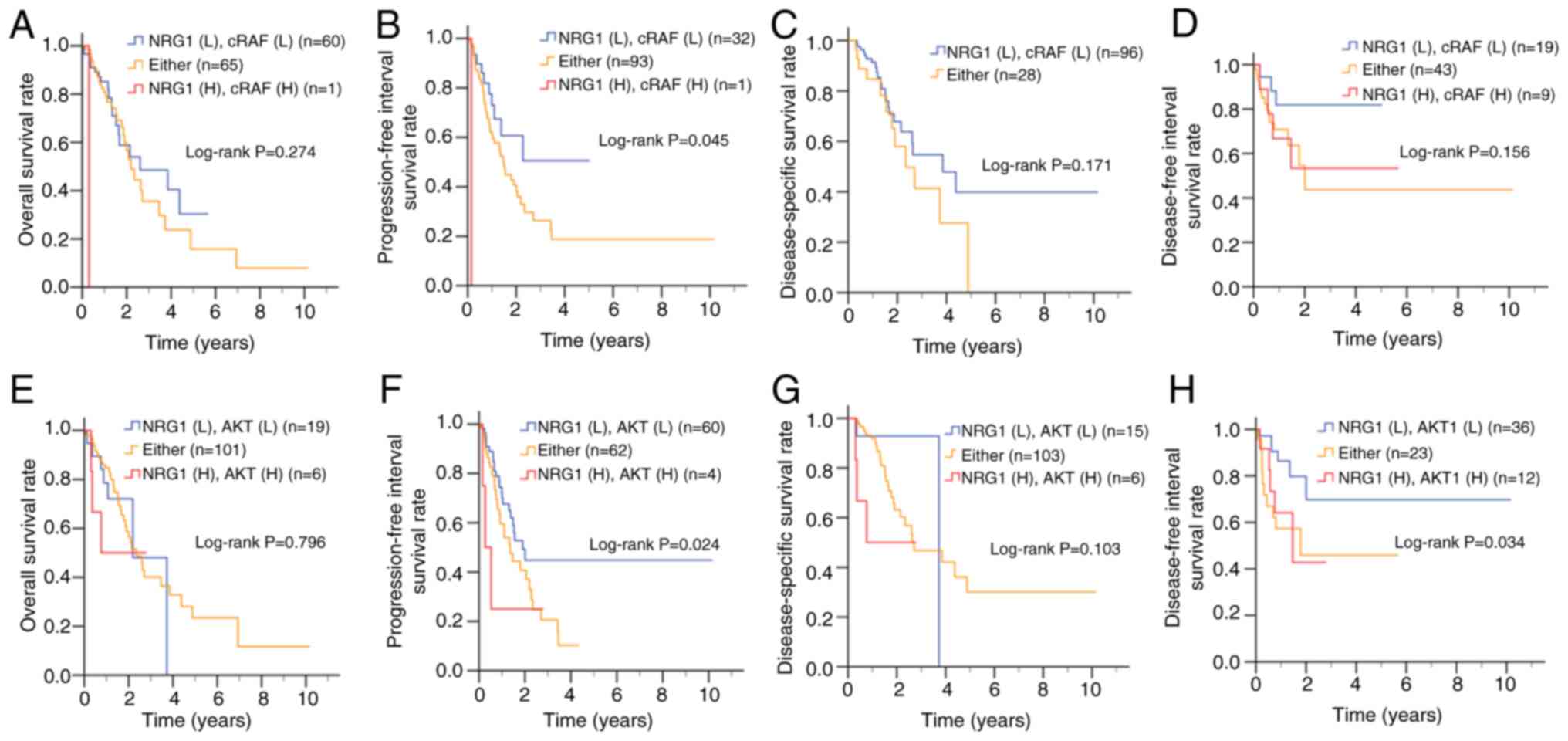
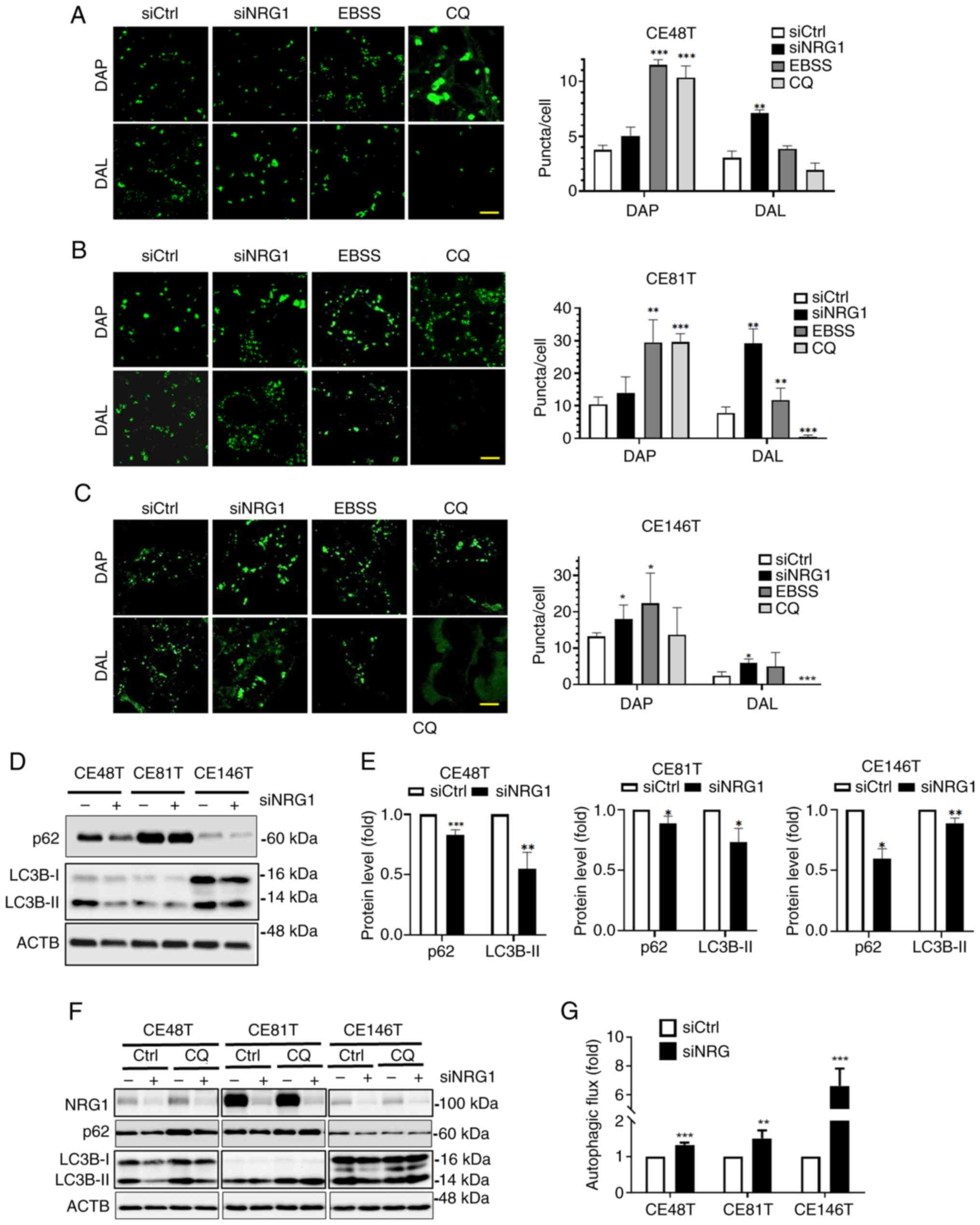
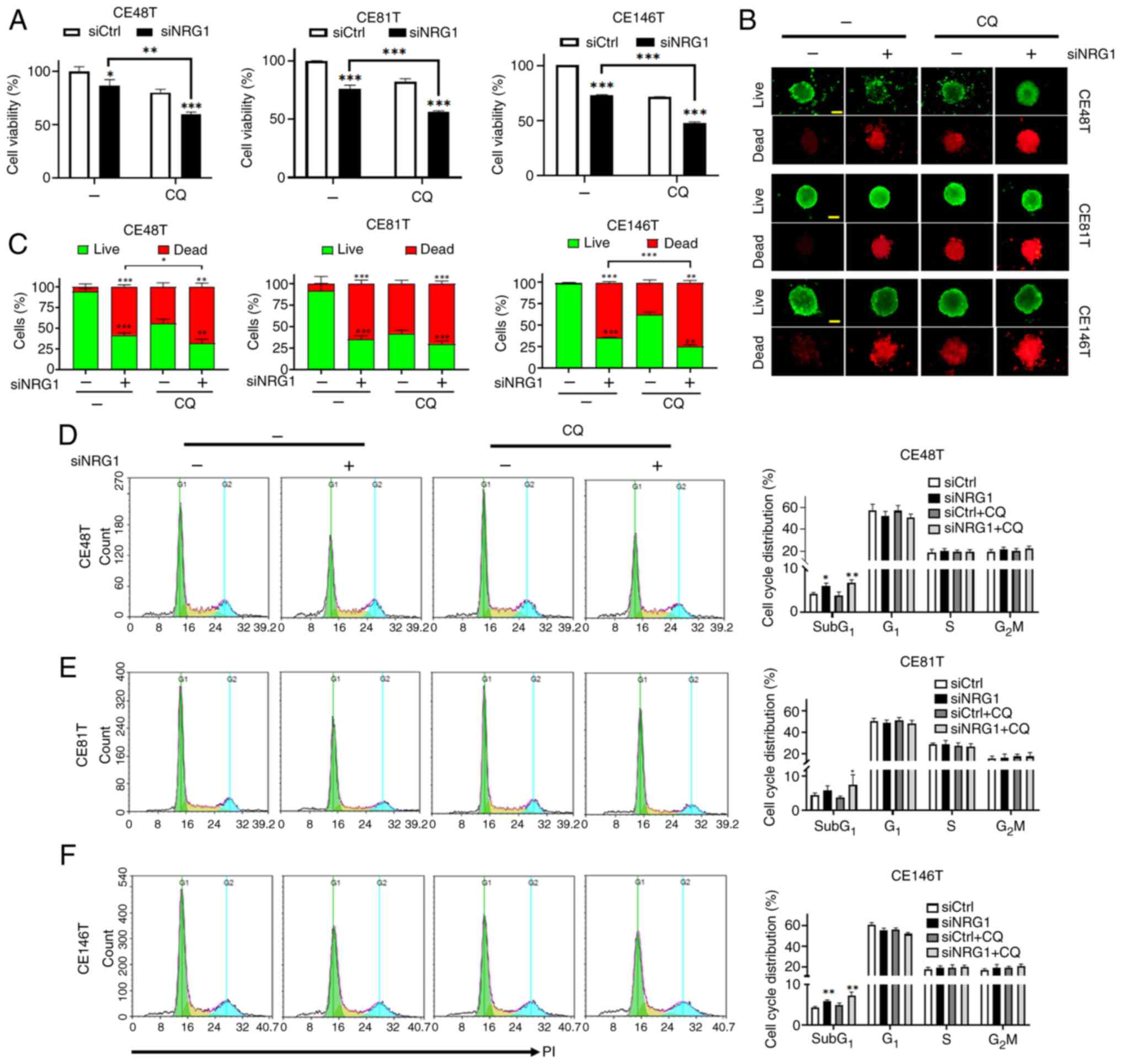
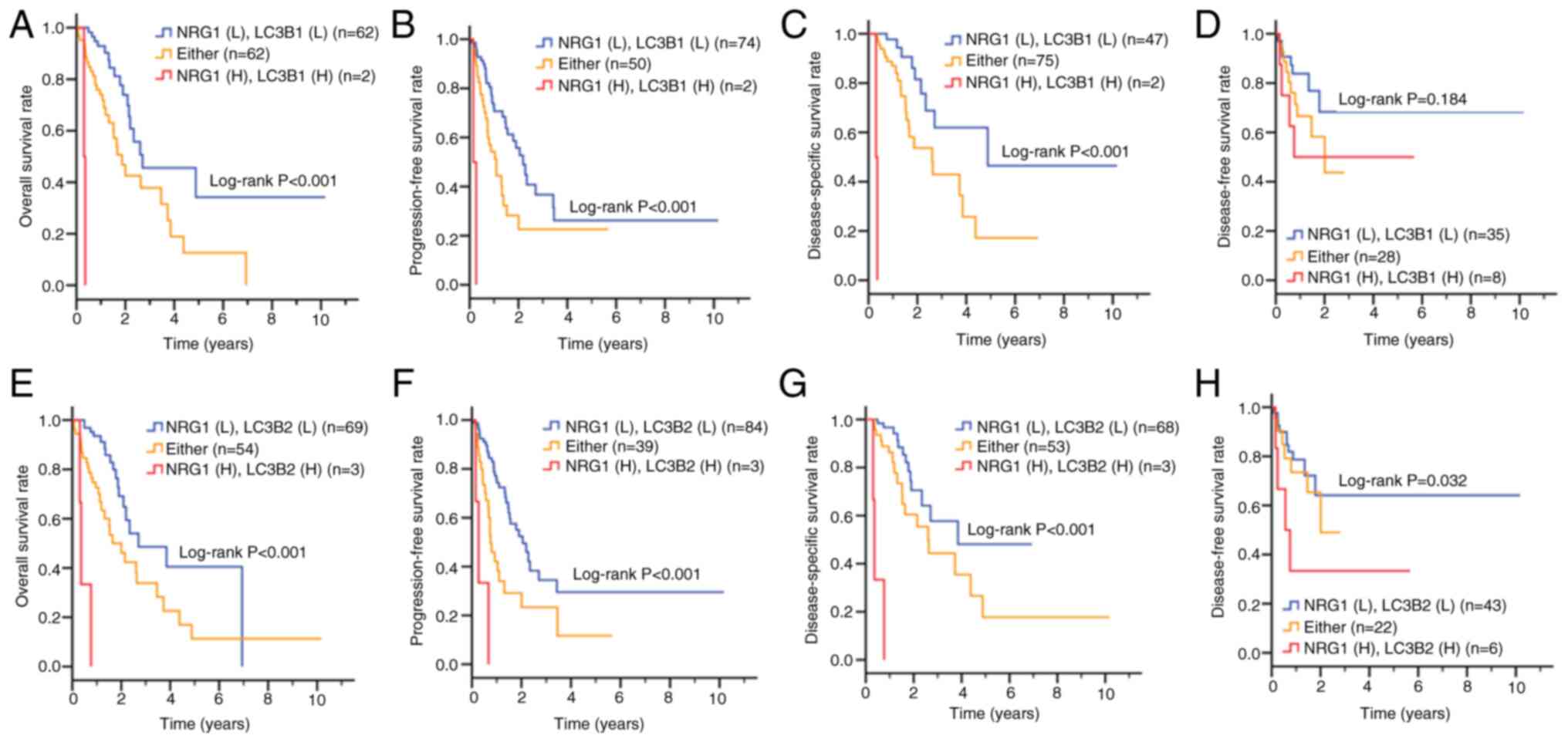
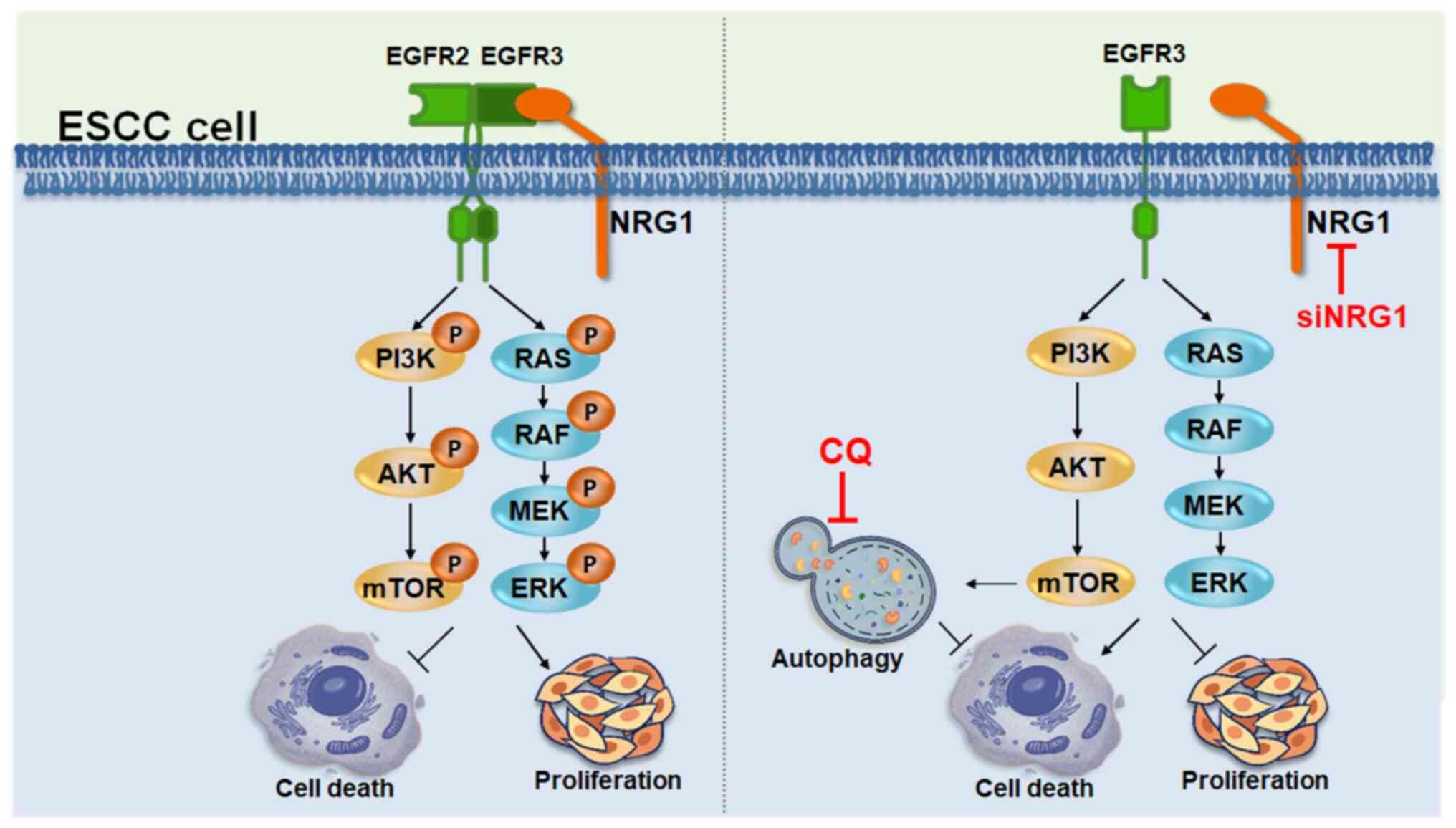
|
1
|
Bray F, Laversanne M, Sung H, Ferlay J,
Siegel RL, Soerjomataram I and Jemal A: Global cancer statistics
2022: GLOBOCAN estimates of incidence and mortality worldwide for
36 cancers in 185 countries. CA Cancer J Clin. 74:229–263. 2024.
View Article : Google Scholar
|
|
2
|
Rustgi AK and El-Serag HB: Esophageal
carcinoma. N Engl J Med. 371:2499–509. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liu CQ, Ma YL, Qin Q, Wang PH, Luo Y, Xu
PF and Cui Y: Epidemiology of esophageal cancer in 2020 and
projections to 2030 and 2040. Thorac Cancer. 14:3–11. 2023.
View Article : Google Scholar :
|
|
4
|
Napier KJ, Scheerer M and Misra S:
Esophageal cancer: A Review of epidemiology, pathogenesis, staging
workup and treatment modalities. World J Gastrointest Oncol.
6:112–120. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Brown LM, Hoover RN, Greenberg RS,
Schoenberg JB, Schwartz AG, Swanson GM, Liff JM, Silverman DT,
Hayes RB and Pottern LM: Are racial differences in squamous cell
esophageal cancer explained by alcohol and tobacco use? J Natl
Cancer Inst. 86:1340–1345. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Mao WM, Zheng WH and Ling ZQ:
Epidemiologic risk factors for esophageal cancer development. Asian
Pac J Cancer Prev. 12:2461–2466. 2011.
|
|
7
|
Liao HY, Wang GP, Gu LJ, Huang SH, Chen
XL, Li Y and Cai SW: HIF-1α siRNA and cisplatin in combination
suppress tumor growth in a nude mice model of esophageal squamous
cell carcinoma. Asian Pac J Cancer Prev. 13:473–477. 2012.
View Article : Google Scholar
|
|
8
|
Sun L and Yu S: Meta-analysis:
Non-steroidal anti-inflammatory drug use and the risk of esophageal
squamous cell carcinoma. Dis Esophagus. 24:544–549. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Enzinger PC and Mayer RJ: Esophageal
cancer. N Engl J Med. 349:2241–2252. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Tang JC, Chan D, Chung PY, Liu Y, Lam AK,
Law S, Huang W, Chan AS, Lam KH and Zhou Y: Downregulation of
chemokine (C-C motif) ligand 5 induced by a novel
8-hydroxyquinoline derivative (91b1) suppresses tumor invasiveness
in esophageal carcinoma. Int J Mol Med. 54:1112024. View Article : Google Scholar
|
|
11
|
Fernandez-Cuesta L and Thomas RK:
Molecular pathways: Targeting NRG1 fusions in lung cancer. Clin
Cancer Res. 21:1989–1994. 2015. View Article : Google Scholar
|
|
12
|
Meyer D and Birchmeier C: Multiple
essential functions of neuregulin in development. Nature.
378:386–390. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Riaz IB, Naqvi SAA, He H, Asghar N,
Siddiqi R, Liu H, Singh P, Childs DS, Ravi P, Hussain SA, et al:
First-line systemic treatment options for metastatic
castration-sensitive prostate cancer: A living systematic review
and network Meta-analysis. JAMA Oncol. 9:635–645. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Laskin J, Liu SV, Tolba K, Heining C,
Schlenk RF, Cheema P, Cadranel J, Jones MR, Drilon A, Cseh A, et
al: NRG1 fusion-driven tumors: Biology, detection, and the
therapeutic role of afatinib and other ErbB-targeting agents. Ann
Oncol. 31:1693–1703. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Rimer M, Cohen I, Lømo T, Burden SJ and
McMahan UJ: Neuregulins and erbB receptors at neuromuscular
junctions and at agrin-induced postsynaptic-like apparatus in
skeletal muscle. Mol Cell Neurosci. 12:1–15. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Telesco SE, Shih AJ, Jia F and
Radhakrishnan R: A multiscale modeling approach to investigate
molecular mechanisms of pseudokinase activation and drug resistance
in the HER3/ErbB3 receptor tyrosine kinase signaling network. Mol
Biosyst. 7:2066–2080. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Wen D, Peles E, Cupples R, Suggs SV, Bacus
SS, Luo Y, Trail G, Hu S, Silbiger SM, Levy RB, et al: Neu
differentiation factor: A transmembrane glycoprotein containing an
EGF domain and an immunoglobulin homology unit. Cell. 69:559–572.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Alvarado D, Ligon GF, Lillquist JS, Seibel
SB, Wallweber G, Neumeister VM, Rimm DL, McMahon G and LaVallee TM:
ErbB activation signatures as potential biomarkers for anti-ErbB3
treatment in HNSCC. PLoS One. 12:e01813562017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lee C, Kim M, Park C, Jo W, Seo JK, Kim S,
Oh J, Kim CS, Ryu HS, Lee KH and Park J: Epigenetic regulation of
Neuregulin 1 promotes breast cancer progression associated to
hyperglycemia. Nat Commun. 14:4392023. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Yun S, Koh J, Nam SK, Park JO, Lee SM, Lee
K, Lee KS, Ahn SH, Park DJ, Kim HH, et al: Clinical significance of
overexpression of NRG1 and its receptors, HER3 and HER4, in gastric
cancer patients. Gastric Cancer. 21:225–236. 2018. View Article : Google Scholar
|
|
21
|
Adélaïde J, Huang HE, Murati A, Alsop AE,
Orsetti B, Mozziconacci MJ, Popovici C, Ginestier C, Letessier A,
Basset C, et al: A recurrent chromosome translocation breakpoint in
breast and pancreatic cancer cell lines targets the neuregulin/NRG1
gene. Genes Chromosomes Cancer. 37:333–345. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Huang HE, Chin SF, Ginestier C, Bardou VJ,
Adélaïde J, Iyer NG, Garcia MJ, Pole JC, Callagy GM, Hewitt SM, et
al: A recurrent chromosome breakpoint in breast cancer at the
NRG1/neuregulin 1/heregulin gene. Cancer Res. 64:6840–6844. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Prentice LM, Shadeo A, Lestou VS, Miller
MA, deLeeuw RJ, Makretsov N, Turbin D, Brown LA, Macpherson N,
Yorida E, et al: NRG1 gene rearrangements in clinical breast
cancer: Identification of an adjacent novel amplicon associated
with poor prognosis. Oncogene. 24:7281–7289. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Shin DH, Lee D, Hong DW, Hong SH, Hwang
JA, Lee BI, You HJ, Lee GK, Kim IH, Lee YS and Han JY: Oncogenic
function and clinical implications of SLC3A2-NRG1 fusion in
invasive mucinous adenocarcinoma of the lung. Oncotarget.
7:69450–69465. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Drilon A, Somwar R, Mangatt BP, Edgren H,
Desmeules P, Ruusulehto A, Smith RS, Delasos L, Vojnic M,
Plodkowski AJ, et al: Response to ERBB3-directed targeted therapy
in NRG1-rearranged cancers. Cancer Discov. 8:686–695. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Shin DH, Jo JY and Han JY: Dual Targeting
of ERBB2/ERBB3 for the treatment of SLC3A2-NRG1-mediated lung
cancer. Mol Cancer Ther. 17:2024–2033. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hou G, Niu T, Jia A, Zhang Y, Chen X, Wei
H, Jia Y, Xu Y, Li Y, Wang P and Chatterjee A: NRG1 promotes
tumorigenesis and metastasis and afatinib treatment efficiency is
enhanced by NRG1 inhibition in esophageal squamous cell carcinoma.
Biochem Pharmacol. 218:1159202023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kim DW, Schram AM, Hollebecque A, Nishino
K, Macarulla T, Rha SY, Duruisseaux M, Liu SV, Al Hallak MN,
Umemoto K, et al: The phase I/II eNRGy trial: Zenocutuzumab in
patients with cancers harboring NRG1 gene fusions. Future Oncol.
20:1057–1067. 2024. View Article : Google Scholar
|
|
29
|
Yamamoto H, Zhang S and Mizushima N:
Autophagy genes in biology and disease. Nat Rev Genet. 24:382–400.
2023. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Ichimiya T, Yamakawa T, Hirano T, Yokoyama
Y, Hayashi Y, Hirayama D, Wagatsuma K, Itoi T and Nakase H:
Autophagy and autophagy-related diseases: A Review. Int J Mol Sci.
21:89742020. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Liu PF, Farooqi AA, Peng SY, Yu TJ, Dahms
HU, Lee CH, Tang JY, Wang SC, Shu CW and Chang HW: Regulatory
effects of noncoding RNAs on the interplay of oxidative stress and
autophagy in cancer malignancy and therapy. Semin Cancer Biol.
83:269–282. 2022. View Article : Google Scholar
|
|
32
|
Li Z, Zhang Y, Lei J and Wu Y: Autophagy
in oral cancer: Promises and challenges (Review). Int J Mol Med.
54:1162024. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Debnath J, Gammoh N and Ryan KM: Autophagy
and autophagy-related pathways in cancer. Nat Rev Mol Cell Biol.
24:560–575. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Chen HT, Liu H, Mao MJ, Tan Y, Mo XQ, Meng
XJ, Cao MT, Zhong CY, Liu Y, Shan H and Jiang GM: Crosstalk between
autophagy and epithelial-mesenchymal transition and its application
in cancer therapy. Mol Cancer. 18:1012019. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yu TJ, Shiau JP, Tang JY, Yen CH, Hou MF,
Cheng YB, Shu CW and Chang HW: Physapruin a induces reactive oxygen
species to trigger cytoprotective autophagy of breast cancer cells.
Antioxidants (Basel). 11:13522022. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Liu PF, Tsai KL, Hsu CJ, Tsai WL, Cheng
JS, Chang HW, Shiau CW, Goan YG, Tseng HH and Wu CH: Drug
repurposing screening identifies tioconazole as an ATG4 inhibitor
that suppresses autophagy and sensitizes cancer cells to
chemotherapy. Theranostics. 8:830–845. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Liu PF, Chang HW, Cheng JS, Lee HP, Yen
CY, Tsai WL, Cheng JT, Li YJ, Huang WC, Lee CH, et al: Map1lc3b and
sqstm1 modulated autophagy for tumorigenesis and prognosis in
certain subsites of oral squamous cell carcinoma. J Clin Med.
7:4782018. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Serrano-Oviedo L, Ortega-Muelas M,
Garcia-Cano J, Valero ML, Cimas FJ, Pascual-Serra R,
Fernandez-Aroca DM, Roche O, Ruiz-Hidalgo MJ, Belandia B, et al:
Autophagic cell death associated to Sorafenib in renal cell
carcinoma is mediated through Akt inhibition in an ERK1/2
independent fashion. PLoS One. 13:e02008782018. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Huang CY, Lee CH, Tu CC, Wu CH, Huang MT,
Wei PL and Chang YJ: Glucose-regulated protein 94 mediates
progression and metastasis of esophageal squamous cell carcinoma
via mitochondrial function and the NF-kB/COX-2/VEGF axis.
Oncotarget. 9:9425–9441. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Bahar ME, Kim HJ and Kim DR: Targeting the
RAS/RAF/MAPK pathway for cancer therapy: From mechanism to clinical
studies. Signal Transduct Target Ther. 8:4552023. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Kim YC and Guan KL: mTOR: A pharmacologic
target for autophagy regulation. J Clin Invest. 125:25–32. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Pankiv S, Clausen TH, Lamark T, Brech A,
Bruun JA, Outzen H, Øvervatn A, Bjørkøy G and Johansen T:
p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of
ubiquitinated protein aggregates by autophagy. J Biol Chem.
282:24131–24145. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Liu WJ, Ye L, Huang WF, Guo LJ, Xu ZG, Wu
HL, Yang C and Liu HF: p62 links the autophagy pathway and the
ubiqutin-proteasome system upon ubiquitinated protein degradation.
Cell Mol Biol Lett. 21:292016. View Article : Google Scholar
|
|
44
|
Tsai ST, Wang PJ, Liou NJ, Lin PS, Chen CH
and Chang WC: ICAM1 is a potential cancer stem cell marker of
esophageal squamous cell carcinoma. PLoS One. 10:e01428342015.
View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Hickerson RP, Vlassov AV, Wang Q, Leake D,
Ilves H, Gonzalez-Gonzalez E, Contag CH, Johnston BH and Kaspar RL:
Stability study of unmodified siRNA and relevance to clinical use.
Oligonucleotides. 18:345–354. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Cheng JS, Tsai WL, Liu PF, Goan YG, Lin
CW, Tseng HH, Lee CH and Shu CW: The MAP3K7-mTOR axis promotes the
proliferation and malignancy of hepatocellular carcinoma cells.
Front Oncol. 9:4742019. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Liu PF, Chen CF, Ger LP, Tsai WL, Tseng
HH, Lee CH, Yang WH and Shu CW: MAP3K11 facilitates autophagy
activity and is correlated with malignancy of oral squamous cell
carcinoma. J Cell Physiol. 237:4275–4291. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar
|
|
49
|
Liu PF, Hu YC, Kang BH, Tseng YK, Wu PC,
Liang CC, Hou YY, Fu TY, Liou HH, Hsieh IC, et al: Expression
levels of cleaved caspase-3 and caspase-3 in tumorigenesis and
prognosis of oral tongue squamous cell carcinoma. PLoS One.
12:e01806202017. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Liu PF, Chen HC, Cheng JS, Tsai WL, Lee
HP, Wang SC, Peng WH, Lee CH, Ger LP and Shu CW: Association of
ATG4B and phosphorylated ATG4B proteins with tumorigenesis and
prognosis in oral squamous cell carcinoma. Cancers (Basel).
11:18542019. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Rajendran V and Jain MV: In vitro
tumorigenic assay: Colony forming assay for cancer stem cells.
Methods Mol Biol. 1692:89–95. 2018. View Article : Google Scholar
|
|
52
|
Singh SK, Abbas S, Saxena AK, Tiwari S,
Sharma LK and Tiwari M: Critical role of three-dimensional
tumorsphere size on experimental outcome. Biotechniques.
69:333–338. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Xu Z, Han X, Ou D, Liu T, Li Z, Jiang G,
Liu J and Zhang J: Targeting PI3K/AKT/mTOR-mediated autophagy for
tumor therapy. Appl Microbiol Biotechnol. 104:575–587. 2020.
View Article : Google Scholar
|
|
54
|
Huang Y, Zhen Y, Chen Y, Sui S and Zhang
L: Unraveling the interplay between RAS/RAF/MEK/ERK signaling
pathway and autophagy in cancer: From molecular mechanisms to
targeted therapy. Biochem Pharmacol. 217:1158422023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chang KC, Liu PF, Chang CH, Lin YC, Chen
YJ and Shu CW: The interplay of autophagy and oxidative stress in
the pathogenesis and therapy of retinal degenerative diseases. Cell
Biosci. 12:12022. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Jiang TX, Zou JB, Zhu QQ, Liu CH, Wang GF,
Du TT, Luo ZY, Guo F, Zhou LM, Liu JJ, et al: SIP/CacyBP promotes
autophagy by regulating levels of BRUCE/Apollon, which stimulates
LC3-I degradation. Proc Natl Acad Sci USA. 116:13404–13413. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Liu J and Kern JA: Neuregulin-1 activates
the JAK-STAT pathway and regulates lung epithelial cell
proliferation. Am J Respir Cell Mol Biol. 27:306–313. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Meetze K, Vincent S, Tyler S, Mazsa EK,
Delpero AR, Bottega S, McIntosh D, Nicoletti R, Winston WM, Weiler
S, et al: Neuregulin 1 expression is a predictive biomarker for
response to AV-203, an ERBB3 inhibitory antibody, in human tumor
models. Clin Cancer Res. 21:1106–1114. 2015. View Article : Google Scholar
|
|
59
|
Montero JC, Rodríguez-Barrueco R, Ocaña A,
Díaz-Rodríguez E, Esparís-Ogando A and Pandiella A: Neuregulins and
cancer. Clin Cancer Res. 14:3237–3241. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Krivosheya D, Tapia L, Levinson JN, Huang
K, Kang Y, Hines R, Ting AK, Craig AM, Mei L, Bamji SX and
El-Husseini A: ErbB4-neuregulin signaling modulates synapse
development and dendritic arborization through distinct mechanisms.
J Biol Chem. 283:32944–32956. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Hegde GV, de la Cruz CC, Chiu C, Alag N,
Schaefer G, Crocker L, Ross S, Goldenberg D, Merchant M, Tien J, et
al: Blocking NRG1 and other ligand-mediated Her4 signaling enhances
the magnitude and duration of the chemotherapeutic response of
non-small cell lung cancer. Sci Transl Med. 5:171ra182013.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Chua YL, Ito Y, Pole JC, Newman S, Chin
SF, Stein RC, Ellis IO, Caldas C, O'Hare MJ, Murrell A and Edwards
PA: The NRG1 gene is frequently silenced by methylation in breast
cancers and is a strong candidate for the 8p tumour suppressor
gene. Oncogene. 28:4041–452. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
63
|
Pole JC, Courtay-Cahen C, Garcia MJ, Blood
KA, Cooke SL, Alsop AE, Tse DM, Caldas C and Edwards PA:
High-resolution analysis of chromosome rearrangements on 8p in
breast, colon and pancreatic cancer reveals a complex pattern of
loss, gain and translocation. Oncogene. 25:5693–5706. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Wang Y, Ning Z, Zhou X, Yang Z, Tang H, Xu
M, Wang X, Zhao J and Bai Y: Neuregulin1 acts as a suppressor in
human lung adenocarcinoma via AKT and ERK1/2 pathway. J Thorac Dis.
10:3166–3179. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Adashek JJ, Pandya C, Maragakis NJ, De P,
Cohen PR, Kato S and Kurzrock R: Neuregulin-1 and ALS19 (ERBB4): At
the crossroads of amyotrophic lateral sclerosis and cancer. BMC
Med. 22:742024. View Article : Google Scholar : PubMed/NCBI
|
|
66
|
Dhanasekaran SM, Balbin OA, Chen G, Nadal
E, Kalyana-Sundaram S, Pan J, Veeneman B, Cao X, Malik R, Vats P,
et al: Transcriptome meta-analysis of lung cancer reveals recurrent
aberrations in NRG1 and Hippo pathway genes. Nat Commun.
5:58932014. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Duruisseaux M, McLeer-Florin A, Antoine M,
Alavizadeh S, Poulot V, Lacave R, Rabbe N, Cadranel J and Wislez M:
NRG1 fusion in a French cohort of invasive mucinous lung
adenocarcinoma. Cancer Med. 5:3579–3585. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
68
|
Shamir A and Buonanno A: Molecular and
cellular characterization of Neuregulin-1 type IV isoforms. J
Neurochem. 113:1163–1176. 2010. View Article : Google Scholar : PubMed/NCBI
|