Introduction
Osteosarcoma is the most common primary malignant
bone tumor, accounting for approximately 20% of all primary
sarcomas in bone. Although treatment modalities have been improved
over the past decades, it is still a tumor with a high mortality
rate ascribed to tumor cell invasion and metastasis (1). Bone morphogenetic proteins (BMPs), a
group of the most powerful bone inductive factors enriched in the
bone matrix, have been indicated in osteosarcoma and its
metastasis.
BMP belongs to the transforming growth factor-β
super-family. BMP signaling in diseases is the subject of a large
quantity of studies, and has already been shown to play a vital
role in the development and progression of cancer (2,3). The
presence of BMPs in osteosarcoma has been verified and the role of
BMPs in osteosarcoma has also been widely investigated. Studies
demonstrated that seven BMPs (BMP 2-8) and all their receptors were
simultaneously expressed in osteosarcoma cells (4) and osteosarcomas producing BMPs
contained less-differentiated mesenchymal cells, resulting in a
poorer prognosis for those patients (5). BMP 2–8 were all expressed in
osteosarcomas, of which BMP 6–8 were highly expressed and BMP-6
correlated with a chondroid differentiation (6). BMP-2 promoted migration of
osteosarcom cells by enhancing incorporation of integrin-β
(7), and may be an important
factor in the metastasis of osteosarcoma. A recent study indicated
that inhibition of BMPs could be investigated as a possible
strategy for decreasing the incidence of pulmonary metastases in
osteosarcoma (8).
BMP-9 has been shown as a pleiotropic cytokine,
which is implicated in a number of physiologic events. These
include bone morphogenesis, functions of hepatic
reticuloendothelial system, hematopoiesis, neuronal
differentiation, glucose homeostasis, iron homeostasis, and
angiogenesis (9–15). Although the expression of BMP-9 was
initially thought to be restricted to the liver, it has been shown
that BMP-9 binds with high affinity to activin receptor-like kinase
1 (ALK1) and endoglin in endothelial cells, and induces
phosphorylation of Smad-1/5/8 (16). This signaling inhibits basic
fibroblast growth factor-stimulated proliferation and migration of
bovine aortic endothelial cells and VEGF-induced angiogenesis
(17). BMP-9 also inhibits the
migration and growth of human dermal microvascular endothelial
cells (15) and the DNA synthesis
of human aortic endothelial cells via ALK-1 (18). BMP-9 has been reported to induce
apoptosis in prostate cancer cells and prevent the migration and
invasiveness of prostate cancer (19). However, the roles played by this
cytokine in osteosarcoma have not been explored. In the present
study, the endogenous expression of BMP-9 was examined in
osteosarcoma cell lines. The biological function of this molecule
was investigated in cells that overexpress this molecule in order
to establish the functional role of BMP-9 in osteosarcoma cells.
Finally, the underlying signaling pathway involved in the
progression of osteosarcoma was explored.
Materials and methods
Materials
MG-63, U-2 and HOS osteosarcoma cell lines used in
the experiment were from Institute of Biochemistry and Cell Biology
(Shanghai, China). Fifteen 6-week-old female immune-deficient nude
mice (BALB/c-nu) were purchased from Shanghai SLAC Laboratory
Animal Co. Ltd (Shanghai, China). AdBMP-9 adenovirus vector,
negative control vector ad-GFP and virion-packaging elements were
from Genechem (Shanghai, China). The primers of BMP-9, PI3Kp85α and
p-AKT, GAPDH, were synthesized by ABI Co. Ltd (USA). All antibodies
were from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Drugs and reagents
3-(4,5)-Dimethylthiahiazo(-z-yl)-3,5-diphenyltetrazolium bromide
(MTT) was from Dingguo biology (Shanghai, China); Dulbecco’s
modified Eagle’s medium (DMEM) and fetal bovine serum (FBS) were
from Thermo Fisher Scientific Inc. (Waltham, MA, USA); TRIzol
Reagent and Lipofectamine 2000 were from Invitrogen (Carlsbad, CA,
USA); M-MLV Reverse Transcriptase was from Promega (Madison, WI,
USA); SYBR-Green Master mixture was from Takara (Otsu, Japan);
RNase A and double stain apoptosis detection kit (Hoechst 33342/PI)
were from Roche Diagnostics (Mannheim, Germany) and Annexin V-FITC
apoptosis detection kit I from BD Pharmingen (San Diego, CA, USA).
ECL-PLUS/Kit was from GE Healthcare (Piscataway, NJ, USA).
Cell culture and adenovirus
transfection
MG-63 and HOS osteosarcoma cell lines were cultured
in DMEM medium supplemented with 10% heat-inactivated FBS, 100 U/ml
of penicillin and 100 μg/ml of streptomycin. They were all placed
in a humidified atmosphere containing 5% CO2 at 37°C.
Recombinant adenovirus vector adBMP-9 and negative control ad-GFP
were transfected into MG-63 and HOS osteosarcoma cell lines. Cells
were subcultured at a 1:5 dilution in 300 μg/ml G418-containing
medium. Positive stable transfectants were selected and expanded
for further study. The clone in which the adBMP-9 adenovirus
vectors were transfected was named as adBMP-9 group, the negative
control vectors as the ad-GFP group and MG-63 or HOS osteosarcoma
cells without gene transfection as the CON group.
Real-time PCR
To quantitatively determine the mRNA expression
levels of BMP-9, PI3Kp85α and p-AKT in osteosarcoma cell lines,
real-time PCR was used. Total-RNA of each clone was extracted with
TRIzol according to the manufacturer’s protocol.
Reverse-transcription was carried out using M-MLV and cDNA
amplification was carried out using SYBR-Green Master mix kit
according to the manufacturer’s protocol. The genes were amplified
using specific oligonucleotide primer and human
glyceraldehyde-3-phosphate dehydrogenase (GAPDH) gene was used as
an endogenous control. The PCR primer sequences were as follows:
BMP-9, 5′-TTCCTTCAGAGC AAACAGCA-3′ and 5′-GTTGTGCTCAAATCCCCATT-3′;
PI3Kp85α, 5′-GAAAGGAGGAAAUAACAAAdtdt-3′ and
5′-UUUGUUAUUUCCUCCUUUCdtdt-3′; p-AKT, 5′-GGAG
AUCAUGCAGCAUCGCdtdt-3′ and 5′-GCGAUGCUGCA UGAUCUCCdtdt-3′; GAPDH,
5′-CAACGAATTTGGCTAC AGCA-3′ and 5′-AGGGGTCTACATGGCAACTG-3′. Data
were analyzed using the comparative Ct method (2−ΔΔCt).
Three separate experiments were performed for each clone.
Western blot assay
Osteosarcoma cells were harvested and extracted
using lysis buffer (Tris-HCl, SDS, mercaptoethanol, glycerol). Cell
extracts were boiled for 5 min in loading buffer and then equal
amount of cell extracts were separated on 15% SDS-PAGE gels.
Separated protein bands were transferred into polyvinylidene
fluoride (PVDF) membranes and the membranes were blocked in 5% skim
milk powder. The primary antibodies against BMP-9, PI3Kp85α, p-AKT,
Ki-67 and MMP-9 were diluted according to the instructions of
antibodies and incubated overnight at 4°C. Then, horseradish
peroxidase-linked secondary antibodies were added at a dilution
ratio of 1:1,000 and incubated at room temperature for 2 h. The
membranes were washed with PBS three times and the immunoreactive
bands were visualized using ECL-PLUS/Kit according to the kit
instructions. The relative protein level in different cell lines
was normalized to GAPDH concentration. Three separate experiments
were performed for each clone.
Cell proliferation assay
Cell proliferation was analyzed with the MTT assay.
Briefly, cells infected with AdBMP-9 were incubated in 96-well
plates at a density of 1×105 cells per well with DEME
medium supplemented with 10% FBS. Cells were treated with 20 μl MTT
dye at 0, 24, 48, 72 h and then incubated with 150 μl of DMSO for 5
min. The color reaction was measured at 570 nm with enzyme
immunoassay analyzer (Bio-Rad, Hercules, CA, USA). The
proliferative activities were calculated for each clone.
Wound-healing assay
MG-63 and HOS osteosarcoma cells were plated in each
well of a 6-well culture plate and allowed to grow to 90%
confluence. Treatment with adBMP-9 was then performed. The next
day, a wound was created using a micropipette tip. The migration of
cells towards the wound was monitored daily, and images were
captured at time intervals of 24 h.
Hoechst 33342/PI assay
This assay was performed according to a combination
of methods with modifications as detailed: 50 μl/well of a 200
μg/ml PI stock and 2.5 μl of a 100 μg/ml Hoechst 33342 was added to
a microtitre plate volume of 100 μl and incubated in the dark for
60 and 15 min, respectively. A 100 μl of methanol:acetic acid (3:1)
fixative was then added directly to each well. Cells were viewed
under a UV microscope with DAPI filter. Six replicate wells were
analyzed for each treatment by quantitative and qualitative
examination of four random fields in each well.
Detection of apoptosis incidence by flow
cytometry
Apoptosis incidence was detected by the Annexin
V-FITC apoptosis detection kit I (BD Pharmingen). Briefly, cells
that were attached to the plate as well as those present in the
super-natant were collected together and re-suspended in binding
buffer at a concentration of 1×106 cells/ml. A 100 μl
sample of solution containing 1×105 cells was incubated
with 5 μl of Annexin V-FITC and 5 μl of propidium iodide for 15 min
at room temperature in the dark, followed by addition of 400 μl of
binding buffer. Samples were analyzed by a fluorescence-activated
cell sorter (Beckman Coulter, Miami, FL, USA) within 1 h. Apoptotic
cells, including those staining positive for Annexin V-FITC and
negative for propidium iodide and those that were double positive,
were counted and reported as a percentage of the total cell
count.
In vivo tumor xenograft studies
The animal experiments and protocols were reviewed
and approved by the Institutional Animal Care and Use Committee of
the Shanghai Jiao Tong University School of Medicine. Six mice were
injected subcutaneously with 1×108 MG-63 and HOS
osteosarcoma cells in 50 μl of PBS pre-mixed with an equal volume
of Matrigel matrix (Becton-Dickinson). Mice were monitored daily
and all mice developed a subcutaneous tumor. When the tumor size
reached approximately 5 mm in length, they were surgically removed,
cut into 1–2 mm3 pieces, and re-seeded individually into
other twelve mice. When tumor size reached approximately 5 mm in
length, the mice were randomly assigned to ad-BMP-9 and ad-GFP
groups, in which 15 μl of adenovirus was injected into the
subcutaneous tumors using a multi-site injection format. Injections
were repeated on the third day after initial treatment. The tumor
volume every three days was measured with a caliper, using the
formula volume = (length × width)2/2.
Statistical analysis
The results obtained were expressed as the mean ± SE
from at least three independent experiments. One-way analysis of
variance (ANOVA) was used to analyze the differences between
groups. The LSD method of multiple comparisons was used when the
probability for ANOVA was statistically significant. Statistical
significance was set at P<0.05.
Results
The endogenous expression of BMP-9 in
osteosarcoma cell lines
The endogenous expression of BMP-9 in human MG-63,
U-2 and HOS osteosarcoma cell lines was evaluated using RT-PCR and
western blot analysis. As shown in Fig. 1, there were different levels of
mRNA and protein expression of BMP-9 in MG-63, U-2 and HOS
osteosarcoma cell lines, but the expression levels of BMP-9 were
significantly higher in U-2 cell line than in MG-63 and HOS cell
lines. Thus, we chose MG-63 and HOS osteosarcoma cell lines for
infection by the adBMP-9 adenovirus.
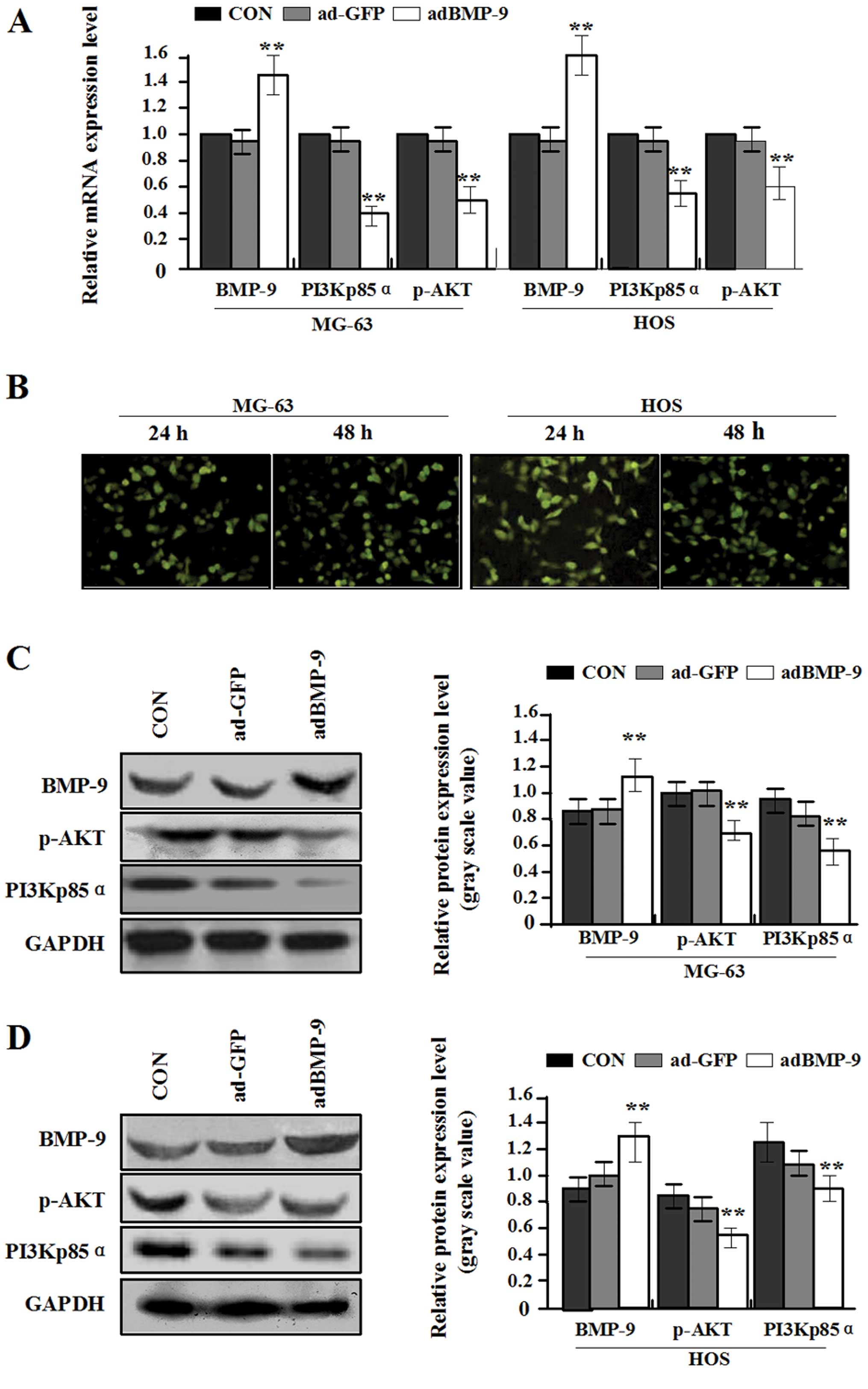
The expression of BMP-9, PI3Kp85α and
p-AKT after infection with adBMP-9
In order to enhance the exogenous expression of
BMP-9 in MG-63 and HOS osteosarcoma cells with relatively low
endogenous BMP-9 expression, constructed adenovirus vector adBMP-9
was used for infecting the MG-63 and HOS cell lines. In pilot
studies, the infection efficiency of adBMP-9 (MOI=100) in MG-63 and
HOS cell lines was greater than 80% under fluorescence microscopy
(Fig. 2B). Real-time PCR and
western blot assays were performed at 48 h recovery to measure the
exogenous expression of BMP-9, PI3Kp85α and p-AKT. An obvious
increase of BMP-9 expression was observed in adBMP-9 group compared
with the ad-GFP group and CON group (**P<0.01), while
a marked decrease of PI3Kp85α and p-AKT expression was found in
adBMP-9 group compared with the ad-GFP group and CON group in MG-63
and HOS cell lines (**P<0.01) (Fig. 2A, C and D), but no difference was
found between ad-GFP group and CON group in MG-63 and HOS cell
lines (P>0.05). These data indicated that overexpression of
BMP-9 might block the PI3K/AKT signaling pathway in osteosarcoma
cells.
Inhibition of osteosarcoma cell
proliferation by adBMP-9
Deregulated cell proliferation is a hallmark of
cancer (20). In order to detect
the effect of adBMP-9 on osteosarcoma cell proliferation, we
investigated the proliferative activities of MG-63 and HOS cell
lines by MTT. As a result, it was indicated that adBMP-9
significantly reduced the proliferative activities of MG-63 and HOS
cell lines in a time-dependent manner compared with the ad-GFP
group and CON group. No difference was found between ad-GFP group
and CON group in MG-63 and HOS cell lines (Fig. 3A and B). In addition, Ki-67 is at
the very heart of many essential cellular processes and determines
the tumor progression and the outcome of anticancer treatment. To
determine whether BMP-9 overexpression suppressed the endogenous
Ki-67 through translational repression, the expression of Ki-67 was
examined by western blot assay, indicating that the amount of Ki-67
expression was significantly decreased in adBMP-9 group compared
with the ad-GFP group and CON group in MG-63 and HOS cell lines
(Fig. 3C and D). No difference was
found between ad-GFP group and CON group in MG-63 and HOS cell
lines. The results suggested that over-expression of BMP-9 might
inhibit osteosarcoma cell proliferation via downregulation of Ki-67
expression.
Inhibition of osteosarcoma cell migration
by adBMP-9
To determine the effect of adBMP-9 on MG-63 and HOS
cell migration, wound-healing assay was performed which showed that
the migration capacity of the osteosarcoma cells in adBMP-9 group
were markedly lower than those in ad-GFP group and CON group, but
no significant changes were found between ad-GFP group and CON
group (Fig. 4A). Furthermore,
western blot assay was used to examine the effect of adBMP-9 on
MMP-9 expression. As shown in Fig. 4B
and C, the expression of MMP-9 protein was significantly
inhibited in adBMP-9 group compared with the ad-GFP group and CON
group in MG-63 and HOS cell lines. No difference was found between
ad-GFP group and CON group. These data suggested that
overexpression of BMP-9 might inhibit cell migration via
down-regulation of MMP-9 expression.
Induction of osteosarcoma cell apoptosis
by adBMP-9
It has been shown that the action mechanism of many
anticancer factors is based on their ability to induce apoptosis.
Therefore, it was desirable that MG-63 and HOS osteosarcoma cells
treated with adBMP-9 underwent apoptosis as their mode of cell
death. At the end of the incubation period for 24, 48 and 72 h, the
osteosarcoma cells were stained with Hoechst 33342/PI double
staining and their cell nucleus was observed under the microscope
for apoptosis. As shown in Fig.
5A, the number of MG-63 and HOS apoptotic cells (weak red and
strong blue staining) and necrotic cells (strong red and strong
blue staining) in adBMP-9 group significantly increased compared
with that in the ad-GFP group and CON group (weak red and weak blue
staining) in a time-dependent manner. As shown in Fig. 5B, the apoptotic indexes of MG-63
and HOS cells in adBMP-9 group were also significantly higher than
those in ad-GFP group and CON group (*P<0.05,
**P<0.01). However, no difference was found between
ad-GFP group and CON group (P>0.05). The apoptotic incidence was
also observed by flow cytometry at 24 h after the transfection
(Fig. 6A and B) and the result
showed that the incidence of apoptotic cells significantly
increased in adBMP-9 group compared with that in the ad-GFP group
and CON group (**P<0.01), but no difference was found
between ad-GFP group and CON group (P>0.05). These data
suggested that over-expression of BMP-9 could induce osteosarcoma
cell apoptosis.
Antitumor effect of adBMP-9 in the
osteosarcoma xenograft model
Our in vitro experiments confirmed that
adBMP-9 efficiently inhibited the growth and migration of MG-63 and
HOS cells. Whether BMP-9 has the same inhibitory effect on in
vivo osteosarcoma was not known. Therefore, we further
investigated the antitumor effect of adBMP-9 in vivo using
MG-63 and HOS xenograft models. The mean volumes of MG-63 and HOS
xenograft tumors were 70.21±15.30 mm3 and 75.45±18.29
mm3 in the experimental mice before treatment. At the
14th day, the average volumes of MG-63 and HOS xenograft tumors
were measured and found significantly smaller in adBMP-9 group than
those in the ad-GFP group (Fig. 7A and
B). During the whole tumor growth period, the tumor growth
activity was measured. The tumors treated with adBMP-9 grew
substantially slower than the ad-GFP group (Fig. 7C and D). When the tumors were
harvested, the average weights of MG-63 and HOS xenograft tumors in
adBMP-9 group were significantly lighter than those in the ad-GFP
group (**P<0.01) (Fig.
7E and F). These results in vivo indicated that
overexpression of BMP-9 could inhibit MG-63 and HOS cell
growth.
Discussion
BMPs are multifunctional cytokines that regulate the
growth, differentiation and apoptosis of various cell types
(21). BMP has also been proved to
be one of the significant factors in the prognosis of bone tumors
(22), and detection of BMPs in
osteosarcoma has been reported in several studies (23–25).
The BMP-2/4, BMP-3, BMP-5, BMP-6, BMP-7 and BMP-8
were previously found highly expressed in osteosarcoma tissues
(6), but BMP-9 expression in
osteosarcoma has not been reported. Our study documented for the
first time the endogenous expression of BMP-9 in MG-63, U-2 and HOS
osteosarcoma cells, and demonstrated that the expression level of
BMP-9 was significantly higher in U-2 cell line than that in MG-63
and HOS cell lines, which were chosen for infection by the adBMP-9
adenovirus. In pilot studies, the infectious efficiency of adBMP-9
in MG-63 and HOS cell lines was very high, and an obvious increase
of BMP-9 expression was observed in adBMP-9 group compared with the
ad-GFP group and CON group.
Studies have shown that BMP-9 is also produced by
hepatocytes and circulates mainly in an active mature form
complexed to its prodomain (26),
and induces proliferation of multiple types of endothelial cells
in vitro and in vivo (27). In contrast, we found in our study
that the overexpression of BMP-9 significantly reduced the
proliferative activities of MG-63 and HOS cell lines in a
time-dependent manner. More importantly, BMP-9 has been confirmed
to prevent the migration and invasiveness and induce apoptosis in
prostate cancer, which suggests that BMP-9 may be considered as a
tumor suppressor and apoptosis regulator in prostate cancer
(19). Additionally, soluble
endoglin can specifically binds BMP-9 via its orphan domain,
inhibits blood vessel formation and suppresses tumor growth
(28). By contrast, BMP-9 was also
reported to promote ovarian cancer cell proliferation through
ALK-2/Smad1/Smad4 pathway (29).
We think that the response to BMP-9 is not accordant among all
cancers. The biological response of cancer cells to BMP-9 may
depend not only on the particular cell type or concentration of BMP
used, but also on the presence of other factors that are not yet
defined. Our gain-of-function studies in vitro and in
vivo using adBMP-9 revealed significant decrease in growth and
migration and increase in apoptosis in MG-63 and HOS cells,
suggesting that BMP-9 may function as a tumor suppressor in
osteosarcoma.
PI3K/AKT is a major pathway for malignant
progression in various tumors. It is involved in mediating survival
signals that rescue Ewing tumor from fibroblast growth factor
2-induced cell death (30).
Blockade of Ras/PI3K/AKT pathways by statins reduces the expression
of TGF-β as angiogenic factors in mouse osteosarcoma (31). While grifolin or celecoxib, a
cyclooxygease-2 inhibitor, induces apoptosis via inhibition of
PI3K/AKT signaling pathway in human osteosarcoma cells (32,33),
overexpression of BMI-1 promotes cell growth and resistance to
cisplatin treatment via activation of PI3K/AKT pathway in
osteosarcoma (34). However, our
study indicated a marked decrease of PI3Kp85α and p-AKT expression
in adBMP-9 group compared with the ad-GFP group and CON group in
MG-63 and HOS cell lines, suggesting that overexpression of BMP-9
inhibited osteosarcoma cell growth and migration via blockade of
the PI3K/AKT signaling.
Ki-67 is a nuclear protein that is expressed in
proliferating cells and may be required for maintaining cell
proliferation, used as a marker for cell proliferation of
osteosarcoma (35). MMP-9 is
thought to be key enzymes involved in the degradation of type IV
collagen and high level of MMP-9 in tissues is interralated with
tumor growth and invasion (36,37).
It is reported that PI3K/AKT pathway promotes the growth and
metastasis of malignant tumors via upregulation of Ki-67 and MMP-9
(38). Our results showed a marked
decrease of Ki-67 and MMP-9 expression in adBMP-9 group compared
with the ad-GFP group and CON group in MG-63 and HOS cell lines,
suggesting that BMP-9 might downregulate the expression of Ki-67
and MMP-9 via blockade of the PI3K/AKT signaling pathway in
osteosarcoma cells.
To our knowledge, this is the first report
investigating the role of BMP-9 in the growth and migration of
osteosarcoma cells. Our investigation revealed that the enhanced
expression of BMP-9 in osteosarcoma cells by adBMP-9 exerted
inhibitory effects on growth and migration of osteosarcoma cells
possibly via blockade of the PI3K/AKT signaling. BMP-9 as a tumor
suppressor may provide a novel approach to human osteosarcoma
treatment.
References
|
1.
|
Jemal A, Bray F, Center MM, Ferlay J, Ward
E and Forman D: Global cancer statistics. CA Cancer J Clin.
61:69–90. 2011. View Article : Google Scholar
|
|
2.
|
Kim M and Choe S: BMPs and their clinical
potentials. BMB Rep. 44:619–634. 2011. View Article : Google Scholar
|
|
3.
|
Boon MR, van der Horst G, van der Pluijm
G, et al: Bone morphogenetic protein 7: a broad-spectrum growth
factor with multiple target therapeutic potency. Cytokine Growth
Factor Rev. 22:221–229. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
4.
|
Gobbi G, Sangiorgi L, Lenzi L, et al:
Seven BMPs and all their receptors are simultaneously expressed in
osteosarcoma cells. Int J Oncol. 20:143–147. 2002.PubMed/NCBI
|
|
5.
|
Yoshikawa H, Nakase T, Myoui A and Ueda T:
Bone morphogenetic proteins in bone tumors. J Orthop Sci.
9:334–340. 2004. View Article : Google Scholar
|
|
6.
|
Sulzbacher I, Birner P, Trieb K,
Pichlbauer E and Lang S: The expression of bone morphogenetic
proteins in osteosarcoma and its relevance as a prognostic
parameter. J Clin Pathol. 55:381–385. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7.
|
Sotobori T, Ueda T, Myoui A, et al: Bone
morphogenetic protein-2 promotes the haptotactic migration of
murine osteoblastic and osteosarcoma cells by enhancing
incorporation of integrin beta1 into lipid rafts. Exp Cell Res.
312:3927–3938. 2006. View Article : Google Scholar
|
|
8.
|
Weiss KR, Cooper GM, Jadlowiec JA, McGough
RL III and Huard J: VEGF and BMP expression in mouse osteosarcoma
cells. Clin Orthop Relat Res. 450:111–117. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
9.
|
Song JJ, Celeste AJ, Kong FM, Jirtle RL,
Rosen V and Thies RS: Bone morphogenetic protein-9 binds to liver
cells and stimulates proliferation. Endocrinology. 136:4293–4297.
1995.PubMed/NCBI
|
|
10.
|
Ploemacher RE, Engels LJ, Mayer AE, Thies
S and Neben S: Bone morphogenetic protein 9 is a potent synergistic
factor for murine hemopoietic progenitor cell generation and colony
formation in serum-free cultures. Leukemia. 13:428–437. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
11.
|
Helm GA, Alden TD, Beres EJ, et al: Use of
bone morphogenetic protein-9 gene therapy to induce spinal
arthrodesis in the rodent. J Neurosurg. 92:191–196. 2000.PubMed/NCBI
|
|
12.
|
López-Coviella I, Berse B, Krauss R, Thies
RS and Blusztajn JK: Induction and maintenance of the neuronal
cholinergic phenotype in the central nervous system by BMP-9.
Science. 289:313–316. 2000.
|
|
13.
|
Chen C, Grzegorzewski KJ, Barash S, et al:
An integrated functional genomics screening program reveals a role
for BMP-9 in glucose homeostasis. Nat Biotechnol. 21:294–301. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
14.
|
Truksa J, Peng H, Lee P and Beutler E:
Bone morphogenetic proteins 2, 4, and 9 stimulate murine hepcidin 1
expression independently of Hfe, transferrin receptor 2 (Tfr2), and
IL-6. Proc Natl Acad Sci USA. 103:10289–10293. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15.
|
David L, Mallet C, Mazerbourg S, Feige JJ
and Bailly S: Identification of BMP9 and BMP10 as functional
activators of the orphan activin receptor-like kinase 1 (ALK1) in
endothelial cells. Blood. 109:1953–1961. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16.
|
Brown MA, Zhao Q, Baker KA, et al: Crystal
structure of BMP-9 and functional interactions with pro-region and
receptors. J Biol Chem. 280:25111–25118. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17.
|
Scharpfenecker M, van Dinther M, Liu Z, et
al: BMP-9 signals via ALK1 and inhibits hFGF-induced endothelial
cell proliferation and VEGF-stimulated angiogenesis. J Cell Sci.
120:964–972. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18.
|
Upton PD, Davies RJ, Trembath RC and
Morrell NW: Bone morphogenetic protein (BMP) and activin type II
receptors balance BMP9 signals mediated by activin receptor-like
kinase-1 in human pulmonary artery endothelial cells. J Biol Chem.
284:15794–15804. 2009. View Article : Google Scholar
|
|
19.
|
Ye L, Kynaston H and Jiang WG: Bone
morphogenetic protein-9 induces apoptosis in prostate cancer cells,
the role of prostate apoptosis response-4. Mol Cancer Res.
6:1594–1606. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
20.
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: the next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
21.
|
Yang LJ and Jin Y: Immunohistochemical
observation of bone morphogenetic protein in normal and abnormal
conditions. Clin Orthop Relat Res. 257:249–256. 1990.PubMed/NCBI
|
|
22.
|
Laiitnen M, Jortikka L, Halttunen T, et
al: Measurement of total and local bone morphogenetic protein
concentration in bone tumours. Int Orthop. 21:188–193. 1997.
View Article : Google Scholar : PubMed/NCBI
|
|
23.
|
Yoshikawa H, Shimizu K, Nakase T and
Takaoka K: Periosteal sunburst speculation in osteosarcoma. A
possible role for bone morphogenetic protein. Clin Orthop Relat
Res. 308:213–219. 1994.PubMed/NCBI
|
|
24.
|
Yoshikawa H, Retting WJ, Lane JM, et al:
Immunohistochemical detection of bone morphogenetic protein in bone
and soft-tissue sarcomas. Cancer. 74:842–847. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
25.
|
Yoshikawa H, Retting WJ, Takaoka K, et al:
Expression of bone morphogenetic proteins in human osteosarcoma.
Immunohistochemical detection with monoclonal antibody. Cancer.
73:85–91. 1994. View Article : Google Scholar
|
|
26.
|
Bidart M, Ricard N, Levet S, et al: BMP9
is produced by hepatocytes and circulates mainly in an active
mature form complexed to its prodomain. Cell Mol Life Sci.
69:313–324. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
27.
|
Suzuki Y, Ohga N, Morishita Y, et al:
BMP-9 induces proliferation of multiple types of endothelial cells
in vitro and in vivo. J Cell Sci. 123:1684–1692. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
28.
|
Castonguay R, Werner ED, Matthews RG, et
al: Soluble endoglin specifically binds bone morphogenetic proteins
9 and 10 via its orphan domain, inhibits blood vessel formation,
and suppresses tumor growth. J Biol Chem. 286:30034–30046. 2011.
View Article : Google Scholar
|
|
29.
|
Herrera B, van Dinther M, Ten Dijke P and
Inman GJ: Autocrine bone morphogenetic protein-9 signals through
activin receptor-like kinase-2/Smad1/Smad4 to promote ovarian
cancer cell proliferation. Cancer Res. 69:9254–9262. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
30.
|
Hotfilder M, Sondermann P, Senss A, et al:
PI3K/AKT is involved in mediating survival signals that rescue
Ewing tumor cells from fibroblast growth factor 2-induced cell
death. Br J Cancer. 92:705–710. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
31.
|
Tsubaki M, Yamazoe Y, Yanae M, et al:
Blockade of the Ras/MEK/ERK and Ras/PI3K/Akt pathways by statins
reduces the expression of bFGF, HGF, and TGF-β as angiogenic
factors in mouse osteosarcoma. Cytokine. 54:100–107.
2011.PubMed/NCBI
|
|
32.
|
Jin S, Pang RP, Shen JN, et al: Grifolin
induces apoptosis via inhibition of PI3K/AKT signaling pathway in
human osteosarcoma cells. Apoptosis. 12:1317–1326. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
33.
|
Liu B, Shi ZL, Feng J and Tao HM:
Celecoxib, a cyclooxygenase-2 inhibitor, induces apoptosis in human
osteosarcoma cell line MG-63 via down-regulation of PI3K/Akt. Cell
Biol Int. 32:494–501. 2008. View Article : Google Scholar
|
|
34.
|
Wu Z, Min L, Chen D, et al: Overexpression
of BMI-1 promotes cell growth and resistance to cisplatin treatment
in osteosarcoma. PLoS One. 6:e146482011. View Article : Google Scholar : PubMed/NCBI
|
|
35.
|
Hernández-Rodríguez NA, Correa E, Sotelo
R, et al: Ki-67: a proliferative marker that may predict pulmonary
metastases and mortality of primary osteosarcoma. Cancer Detect
Prev. 25:210–215. 2001.PubMed/NCBI
|
|
36.
|
Loukopoulos P, O’Brien T, Ghoddusi M,
Mungall BA and Robinson WF: Characterization of three novel canine
osteosarcoma cell lines producing high levels of matrix
metal-loproteinases. Res Vet Sci. 77:131–141. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
37.
|
Cawston TE and Wilson AJ: Understanding
the role of tissue degrading enzymes and their inhibitors in
development and disease. Best Pract Res Clin Rheumatol.
20:983–1002. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
38.
|
Fu Y, Zhang Q, Kang C, et al: Inhibitory
effects of adenovirus mediated COX-2, Akt1 and PIK3R1 shRNA on the
growth of malignant tumor cells in vitro and in vivo.
Int J Oncol. 35:583–591. 2009.
|