Introduction
Primary central nervous system lymphoma (PCNSL) is
an aggressive form, confined to the CNS, including the brain,
leptomeninges, spinal cord and intraocular structure (1,2).
Currently, it is individualized in the new World Health
Organization classification of non-Hodgkin's lymphoma (3). Almost 95% of PCNSL are diffuse large
B-cell lymphoma (DLBCL). Although PCNSL shares certain common
characteristics with system DLBCL in morphology, it has its own
unique features. Its incidence has significantly increased among
immunocompetent patients over the last few decades. However, due to
the development of highly active antiretroviral therapies, the
incidence appears to be decreasing in immunocompromised patients
(4).
PCNSLs are sensitive to chemotherapy and
radiotherapy, and they are potentially curable tumors. However,
until recently, the overall outcome and prognosis for PCNSL
patients remains poor, particularly when compared to that of
systemic lymphoma. The level of scientific evidence supporting the
therapeutic choices for this disease is extremely low and the
different opinions on numerous therapeutic aspects results in no
consensus regarding the overall strategy (5,6). A
previous study has shown that the methotrexate (MTX)-based regimens
were more effective compared to high-dose methotrexare (HD-MTX)
alone, but the toxicity is serious (7).
In the present study, the characteristics of 18
PCNSL patients were retrospectively analyzed and the efficacy and
safety of the rituximab, MTX, cytarabine (Ara-C) and dexamethasone
(R-MAD) regimen was examined as first-line treatment in clinical
practice.
Patients and methods
Clinical characteristics
Between January 2010 and March 2014, 18 patients
were newly diagnosed with PCNSL at the Beijing Tiantan Hospital
(Beijing, China) and met the inclusion criteria. All the PCNSL
patients were immunocompetent patients and they were proven to be
DLBCL by pathological examination. The Beijing Tiantan Hospital
Ethics Committee approved the study protocol and all the patients
provided written informed consent.
Among these patients, there were 11 men and 7 women
whose median age was 51 years old (range, 34–83 years, 8 were
<60 and 10 were >60 years old). For the performance status of
the patients according to Eastern Cooperative Oncology Group, there
were 3 in grade 0–1 and 15 in grade 2–4 (Table I).
 | Table I.Clinical characteristics of all the
patients. |
Table I.
Clinical characteristics of all the
patients.
| Characteristics | Patients (n=18) |
|---|
| Age, median years
(range) | 51 (34–83) |
| Gender, no. (%) |
|
| Male | 11 (61) |
|
Female | 7 (39) |
| ECOG, no. (%) |
|
| 0–1 | 3 (17) |
| 2–4 | 15 (83) |
| IELSG, no. (%) |
|
| 0–1 | 5 (28) |
| 2–3 | 11 (61) |
| 4–5 | 2 (11) |
| LDH, no. (%) |
|
|
Normal | 13 (72) |
|
Elevated | 5 (28) |
| CSF cytology, no.
(%) |
|
|
Positive | 4 (22) |
|
Negative | 14 (78) |
| CSF protein, no.
(%) |
|
|
Normal | 12 (67) |
|
Elevated | 6 (33) |
| Pathology, no.
(%) |
|
|
DLBCL | 18 (100) |
Neuroimaging
On computed tomography (CT) the lesions of PCNSL
were hypodense, hyperdense or mixed dense. In general, the shape of
the tumor was irregular and the edge was blurred. The lesions
should be considered in the differential diagnosis of subacute
cerebral infarction and metastases.
On magnetic resonance imaging (MRI), T1-weighted
imaging (WI) lesions were usually isointense or hypointense, and
T2WI lesions were isointense or hyperintense. The tumor was single
in 6 (33.3%) patients and multiple in 12 (66.7%) patients. There
were 35 lesions in total. There were 6 (17.1%) lesions in the
supratentorial, 28 (80.0%) in the subtentorial and 1 (2.9%) in the
lumbar spine. Ten (55.6%) PCNSL cases were involved in deep regions
of the brain. Enhancing lesions were observed in all the
immunocompetent patients with PCNSL (100%). There were 5 (14.3%)
lesions with enhancement near the meninges (Table II).
 | Table II.Examination of MRI. |
Table II.
Examination of MRI.
| MRI | No. (%) |
|---|
| Lesions, n |
|
|
Single | 6 (33.3) |
|
Multiple | 12 (66.7) |
| Strengthen the
scan | 18 (100.0) |
| Deep brain
lesions | 10 (55.6) |
| Lesions location |
|
|
Supratentorial | 6 (17.1) |
|
Subtentorial | 28 (80.0) |
| Lumbar spine | 1 (2.9) |
| Midline shift | 4 (22.2) |
| Enhancement near the
meninges | 5 (14.3) |
Among 18 patients, 6 were examined by
18F-fludeoxyglucose (FDG)-positron emission
tomography-CT. 18F-FDG uptake values measured by maximum
standardized uptake value (SUV) were 7.2–31.9 in 6 PCNSL patients
and this value was ~2.5 times higher than the average SUV in the
normal gray matter.
Clinical presentation
The patients' clinical presentation is relevant to
the tumor location, including cognitive dysfunction, psychomotor
slowing, personality changes and disorientation; and raised
intracranial pressure, headache and focal symptoms. Early
presentation usually presents a combination of generalized symptoms
such as headache, confusion, lethargy and vomiting. Visual symptoms
are typically blurred vision or floaters.
Treatment
The R-MAD as first-line treatment for 18 patients
was 375 mg/m2 rituximab administered on day 0; 3.5
g/m2 MTX administered on day 1, intravenous infusion in
3 h; 1 g/m2 Ara-C administered on day 2 and 10 mg
dexamethasone administered on day 1–3, every 3 weeks. To decrease
the side effects of MTX, calcium folinate salvage was started after
24 h of MTX administration every 6 h for a total of 8 times. When
the cerebrospinal fluid (CSF) was involved, intrathecal injection
with either 10 mg MTX plus 10 mg dexamethasone, or 50 mg Ara-C plus
10 mg dexamethasone occurred once a week until CSF negative.
Evaluation
According to the curative effect evaluation standard
of the International Primary CNS Lymphoma Collaborative Group,
patients were assessed after 6 cycles. Complete response (CR),
partial response (PR) or progressive disease (PD) were defined by
MRI. CR was complete disappearance of all the measurable disease
and no new lesions. PR was defined as ≥50% reduction of all the
measurable lesions. PD was a 25% increase of measurable lesions or
appearance of new lesions. Toxicity was assessed according to the
National Cancer Institute-Common Toxicity Criteria (8).
Statistical analysis
Overall survival (OS) was calculated from the start
of diagnosis to the time of mortality from any cause or the last
follow-up. Progression-free survival (PFS) was calculated from the
start of diagnosis to the time of first disease progression or
relapse, or fatality resulting from any cause. Time to PCNSL,
survival following PCNSL, PSF and OS were estimated according to
Kaplan-Meier method. SPSS 18.0 software (SPSS, Inc., Chicago, IL,
USA) was used to conduct statistical analysis. P<0.05 was
considered to indicate a statistically significant difference.
Results
Evaluation of patients following
immunochemotherapy
Following 6 cycles of the immunochemotherapy,
including R-MAD, 10 (55.6%) PCNSL patients achieved CR, 7 (38.9%)
achieved PR and 1 (5.5%) achieved PD. The OS rate was 94.5% and PFS
rate was 94.5% (Fig. 1). The median
survival was 22 months (95% confidence interval, 19.4–24.6). None
of the patients experienced any grade 4 bone marrow toxicity.
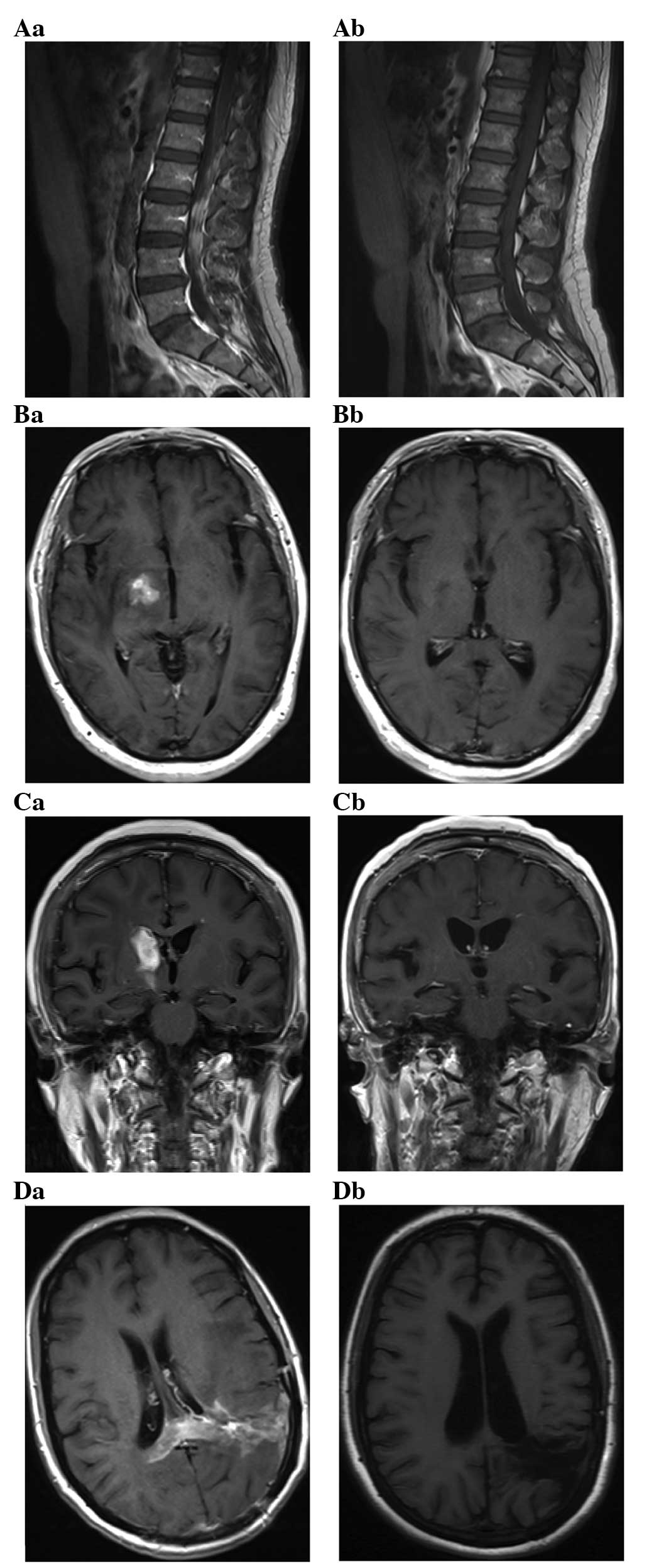
MRI analysis
MRI following successful treatment of PCNSL showed
small enhancing lesions in regions of the initial tumor or surgical
manipulation. Complete disappearance of all the measurable disease
and no new lesions following treatment were classified as CR. T1
axial, post-gadolinium MRI of PCNSLs at diagnosis (Fig. 2Aa–Da) and a CR after 6 cycles of
immunochemotherapy (Fig. 2Ab–Db) were
identified. A 42-year-old man exhibited enhancing lesions in the
L1–5 vertebral levels of the spinal canal (Fig. 2A). A 68-year-old man and a 63-year-old
woman exhibited a solid enhancing lesion involving the right basal
ganglia (Fig. 2B and C). A
39-year-old woman exhibited a diffuse involvement of the left
temporal lobe and parietal lobe with enhancing masses (Fig. 2D).
Toxicity
Eighteen patients experienced 6 cycles, and none of
the patients exhibited serious side effects in the retrospective
review. According to the National Cancer Institute-Common Toxicity
Criteria, for the toxicity of bone marrow suppression, 6 (33.3%)
patients experienced grade 1, 4 (22.2%) experienced grade 2 and 2
(11.1%) experienced grade 3. For the toxicity of gastrointestinal
reaction, 2 (11.1%) patients experienced grade 1. For the toxicity
of liver function damage, 3 (16.7%) patients experienced grade 1
and 1 (0.06%) experienced grade 3. For the toxicity of renal
function damage, 1 (0.06%) patient experienced grade 1. For the
toxicity of oral ulcer, 1 (0.06%) patient experienced grade 1 and 1
(0.06%) experienced grade 3. None of the 18 patients experienced
grade 4 toxicities and there were no treatment-related mortalities
(Table III).
 | Table III.Rituximab, methotrexate, cytarabine
and dexamethasone (R-MAD). |
Table III.
Rituximab, methotrexate, cytarabine
and dexamethasone (R-MAD).
| Toxicity | Grade 1, no. (%) | Grade 2, no. (%) | Grade 3, no. (%) | Grade 4, no. (%) |
|---|
| Bone marrow
suppression | 6 (33.3) | 4 (22.2) | 2 (11.1) | 0 (0.0) |
| Gastrointestinal
reaction | 2 (11.1) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Liver function
damage | 3 (16.7) | 0 (0.0) | 1 (0.06) | 0 (0.0) |
| Renal function
damage | 1 (0.06) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Oral ulcer | 1 (0.06) | 0 (0.0) | 1 (0.06) | 0 (0.0) |
Discussion
PCNSL is rare and fatal. The prognosis of untreated
PCNSL patients is extremely poor and their median survival time is
1.5–3.3 months (9). It was previously
demonstrated that the MTX-based protocols could be effective
compared with HD-MTX alone, but the overall response rate is poor
and the toxicity is serious (10,11).
Pels et al (7)
reported the results of a combination of HD-MTX, high-dose of Ara-C
(HD-Ara-C), vinca-alkaloids and alkylating agents treated with
PCNSL. The CR rate was 61%, PR rate was 10% and there were 9%
treatment-related fatalities. In a previous trial, 52 patients were
treated with the combination of MTX, teniposide, carmustine and
methylprednisolone plus intrathecal chemotherapy followed by
whole-brain radiation therapy (WBRT) (12). This regimen resulted in an overall
response rate of 81%, a 2-year OS of 69% and 10% of patients
succumbed. Ferreri et al (13)
treated 41 patients with HD-MTX, HD-Ara-C, thiotepa and idarubicin,
and subsequent WBRT. The overall response rate was 83%. In a
single-institution study, the overall response rate for HD-MTX plus
temozolomide was 70% and the CR rate was 45%. One treatment-related
fatality was observed (14). In the
present study, 18 PCNSL patients were treated with R-MAD, the
overall response rate was 94.5% and none of the patients
experienced any grade 4 bone marrow toxicity.
Ara-C increases the incorporation of
cytarabinecytidine triphosphate formation and DNA, which killed
more proliferating cells of the S-phase. The combination of MTX and
HD-Ara-C increased the rate of CR from 18 to 46% (P=0.006).
Preliminary results of a randomized phase II study provided
evidence for the benefit of the addition of HD-Ara-C to HD-MTX
(15). In a large randomized phase II
study that evaluated HD-MTX-based induction, with or without
HD-Ara-C (2 g/m2), the median failure-free survival in
patients who received the combination of HD-MTX and HD-Ara-C
induction was 8 months. By contrast, the median failure-free
survival in patients who were administered HD-MTX alone was only 4
months (16). In Western countries, 2
g/m2 Ara-C was administered twice a day, on days 2 and 3
(15). In the present study, 18
patients received 1 g/m2 Ara-C on day 1 in order to
decrease the toxicity. None of the patients experienced any grade 4
toxicity. The optimal dose of Ara-C remains to be further
studied.
Rituximab is an anti-cluster of differentiation 20
monoclonal antibody. Currently, the addition of rituximab to the
cyclophosphamide, doxorubicin, vincristine and prednisone (CHOP)
regimen is widely accepted as the standard chemotherapy for
systemic DLBCL. Approximately 95% of PCNSL are DLBCL. However,
rituximab is a large protein and has poor penetration to cross the
blood-brain barrier (BBB). Addition of rituximab to CHOP was
reported to prevent the CNS dissemination of DLBCL in a
retrospective German analysis of 1,222 patients >60 years of
age, which, however, was not confirmed in a French randomized trial
including 399 patients (17). A
recent study provided evidence that HD-MTX plus rituximab compared
with HD-MTX alone in immunocompetent patients with PCNSL improves
the CR and OS rates (18). In CNS
lymphoma, rituximab may induce the response of contrast-enhancement
lesions, possibly in local where there is disruption of the BBB
(19). In the present retrospective
study, the addition of rituximab to HD-MTX-based chemotherapy
proved feasible in 18 PCNSL patients, with a CR rate of 55.6%.
These results encouraged further research of the precise role of
rituximab in the treatment of PCNSL.
Lymphoma cells are sensitive to steroids, however,
high-dose steroids cause immunosuppressive effects. In the present
retrospective study, we applied short-term and small doses of
dexamethasone and achieved an effective outcome.
The serum lactate dehydrogenase (LDH) concentration
has been reported to be an independent prognostic marker in PCNSL
(20,21). In the present retrospective study,
there were 5 patients' LDH concentrations elevated, and 1 of these
patients achieved CR, 3 had PR and 1 had PD. These results
suggested that the concentration of LDH may be an indicator of poor
prognosis.
In conclusion, R-MAD as first-line treatment
performs a good effect on PCNSL patients and this regimen is
well-tolerated in PCNSL, including elderly patients. Further
research of clinical trials is required that focuses on
optimization of combination based on high-dose MTX. PCNSL is a rare
disease, despite its increasing incidence. The present study was
limited by its small sample size and its shorter follow-up time.
This requires cooperation of multi-center studies and more evidence
is required in further research. The poor treatment and prognosis
of PCNSL stimulated the requirement to provide the certain
pathogenesis and the pathological and biological features of PCNSL,
which may help to stratify therapy with specific regimens for
patients, and thus, to improve the outcome.
Acknowledgements
The present study was supported by the National
Natural Science Foundation of China General Program (grant no.
81272842).
References
|
1
|
Batchelor T and Loeffler JS: Primary CNS
lymphoma. J Clin Oncol. 24:1281–1288. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Abrey LE: Primary central nervous system
lymphoma. Curr Opin Neurol. 22:675–680. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Swerdlow SH, Campo E, Harris NL, et al:
World Health Organization Classification of tumours. Pathology and
genetics of tumours of haematopoietic and lymphoid tissuesWHO
press: WHO classification of tumours of haematopoietic and lymphoid
tissues. 4th. IARC; Lyon: pp. 236–237. 2008
|
|
4
|
Corn BW, Marcus SM, Topham A, Hauck W and
Curran WJ Jr: Will primary central nervous system lymphoma be the
most frequent brain tumor diagnosed in the year 2000? Cancer.
79:2409–2413. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Morris PG and Abrey LE: Therapeutic
challenges in primary CNS lymphoma. Lancet Neurol. 8:581–592. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nayak L and Batchelor TT: Recent advances
in treatment of primary central nervous system lymphoma. Curr Treat
Options Oncol. 14:539–552. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Pels H, Schmidt-Wolf IG, Glasmacher A,
Schulz H, Engert A, Diehl V, Zellner A, Schackert G, Reichmann H,
Kroschinsky F, et al: Primary central nervous system lymphoma:
Results of a pilot and phase II study of systemic and
intraventricular chemotherapy with deferred radiotherapy. J Clin
Oncol. 21:4489–4495. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Abrey LE, Batchelor TT, Ferreri AJM,
Gospodarowicz M, Pulczynski EJ, Zucca E, Smith JR, Korfel A,
Soussain C, DeAngelis LM, et al: International Primary CNS Lymphoma
Collaborative Group: Report of an international workshop to
standardize baseline evaluation and response criteria for primary
CNS lymphoma. J Clin Oncol. 23:5034–5043. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Reni M, Ferreri AJ, Garancini MP and Villa
E: Therapeutic management of primary central nervous system
lymphoma in immunocompetent patients: Results of a critical review
of the literature. Ann Oncol. 8:227–234. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cobert J, Hochberg E, Woldenberg N and
Hochberg F: Monotherapy with methotrexate for primary central
nervous lymphoma has single agent activity in the absence of
radiotherapy: a single institution cohort. J Neurooncol.
98:385–393. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ferreri AJ, Reni M, Foppoli M, et al:
International Extranodal Lymphoma Study Group (IELSG): High-dose
cytarabine plus high-dose methotrexate versus high-dose
methotrexate alone in patients with primary CNS lymphoma: a
randomised phase 2 trial. Lancet. 374:1512–1520. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Poortmans PM, Kluin-Nelemans HC,
Haaxma-Reiche H, Van't Veer M, Hansen M, Soubeyran P, Taphoorn M,
Thomas J, Van den Bent M, Fickers M, et al: European Organization
for Research and Treatment of Cancer Lymphoma Group: High-dose
methotrexate-based chemotherapy followed by consolidating
radiotherapy in non-AIDS-related primary central nervous system
lymphoma: European Organization for Research and Treatment of
Cancer Lymphoma Group Phase II Trial 20962. J Clin Oncol.
21:4483–4488. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ferreri AJ, Dell'Oro S, Foppoli M,
Bernardi M, Brandes AA, Tosoni A, Montanari M, Balzarotti M, Spina
M, Ilariucci F, et al: MATILDE regimen followed by radiotherapy is
an active strategy against primary CNS lymphomas. Neurology.
66:1435–1438. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Wang XX, Huang HQ, Bai B, Cai QQ, Cai QC,
Gao Y, Xia YF, Xia ZJ and Jiang WQ: Clinical outcomes of patients
with newly diagnosed primary central nervous system lymphoma are
comparable on treatment with high-dose methotrexate plus
temozolomide and with high-dose methotrexate plus cytarabine: A
single-institution experience. Leuk Lymphoma. 55:2497–2501. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ferreri AJM, Reni M, Foppoli M, et al:
Randomized phase II trial on primary chemotherapy with high-dose
methotrexate alone or associated with highdose cytarabine for
patients with primary CNS lymphoma (IELSG #20 Trial): Tolerability,
activity and event-free survival analysis. Blood. 112:5802008.
|
|
16
|
Ferreri AJ, Reni M, Foppoli M, Martelli M,
Pangalis GA, Frezzato M, Cabras MG, Fabbri A, Corazzelli G,
Ilariucci F, et al: International Extranodal Lymphoma Study Group
(IELSG): High-dose cytarabine plus high-dose methotrexate versus
high-dose methotrexate alone in patients with primary CNS lymphoma:
A randomised phase 2 trial. Lancet. 374:1512–1520. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Feugier P, Virion JM, Tilly H, Haioun C,
Marit G, Macro M, Bordessoule D, Recher C, Blanc M, Molina T, et
al: Incidence and risk factors for central nervous system
occurrence in elderly patients with diffuse large-B-cell lymphoma:
Influence of rituximab. Ann Oncol. 15:129–133. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Holdhoff M, Ambady P, Abdelaziz A, Sarai
G, Bonekamp D, Blakeley J, Grossman SA and Ye X: High-dose
methotrexate with or without rituximab in newly diagnosed primary
CNS lymphoma. Neurology. 83:235–239. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Batchelor TT, Grossman SA, Mikkelsen T, Ye
X, Desideri S and Lesser GJ: Rituximab monotherapy for patients
with recurrent primary CNS lymphoma. Neurology. 76:929–930. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ghesquières H, Drouet Y, Sunyach MP,
Sebban C, Chassagne-Clement C, Jouanneau E, Honnorat J, Biron P and
Blay JY: Evidence of time-dependent prognostic factors predicting
early death but not long-term outcome in primary CNS lymphoma: A
study of 91 patients. Hematol Oncol. 31:57–64. 2013. View Article : Google Scholar
|
|
21
|
Liu BL, Cheng JX, Zhang X, Zhang W and
Cheng H: Limited role of surgery in the management of primary
central nervous system lymphoma (Review). Oncol Rep. 22:439–449.
2009.PubMed/NCBI
|