Introduction
Post-transplant lymphoproliferative disorder (PTLD)
is a known complication of both solid organ and stem cell
transplantation. The majority of PTLD cases occur within the first
year after the transplant and ~90% of the cases are Epstein-Barr
virus (EBV)-related, CD20-positive B-cell neoplasms (1). PTLD is associated with a variety of
clinical presentations, but is rarely found in the skin (2). Reduction of immunosuppression is
currently the preferred initial treatment for PTLD. Due to comorbid
diseases and chronic immunosuppression, high morbidity and
mortality rates are observed in PTLD patients treated with
cytotoxic drugs. The risk of infection is also high.
We herein report a case of PTLD in a renal
transplant recipient, presenting with skin ulceration that was
complicated with cryptococcal infection following treatment with
chemotherapy. Antifungal agents were administered to control the
fungal infection. The patient later developed recurrence of the
lymphoma and was successfully treated with single-agent
rituximab.
Case report
A 41-year-old man with a past history of hepatitis B
and chronic renal failure of unknown etiology had received a
cadaveric renal transplant in 1997. The maintenance
immunosuppressive therapy included prednisolone, mycophenolate
mofetil and tacrolimus. The patient presented to the Tuen Mun
Hospital (Hong Kong, China) in June, 2009 with an ulcer in the
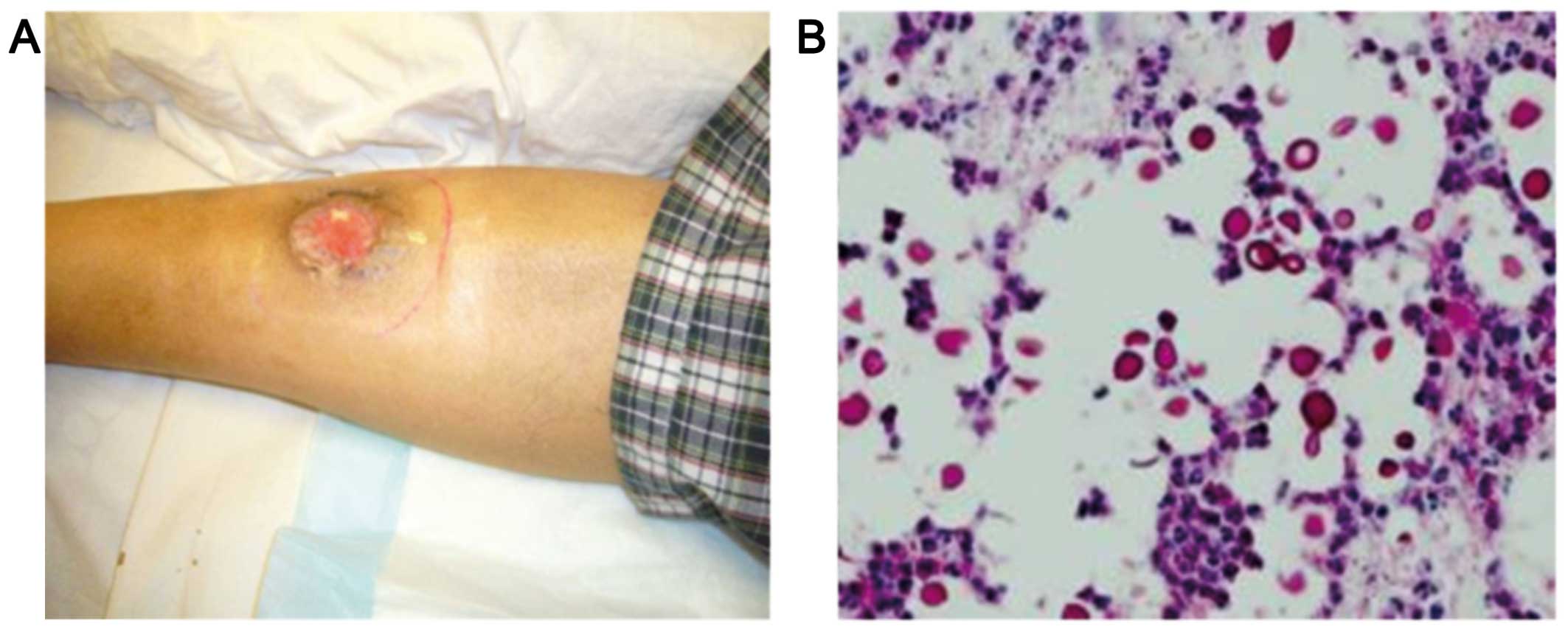
right leg 12 years after receiving the renal transplant (Fig. 1A). There was no fever, night sweats
or weight loss. There was also no lymphadenopathy or
hepatosplenomegaly. Laboratory investigations revealed a white
blood cell count of 5.2×109/l, a haemoglobin level of
9.9 g/dl, a platelet count of 151×109/l and a creatinine
level of 340 µmol/l. The lactate dehydrogenase level was elevated
to 1,063 U/l (normal, 106–218 U/l). A computed tomography scan of
the neck, thorax, abdomen and pelvis did not reveal any enlarged
lymph nodes and bone marrow examination did not show any evidence
of lymphoma. A biopsy of the leg ulcer revealed the presence of
ulcerated skin tissue with diffuse infiltration of the dermis by
monomorphous lymphoid cells, associated with large areas of
necrosis. The cells were medium to large in size, with irregular
nuclei, several of which contained large prominent nucleoli. The
mitotic activity was brisk. The lymphoid cells were positive for
CD20, CD30, CD79a, B-cell lymphoma (Bcl)-2, Bcl-6 and multiple
myeloma oncogene 1, and negative for CD3, CD4, CD5, CD8, CD10,
CD43, CD56, cyclin D1 and anaplastic lymphoma kinase. Epstein Barr
virus (EBV)-encoded RNA in situ hybridization and latent
membrane protein-1 were both positive. The pathological diagnosis
was post-transplant lymphoproliferative disorder, namely diffuse
large B-cell lymphoma.
Following the diagnosis of PTLD, the patient was
treated with immunosuppression reduction, but the skin ulcer
persisted. He was then administered two courses of CEOP
chemotherapy, consisting of cyclophosphamide 750 mg/m2
intravenously (i.v.) on day 1, epirubicin 50 mg/m2 i.v.
on day 1, vincristine 1.4 mg/m2 i.v. on day 1, and
prednisolone 40 mg/m2 per os on days 1–5. The
patient also received antibiotic prophylaxis with levofloxacin. The
circulating EBV-DNA level was measured at diagnosis and serially
every 2 months after treatment initiation using quantitative
polymerase chain reaction. The results were expressed as copies/ml
of total EBV DNA calculated using a standard curve, and log scale
interpretation was used.
The patient developed cellulitis of the right leg 1
week after the second course of chemotherapy. A biopsy of the
affected area revealed cryptococcal infection (Fig. 1B). Chemotherapy was discontinued and
the patient was administered a course of antifungal agents
(amphotericin B followed by fluconazole). Debridement of the
infected tissue was also performed and the cellulitis gradually
subsided. There was recurrence of leg ulcer 2 months later and the
plasma EBV-DNA was elevated. The patient was then administered four
courses of rituximab 375 mg/m2 delivered i.v. once a
week for 4 weeks. The skin ulcer healed and the plasma EBV-DNA
titre decreased to undetectable levels. The patient remains well 6
years after treatment, with no evidence of disease relapse at the
last follow-up in April, 2016.
Discussion
Post-transplant lymphoproliferative disorder is a
well-known complication of solid organ or allogenic stem cell
transplantation. The risk of PTLD depends on the type of
transplant, with a likely direct association with the intensity and
duration of the immunosuppression (1). Approximately 90% of the cases are
EBV-related CD20-positive B-cell neoplasms, which proliferate in an
environment of impaired T-cell immunity. EBV induces uncontrolled
B-cell proliferation, resulting in increased levels of interleukin
and tumour growth factors. The dysregulated and uncontrolled B-cell
proliferation ultimately causes PTLD (3).
The World Health Organization (WHO) classifies PTLDs
into four major categories: i) Early lesions, which encompass
reactive plasmacytic hyperplasia and infectious mononucleosis-like
lesions; ii) polymorphous PTLD; iii) monomorphic PTLD, which is
classified according to the WHO classification of lymphoma; and iv)
classical Hodgkin lymphoma-like PTLD (4).
The risk factors for developing PTLD include EBV
serostatus (higher risk of developing PTLD when an EBV seronegative
organ recipient receives an organ from a seropositive donor), type
of organ transplant, intensity of immunosuppression and age: There
is a higher incidence of PTLD among extrarenal transplant
recipients (5–7); the incidence of PTLD is highest with
haploidentical hematopoietic stem cell transplant (HSCT),
heart/lung and multivisceral transplants (≤20%), followed by liver
(4.5%), combination heart-lung (2.5%), pancreas (2%), kidney
(1–1.5%) and matched related and unrelated HSCT (0.5–1%) (7,8); in
addition, pediatric patients are at greater risk compared with
adults (5).
The presentation of PTLD is variable, and early
symptoms may be non-specific, such as fever, malaise and weight
loss. Extranodal involvement by PTLD is also common, but it rarely
affects the skin. Skin lesions in PTLD may be solitary or multiple
papules, nodules, plaques with ulceration, comedo-like lesions,
localized alopecia and follicular keratotic papules (2). It was reported that cutaneous T-cell
PTLD was more common compared with cutaneous B-cell PTLD, with
mycosis fungoides as the most common cutaneous T-cell lymphoma
subtype. EBV-associated cutaneous B-cell PTLD predominates among
organ transplant recipients (5). Our
patient presented with a leg ulceration and awareness of this rare
presentation is crucial for early diagnosis. Our patient also had
late-onset PTLD, 12 years after the renal transplant. Relative to
early-onset PTLD, late-onset PTLD is often associated with more
monoclonal lesions and a worse prognosis.
Treatment modalities for PTLD include reduction of
immunosuppression, antiviral agents, chemotherapy and monoclonal
antibodies. Reduction of immunosuppression is the preferred initial
management of PTLD. The goal is to restore EBV-specific cellular
immunity without graft rejection. Chemotherapy is used to treat
patients who do not respond to reduction of immunosuppression.
Cytotoxic drugs are usually effective and have a rapid response
rate, but at a considerable cost. Due to comorbid diseases and
chronic immunosuppression, high morbidity and mortality rates are
observed in PTLD patients compared with non-Hodgkin lymphoma
patients treated with similar regimens. There is also a higher risk
of infection in PTLD patients. Cyclophosphamide, doxorubicin,
vincristine and prednisolone (CHOP)-based therapy was shown to
achieve a complete remission rate of 50% and a 5-year progression
free survival of 43%, but the treatment-related mortality was high
at 31% (9).
Additional treatment options include immunotherapy
with the anti-CD20 monoclonal antibody rituximab. Single-agent
rituximab therapy, following failure of immunosuppression
reduction, achieved overall response rates of 44–70.5% and complete
response rates of 28–53% (10,11).
Sequential immunochemotherapy with rituximab followed by
chemotherapy was also proven to be safe and effective in adult
B-cell and Burkitt PTLD. Trappe et al reported an overall
response rate of 90%, a complete remission rate of 68% and a median
overall survival of 6.6 years after sequential therapy with
rituximab followed by chemotherapy in patients with adult B-cell
PTLD (12).
Quantification of the plasma EBV-DNA level may
facilitate the monitoring of treatment response and it has been
found to be effective in different types of lymphoproliferative
diseases (13–15). For cases responding to treatment,
circulating EBV-DNA falls to undetectable levels when complete
remission is achieved (13). The
disease activity in our patient correlated well with the plasma
EBV-DNA level.
In conclusion, skin lesions are a rare manifestation
of PTLD and awareness of this presentation is crucial. Physicians
must be aware of infective complications if cytotoxic chemotherapy
is used for the treatment of PTLD. Rituximab appears to be a
feasible option for treatment of PTLD in patients who do not
respond to reduction of immunosuppression.
References
|
1
|
Landgren O, Gilbert ES, Rizzo JD, Socié G,
Banks PM, Sobocinski KA, Horowitz MM, Jaffe ES, Kingma DW, Travis
LB, et al: Risk factors for lymphoprolipherative disorders after
allogeneic hematolopoietic cell transplantation. Blood.
113:4992–5001. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Seçkin D, Barete S, Euvrard S, Francès C,
Kanitakis J, Geusau A, Del Marmol V, Harwood CA, Proby CM, Ali I,
et al: Primary cutaneous posttransplant lymphoproliferative
disorders in solid organ transplant recipients: A multicenter
European case series. Am J Transplant. 13:2146–2153. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Tanner JE and Alfieri C: The Epstein-Barr
virus and post-transplant lymphoproliferative diseases: Interplay
of immunosuppression, EBV, and the immune system in disease
pathogenesis. Transpl Infect Dis. 3:60–69. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Swerdlow SH, Webber SA, Chadburn A and
Ferry JA: Post-transplant lymphoproliferative disordersWHO
Classification of Tumours of Haematopoietic and Lymphoid Tissues.
Swerdlow SH, Campo E, Harris NL, Jafffe ES, Pileri SA, Stein H,
Thiele J and Vardiman JW: 4th. International Agency for Research on
Cancer; Lyon: pp. 343–349. 2008
|
|
5
|
Trofe J, Beebe TM, Buell JF, Hanaway MJ,
First MR, Alloway RR, Gross TG and Woodle ES: Posttransplant
malignancy. Prog Transplant. 14:193–200. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Nassi L and Gaidano G: II. Challenges in
the management of post-transplant lymphoproliferative disorder.
Hematol Oncol. 33(Suppl 1): S96–S99. 2015. View Article : Google Scholar
|
|
7
|
Dierickx D, Tousseyn T and Gheysens O: How
I treat posttransplant lymphoproliferative disorders. Blood.
126:2274–2283. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Dierickx D, Tousseyn T, Sagaert X, Fieuws
S, Wlodarska I, Morscio J, Brepoels L, Kuypers D, Vanhaecke J,
Nevens F, et al: Single-center analysis of biopsy-confirmed
posttransplant lymphoproliferative disorder: Incidence,
clinicopathological characteristics and prognostic factors. Leuk
Lymphoma. 54:2433–2440. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Choquet S, Trappe R, Leblond V, Jäger U,
Davi F and Oertel S: CHOP-21 for the treatment of post-transplant
lymphoproliferative disorders (PTLD) following solid organ
transplantation. Haematologica. 92:273–274. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Choquet S, Leblond V, Herbrecht R, Socié
G, Stoppa AM, Vandenberghe P, Fischer A, Morschhauser F, Salles G,
Feremans W, et al: Efficacy and safety of rituximab in B-cell
post-transplantation lymphoproliferative disorders: Results of a
prospective multicenter phase 2 study. Blood. 107:3053–3057. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Oertel SH, Verschuuren E, Reinke P,
Zeidler K, Papp-Váry M, Babel N, Trappe RU, Jonas S, Hummel M,
Anagnostopoulos I, et al: Effect of anti-CD 20 antibody rituximab
in patients with post-transplant lymphoproliferative disorder
(PTLD). Am J Transplant. 5:2901–2906. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Trappe R, Oertel S, Leblond V, Mollee P,
Sender M, Reinke P, Neuhaus R, Lehmkuhl H, Horst HA, Salles G, et
al: Sequential treatment with rituximab followed by CHOP
chemotherapy in adult B-cell post-transplant lymphoproliferative
disorder (PTLD): The prospective international multicentre phase 2
PTLD-1 trial. Lancet Oncol. 13:196–206. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Au WY, Pang A, Choy C, Chim CS and Kwong
YL: Quantification of circulating Epstein-Barr virus (EBV) DNA in
the diagnosis and monitoring of natural killer cell and
EBV-positive lymphomas in immunocompetent patients. Blood.
104:243–249. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Gulley ML and Tang W: Using Epstein-Barr
viral load assays to diagnose, monitor, and prevent posttransplant
lymphoproliferative disorder. Clin Microbiol Rev. 23:350–366. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Law MF, Poon WL, Ng KS, Chan HN, Lai HK,
Ha CY, Ng C, Yeung YM and Yip SF: Quantification of plasma
Epstein-Barr virus DNA for assessing treatment response in a
patient with plasmablastic lymphoma. Ann Hematol. 91:789–791. 2012.
View Article : Google Scholar : PubMed/NCBI
|