Introduction
Leptin, mainly synthesized by adipocytes, is a
pleiotropic regulator involving food intake, inflammatory and
immune response, cellular apoptosis, angiogenesis and proliferation
of different cell types (1–3). However, the effects of leptin on colon
cancer remain a matter of debate (4–8).
Moreover, the majority of previous studies only included
non-overweight patients and focused on Western individuals. The
serum leptin levels in overweight Chinese patients with colon
carcinoma and the changes in serum leptin levels following
treatment have rarely been documented. Thus, the present study was
conducted to compare the serum leptin levels between overweight
colon cancer Chinese patients and colonoscopy-negative controls, as
well as in preoperative and postoperative cancer patients.
Materials and methods
Patient characteristics and inclusion
criteria
Electronic colonoscopy was conducted in all the
participants. Patients with suspected cancer were confirmed
histologically and cancer-free subjects were identified as
controls. The clinical records of all the participants were
reviewed. The inclusion criteria were Chinese individuals aged
>18 years with a body mass index (BMI) of ≥25 kg/m2,
whereas the exclusion criteria were dyslipidemia, diabetes mellitus
and hypertension. Finally, 63 patients with histologically
confirmed colon cancer and 40 control volunteers free of colon
cancer were included in the present study.
The weight and height of all the participants were
determined and the BMI of each participant was calculated using the
quotation: BMI = weight (kg) / height (m)2. Blood
samples (3 ml) were collected from the control subjects and from
the patients 3–7 days preoperatively and on the 21th postoperative
day. The sera of the blood samples were obtained after
centrifugation at 1,000 × g for 20 mins. Thereafter, the
samples were stored at −80°C until further analysis. The serum
leptin levels were measured using a human leptin ELISA kit
(TiterZyme® Enzyme Immunometric Assay Kit; Assay Designs
Inc., Ann Arbor, MI, USA).
This study was approved by the Hunan Subject Ethics
Subcommittee of the hospital. Informed and written consents were
obtained from each subject prior to the commencement of the
examinations of the study.
Immunohistochemistry
The resected specimens were fixed in 10% formalin,
cut into 4-µm sections and mounted onto adhesive-coated slides. The
slides were deparaffinized in xylene twice for 10 min and
rehydrated through descending concentrations of ethanol. Antigen
retrieval was performed in 0.01 M citrate buffer (pH 6.0) for 2 min
and 30 sec at 100°C using a microwave oven. Endogenous peroxidase
activity was blocked with 0.3% hydrogen peroxidase for 10 min.
After washing with Tris-buffered saline, the sections were
incubated with blocking serum for 1 h. The sections were then
incubated with the following primary antibodies: H-146 rabbit
polyclonal antibody for leptin (dilution, 1:200; cat. no. SC-9014,
Santa Cruz Biotechnology, Dallas, TX, USA), D9E rabbit polyclonal
antibody for phosphorylated (p)-Akt (Ser 473) (dilution, 1:100;
cat. no. SC-135651, Santa Cruz Biotechnology), rabbit polyclonal
antibody for P-70S6 Kinase (Thr 389) (dilution, 1:100; cat. no.
LS-C416604-100, LifeSpan BioSciences, Seattle, WA, USA) and rabbit
polyclonal antibody () for p-mammalian target of rapamycin (mTOR)
(Ser 2448) (dilution, 1:250; cat. no. SC-101738, Santa Cruz
Biotechnology). The sections were then washed and incubated with
biotinylated secondary antibody (cat. no. AP21490BT-N, Acris
Antibodies GmbH, San Diego, CA, USA) for 30 mins. The
immunohistochemical (IHC) staining was evaluated and photographed
using an Olympus CX42 microscope (Olympus, Tokyo, Japan). Two
independent investigators scored the expression of leptin, p-Akt,
p-mTOR and P-70S6K. A scale of 0 (negative), + (weak
inmmunoreactivity), ++ (moderate immunoreactivity) and +++ (robust
immunoreactivity) was applied according to the intensity of
immunolabelling and number of positive cells. Tissues scored as ≥1+
(weak, moderate or robust immunoreactivity) were considered to be
positive, whilst those those scoring 0 were considered to be
negative. In the IHC analysis, there was no discrepancy between the
two observers regarding the patterns of biomarker expression and
the scores assigned to analyzed sections.
Statistical analysis
Statistical analyses were performed using SPSS 12.0
software (SPSS Inc., Chicago, IL, USA). Continuous data were
expressed as mean ± standard deviation. The serum leptin levels
between groups were compared using the Mann-Whitney U test. The
bivariate Pearson correlation was applied to determine the
correlation between serum leptin levels and other markers and
parameters. P-values <0.05 were considered to indicate
statistically significant differences.
Results
Patient characteristics
A total of 63 patients with colon cancer and 40
cancer-free controls were analyzed. All the patients underwent
total colectomy, and none of the patients had received chemotherapy
or radiotherapy preoperatively. The 63 patients included 29 men and
34 women with an age ranging from 27 to 78 years (median, 61 year
mean, 58.4 years). Based on histological grading (9), 13 of the patients had grade 1, 22 had
grade 2, and 28 had grade 3 tumors, whilst based on the TNM staging
system (http://www.cancer.org/cancer/colonandrectumcancer/detailedguide/colorectal-cancer-staged),
2 had stage I, 14 had stage II, 46 had stage III, and 1 had stage
IV disease.
The mean BMI was 27.32±2.3 kg/m2 in the
control group, and 27.24±2.0 and 27.31±2.1 kg/m2 before
and after colectomy, respectively, in the patient group (Table I). No significant difference in BMI
was observed between the patients and the controls, and between
patients before and after colectomy.
 | Table I.Serum leptin levels and BMI in colon
cancer patients and cancer-free controls. |
Table I.
Serum leptin levels and BMI in colon
cancer patients and cancer-free controls.
| Participants | Serum leptin level
(ng/ml) | BMI
(kg/m2) |
|---|
| Preoperative patients
(n=63) |
22.67±12.56 | 27.24±2.0 |
| Postoperative
patients (n=63) |
18.67±8.54 | 27.31±2.1 |
| Cancer-free controls
(n=40) | 12.68±7.80 | 27.32±2.3 |
Colon cancer patients had significantly higher serum
leptin levels compared with those in the cancer-free controls
(22.67±12.56 vs. 12.68±7.8 ng/ml, respectively, P<0.05; Table I). In the colon cancer patients, the
serum leptin levels were measured prior to and 21 days after
colectomy. The serum leptin level significantly decreased after the
operation (18.67±8.54 vs. 22.67±12.56 ng/ml, respectively,
P<0.05; Table I).
Associations between preoperative
serum leptin level and clinicopathological factors in colon
cancer
In the colon cancer group, there was a significant
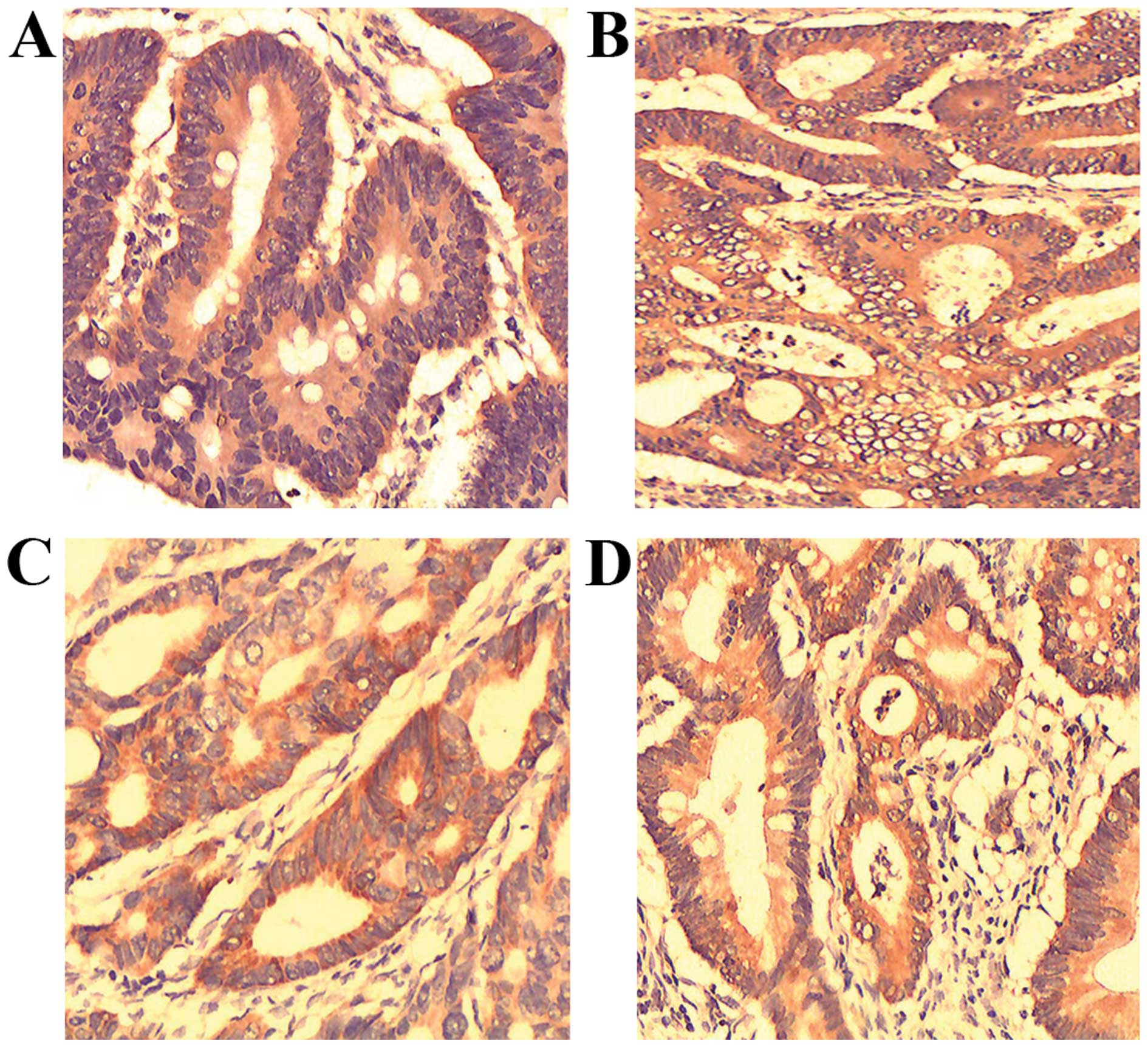
correlation of the leptin, p-Akt, p-mTOR and P-70S6K status with
the serum leptin levels (P<0.05; Fig.
1 and Table II). However, there
was no significant association of the serum leptin level with age,
gender, or lymph node involvement (P>0.05, Table II).
 | Table II.Associations between preoperative
serum leptin levels and clinicopathological factors in colon
cancer. |
Table II.
Associations between preoperative
serum leptin levels and clinicopathological factors in colon
cancer.
| Factors | No. of cases (%) | Serum leptin level
(ng/ml) | P-values |
|---|
| Gender |
|
|
|
| Male | 29 (46.0) | 22.18±11.34 | >0.05a |
|
Female | 34 (54.0) | 23.09±13.61 |
|
| Age (years) |
|
|
|
| <60 | 25 (39.7) | 22.06±12.02 | >0.05a |
| ≥60 | 38 (60.3) | 23.07±13.10 |
|
| T stage |
|
|
|
|
T1/T2 | 26 (41.3) | 23.01±14.25 | >0.05b |
|
T3/T4 | 37 (58.7) | 23.67±11.37 |
|
| TNM stage |
|
|
|
| I/II | 16 (25.4) | 16.89±9.53 | <0.05b |
| III | 47 (74.6) | 24.64±13.59 |
|
| Differentiation |
|
|
|
| High | 13 (20.6) | 18.98±11.35 | <0.05b |
|
Moderate | 22 (34.9) | 21.78±13.91 |
|
| Poor | 28 (44.5) | 25.08±12.06 |
|
| p-mTOR |
|
|
|
|
Negative | 18 (28.6) | 15.97±8.97 | <0.05b |
|
Positive | 45 (71.4) | 25.35±14.00 |
|
| p-70S6 kinase |
|
|
|
|
Negative | 19 (30.2) | 17.89±10.97 | <0.05b |
|
Positive | 44 (69.8) | 24.74±13.25 |
|
| p-Akt |
|
|
|
|
Negative | 16 (25.4) | 16.93±9.82 | <0.05b |
|
Positive | 47 (74.6) | 24.62±13.49 |
|
| Leptin |
|
|
|
|
Negative | 15 (23.8) | 15.78±8.21 | <0.05b |
|
Positive | 48 (76.2) | 24.82±13.91 |
|
Discussion
Colorectal cancer is a major cause of cancer-related
mortality and morbidity in Western countries and an increasing
health concern in China (10).
Epidemiological studies have demonstrated that obesity is
associated with an increasing risk of colorectal cancer development
and death from colon cancer (11).
Leptin may represent a biological link between colon cancer and
obesity (6).
The primary aim of the present study was to evaluate
serum leptin levels in colon carcinoma patients and compare them to
those of cancer-free individuals. A significant increase in serum
leptin was observed in colon carcinoma patients. Consistent with
our findings, Stattin et al found that higher serum levels
of leptin increased the risk of colon cancer (6). In addition, Chia et al
demonstrated that, among men, those in the highest tertile of
leptin concentrations had a 3.3-fold (95% confidence interval:
1.2–8.7) increased colorectal adenoma risk compared with those in
the lowest tertile (5). However,
Nakajima et al and Kumor et al reported that colon
cancer patients had lower or similar serum levels of leptin
compared with cancer-free controls (7,8).
Leptin is mainly synthesized and secreted by
adipocytes and its plasma levels in humans are strongly correlated
with BMI. In the study of Chia et al (5), the majority of the cancer patients were
overweight, whereas Kumor et al (7) and Nakajima et al (8) investigated colon cancer patients with
normal body weight. Thus, overweight colon patients may have a
different leptin status, and the serum leptin levels may differ
among various races. The results of our study suggested that, in
overweight Chinese patients with colon carcinoma, the serum levels
of leptin were significantly higher when compared with those of
cancer-free controls, and the level decreased significantly
following colectomy.
Akt/mTOR/70S6K is a critical pathway for tumor
growth and progression. p-Akt activates mTOR, which subsequently
phosphorylates 70S6K, inducing translation of mRNA and finally cell
growth (12). In the present study,
the serum level of leptin was significantly associated with focal
expression of leptin, p-Akt, p-mTOR and P-70S6K. In addition,
significant differences in serum leptin levels between various
histopathological grades of colon carcinoma were observed. Previous
studies also demonstrated that leptin may regulate proliferation
and apoptosis of colorectal carcinoma through the phosphoinositide
3-kinase/Akt/mTOR signalling pathway (12). These results suggested that leptin
may promote progression of colon cancer through the Akt/mTOR/70S6K
signaling pathway. However, further studies are required to
establish an association between the leptin and Akt/mTOR/70S6K
pathways in colon carcinogenesis.
In conclusion, the serum level of leptin was found
to be higher in overweight Chinese colon cancer patients, and
decreases following colectomy. In addition, the serum leptin level
was found to be significantly associated with focal expression of
leptin and, factors promoting carcinogenesis, such as p-Akt, p-mTOR
and P-70S6K. Thus, leptin may be associated with colon
carcinogenesis, and the serum leptin level may be used for early
diagnosis and monitoring of the response to treatment of colon
carcinoma in overweight Chinese patients.
Acknowledgements
This study was supported by grants from the Health
Department of Hunan Province (B2014-144), the Changsha Municipal
Science and Technology Bureau (k1406015-61) and the Key Research
and Development Program of Hunan Province (2016SK2066).
References
|
1
|
Considine RV, Sinha MK, Heiman ML,
Kriauciunas A, Stephens TW, Nyce MR, Ohannesian JP, Marco CC, McKee
LJ, Bauer TL, et al: Serum immunoreactive-leptin concentrations in
normal-weight and obese humans. N Engl J Med. 334:292–295. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Maffei M, Halaas J, Ravussin E, Pratley R,
Lee G, Zhang Y, Fei H, Kim S, Lallone R, Ranganathan S, et al:
Leptin levels in human and rodent: Measurement of plasma leptin and
ob RNA in obese and weight-reduced subjects. Nat Med. 1:1155–1161.
1995. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Baratta M: Leptin-from a signal of
adiposity to a hormonal mediator in peripheral tissues. Med Sci
Moni. 8:RA282–RA292. 2002.
|
|
4
|
Garofalo C and Surmacz E: Leptin and
cancer. J Cell Physiol. 207:12–22. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Chia VM, Newcomb PA, Lampe JW, White E,
Mandelson MT, McTiernan A and Potter JD: Leptin concentrations,
leptin receptor polymorphisms and colorectal adenoma risk. Cancer
Epidemiol Biomarkers Prev. 16:2697–2703. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Stattin P, Lukanova A, Biessy C, Söderberg
S, Palmqvist R, Kaaks R, Olsson T and Jellum E: Obesity and colon
cancer: Does leptin provide a link? Int J Cancer. 109:149–152.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kumor A, Daniel P, Pietruczuk M and
Malecka-Panas E: Serum leptin, adiponectin, and resistin
concentration in colorectal adenoma and carcinoma (CC) patients.
Int J Colorectal Dis. 24:275–281. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nakajima TE, Yamada Y, Hamano T, Furuta K,
Matsuda T, Fujita S, Kato K, Hamaguchi T and Shimada Y:
Adipocytokines as new promising markers of colorectal tumors:
Adiponectin for colorectal adenoma and resistin and visfatin for
colorectal cancer. Cancer Sci. 101:1286–1291. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sung JJ, Lau JY, Goh KL and Leung WK: Asia
Pacific Working Group on Colorectal Cancer: Increasing incidence of
colorectal cancer in Asia: Implications for screening. Lancet
Oncol. 6:871–876. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Calle EE and Kaaks R: Overweight, obesity
and cancer: Epidemiological evidence and proposed mechanisms. Nat
Rev Cancer. 4:579–591. 2004. View
Article : Google Scholar : PubMed/NCBI
|
|
11
|
Wang D, Chen J, Guo F, Chen H, Duan Z, Wei
MY, Xu QM, Wang LH and Zhong MZ: Clinical significance of mTOR and
p-mTOR protein expression in human colorectal carcinomas. Asian Pac
J Cancer Prev. 12:2581–2584. 2011.PubMed/NCBI
|
|
12
|
Wang D, Chen J, Chen H, Duan Z, Xu Q, Wei
M, Wang L and Zhong M: Leptin regulates proliferation and apoptosis
of colorectal carcinoma through PI3K/Akt/mTOR signalling pathway. J
Biosci. 37:91–101. 2012. View Article : Google Scholar : PubMed/NCBI
|