Introduction
Ocular toxicity induced by cancer chemotherapy
includes a broad spectrum of disorders, such as cortical blindness,
blurred vision and maculopathy, reflecting the unique anatomical,
physiological and biochemical characteristics of the eye. The
ocular side effects may be grouped into adnexal, anterior segment,
posterior segment and neuro-ophthalmic. Posterior segment lesions
are important due to the marked visual loss that may occur
(1). Visual loss secondary to
secondary retinitis pigmentosa (RP) is an uncommon complication of
cytotoxic chemotherapy. Docetaxel and platinum have been frequently
used in combination to treat various solid tumors, and are
relatively rarely associated with severe side effects (2). Although secondary RP with bilateral
blindness following docetaxel and platinum combination chemotherapy
is extremely rare, this complication warrants attention due to its
devastating impact on the quality of life of patients who receive
various anticancer therapies. We herein report a case of a patient
with small-cell carcinoma (SCC) of the endometrium who suffered
from bilateral blindness during postoperative chemotherapy at the
recommended dose, and review docetaxel- and/or platinum-induced
retinal toxicity in terms of clinical manifestations, diagnostic
and preventive strategies, with the aim of improving patient safety
during anticancer therapy.
Case report
On 13th October 2013, a 48-year-old Chinese woman
was admitted to the Department of Obstetrics and Gynecology of the
First Affiliated Hospital of Jinan University (Guangzhou, China)
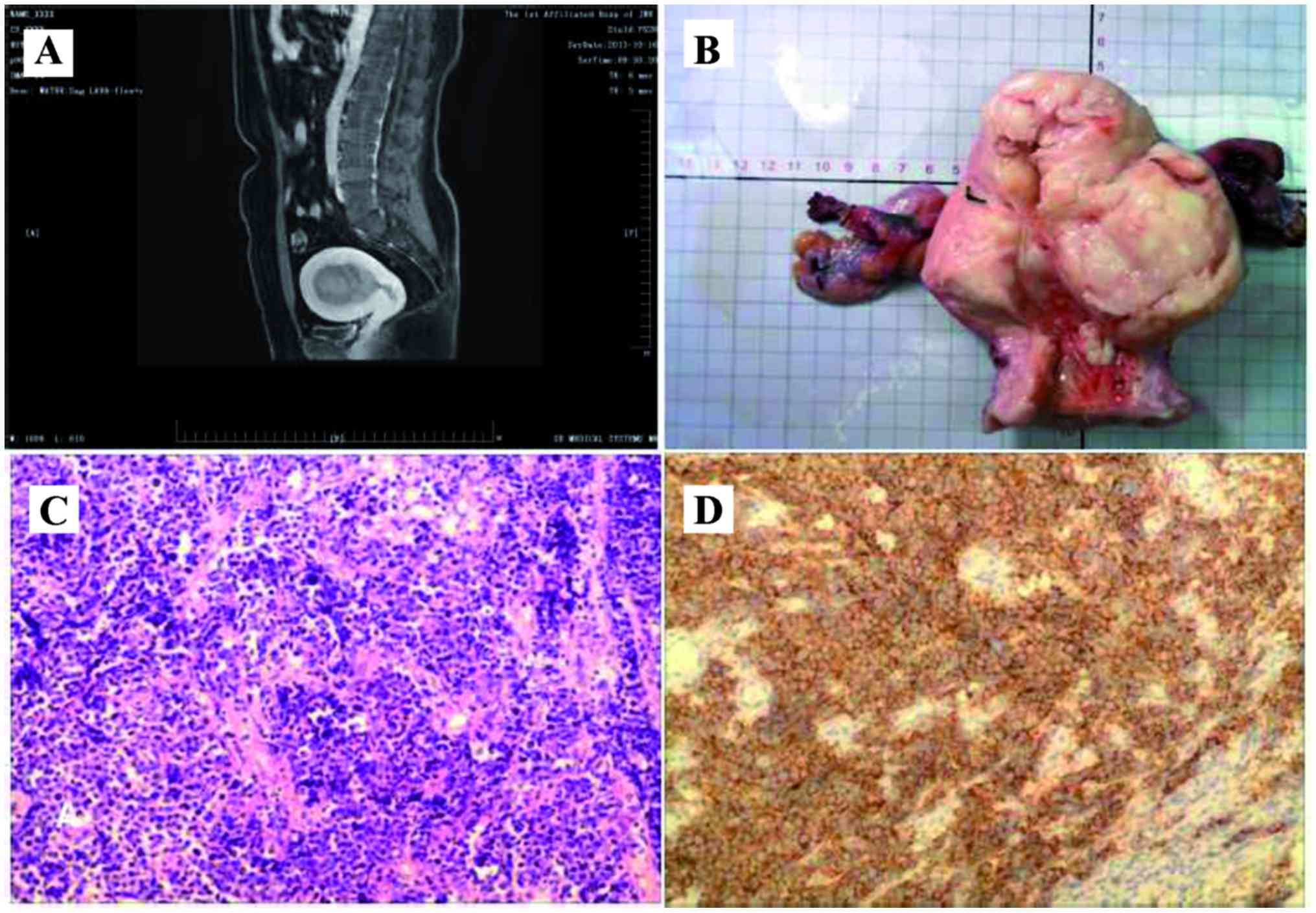
complaining of irregular vaginal bleeding for 1 month. Magnetic
resonance imaging (MRI) revealed a 5.0-cm mass in the endometrial
cavity and cervical stromal invasion. Dilation and curettage and
histological examination revealed SCC of the endometrium involving
the cervix. Immunohistochemical examination showed positive
staining for synaptophysin, chromogranin A and neuron-specific
enolase in the tumor cells. The patient also had a 1-year history
of diabetes mellitus type 2, treated with 50 mg acarbose per day.
The blood pressure and blood sugar levels were normal, there were
no visual abnormalities, and other systemic investigations were
also negative. After systemic metastasis of SCC was excluded, the
patient underwent open extensive hysterectomy, bilateral
salpingo-oophorectomy and pelvic lymph node dissection.
Pathological evaluation revealed that ~90% of the tumor in the
corpus uteri was SCC with myometrial invasion and cervical
involvement, and 10% was endometrioid adenocarcinoma (Fig. 1). Metastatic SCC was also observed in
the left external iliac and obturator lymph nodes. The patient was
diagnosed with SCC of the endometrium, International Federation of
Gynecology and Obstetrics stage IIIC1 (3). Docetaxel and platinum combination
adjuvant chemotherapy was administered after the operation. After
the first four courses of chemotherapy with docetaxel 120 mg (70
mg/m2) and cisplatin 90 mg (50 mg/m2) at
3-week intervals, the patient complained of visual impairment,
particularly farsightedness. The visual acuities were 0.3 on the
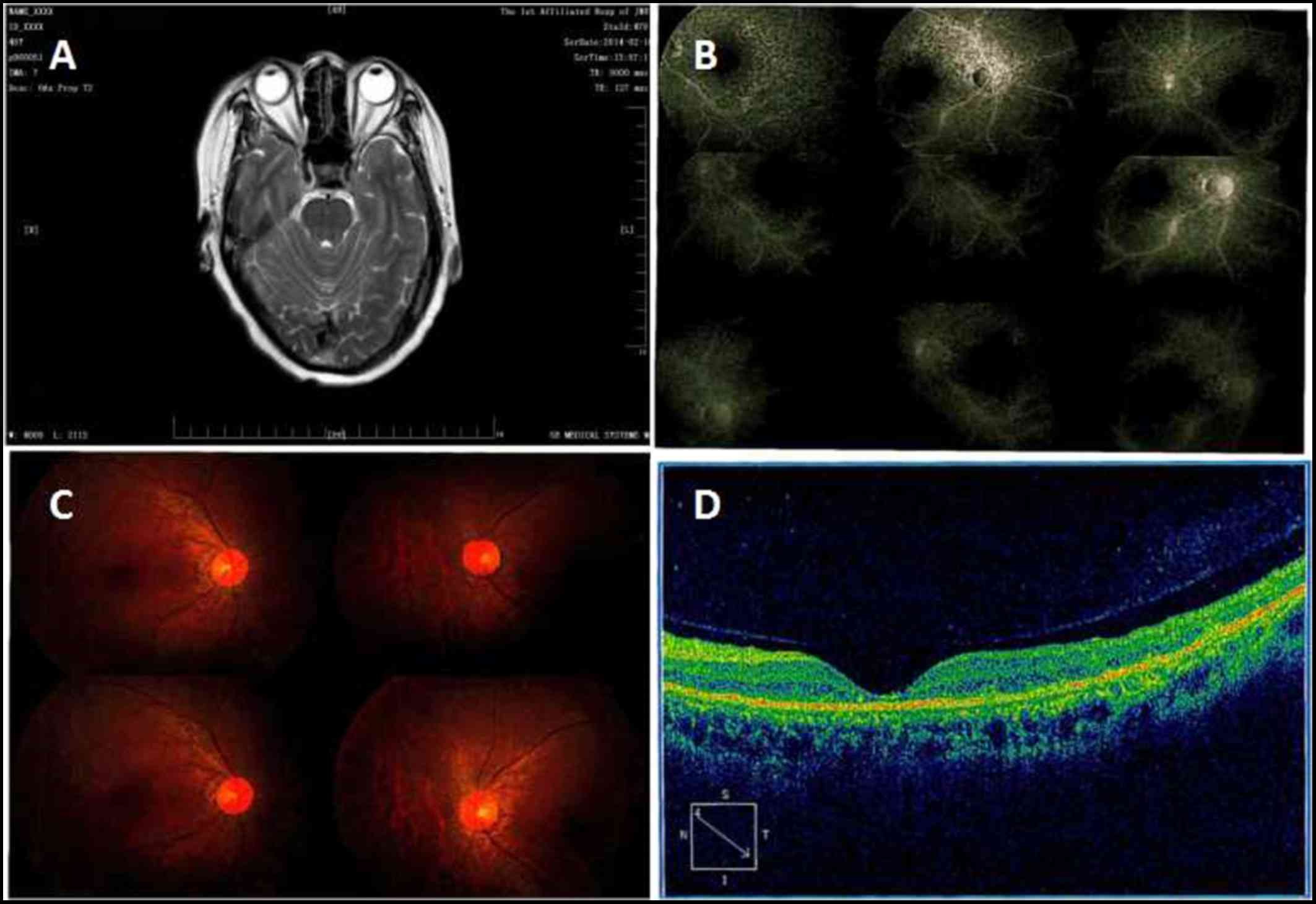
right and 0.5 on the left side, and MRI revealed no intracranial
space-occupying lesion. Following exclusion of ophthalmological
contraindications, the fifth course of chemotherapy consisted of
docetaxel 120 mg (70 mg/m2) and reduced lobaplatin 45 mg
(25 mg/m2) was administered. After 2 weeks, the patient
was admitted to the hospital for worsening vision with light
perception. Fundus fluorescence angiography revealed early-stage
diffuse depigmentation of the retinal pigment epithelium and a
window defect (hyperfluorescence) involving the macular area in
both eyes. Electroretinography revealed loss of function of rod and
cone cells bilaterally. Optical coherence tomography showed
thinning of the macular retina (Fig.
2). Retinal current map examination showed that the binocular
dark optic rod b-wave response and dark optic mixed reaction
b-waves were not recorded, and absence of photopic a- and b-wave
cone response. Fundus angiography showed secondary RP. Treatments
aimed at improving fundus microcirculation were performed,
including administration of enteric-coated aspirin and tanshinone.
Unfortunately, the visual acuity of the patient did not improve
following cessation of chemotherapy. There was no tumor recurrence
during the 33-month follow-up, but the patient developed bilateral
blindness.
The patient provided written informed consent to the
publication of this case report and associated images.
Discussion
Although postoperative adjuvant chemotherapy with
docetaxel and platinum is an increasingly used regimen for
advanced/metastatic or recurrent endometrial cancer with lower
toxicity and good tolerability (3),
some of these toxicities are irreversible and may adversely affect
the patient's quality of life, or even cause a permanent disability
(2). To the best of our knowledge,
RP with bilateral blindness following docetaxel and platinum
combination chemotherapy has not yet been reported in the English
literature.
Of note, paraneoplastic retinopathy-related
bilateral blindness from SCC of the endometrium should be excluded.
The initial symptoms of paraneoplastic retinopathy-related
bilateral blindness were decreased visual acuity and narrowing of
the visual field, or blindness on exposure to bright light and
total achromatopsia. In the present case, the patient first
presented with visual disturbances >2 months after the diagnosis
of SCC of the endometrium. Visual disturbances associated with
malignant neoplasms are more often caused by metastasis to the
brain, meninges, optic nerve, orbit, choroid, or retina. MRI
revealed no intracranial space-occupying lesion and metastatic
brain tumor from SCC of the endometrium was also excluded.
A total of 35 cases involving docetaxel- and/or
platinum-related retinal toxicity have been reported and are
summarized in Table I. The mean age
of the patients was 51.3 years. Vision loss developed immediately
after treatment in 2 cases, whereas in the remaining cases it
developed after 10 days to 4 cycles of chemotherapy. A total of 7
cases suffered from bilateral blindness or hemianopia. A total of
24 cases were reversible and 8 cases were irreversible at
follow-up. Retinal toxicity was more severe in 4 patients who
received intra-arterially administered cisplatin chemotherapy for
brain malignancy.
 | Table I.Summarized reported cases of
docetaxel- and/or platinum-induced retinal toxicity. |
Table I.
Summarized reported cases of
docetaxel- and/or platinum-induced retinal toxicity.
| Author(s) | Age, years | No. of cases | Toxicity | Drug | Diagnosis | Ophthalmic
evaluation | Occurrence after
chemotherapy | Follow-up | (Refs.) |
|---|
| Berman and Mann | 30 | 1 | Cortical
blindness | Cisplatin,
vinblastine and bleomycin | Embryonic cell
carcinoma of the testicle | The patient could not
perceive light; an optokinetic examination was positive for
blindness | 1 cycle | Reversible | (4) |
| Wilding et
al | ND | 11 | Blurred vision (n=8)/
altered color perception (n=3) | Cisplatin (high-dose
cisplatin (200 mg/m2 in five divided daily doses) | Ovarian
carcinoma | Retinal toxicity in
the form of cone dysfunction was documented by ERG and color vision
testing | 2–4 cycles | Reversible | (5) |
| Kupersmith et
al | ND | 3 | Maculopathy (severe
macular retinal pigment abnormality) | Cisplatin
(intra-arterially administered), carmustine | Malignant
gliomas | Localized retinal
pigment disturbance in the macula | ND | ND | (6) |
| Khawly et
al | ND | 8 | Cotton-wool spots,
intraretinal hemorrhages and macular exudate | Cisplatin,
cyclophosphamide, carmustine and autologous bone marrow
transplantation | Breast cancer | ND | 1–5 months | Reversible | (7) |
| Hilliard et
al | ND | 2 (pediatric
patients) | Symptomatic
retinopathy with abnormal ERG and VER | Cisplatin, etoposide
(both patients had abnormal renal function) | ND | Retinal toxicity
documented by VER and ERG | ND | Irreversible | (8) |
| Tan and Walsh | 65/45 | 2 | Blurred vision with
flashing lights; photopsia | Cisplatin,
paclitaxel | Lung cancer | ND | 2 cycles/1 cycle | Reversible | (9) |
| Wang et
al | 47 | 1 | Bilateral
blindness | Cisplatin,
carmustine | Breast cancer | Histopathological
examination of the eye and optic nerves at autopsy revealed nerve
fiber layer infarction secondary to right inferior temporal retinal
artery thrombosis | 1 cycle | Irreversible | (10) |
| Gonzalez et
al | ND | 1 | Acute blindness in
the left eye | Cisplatin | Lung cancer | ND | Immediately after
treatment | Irreversible | (11) |
| Watanabe et
al | 58 | 1 | Visual disturbance,
vision loss in left eye | Carboplatin
(intracarotid injection) | Glioblastoma | Diffuse chorioretinal
atrophy with optic atrophy | 30 h | Irreversible | (12) |
| Katz et
al | 55 | 1 | Bilateral visual
loss | Four times the
intended dose of intravenous cisplatin | Non-Hodgkin
lymphoma | Visual fields showed
central scotomas bilaterally; an ERG showed markedly reduced a-wave
amplitudes and absent b-waves | Immediately after
treatment | Irreversible | (13) |
| Kwan et
al | 31 | 1 | Vision loss | Cisplatin, bleomycin,
in left eye | Non-seminomatous
etoposide testicular tumor | Fluorescein
angiography germ cell retinal ischemia and left retinal
neovascularization | 10 weeks revealed
bilateral | Irreversible | (14) |
| Li et al | 56 | 1 | Bilateral
blindness | Cisplatin,
paclitaxel | Nasopharyngeal
cancer | Abnormal
visual-evoked potentials and transient, flash ERGs | 10 days | Irreversible | (2) |
| Kord Valeshabad et
al | 78 | 1 | Blurred vision | Gemcitabine,
docetaxel | Sarcoma | Uveal effusion and
outer retinal disruption | 2 cycles | Reversible | (15) |
| Tang et
al | 48 | 1 | Bilateral
blindness | Platinum,
docetaxel | Small-cell carcinoma
of the endometrium | Retinal current map
examination showed the binocular dark optic rod b-wave response and
dark optic mixed reaction b-waves; secondary retinitis pigmentosa
was diagnosed | 4 cycles | Irreversible | Present study |
Retinal toxicity includes maculopathy in the form of
pigmentary changes attributable to localized retinal pigment
disturbance, altered color perception attributable to cone
dysfunction, and mild retinal ischemic changes, such as cotton-wool
spots and intraretinal hemorrhages in the posterior pole (14).
The mechanism underlying ocular neurotoxicity
remains unknown and the ischemic and electrophysiological
hypotheses may be involved in the pathogenesis of ocular toxicity.
Cisplatin-associated neurotoxicity is dose-dependent. Severe
neurotoxicity is extremely uncommon at a cumulative dose <400
mg/m2, but the incidence increases at a cumulative dose
of 600–800 mg/m2 (2).Occlusion of a retinal artery branch,
severe macular ischemia or retinal neovascularization are
associated with high-dose platinum. Cisplatin increases human
platelet reactivity (onset of platelet aggregation wave and
thromboxane production) to non-aggregating concentrations of the
agonists involving arachidonic acid metabolism (16). In addition, a study on autopsy
specimens also identified focal small-vessel thrombosis and vessel
occlusion as the cause of blindness in a patient on high-dose
carmustine and cisplatin therapy (10).
The visual symptoms and electrophysiological changes
following intravenous paclitaxel administration were likely caused
by retinal vascular dysregulation or optic nerve ischemia (17). As the cystoid macular edema occurred
following treatment with paclitaxel, one possible theory is that
Müller cell toxicity results from intracellular fluid accumulation
and subclinical extracellular fluid leakage. Reversible uveal
effusion and outer retinal disruption were reported following
gemcitabine and docetaxel chemotherapy (15).
Bakbak et al (18) reported that systemic administration
of cisplatin and paclitaxel affected the peripapillary retinal
nerve fibre layer thickness and visual field index, as revealed by
frequency-doubling technology (FDT) perimetry. Optical coherence
tomography and FDT perimetry may be adjunctive tools for the
screening of ocular toxicity in patients treated with these
agents.
It has been reported that children and those
patients with renal dysfunc-tion or diabetes mellitus appear to be
the highest risk groups for cisplatin-related neurotoxicity
(19,20). Patients with diabetic complications
may opt to avoid paclitaxel plus platinum combination therapies if
there are alternative effective treatment options available
(20). Unfortunately, our patient
suffered from diabetes and developed an apparent accelerated
decline in visual function during conventional adjuvant
chemotherapy; an alternative regimen of adjuvant radiotherapy was
not applied.
Due to its rarity, retinal toxicity may be
underes-timated or considered as a minor complication when compared
with other life-threatening complications. Understanding the ocular
side effects of chemotherapy may assist ophthalmologists and
oncologists with early identification and timely intervention,
before blindness becomes established.
References
|
1
|
Omoti AE and Omoti CE: Ocular toxicity of
systemic anticancer chemotherapy. Pharm Pract (Granada). 4:55–59.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Li Y, Li Y, Li J, Pi G and Tan W:
Paclitaxel- and/or cisplatin-induced ocular neurotoxicity: A case
report and literature review. Onco Targets Ther. 7:1361–1366.
2014.PubMed/NCBI
|
|
3
|
Uterine Neoplasms: NCCN Guidelines Version
2 MS. 19:2016.
|
|
4
|
Berman IJ and Mann MP: Seizures and
transient cortical blindness associ-ated with cisplatinum (II)
diamminedichloride (PDD) therapy in a thirty-year-old man. Cancer.
45:764–766. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Wilding G, Caruso R, Lawrence TS, Ostchega
Y, Ballintine EJ, Young RC and Ozols RF: Retinal toxicity after
high dose cisplatin therapy. J Clin Oncol. 3:1683–1689. 1985.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Kupersmith MJ, Seiple WH, Holopigian K,
Noble K, Hiesiger E and Warren F: Maculopathy caused by
intra-arterially administered cisplatin and intravenously
administered carmustine. Am J Ophthalmol. 113:435–438. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Khawly JA, Rubin P, Petros W, Peters WP
and Jaffe GJ: Retinopathy and optic neuropathy in bone marrow
transplantation for breast cancer. Ophthalmology. 103:87–95. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Hilliard LM, Berkow RL, Watterson J,
Ballard EA, Balzer GK and Moertel CL: Retinal toxicity associated
with cisplatin and etoposide in pediatric patients. Med Pediatr
Oncol. 28:310–313. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Tan WW and Walsh T: Ocular toxicity
secondary to paclitaxel in two lung cancer patients. Med Pediatr
Oncol. 31:1771998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wang MY, Arnold AC, Vinters HV and Glasgow
BJ: Bilateral blindness and lumbosacral myelopathy associated with
high-dose carmustine and cisplatin therapy. Am J Ophthalmol.
130:367–368. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Gonzalez F, Menendez D and Gomez-Ulla F:
Monocular visual loss in a patient undergoing cisplatin
chemotherapy. Int Ophthalmol. 24:301–304. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Watanabe W, Kuwabara R, Nakahara T,
Hamasaki O, Sakamoto I, Okada K, Minamoto A and Mishima HK: Severe
ocular and orbital toxicity after intracarotid injection of
carboplatin for recurrent glioblastomas. Graefes Arch Clin Exp
Ophthalmol. 240:1033–1035. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Katz BJ, Ward JH, Digre KB, Creel DJ and
Mamalis N: Persistent severe visual and electroretinographic
abnormalities after intravenous Cisplatin therapy. J
Neuroophthalmol. 23:132–135. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kwan AS, Sahu A and Palexes G: Retinal
ischemia with neovascularization in cisplatin related retinal
toxicity. Am J Ophthalmol. 141:196–197. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Valeshabad A Kord, Mieler WF, Setlur V,
Thomas M and Shahidi M: Posterior segment toxicity after
gemcitabine and docetaxel chemotherapy. Optom Vis Sci.
92:e110–e113. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Togna GI, Togna AR, Franconi M and Caprino
L: Cisplatin triggers platelet activation. Thromb Res. 99:503–509.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Scaioli V, Caraceni A, Martini C, Curzi S,
Capri G and Luca G: Electrophysiological evaluation of visual
pathways in paclitaxel-treated patients. J Neurooncol. 77:79–87.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Bakbak B, Gedik S, Koktekir BE, Yavuzer K,
Tulek B, Kanat F and Pancar E: Assessment of ocular neurotoxicity
in patients treated with systemic cancer chemotherapeutics. Cutan
Ocul Toxicol. 33:7–10. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Walsh TJ, Clark AW, Parhad IM and Green
WR: Neurotoxic effects of cisplatin therapy. Arch Neurol.
39:719–720. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hershman DL, Till C, Wright JD, Awad D,
Ramsey SD, Barlow WE, Minasian LM and Unger J: Comorbidities and
risk of chemotherapy-induced peripheral neuropathy among
participants 65 years or older in southwest oncology group clinical
trials. J Clin Oncol. 34:3014–3022. 2016. View Article : Google Scholar : PubMed/NCBI
|