Introduction
The incidence of convulsive seizures following
surgery for non-traumatic supratentorial brain lesions is estimated
to range between 15 and 20% (1).
However, depending on the underlying pathology, seizure risk shows
marked variations, ranging from 3 to 92% (2). Postoperative seizures generally
manifest in the first month after surgery and may herald the onset
of refractory epilepsy (3,4).
The prophylactic use of antiseizure medication (ASM)
in patients undergoing neurosurgical procedures is still being
debated (4). Randomized controlled
trials have yielded conflicting results; certain trials have
indicated a significant reduction in the incidence of early
postoperative seizures due to ASM prophylaxis (3,5),
while others do not agree (6).
Around 35-70% of patients with brain tumor suffer from seizures
(7,8). Perioperative levetiracetam (LEV)
appears to be effective in treating these patients; however, the
evidence regarding perioperative prophylaxis remains conflicting
(9-11).
LEV binds to synaptic vesicle glycoprotein 2A to modulate
presynaptic neurotransmitter release (12) and inhibits presynaptic
Ca2+ channels to reduce neuronal excitability (12,13).
Though LEV is a relatively safe and effective ASM with fewer side
effects than carbamazepine, phenytoin and valproic acid, its side
effects include somnolence, asthenia, mood disorder and behavioral
disturbance (14), which cannot be
tolerated by certain patients. In addition, LEV monotherapy does
not adequately control seizures in all patients.
Perampanel (PER) is a novel antiepileptic agent that
selectively and non-competitively inhibits
α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA)-type
glutamate receptors present on the postsynaptic membrane. It can
suppress neuronal excitability irrespective of the glutamate
concentration in the synaptic cleft (15). PER was approved in Europe and the
US in 2012, while in Japan, in 2016, it was approved for use in
combination with other ASMs for treatment-resistant focal seizures
(including those with secondary generalization) and tonic-clonic
seizures, and in 2020, it was approved in Japan as monotherapy for
treating focal seizures (including those with secondary
generalization) (16,17).
Several studies have reported the use of PER in
patients with brain tumors (18,19).
These reports have demonstrated the efficacy of PER combined with
other ASM in controlling seizures. Only one study has examined PER
monotherapy, and has demonstrated its efficacy in preventing early
seizures in patients with supratentorial brain tumors undergoing
craniotomy (20). The present
study aimed to further investigate PER monotherapy in patients with
brain tumor and determine its safety and effectiveness.
Materials and methods
Patients and study design
This retrospective case-control study analyzed the
cases of 25 patients with brain tumor who underwent surgery and
received PER monotherapy at Keio University Hospital, (Tokyo,
Japan) between April 2020 and September 2022. Furthermore, 45
patients with brain tumor who underwent surgery and received LEV
monotherapy from April 2018 to October 2018 were used as controls.
All of the patients in both the PER and LEV groups received
standard clinical treatment. In the PER group, the patients were
prescribed an initial dose of 2 mg/day of PER. The daily dosage was
increased by 2 mg at 2-week intervals based on each patient's
clinical response and tolerance, as per the label instructions. In
the LEV group, the patients were initially prescribed a dosage of
1,000 mg/day of LEV, and the dosage was subsequently increased by
1,000 mg/day until the epileptic seizures were resolved.
The present study was approved by the Keio
University School of Medicine Ethics Committee (Tokyo, Japan;
approval no., 20050002). At the time of surgery, all patients
provided written consent for the possibility of subsequent
retrospective review of clinical and other outcomes. If the patient
was a minor or was incapable of making decisions due to the effects
of a brain tumor, the next of kin signed the form.
Data collection
Clinical data, including age, sex, diagnosis, date
of surgery, date of initiation of ASM, details of seizures before
initiation of treatment, details of seizures after initiating
administration, side effects and dosage were obtained from the
medical records. Inclusion criteria were as follows: Patients who
had undergone craniotomy for tumor resection or a biopsy along with
a histopathological diagnosis; and had received either PER or LEV
during the above-mentioned period. Patients with a previous history
of epilepsy were also included, since patients who had surgery at
our hospital during the relevant period were included, regardless
of their previous epilepsy history. Patients were excluded if they
were receiving concomitant therapy with other antiepileptic drugs
or if any essential variables were missing from the records. Tumors
were graded in accordance with the World Health Organization
classification (21). Adverse
events were graded in accordance with the Common Terminology
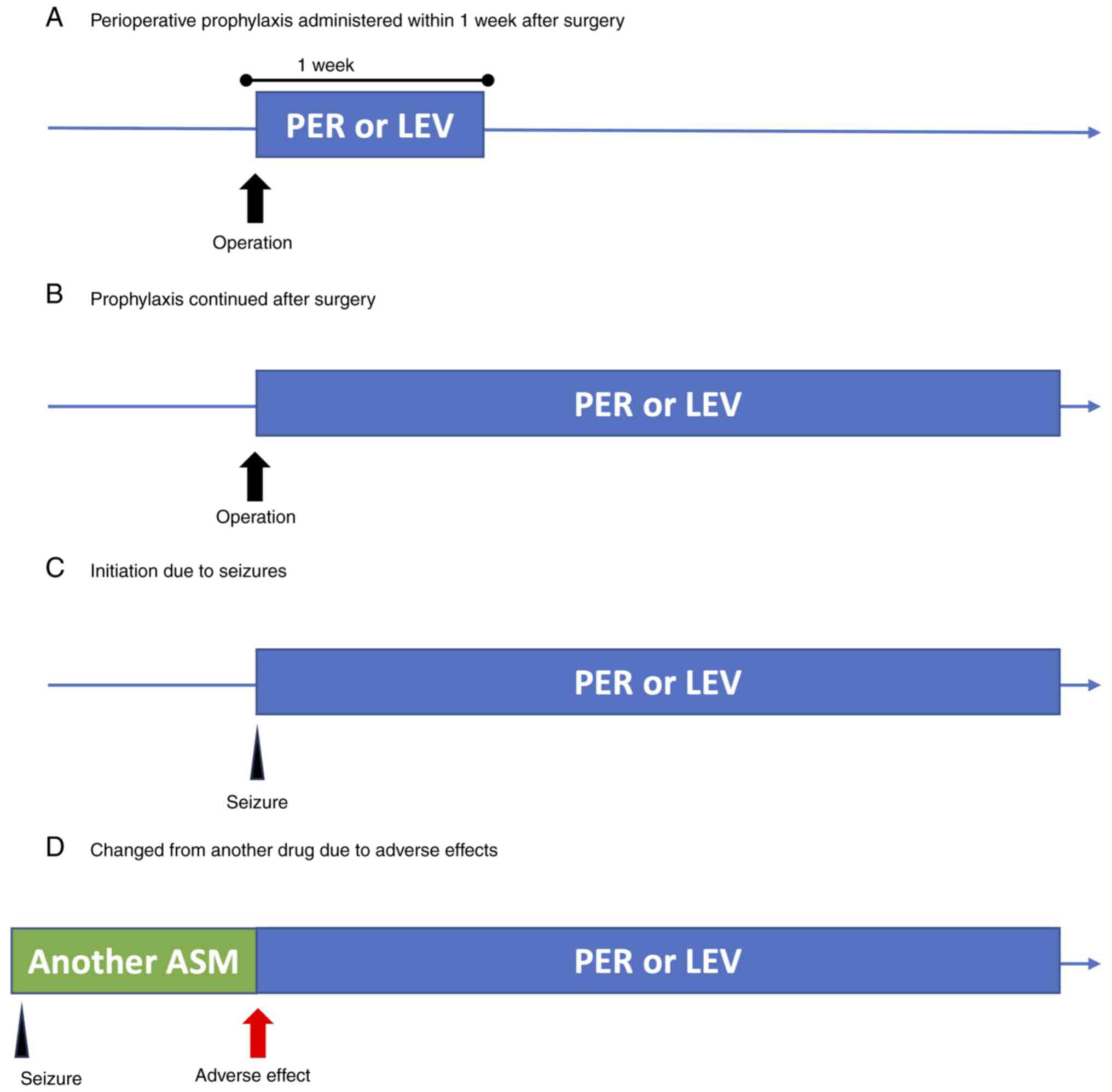
Criteria for Adverse Events, version 5.0(22). The ASM administration patterns were
categorized as follows: Perioperative prophylaxis administered
within one week post-surgery, perioperative prophylaxis
administered for more than one week, initiation of ASM due to
seizures, switch from another ASM due to adverse events and others
(Fig. 1). Epileptic seizures were
defined in accordance with the International League Against
Epilepsy 2017 seizure classification (23).
Statistical analysis
Fisher's exact test or the Mann-Whitney U-test were
used as appropriate for comparison of the clinical and demographic
data. Rates of seizures and adverse events between the PER and LEV
groups were compared using Fisher's exact test. P<0.05 was
considered significant.
Compliance with standards
The present study followed The Strengthening the
Reporting of Observational Studies in Epidemiology checklist
(24).
Results
Patient characteristics
The 25 patients in the PER group included 11 males
and 14 females. The mean age at initiation of PER therapy was 46.2
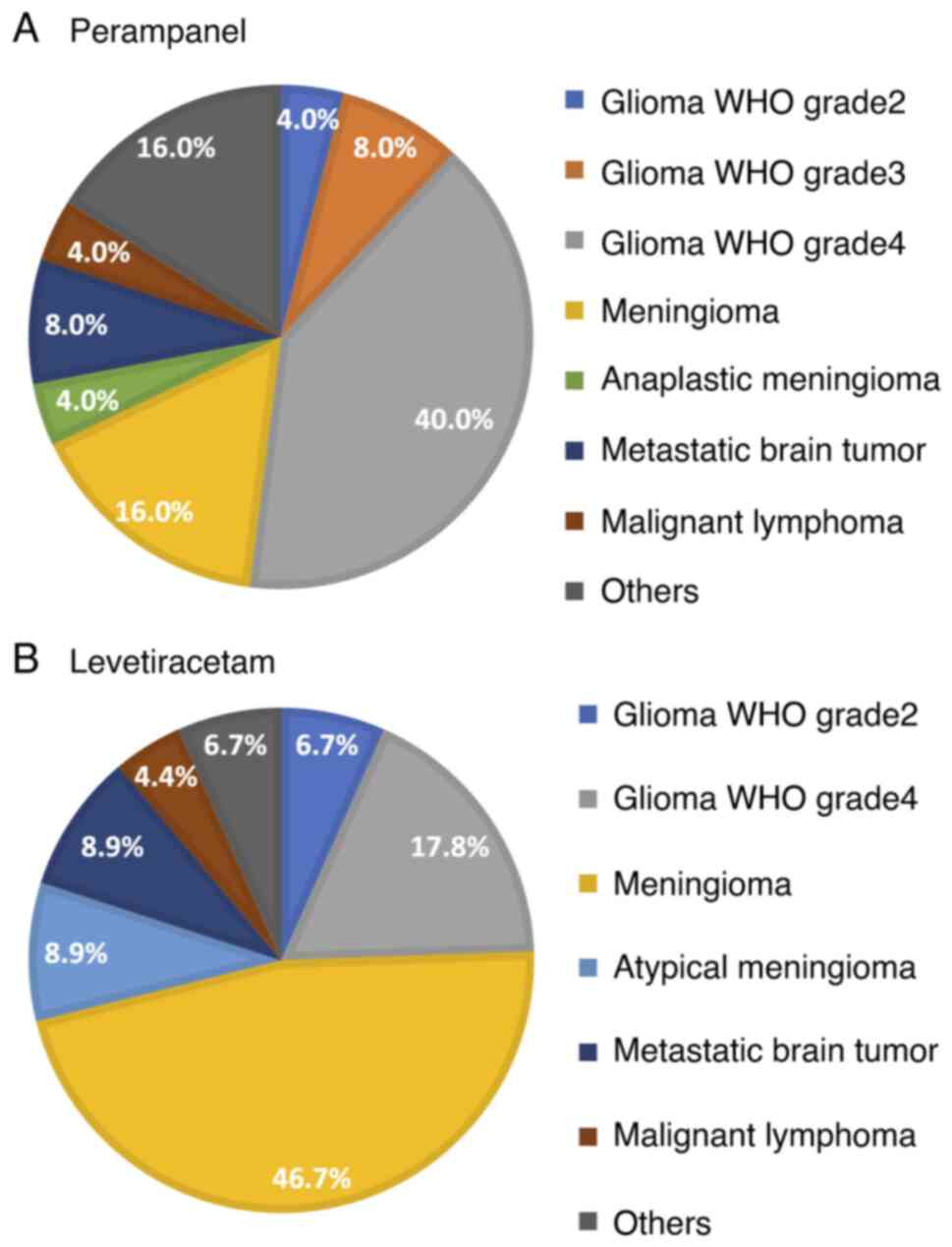
years (range, 17-83 years). The different types of brain tumor were
as follows: Glioma [grade 4 (10 patients, 40.0%); grade 3 (two
patients, 8.0%); grade 2 (one patient (4.0%)]; meningioma [grade 3
(one patient, 4.0%); grade 1 (four patients, 16.0%)]; malignant
lymphoma (one patient, 4.0%); metastatic brain tumor (two patients,
8.0%); and other tumor types (four patients, 16.0%). The tumor
distribution in the PER group is depicted in Fig. 2A. Glioma was the most prevalent
tumor type (13 patients, 52.0%), with the majority of patients (10
patients, 77%) having grade 4 glioma.
Among the 45 patients in the LEV group, 23 were
males and 22 were females. The mean age at initiation of LEV
therapy was 60.6 years (range, 21-85 years). The following brain
tumor types were present in this group: Glioma [grade 4 (eight
patients, 17.8%); grade 2 (three patients, 6.7%)]; meningioma
[grade 2 (four patients, 8.9%); grade 1 (21 patients, 46.7%)];
malignant lymphoma (two patients, 4.4%); metastatic brain tumor
(four patients, 8.9%); and other tumors (three patients, 6.7%). The
tumor distribution in the LEV is depicted in Fig. 2B.
While the PER and LEV groups differed significantly
with respect to age and tumor type, the differences with regard to
sex were not significant. The mean age was significantly lower and
gliomas were significantly more frequent in the PER group (Table I).
 | Table IPatients' characteristics. |
Table I
Patients' characteristics.
| Item | PER (n=25) | LEV (n=45) | P-value |
|---|
| Female sex | 14 (56.0) | 22 (48.9) | 0.568 |
| Mean age, years
(range) | 46.2 (17-83) | 60.6 (21-85) | 0.002 |
| Type of tumor | | | 0.017 |
|
Glioma WHO
grade 2 | 1 (4.0) | 3 (6.7) | |
|
Glioma WHO
grade 3 | 2 (8.0) | 0 (0) | |
|
Glioma WHO
grade 4 | 10 (40.0) | 8 (17.8) | |
|
Meningioma | 4 (16.0) | 21 (46.7) | |
|
Atypical
meningioma | 0 (0) | 4 (8.9) | |
|
Anaplastic
meningioma | 1 (4.0) | 0 (0) | |
|
Metastatic
brain tumor | 2 (8.0) | 4 (8.9) | |
|
Malignant
lymphoma | 1 (4.0) | 2 (4.4) | |
|
Others | 4 (16.0) | 3 (6.7) | |
| Antiseizure
medication administration pattern | | | 0.010 |
|
Prophylactic
use within 1 week after surgery | 3(12) | 17 (37.8) | |
|
Prophylaxis
continued after surgery | 12(48) | 14 (31.1) | |
|
Initiation
due to seizures | 6(24) | 14 (31.1) | |
|
Changed from
another drug due to adverse effects | 2(8) | 0 | |
|
Others | 2(8) | 0 | |
ASM administration patterns
Patients were classified based on the PER/LEV
administration pattern (Fig. 1) as
follows: Perioperative prophylaxis administered for one week
following surgery, perioperative prophylaxis administered for more
than one week, initiation prompted by seizure/s, switch from
another ASM due to adverse events and others. In the PER group, the
number of patients in each category was 3 (12%), 12 (48%), 6 (24%),
2 (8%) and 2 (8%), respectively. Six of the 12 patients (50%) who
received prophylactic PER for more than one week had a grade 3 or 4
glioma (data not shown). In the LEV group, 17 patients had received
perioperative prophylaxis for one week, 14 received prophylaxis for
more than one week and 14 had received LEV because of seizures
(Table I).
Dosage
PER was initiated at 2 mg/day; the daily dose was
increased in 2-mg increments at 2-week intervals if needed, based
on clinical response and tolerability and in accordance with the
label instructions. In patients receiving PER for seizure
treatment, the dose was increased until the cessation of seizures.
Prophylactic PER was administered at maintenance doses of 2 or 4 mg
based on tolerability. The mean dose administered was 3.12 mg/day
(range, 2-8). In two patients, the dosage had to be reduced because
of side effects. LEV was initiated at 1,000 mg/day, and when used
as a treatment regime, the dose was increased by 1,000 mg/day until
resolution of epileptic seizures. The mean dose was 1,066.7 mg/day
(range, 1,000-2,000) (Table
II).
 | Table IIOutcomes regarding dose, seizures and
adverse effects. |
Table II
Outcomes regarding dose, seizures and
adverse effects.
| Item | PER (n=25) | LEV (n=45) | P-value |
|---|
| Mean dosage, mg/day
(range) | 3.12 (2-8) | 1066.7
(1,000-2,000) | |
| Seizure, n (%) | 2(8) | 5 (11.1) | 1.000 |
| Adverse effect, n
(%) | 3(12) | 1 (2.2) | 0.127 |
|
Liver
dysfunction | 2(8) | 0 (0) | 0.124 |
|
Drug
rash | 1(4) | 1 (2.2) | 1.000 |
Seizure control and adverse
events
Two patients in the PER group and five in the LEV
group suffered from seizures during ASM administration (8.0 and
11.1%, respectively; P=1.000) (Table
II). Increasing the dose or adding another ASM successfully
controlled the seizures in all patients in each group. Both
patients from the PER group who experienced seizures had focal
seizures and had a history of seizures prior to PER administration.
One of the patients had a grade 4 glioma and the other had a grade
1 meningioma. None of the patients who received PER for prophylaxis
experienced seizures.
All five patients in the LEV group with seizures
also experienced focal seizures. Of these, three patients had a
history of seizures and two patients experienced seizures while on
prophylactic LEV. Tumor pathology in these five patients was as
follows: One patient had a grade 4 glioma, two patients had grade 1
meningioma and the remaining two had other types.
A total of three patients in the PER group (12.0%)
taking PER at 2 mg/day experienced adverse events, with two
patients suffering from grade 3 liver dysfunction and one patient
having a grade 2 drug rash. In these patients, PER was either
discontinued or they were switched to another ASM. In the LEV
group, only one patient (2.2%) taking 1,000 mg per day experienced
an adverse event (drug rash, grade was not documented). The PER and
LEV groups did not differ significantly with regard to incidence of
adverse events (P=0.127) (Table
II).
Discussion
In the present study, the results of PER monotherapy
in patients with brain tumor were summarized and the results were
compared with those of LEV monotherapy. Both groups were similar
with respect to the rates of seizures and adverse events. Adverse
events were observed in 12% of patients in the PER group, which is
lower than previously reported rates for PER monotherapy
(20.0-45.9%) (25-28).
Since in the present study, ASM was mostly used in both groups in
the perioperative period, one of the many other drugs administered
during this period may have been the underlying cause of the
adverse events instead of PER or LEV. Reported incidence rates of
adverse events that result in discontinuation of PER monotherapy
range from 6.7 to 16.3% (16),
which is similar to that observed in the present study (12%).
Approximately 20% of patients undergoing craniotomy
for a supratentorial non-traumatic lesion experience seizures
(1). Several randomized controlled
trials and systematic reviews have reported that ASM prophylaxis
does not result in a significant reduction in postoperative
seizures in these patients (6,10,29).
However, a meta-analysis that specifically included patients
undergoing craniotomy for brain tumors has reported a significant
reduction in early postoperative seizures caused by prophylactic
ASM (30). While recent surgical
techniques, such as awake surgery, can induce intraoperative
seizures (31), the efficacy of
prophylactic administration for such seizures has not yet been
adequately debated.
Although the efficacy of prophylactic ASMs in the
perioperative period remains a debatable topic (29), they are still frequently used
(32). In our institution,
prophylactic ASM is routinely used in the first week after
craniotomy, even in patients who have not suffered preoperative
seizures. One study has reported that a combination of PER and LEV
in patients undergoing awake surgery for glioma is associated with
a significantly lower risk of intraoperative seizures compared with
LEV monotherapy (33).
Prophylactic administration of PER during the first six days
following a brain tumor surgery may be associated with a similar
seizure prevention rate and safety profile to that of LEV (20). Only 15 patients in the present
study received PER as perioperative prophylaxis and none
experienced postoperative seizures. Also, 12 of them continued PER
monotherapy beyond one week and six of these patients suffered from
grade 3 or 4 gliomas. Seizure-free patients with grade 3 or 4
gliomas at the time of initial surgery may experience tumor
recurrence, and recurrence can further be associated with new
seizures (34). Malignant tumors
remain incurable even when combinations of various treatments are
used, and although substantial basic research has been conducted
(35,36), only a limited number of treatments
have actually been put to practical use. Drug repositioning, which
has been attracting attention in recent years, is a method of
discovering new drug effects from existing drugs with proven safety
and pharmacokinetics in humans (37). In addition, PER has been observed
to inhibit glioma cell growth in vitro (38). In a phase II trial, talampanel, an
AMPA receptor inhibitor, was observed to extend median survival in
patients with glioblastoma treated with radiation and temozolomide
(39). Salmaggi et al
(40) demonstrated that PER in
combination with temozolomide exerts a synergistic effect in
promoting apoptosis in human glioblastoma cell lines. A phase II
trial to assess the efficacy and tolerability of PER in recurrent
glioblastoma is ongoing (41).
Numerous studies have demonstrated the safety and efficacy of PER
in suppressing seizures in patients with brain tumors (42), particularly when used in
combination with other ASMs.
The expression of GRIA2 and GRIA4,
which encode separate subunits of the AMPA receptor, is upregulated
in certain brain tumors besides gliomas (43,44).
AMPA receptor inhibitors exhibit antitumor effects in small-cell
lung cancer, but not in the brain microenvironment (45). GRIA2 upregulation has also
been detected in melanoma brain metastases (46). Hence, the AMPA receptor may be an
effective therapeutic target for brain tumors, leading to the drug
repositioning strategy.
The present study indicates the safety and efficacy
of PER monotherapy in patients with brain tumor. Although the
evidence supporting prophylactic ASM administration in patients
with brain tumor who are seizure-free is limited, PER can be
considered if the side effects are tolerable. PER monotherapy may
be preferable to combination therapy for prophylactic treatment, in
terms of side effects. Currently, the most specific drug option via
drug repositioning for refractory brain tumor remains elusive. If
the side effects of the drug repositioning strategy using PER are
less severe than those of chemotherapy and molecular targeted
therapy, PER can represent a meaningful option in terms of both
seizure control and tumor control, which may lead to an improvement
in symptoms and enhanced quality of life for patients with brain
tumors. Further studies on the effects of PER on seizures and tumor
growth are warranted.
This study has several limitations. The
retrospective design and small sample size may have resulted in a
selection bias, which may affect the stability and generalizability
of the results. Furthermore, patient age and tumor type were not
homogeneous between the PER and LEV groups, which may have
introduced certain inaccuracies in the results. Due to the small
number of cases and the variety of tumors included, classification
by brain tumor grade did not allow for a statistically powerful
analysis. Detailed information regarding the usage of analgesic and
antiemetic medications during the perioperative period may also be
necessary for further discussion regarding adverse events.
Comprehensive and in-depth comparisons of serum biological
indicators and electroencephalograms are warranted for obtaining
more convincing results. Although gliomas are the most common type
of brain tumor causing epilepsy (47,48)
and the glioma prevalence was much higher in the PER group, the
seizure rates in the PER and LEV groups were comparable. A
difference between the groups would have been detectable if the
groups had been more homogeneous. Various types of tumor were
included in this analysis, and thus, factors such as the extent of
tumor resection, which are affected by the tumor type, were not
included because they were not standardized. In future analyses, we
would like to standardize the tumor types and include other
variables, such as the extent of tumor resection.
In conclusion, PER monotherapy may be safe and
effective for seizure treatment or prophylaxis in patients with
brain tumor. Recent studies have revealed that PER may also have
antitumor effects, which could potentially make it an effective
treatment option for epilepsy related to brain tumors. Further
accumulation of case studies is necessary to confirm its efficacy
in the future.
Acknowledgements
Not applicable.
Funding
Funding: No funding was received.
Availability of data and materials
The data generated in the present study may be
requested from the corresponding author.
Authors' contributions
UH and RT conceptualized, designed and performed the
study and wrote the manuscript. TE, KKa, KY and AT assisted in the
acquisition of data. The authenticity of the raw data was checked
and confirmed by UH and RT. KKo and MT assisted with the discussion
and review of the manuscript. All authors have read and approved
the final version.
Ethics approval and consent to
participate
The studies involving humans have been approved by
the Keio University School of Medicine Ethics Committee (Tokyo,
Japan; approval no. 20050002). The patients or their next of kin
provided written informed consent to participate in the study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Foy PM, Copeland GP and Shaw MD: The
incidence of postoperative seizures. Acta Neurochir (Wien).
55:253–264. 1981.PubMed/NCBI View Article : Google Scholar
|
|
2
|
Manaka S, Ishijima B and Mayanagi Y:
Postoperative seizures: Epidemiology, pathology, and prophylaxis.
Neurol Med Chir (Tokyo). 43:589–600; discussion 600.
2003.PubMed/NCBI View Article : Google Scholar
|
|
3
|
North JB, Penhall RK, Hanieh A, Frewin DB
and Taylor WB: Phenytoin and postoperative epilepsy. A double-blind
study. J Neurosurg. 58:672–677. 1983.PubMed/NCBI View Article : Google Scholar
|
|
4
|
Temkin NR: Prophylactic anticonvulsants
after neurosurgery. Epilepsy Curr. 2:105–107. 2002.PubMed/NCBI View Article : Google Scholar
|
|
5
|
Lee ST, Lui TN, Chang CN, Cheng WC, Wang
DJ, Heimburger RF and Lin CG: Prophylactic anticonvulsants for
prevention of immediate and early postcraniotomy seizures. Surg
Neurol. 31:361–364. 1989.PubMed/NCBI View Article : Google Scholar
|
|
6
|
Wu AS, Trinh VT, Suki D, Graham S, Forman
A, Weinberg JS, McCutcheon IE, Prabhu SS, Heimberger AB, Sawaya R,
et al: A prospective randomized trial of perioperative seizure
prophylaxis in patients with intraparenchymal brain tumors. J
Neurosurg. 118:873–883. 2013.PubMed/NCBI View Article : Google Scholar
|
|
7
|
Rudà R, Bello L, Duffau H and Soffietti R:
Seizures in low-grade gliomas: Natural history, pathogenesis, and
outcome after treatments. Neuro Oncol. 14 (Suppl 4):iv55–iv64.
2012.PubMed/NCBI View Article : Google Scholar
|
|
8
|
Vecht CJ and Wilms EB: Seizures in low-
and high-grade gliomas: Current management and future outlook.
Expert Rev Anticancer Ther. 10:663–669. 2010.PubMed/NCBI View Article : Google Scholar
|
|
9
|
Walbert T, Harrison RA, Schiff D, Avila
EK, Chen M, Kandula P, Lee JW, Le Rhun E, Stevens GHJ, Vogelbaum
MA, et al: SNO and EANO practice guideline update: Anticonvulsant
prophylaxis in patients with newly diagnosed brain tumors. Neuro
Oncol. 23:1835–1844. 2021.PubMed/NCBI View Article : Google Scholar
|
|
10
|
Iuchi T, Kuwabara K, Matsumoto M, Kawasaki
K, Hasegawa Y and Sakaida T: Levetiracetam versus phenytoin for
seizure prophylaxis during and early after craniotomy for brain
tumours: A phase II prospective, randomised study. J Neurol
Neurosurg Psychiatry. 86:1158–1162. 2015.PubMed/NCBI View Article : Google Scholar
|
|
11
|
Konrath E, Marhold F, Kindler W, Scheichel
F, Popadic B, Blauensteiner K, Calabek B, Freydl E, Weber M, Ristl
R, et al: Perioperative levetiracetam for seizure prophylaxis in
seizure-naive brain tumor patients with focus on neurocognitive
functioning. BMC Neurol. 22(250)2022.PubMed/NCBI View Article : Google Scholar
|
|
12
|
Dircio-Bautista M, Colín-González AL,
Aguilera G, Maya-López M, Villeda-Hernández J, Galván-Arzate S,
García E, Túnez I and Santamaría A: The antiepileptic drug
levetiracetam protects against quinolinic acid-induced toxicity in
the rat striatum. Neurotox Res. 33:837–845. 2018.PubMed/NCBI View Article : Google Scholar
|
|
13
|
Vogl C, Mochida S, Wolff C, Whalley BJ and
Stephens GJ: The synaptic vesicle glycoprotein 2A ligand
levetiracetam inhibits presynaptic Ca2+ channels through an
intracellular pathway. Mol Pharmacol. 82:199–208. 2012.PubMed/NCBI View Article : Google Scholar
|
|
14
|
Fuller KL, Wang YY, Cook MJ, Murphy MA and
D'Souza WJ: Tolerability, safety, and side effects of levetiracetam
versus phenytoin in intravenous and total prophylactic regimen
among craniotomy patients: A prospective randomized study.
Epilepsia. 54:45–57. 2013.PubMed/NCBI View Article : Google Scholar
|
|
15
|
Hanada T, Hashizume Y, Tokuhara N,
Takenaka O, Kohmura N, Ogasawara A, Hatakeyama S, Ohgoh M, Ueno M
and Nishizawa Y: Perampanel: A novel, orally active, noncompetitive
AMPA-receptor antagonist that reduces seizure activity in rodent
models of epilepsy. Epilepsia. 52:1331–1340. 2011.PubMed/NCBI View Article : Google Scholar
|
|
16
|
Yamamoto T, Gil-Nagel A, Wheless JW, Kim
JH and Wechsler RT: Perampanel monotherapy for the treatment of
epilepsy: Clinical trial and real-world evidence. Epilepsy Behav.
136(108885)2022.PubMed/NCBI View Article : Google Scholar
|
|
17
|
Eisai News Release. Approval of
antiepileptic drug Fycompa® in Japan for monotherapy and
pediatric indications for partial-onset seizures, as well as a new
formulation. Available at: https://www.eisai.com/news/2020/news202004.html.
Accessed April 12, 2024.
|
|
18
|
Heugenhauser J, Iglseder S, Muigg A,
Kerschbaumer J, Stockhammer G, Nowosielski M and Unterberger I:
Perampanel in brain tumor and SMART-syndrome related epilepsy-A
single institutional experience. J Neurol Sci.
423(117386)2021.PubMed/NCBI View Article : Google Scholar
|
|
19
|
Maschio M, Zarabla A, Maialetti A,
Giannarelli D, Koudriavtseva T, Villani V and Zannino S: Perampanel
in brain tumor-related epilepsy: Observational pilot study. Brain
Behav. 10(e01612)2020.PubMed/NCBI View Article : Google Scholar
|
|
20
|
Kusakabe K, Inoue A, Watanabe H, Nakamura
Y, Nishikawa M, Ohtsuka Y, Ogura M, Shigekawa S, Taniwaki M,
Kitazawa R and Kunieda T: Perioperative perampanel administration
for early seizure prophylaxis in brain tumor patients. Surg Neurol
Int. 14(287)2023.PubMed/NCBI View Article : Google Scholar
|
|
21
|
WHO Classification of Tumours Editorial
Board. World Health Organization Classification of Tumours of the
Central Nervous System. 5th edition. International Agency for
Research on Cancer, 2021.
|
|
22
|
Common Terminology Criteria for Adverse
Events (CTCAE). Version 5. Published: November 27. US Department of
Health and Human Services, National Institutes of Health, National
Cancer Institute, 2017.
|
|
23
|
Fisher RS, Cross JH, D'Souza C, French JA,
Haut SR, Higurashi N, Hirsch E, Jansen FE, Lagae L, Moshé SL, et
al: Instruction manual for the ILAE 2017 operational classification
of seizure types. Epilepsia. 58:531–542. 2017.PubMed/NCBI View Article : Google Scholar
|
|
24
|
von Elm E, Altman DG, Egger M, Pocock SJ,
Gøtzsche PC and Vandenbroucke JP: STROBE Initiative. The
strengthening the reporting of observational studies in
epidemiology (STROBE) Statement: Guidelines for reporting
observational studies. BMJ. 335:806–808. 2007.PubMed/NCBI View Article : Google Scholar
|
|
25
|
Toledano Delgado R, García-Morales I,
Parejo-Carbonell B, Jiménez-Huete A, Herrera-Ramirez D,
González-Hernández A, Ayuga Loro F, Santamarina E, Toledo M, Ojeda
J, et al: Effectiveness and safety of perampanel monotherapy for
focal and generalized tonic-clonic seizures: Experience from a
national multicenter registry. Epilepsia. 61:1109–1119.
2020.PubMed/NCBI View Article : Google Scholar
|
|
26
|
Chinvarun Y: A retrospective, real-world
experience of perampanel monotherapy in patient with first new
onset focal seizure: A Thailand experience. Epilepsia Open.
7:67–74. 2022.PubMed/NCBI View Article : Google Scholar
|
|
27
|
Gil-Nagel A, Burd S, Toledo M, Sander JW,
Lebedeva A, Patten A and Laurenza A: Study 504 investigator group:
A retrospective, multicentre study of perampanel given as
monotherapy in routine clinical care in people with epilepsy.
Seizure. 54:61–66. 2018.PubMed/NCBI View Article : Google Scholar
|
|
28
|
Wechsler RT, Wheless J, Zafar M, Huesmann
GR, Lancman M, Segal E, Chez M, Aboumatar S, Patten A, Salah A and
Malhotra M: PROVE: Retrospective, non-interventional, phase IV
study of perampanel in real-world clinical care of patients with
epilepsy. Epilepsia Open. 7:293–305. 2022.PubMed/NCBI View Article : Google Scholar
|
|
29
|
Greenhalgh J, Weston J, Dundar Y, Nevitt
SJ and Marson AG: Antiepileptic drugs as prophylaxis for
postcraniotomy seizures. Cochrane Database Syst Rev.
5(CD007286)2020.PubMed/NCBI View Article : Google Scholar
|
|
30
|
Joiner EF, Youngerman BE, Hudson TS, Yang
J, Welch MR, McKhann GM, Neugut AI and Bruce JN: Effectiveness of
perioperative antiepileptic drug prophylaxis for early and late
seizures following oncologic neurosurgery: A meta-analysis. J
Neurosurg. 130:1274–1282. 2018.PubMed/NCBI View Article : Google Scholar
|
|
31
|
Nossek E, Matot I, Shahar T, Barzilai O,
Rapoport Y, Gonen T, Sela G, Korn A, Hayat D and Ram Z: Failed
awake craniotomy: A retrospective analysis in 424 patients
undergoing craniotomy for brain tumor. J Neurosurg. 118:243–249.
2013.PubMed/NCBI View Article : Google Scholar
|
|
32
|
Dewan MC, Thompson RC, Kalkanis SN, Barker
FG II and Hadjipanayis CG: Prophylactic antiepileptic drug
administration following brain tumor resection: Results of a recent
AANS/CNS section on tumors survey. J Neurosurg. 126:1772–1778.
2017.PubMed/NCBI View Article : Google Scholar
|
|
33
|
Motomura K, Chalise L, Shimizu H,
Yamaguchi J, Nishikawa T, Ohka F, Aoki K, Tanahashi K, Hirano M,
Wakabayashi T and Natsume A: Intraoperative seizure outcome of
levetiracetam combined with perampanel therapy in patients with
glioma undergoing awake brain surgery. J Neurosurg. 135:998–1007.
2021.PubMed/NCBI View Article : Google Scholar
|
|
34
|
Hildebrand J, Lecaille C, Perennes J and
Delattre JY: Epileptic seizures during follow-up of patients
treated for primary brain tumors. Neurology. 65:212–215.
2005.PubMed/NCBI View Article : Google Scholar
|
|
35
|
Yin X, Yang J, Zhang M, Wang X, Xu W,
Price CH, Huang L, Liu W, Su H, Wang W, et al: Serum metabolic
fingerprints on bowl-shaped submicroreactor chip for chemotherapy
monitoring. ACS Nano. 16:2852–2865. 2022.PubMed/NCBI View Article : Google Scholar
|
|
36
|
Zhu H, Yang C, Yan A, Qiang W, Ruan R, Ma
K, Guan Y, Li J, Yu Q, Zheng H, et al: Tumor-targeted
nano-adjuvants to synergize photomediated immunotherapy enhanced
antitumor immunity. View. 4(20220067)2023.
|
|
37
|
Tamura R: Drug repositioning for
refractory benign tumors of the central nervous system. Int J Mol
Sci. 24(12997)2023.PubMed/NCBI View Article : Google Scholar
|
|
38
|
Venkataramani V, Tanev DI, Strahle C,
Studier-Fischer A, Fankhauser L, Kessler T, Körber C, Kardorff M,
Ratliff M, Xie R, et al: Glutamatergic synaptic input to glioma
cells drives brain tumour progression. Nature. 573:532–538.
2019.PubMed/NCBI View Article : Google Scholar
|
|
39
|
Grossman SA, Ye X, Chamberlain M,
Mikkelsen T, Batchelor T, Desideri S, Piantadosi S, Fisher J and
Fine HA: Talampanel with standard radiation and temozolomide in
patients with newly diagnosed glioblastoma: A multicenter phase II
trial. J Clin Oncol. 27:4155–4161. 2009.PubMed/NCBI View Article : Google Scholar
|
|
40
|
Salmaggi A, Corno C, Maschio M, Donzelli
S, D'Urso A, Perego P and Ciusani E: Synergistic effect of
perampanel and temozolomide in human glioma cell lines. J Pers Med.
11(390)2021.PubMed/NCBI View Article : Google Scholar
|
|
41
|
Heuer S, Burghaus I, Gose M, Kessler T,
Sahm F, Vollmuth P, Venkataramani V, Hoffmann D, Schlesner M,
Ratliff M, et al: PerSurge (NOA-30) phase II trial of perampanel
treatment around surgery in patients with progressive glioblastoma.
BMC Cancer. 24(135)2024.PubMed/NCBI View Article : Google Scholar
|
|
42
|
Tabaee Damavandi P, Pasini F, Fanella G,
Cereda GS, Mainini G, DiFrancesco JC, Trinka E and Lattanzi S:
Perampanel in brain tumor-related epilepsy: A systematic review.
Brain Sci. 13(326)2023.PubMed/NCBI View Article : Google Scholar
|
|
43
|
Vivero M, Doyle LA, Fletcher CD, Mertens F
and Hornick JL: GRIA2 is a novel diagnostic marker for solitary
fibrous tumour identified through gene expression profiling.
Histopathology. 65:71–80. 2014.PubMed/NCBI View Article : Google Scholar
|
|
44
|
Sarver AE, Sarver AL, Thayanithy V and
Subramanian S: Identification, by systematic RNA sequencing, of
novel candidate biomarkers and therapeutic targets in human soft
tissue tumors. Lab Invest. 95:1077–1088. 2015.PubMed/NCBI View Article : Google Scholar
|
|
45
|
Masumoto N, Kato S, Aichi M, Hasegawa S,
Sahara K, Suyama K, Sano A, Miyazaki T, Okudela K, Kaneko T and
Takahashi T: AMPAR receptor inhibitors suppress proliferation of
human small cell lung cancer cell lines. Thorac Cancer.
14:2897–2908. 2023.PubMed/NCBI View Article : Google Scholar
|
|
46
|
Nygaard V, Prasmickaite L, Vasiliauskaite
K, Clancy T and Hovig E: Melanoma brain colonization involves the
emergence of a brain-adaptive phenotype. Oncoscience. 1:82–94.
2014.PubMed/NCBI View Article : Google Scholar
|
|
47
|
Lynam LM, Lyons MK, Drazkowski JF, Sirven
JI, Noe KH, Zimmerman RS and Wilkens JA: Frequency of seizures in
patients with newly diagnosed brain tumors: A retrospective review.
Clin Neurol Neurosurg. 109:634–638. 2007.PubMed/NCBI View Article : Google Scholar
|
|
48
|
Riva M: Brain tumoral epilepsy: A review.
Neurol Sci. 26 (Suppl 1):S40–S42. 2005.PubMed/NCBI View Article : Google Scholar
|