Introduction
4-hydroxy-l-proline (hydroxyproline) is a
non-proteinogenic amino acid, that has a molecular weight of 131.13
g/mol and is synthesized by post-translational hydroxylation of
proline during collagen biosynthesis (Fig. 1). Investigations of physiological
and pathological collagen metabolism most commonly utilize
measurements of hydroxyproline in the plasma, urine and body
tissue. Therefore, determination of hydroxyproline provides useful
information for the diagnosis and prognosis of diseases caused by
disorders of the collagen metabolism (1). Such conditions include
hyperthyroidism, hyperparathyroidism, acromegaly, Paget’s disease,
osteomalacia, rickets, Marfan syndrome, osteogenesis imperfecta,
sclerodactyly, dermatomyositis and Cushing syndrome (2).
Fibrosis that occurs in the liver, lungs, kidneys,
skin and other organs has the capacity to develop into chronic
hepatitis, liver sclerosis, liver cancer, pulmonary fibrosis and
glomerulonephritis (3). Therefore,
preventing the development and reducing the severity of fibrosis in
patients is highly important. Hydroxyproline acts as an important
diagnostic indicator of the severity of fibrosis.
The measurement of hydroxyproline in plasma, urine
and body tissue is possible by colorimetric methods,
high-performance liquid chromatography (HPLC) and flow injection
analyses (4–6). However, these methods require large
sample volumes, due to their low sensitivity. In addition, HPLC
requires a long separation time for each sample. In recent years,
liquid chromatography-mass spectrometry (LC-MS) has been emerging
as advantageous for its high sensitivity and short separation time.
LC-MS has been utilized to measure low concentrations of drugs in
serum and urine with contaminated substances (7–9). The
present study aimed to apply the novel LC-MS method to the
measurement of hydroxyproline. As an indicator of fibrosis,
hydroxyproline in the liver and lung of a rat model of fibrosis was
measured by LC-MS, and compared with previous results obtained by
HPLC and colorimetric methods.
Materials and methods
Ethics statement
The study protocol was approved by the Ethics
Committee of Harbin Medical University (Harbin, China). A standard
procedure for obtaining written informed consent was included in
the protocol, and was approved by the Ethics Committee of Harbin
Medical University.
Reagents
Dimethylnitrosamine (DMN) and hydroxyproline were
obtained from Nacalai Tesque Inc. (Kyoto, Japan). Nembutal was
purchased from Dai-Nihon Pharmaceuticals Inc. (Osaka, Japan) and
bleomycin was obtained from Nihon Kayaku Inc. (Tokyo, Japan). All
other chemicals were of reagent grade.
Preparation of a model of pulmonary and
liver fibrosis in rats
A model of pulmonary fibrosis was created in
five-week-old Wistar rats by injection of bleomycin (BLM, 0.30
U/100 g, i.p.) in the trachea under nembutal (50 mg/kg) anesthesia.
Healthy rats were injected with 0.9% saline (control).
A model of liver fibrosis was created in
seven-week-old Wistar rats by a single injection of DMN (40 mg/kg,
i.p.). Normal healthy rats were the controls and were injected with
0.9% saline (control). The preparation of the pulmonary liver
fibrosis model and of the collection tissue were based on the
methods described in the previous studies (10,11).
Measurement of hydroxyproline in a rat
model of pulmonary fibrosis by a colorimetric method
The left lung was removed, weighed (~0.3 g) and
homogenized in 5% trichloroacetic acid solution (x10 volume) using
a cell homogenizer (Eilard, Berlin, Germany) at 8,000 × g (4°C) for
2 min in an ice bath. Cells were centrifuged (2,500 × g, 4°C) for
20 min and the supernatant washed twice with distilled water. Then,
6 N HCl was added at 110°C completely at the beginning and reacted
for 16 h. Following completion of the reaction, toluene (3 ml) was
added and the mixture was agitated for 20 min. Following
centrifugation (3,000 × g, 20°C) for 10 min, the organic layer was
collected and p-dimethylaminobenzaldehyde added.
Hydroxyproline in the sample was detected using a Multiplate
Spectrometer (Ultramark; Bio-Rad, Hercules, CA, USA) at 560 nm.
Measurement of hydroxyproline in the
liver by HPLC
The left lung was removed, weighed (~0.3 g) and
homogenized in 5 ml ethanol using a cell homogenizer (Eilard) at
8,000 × g (4°C) for 2 min in an ice bath. The homogenate was
centrifuged (2,500 × g, 4°C) for 20 min and the supernatant
collected. The liquid (1 ml) was obtained and heated for 8 h at
60°C until dry. Following dissolving the residue in 40 μl ethanol
and 80 μl borate buffer (0.1 M, pH 8), 40 μl
4-fluoro-7-nitrobenzofurazan (100 mM) was added as a fluorescence
reagent. The reaction was allowed to continue at room temperature
for 15 h in the dark. Then, 840 μl of hydrochloric acid (6 mol/l)
was added to terminate the reaction. Following centrifugation
(2,500 × g, 20°C) for 20 min, the supernatant was removed for HPLC
analysis.
A Hitachi L6000 HPLC system (Hitachi High
Technologies America, Inc., Schaumburg, IL, USA) was used.
Detection was performed with a Hitachi L7480 fluorescence
spectrometer (Hitachi High-Technologies Corporation, Tokyo, Japan;
excitation at 475 nm, emission at 530 nm). The column (Hitachi
High-Technologies Corporation) was a YMC Pack ODS-AQ (150×6.0 mm
ID) at room temperature. The mobile phase was acetonitrile: water
(35:65–50:50 gradient over 15 min) and the flow rate was 1
ml/min.
Measurement of hydroxyproline in
pulmonary and liver organization by LC-MS
The left lung was removed, weighed (~0.3 g) and
homogenized in 5% trichloroacetic acid solution (x10 volume) using
a cell homogenizer (Eilard) at 8,000 × g (4°C) for 2 min in an ice
bath. Cells were centrifuged (2,500 × g, 4°C) for 20 min. The
supernatant was washed twice with distilled water. Then, 6 N HCl
was added at 110°C for 16 h and the samples were prepared for
measurement. The liver was removed, weighed (~0.3 g) and
homogenized in 5 ml ethanol using a cell homogenizer (Eilard) at
8,000 × g (4°C) for 2 min in an ice bath. The homogenate was
centrifuged (2,500 × g, 4°C) for 20 min, the supernatant collected
and the samples prepared for measurement.
The concentration of hydroxyproline was determined
following an LC-MS method using a LC-MS2020 system (Shimadzu Corp.,
Kyoto, Japan). With respect to LC, the mobile phase was 5%
CH3CN-10 mM CH3COONH4. The column
was Shin-pack VP-ODS (150×2 mm diameter). The flow rate was 0.2
ml/min. With regard to MS, atmospheric chemical ionization (APCI)
was employed. Positive ion detection was also used. The probe
electron voltage was 4.5 kV and the probe current was 4.2 μA. The
APCI probe temperature was 250°C and the curved desolvation line
(CDL) electron voltage was −30.0 V. The CDL temperature was 250°C
and block temperature 20°C. The bias voltage for Q-arrays 1, 2 and
3 were 5.0, 25.0 and 35.0 V, respectively. The Q-array RF electron
voltage was 150.0 V.
Statistical analysis
The differences between groups were examined by
one-way analysis of variance. If a significant difference was
noted, the difference between the groups was examined by the
Bonferroni method. Correlations were examined using a two-sided
t-test. P<0.05 was considered to indicate a statistically
significant difference.
Results
Measurement of the hydroxyproline
concentration by LC-MS
MS chromatogram of hydroxyproline
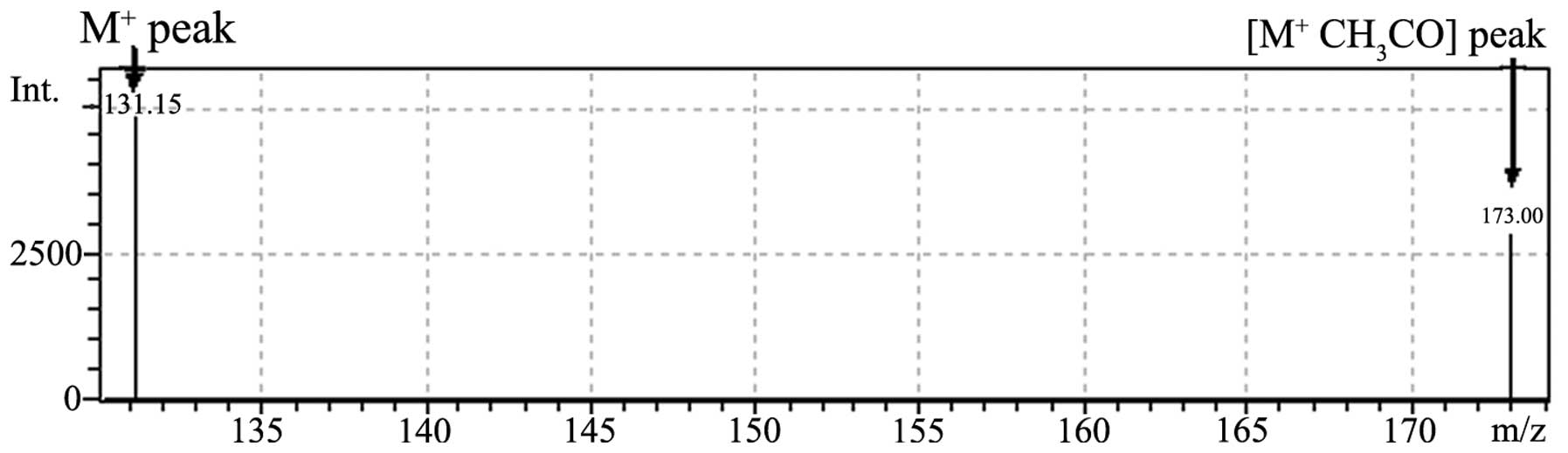
The mass spectrum of hydroxyproline is shown in
Fig. 2. MS of the hydroxyproline
standard (80 pg/ml) resulted in fragment peaks with a molecular
mass of 173.0 (molecular ion + acetic acid-water) generated from
addition to acetic acid ammonium for increasing the number of ionic
molecules (molecular quantity, 131.15) and ionic strength.
LC-MS chromatogram of
hydroxyproline
The LC conditions were as follows: Mobile phase, 5%
CH3CN-10 mM CH3COONH4; column,
Shin-pack VP-ODS (150×2 mm diameter); flow rate, 0.2 ml/min and
ultraviolet (UV) detection at 230 nm. MS was performed using the
APCI ionization method and the ion detection was positive.
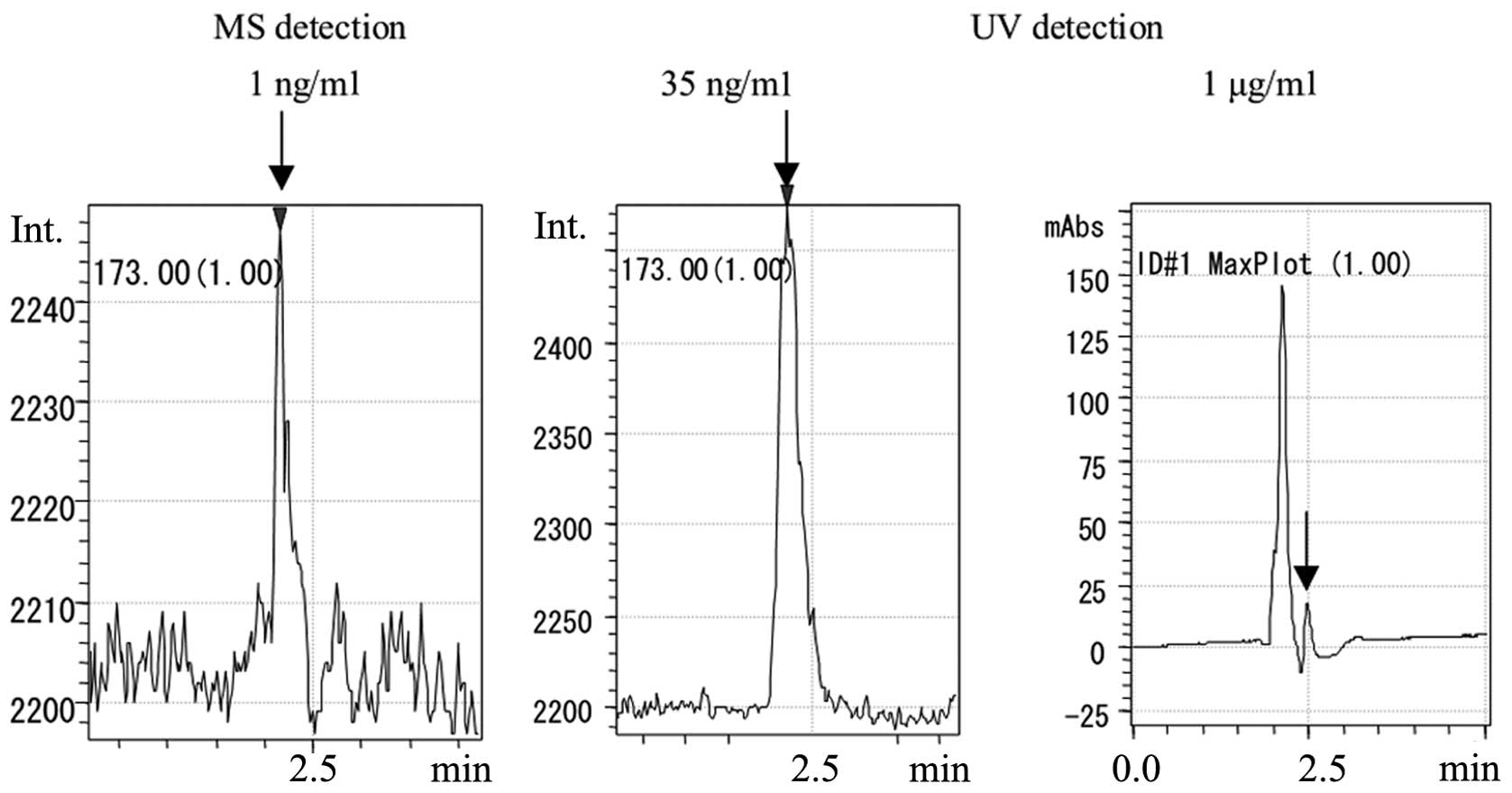
The LC-MS-selected ion monitoring (SIM) chromatogram
of hydroxyproline (m/z, 173.0) is presented in Fig 3. As observed for the hydroxyproline
standard, a peak was observed at 1 ng/ml at a retention time of
2.213 min. A sharp peak was also observed at 50 ng/ml. At 1 μg/ml,
which was 1,000× the standard concentration, a small peak was noted
in the LC-UV spectrum (230 nm).
Sensitivity of LC-MS detection to
hydroxyproline
The LC-MS-SIM standard curve of hydroxyproline
(m/z, 173.0; molecular ion + acetic acid-water) is shown in
Fig. 4. Using the peak area method
for the standard curve, hydroxyproline exhibited a sharp peak at 35
ng/ml (Fig. 3). However, at
concentrations <35 ng/ml, a correlation among the peak areas was
not observed. From 35–560 ng/ml, a correlation among the peak areas
was observed and the standard curve was a straight line. At 60 and
80 μg/ml, the peak areas were almost identical. A correlation among
the peak areas was not observed at concentrations >60 μg/ml.
LC-MS chromatogram and MS spectrum of
hydroxyproline in a model of pulmonary and liver fibrosis in
rats
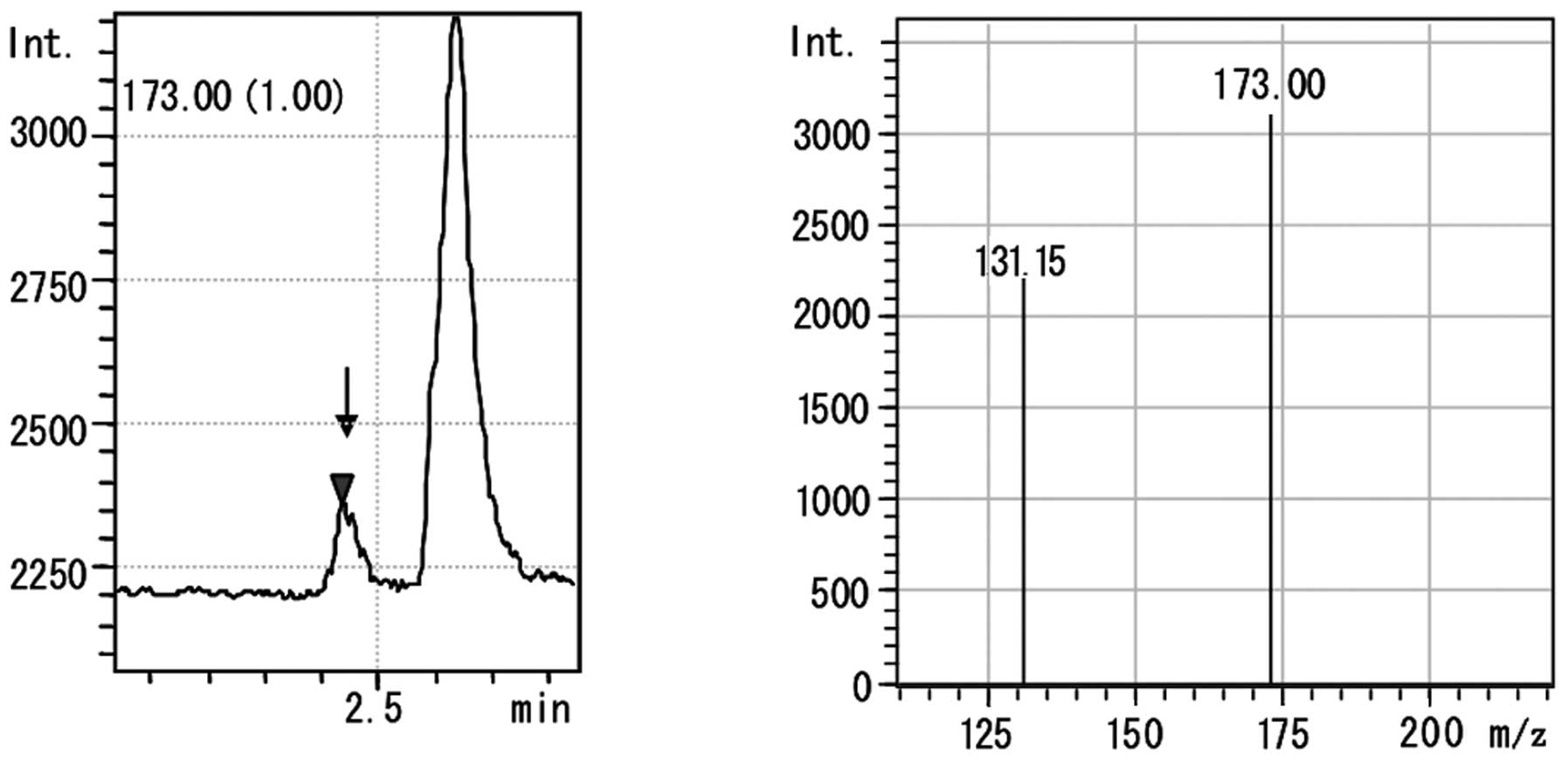
Fig. 5 shows the
LC-MS chromatogram and MS spectrum with SIM detection of
hydroxyproline (m/z 173.0; molecular ion + acetic
acid-water) in a pulmonary model of fibrosis in rats. In the LC-MS
chromatogram of the lung, similar to that of the hydroxyproline
standard, a distinct peak at m/z 173.0 was observed at a
retention time of 2,213 min. Hydroxyproline was identified at
m/z 131.15 in the mass spectrum.
Fig. 6 illustrates
the LC-MS chromatogram and MS spectrum with SIM detection of
hydroxyproline (m/z 173.0; molecular ion + acetic
acid-water) in the fibrotic liver tissue of rats. As observed for
the hydroxyproline standard, a peak was noted at a retention time
of 2.213 min in the LC-MS chromatogram and MS spectrum.
Hydroxyproline concentration in lung
tissue (colorimetric and LC-MS methods)
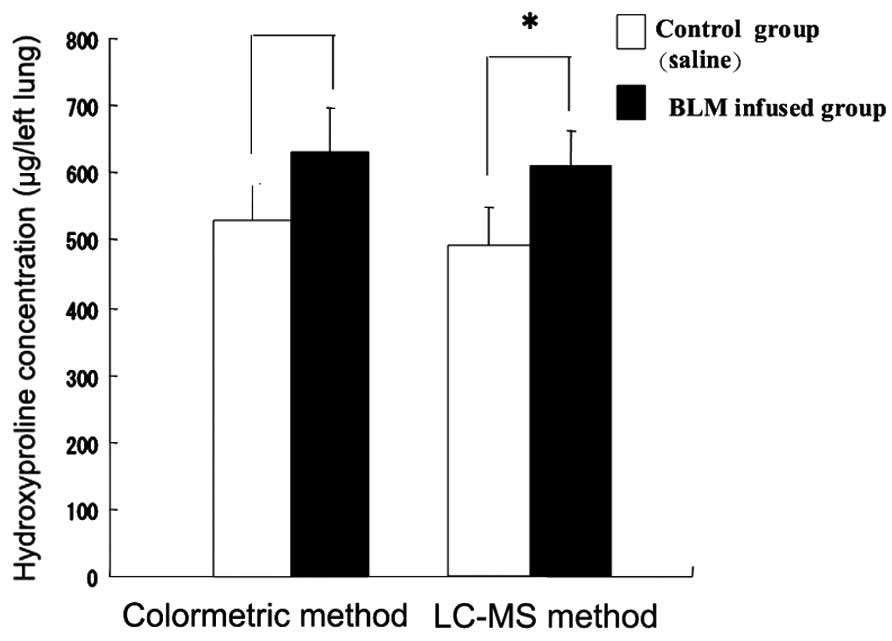
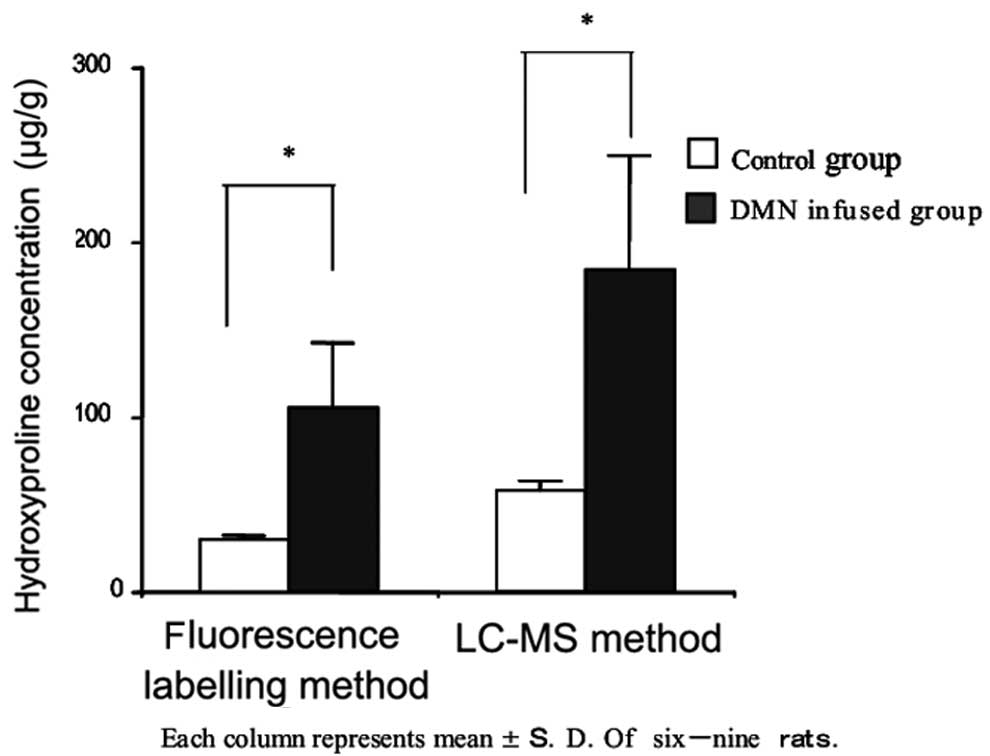
Colorimetric and LC-MS methods employed for the
determination of the hydroxyproline concentration in rat lung
tissue are compared in Fig. 7.
Each column represents the mean ± standard deviation of nine
experiments. According to the colorimetric method, the
hydroxyproline concentration in rat lung tissue in the BLM-infused
group (652.3±70.0 μg/left lung) was significantly higher compared
with that in the control group (547.1±52.3 μg/left lung;
P<0.05). According to the LC-MS method, the BLM-infused group
(610.9±50.3 μg/left lung) had a significantly higher value compared
with the control group (493.3±53.5 μg/left lung; P<0.05).
However, the hydroxyproline concentration in rat lung tissue
measured by the LC-MS method had a lower value compared with that
measured by the colorimetric method.
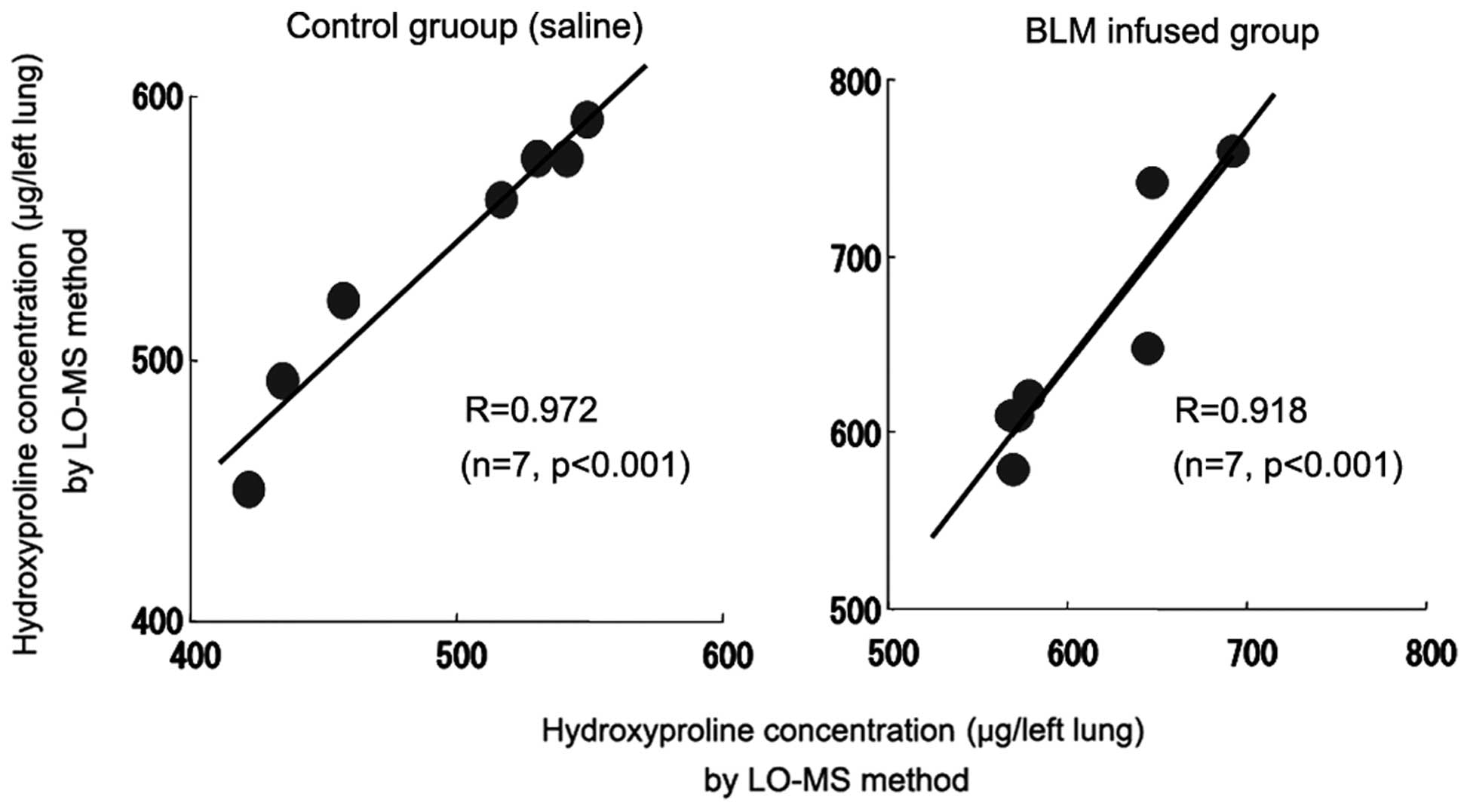
The hydroxyproline concentrations in rat lung tissue
measured by colorimetric and LC-MS methods are compared in Fig. 8. A correlation between the
colorimetric and LC-MS methods for the determination of the
hydroxyproline concentration in the lung tissue of the control
group was identified (r=0.972). Similarly, a correlation between
the colorimetric and LC-MS methods for the determination of the
hydroxyproline concentration in the lung tissue of the BLM-infused
group was identified (r=0.918).
Hydroxyproline concentration in rat liver
tissue by fluorescence labeling and LC-MS methods
The assessment of the hydroxyproline concentration
in rat liver tissue by fluorescence labeling and LC-MS methods is
illustrated in Fig. 9. According
to the fluorescence labeling method, the hydroxyproline
concentration in rat liver tissue of the DMN-infused group
(105.4±36.5 μg/g) was significantly higher compared with that in
the control group (30.3±2.7 μg/g; P<0.05). Also according to
LC-MS analysis, the DMN-infused group (190.4±70.3 μg/g)
demonstrated a significantly higher value compared with the control
group (62.4±6.8 μg/g; P<0.05). However, the hydroxyproline
concentration in rat liver tissue measured by LC-MS was higher
compared with that measured by the fluorescence labeling
method.
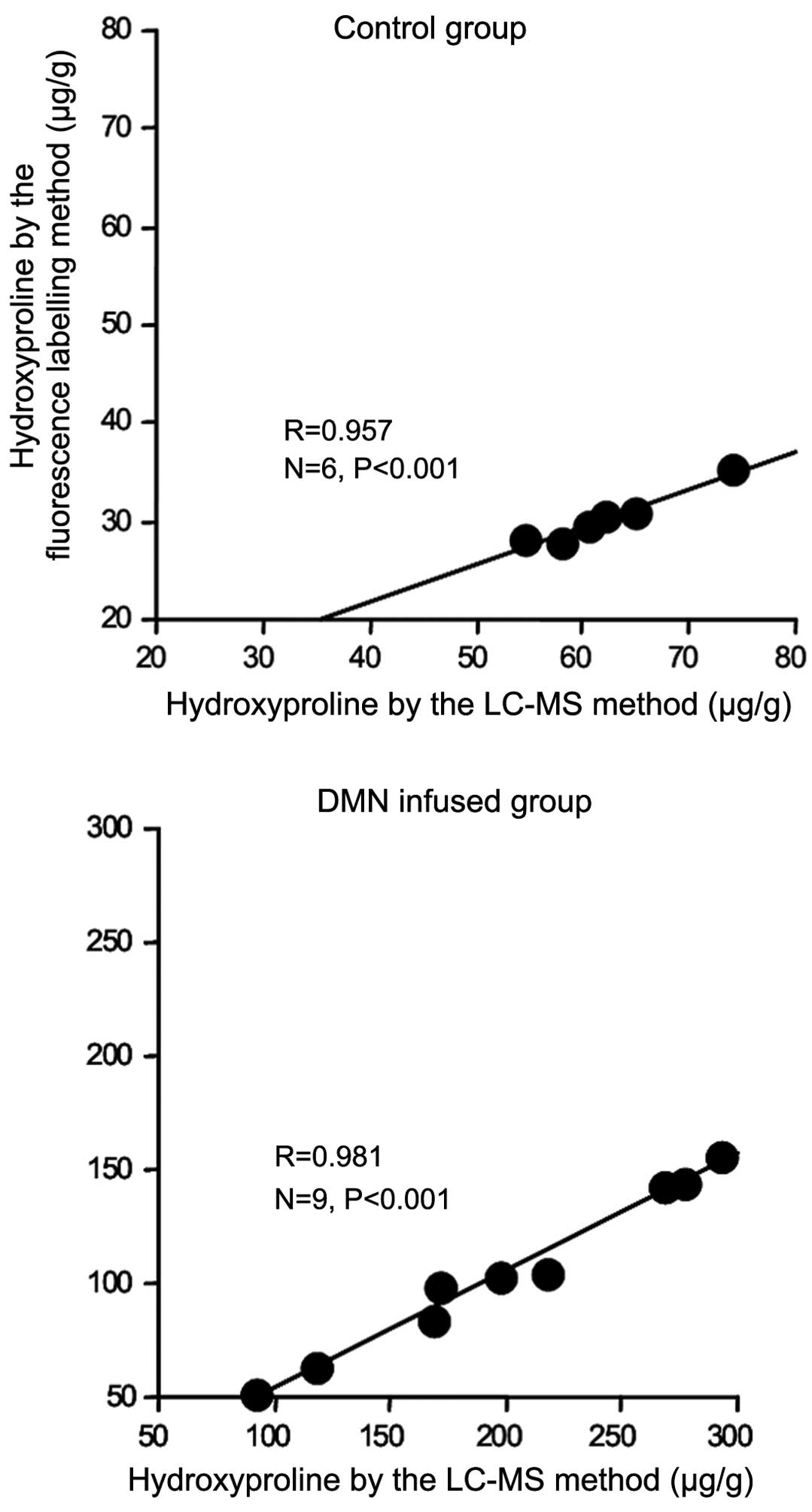
The hydroxyproline concentration in rat liver tissue
assessed by fluorescence labeling and LC-MS methods are correlated
in Fig. 10. The hydroxyproline
concentration in the liver tissue of the control group assessed
using fluorescence labeling correlated with that obtained using the
LC-MS method (r=0.957). Also, the hydroxyproline concentration in
the liver tissue of the DMN-infused group obtained by the
fluorescence labeling method correlated with that obtained using
the LC-MS method (r=0.981).
Discussion
Hydroxyproline is a type of amino acid found in
albumen. The proline residue in the protein is hydroxylated by
4-monooxgenase to generate 4-hydroxy-2-oxoglutal acid, which is
converted to alanine and glycine via 4-glutamic acid in the human
body. In each internal organ of the body, inflammation and fibrosis
may give rise to the accumulation of components of the
extracellular matrix (ECM) (12).
However, under pathological conditions, additional fibrosis may
occur in the liver, lungs and kidneys (13,14).
Chronic hepatitis and liver sclerosis are associated with fibrosis
and also have a close correlation with liver cancer (15).
In the lungs, the severity of fibrosis is dependent
on the type of pneumonia (16).
Usually, pulmonary fibrosis accompanies interstitial pneumonia
(17). Fibrosis in internal organs
is caused by proliferation of ECM components deposited by
myofibroblasts. Understanding the development of fibrosis involves
the examination of collagen concentration. The hydroxyproline
concentration is associated with collagen as an indicator of the
severity of fibrosis (18–20). Hydroxyproline is important as an
index of the disease caused by collagen proliferation (as in
fibrosis) and metabolism malfunction.
In recent years, LC-MS has been demonstrated to be a
highly sensitive detection method (7,8,21).
LC-MS comprises an LC and MS component, and the interface that
connects them. The LC part is identical to conventional HPLC. The
sample is ionized by the interface section. Thermospray ionization
produced unstable (volatile) products, so that APCI was used (which
ionizes material, but not the solvent, following nebulization with
solvent under atmospheric pressure). Electrospray ionization (ESI)
may be used to ionize molecules of high polarity and high molecular
weight, which is ideally suited to the interface component.
However, in the present study, ESI was not adapted to the
measurement of hydroxyproline in body specimens, as ESI was not
accompanied by the thermospray in case of ionization. It was a more
simple procedure to measure hydroxyproline combined with
Na+ by adding additional Na+ to the
sample.
The MS spectrum of hydroxyproline demonstrated peaks
of m/z 173.0 and 131.15. m/z 173.0 was identified as
the peak representing the acetic-acid salt originating from the
acetic acid ammonium that was added to the mobile phase to increase
ionic strength. Furthermore, due to the comparative strength of
m/z 131.15 and 173.0 being approximately identical in the MS
spectrum, the ion of m/z 173.0 was chosen by SIM in the MS
chromatogram in order to avoid the interference peak of the mobile
phase.
In the MS chromatogram of the hydroxyproline
standard at 1 ng/ml, a peak was observed in the SIM measurement
with m/z 173.0. In the UV spectrum (230 nm) of
hydroxyproline at 1 μg/ml, which was 1,000× the standard
concentration, a small peak was observed (measurement was performed
at the same time as the LC measurement).
At concentrations <35 ng/ml, among the peak
areas, no strong correlation was observed between the different
methods, and it was not possible to create a standard curve. It was
hypothesized that the analyte may have been affected by the peak of
the mobile phase, as hydroxyproline has a comparatively low
molecular weight (131.15). In addition, the peak retention time may
have become unstable due to the polarity of the solvent at low
concentrations (hydroxyproline concentration <35 ng/ml).
It was hypothesized that it may be possible to
measure concentrations of substances as low as the pg/ml level. For
the peak of hydroxyproline in the MS chromatogram, the retention
time was short (2.213 min). The capability of hydroxyproline to be
maintained on the ODS column was weaker than originally proposed.
The relative ion strength in the MS spectrum at m/z 131.15
and 173.0 was approximately identical, and accordingly, the
interference peak of the mobile phase chose an ion of m/z
173.0. In the LC-MS chromatogram of hydroxyproline in lung and
liver tissue, a sharp peak of m/z 173.0 was observed at a
retention time of 2.213 min, just as for the hydroxyproline
standard. With respect to the MS spectrum, the molecular ion peak
of hydroxyproline (m/z 131.15) was confirmed.
Measurement of the hydroxyproline concentration in
lung and liver tissues by LC-MS was compared with that obtained by
the colorimetric method as well as a fluorescence method using HPLC
from a previous study by our group. A highly significant positive
correlation was noted. However, the value of the hydroxyproline
concentration in the lung obtained by the colorimetric method was
higher than that obtained by LC-MS. Furthermore, the hydroxyproline
concentration in the liver obtained by the fluorescence method
using HPLC was lower compared with the value obtained by LC-MS
(which was supposed to be a high absorbance of all components in
the sample at a constant wavelength of 560 nm).
In conclusion, hydroxyproline, as an indicator of
fibrosis, was measured by colorimetric and HPLC methods in previous
studies by our group. By comparison, the present study identified
that the LC-MS method was more advantageous, characterized by its
simple process, high sensitivity (pg level) and short separation
time. Further investigations with larger sample sizes are warranted
to confirm these results.
Acknowledgements
The authors are grateful to M. Kusunose, M. Ono and
A. Hamada (Department of Pharmacy, Kochi Medical School, Kochi,
Japan) for providing several reagents used in this study, helpful
suggestions and technical expertise. This work was funded by the
Public Health Department of Heilongjiang Province (no. 2013365) of
China.
References
|
1
|
Kitchener RL and Grunden AM: Prolidase
function in proline metabolism and its medical and biotechnological
applications. J Appl Microbiol. 113:233–247. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Myllyharju J: Prolyl 4-hydroxylases, key
enzymes in the synthesis of collagens and regulation of the
response to hypoxia, and their roles as treatment targets. Ann Med.
40:402–417. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Wick G, Grundtman C, Mayerl C, et al: The
immunology of fibrosis. Annu Rev Immunol. 31:107–135. 2013.
View Article : Google Scholar
|
|
4
|
Hofman K, Hall B, Cleaver H and Marshall
S: High-throughput quantification of hydroxyproline for
determination of collagen. Anal Biochem. 417:289–291. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
McAnulty RJ: Methods for measuring
hydroxyproline and estimating in vivo rates of collagen synthesis
and degradation. Methods Mol Med. 117:189–207. 2005.PubMed/NCBI
|
|
6
|
McCooeye M and Mester Z: Comparison of
flow injection analysis electrospray mass spectrometry and tandem
mass spectrometry and electrospray high-field asymmetric waveform
ion mobility mass spectrometry and tandem mass spectrometry for the
determination of underivatized amino acids. Rapid Commun Mass
Spectrom. 20:1801–1808. 2006. View
Article : Google Scholar
|
|
7
|
Jemal M and Xia YQ: LC-MS Development
strategies for quantitative bioanalysis. Curr Drug Metab.
7:491–502. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Chen G and Pramanik BN: LC-MS for protein
characterization: current capabilities and future trends. Expert
Rev Proteomics. 5:435–444. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Becker S, Kortz L, Helmschrodt C, et al:
LC-MS-based metabolomics in the clinical laboratory. J Chromatogr B
Analyt Technol Biomed Life Sci. 883–884:68–75. 2012.
|
|
10
|
Cui T, Kusunose M, Hamada A, et al:
Relationship between the eosinophilia of bronchoalveolar lavage
fluid (BALF) and the severity of pulmonary fibrosis induced by
bleomycin in rats. Biol Pharm Bull. 26:959–963. 2003. View Article : Google Scholar
|
|
11
|
Kusunose M, Qiu B, Cui T, Hamada A, et al:
Effect of Sho-saiko-to extract on hepatic inflammation and fibrosis
in dimethylnitrosamine induced liver injury rats. Biol Pharm Bull.
25:1417–1421. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Maruyama K: Hydroxproline. Nihon Rinsho.
62(Suppl 12): 220–223. 2004.(In Japanese).
|
|
13
|
Muiznieks LD and Keeley FW: Molecular
assembly and mechanical properties of the extracellular matrix: A
fibrous protein perspective. Biochim Biophys Acta. 1832:866–875.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lu P, Takai K, Weaver VM and Werb Z:
Extracellular matrix degradation and remodeling in development and
disease. Cold Spring Harb Perspect Biol. 3:a0050582011.PubMed/NCBI
|
|
15
|
Pungpapong S, Kim WR and Poterucha JJ:
Natural history of hepatitis B virus infection: an update for
clinicians. Mayo Clin Proc. 82:967–975. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Strieter RM and Mehrad B: New mechanisms
of pulmonary fibrosis. Chest. 136:1364–1370. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Swigris JJ and Brown KK: Acute
interstitial pneumonia and acute exacerbations of idiopathic
pulmonary fibrosis. Semin Respir Crit Care Med. 27:659–267. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ono M, Miyamura M, Kyotani S, et al:
Effects of Sho-saiko-to extract on liver fibrosis in relation to
the changes in hydroxyproline and retinoid levels of the liver in
rats. J Pharm Pharmacol. 51:1079–1084. 1999. View Article : Google Scholar
|
|
19
|
Ono M, Miyamura M, Kyotani S, et al:
Effect of Sho-saiko-to extract on HGF and TGF-beta levels of
intraorgans in liver-injured rats after partial hepatectomy. J
Pharm Pharmacol. 52:111–118. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kusunose M, Qiu B, Cui T, et al: Effect of
Sho-saiko-to extract on hepatic inflammation and fibrosis in
dimethylnitrosamine induced liver injury rats. Biol Pharm Bull.
25:1417–1421. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Holčapek M, Jirásko R and Lísa M: Recent
developments in liquid chromatography-mass spectrometry and related
techniques. J Chromatogr A. 1259:3–15. 2012.
|