Introduction
Acute coronary syndrome is the leading cause of
mortality, not only in developed countries, but also in developing
countries, including China (1).
Observational epidemiological studies have reported an increased
mortality rate from coronary heart disease during winter (2,3).
Previous studies have demonstrated that myocardial infarctions
increase linearly with a decrease in diurnal temperature (4,5). In
addition, a recent study indicated that an extremely cold
temperature increases the risk of mortality from ischemic heart
disease in China (6).
Although an association between cold temperatures
and coronary mortality has been observed, the mechanisms underlying
cold-induced myocardial infarctions remain to be fully elucidated.
Atherosclerotic plaque rupture is most commonly associated with
acute coronary events. The coronary atherosclerotic plaque that is
prone to rupture, termed a vulnerable plaque, is a thin-cap
fibroatheroma, characterized by a large necrotic core covered by a
thin fibrous cap (7). Plaques
vulnerable to rupture contain less structural collagen, which is
induced by increased production of proteases. Matrix
metalloproteinases (MMPs) are the major proteases responsible for
weakening the plaque cap. Additionally, MMPs are negatively
regulated by intrinsic tissue inhibitor of matrix
metalloproteinases (TIMPs). It has been reported that MMPs are
upregulated and activated by exposure to a cold environment in
several tissues (8). Furthermore,
TIMPs appeared to be potent agents in the treatment of cold-induced
injury in multiple tissues, including the brain and liver (9–12).
Thus, it was hypothesized that cold stress was able to promote
atherosclerotic plaque instability associated with an imbalance
between MMPs and TIMPs.
In the present study, apolipoprotein E
(ApoE)-deficient (ApoE−/−) mice were fed a normal diet
and exposed to control conditions or a cold environment for eight
weeks. The lipid profiles, atherosclerotic plaque size and features
of plaque stability were measured at the end of the experiment.
Materials and methods
Animal care
In total, 20 male, 8-week-old, ApoE−/−
mice with a C57BL/6J background, were purchased from Beijing
Yikelihao Biotechnology Co., Ltd (Beijing, China). The animals were
housed under a 12 h/12 h day/night cycle with ad libitum
access to food and water. The mice were fed a normal chow diet.
Experimental procedures were approved by the Animal Care and Use
Committee of the General Hospital of PLA Chengdu Military Area
Command.
Intermittent cold stress
Mice were randomly subjected to room temperature
(control group, n=10) and intermittent cold stress (cold group,
n=10). Mice in the control group were kept in a room with an
ambient temperature of 24±2°C. Mice in the cold group were placed
in a cold room at 4±1°C for 4 h (between 8:00 am and 10:00 am) and
then placed in a room at 24±2°C for the remaining 20 h per day. The
administration period lasted 8 weeks.
Plasma glucose and lipids
Fasting blood samples were obtained at the end of
the experiments. Glucose, triglycerides (TG), total cholesterol
(TC), low-density lipoprotein-cholesterol (LDL-C) and high-density
lipoprotein-cholesterol (HDL-C) were measured by colorimetric
assays using commercially available kits (Nanjing Jiancheng
Bioengineering Institute, Nanjing, Jiangsu, China) in accordance
with the manufacturer’s instructions (13).
Atherosclerotic plaque size
The hearts with the aortic roots were harvested and
fixed in 10% paraformaldehyde overnight and embedded in paraffin.
Atherosclerotic lesions in the aortic roots were examined in
cross-sections of the aortic origin. Six consecutive 5-μm-thick
sections were cut from the aorta where the valve cusp was visible.
The sections were stained with hematoxylin and eosin and images
were captured using an Olympus BX41 microscope (Olympus Corp.,
Tokyo, Japan). The average plaque area was used for statistical
analysis (14).
Collagen analysis
Paraffin-embedded aortic sinus sections were stained
with Masson’s trichrome using a fast Masson dye kit (Nanjing
Jiancheng Bioengineering Institute) according to the manufacturer’s
instructions (15). The percentage
of the positively stained area was calculated using Nikon
NIS-Elements Research Software (Nikon Instruments, Inc., Melville,.
NY, USA).
Immunohistochemistry
Thoracic aortas were fixed in 4% paraformaldehyde
for 12 h, embedded in paraffin and then cut into sections (5 μm).
The sections were incubated with anti-α-smooth muscle-actin (α-SMA;
diluted 1:1,500; Epitomics, San Diego, CA, USA),
anti-macrophages/monocytes antibody (MOMA)-2 (diluted 1:50; Abcam,
Cambridge, MA, USA), anti-cluster of differentiation 3 (CD3;
diluted 1:100; GeneTex Inc., San Antonio, TX, USA) antibodies
overnight at 4°C. Specific binding was detected with biotinylated
goat anti-rabbit IgG secondary antibody-horseradish peroxidase
complexes using an ABC kit (Wuhan Boster Biological Technology,
Ltd., Wuhan, Hubei, China). The antigen-antibody complex was
subsequently visualized with 3′,3′-diaminobenzidine solution. The
sections were viewed under a light microscope (Olympus BX41;
Olympus Corp., Tokyo, Japan) (16). The percentage of the positively
stained area was calculated using Nikon NIS-Elements Research
Software.
Western blot analysis
The proteins from the thoracic aorta were extracted
using a protein extraction kit (Nanjing KeyGen Biotech, Co., Ltd.,
Nanjing, Jiangsu, China). The protein concentrations were
determined using an enhanced BCA Protein Assay kit (Beyotime
Institute of Biotechnology, Shanghai, China). Extracted protein (30
μg) was loaded onto 10% sodium dodecyl sulfate polyacrylamide gels.
The electrophoresis time was decided by the molecular weight of the
protein. The separated proteins were then transferred onto
appropriate nitrocellulose membranes. The membranes were blocked
with Tris-buffered saline with Tween-20 (TBS-T) containing 5%
powdered milk for 1 h and then incubated with anti-MMP-2,
anti-MMP-3, anti-MMP-9, anti-MMP-14, anti-TIMP-1 and anti-TIMP-2
antibodies (Wuhan Boster Biological Technology, Ltd.) at a dilution
of 1:200 in 5% milk/TBS-T overnight. The membranes were rinsed
three times with TBS-T and incubated with horseradish
peroxidase-conjugated goat antimouse antibody (diluted 1:1,000;
Beyotime Institute of Biotechnology) for 1 h. The membranes were
rinsed three times with TBS-T and then rinsed once with TBS.
Chemiluminescence detection reagent (Millipore, Billerica, MA, USA)
was added dropwise onto the membranes. The luminescent signal was
detected by exposure to X-ray film (17).
Statistical analysis
Continuous data are presented as the mean ± standard
error of the mean. Comparisons between the two groups were
determined by the independent samples t-test (SPSS, Inc., Chicago,
IL, USA). P<0.05 was considered to indicate a statistically
significant difference.
Results
Metabolic profiles
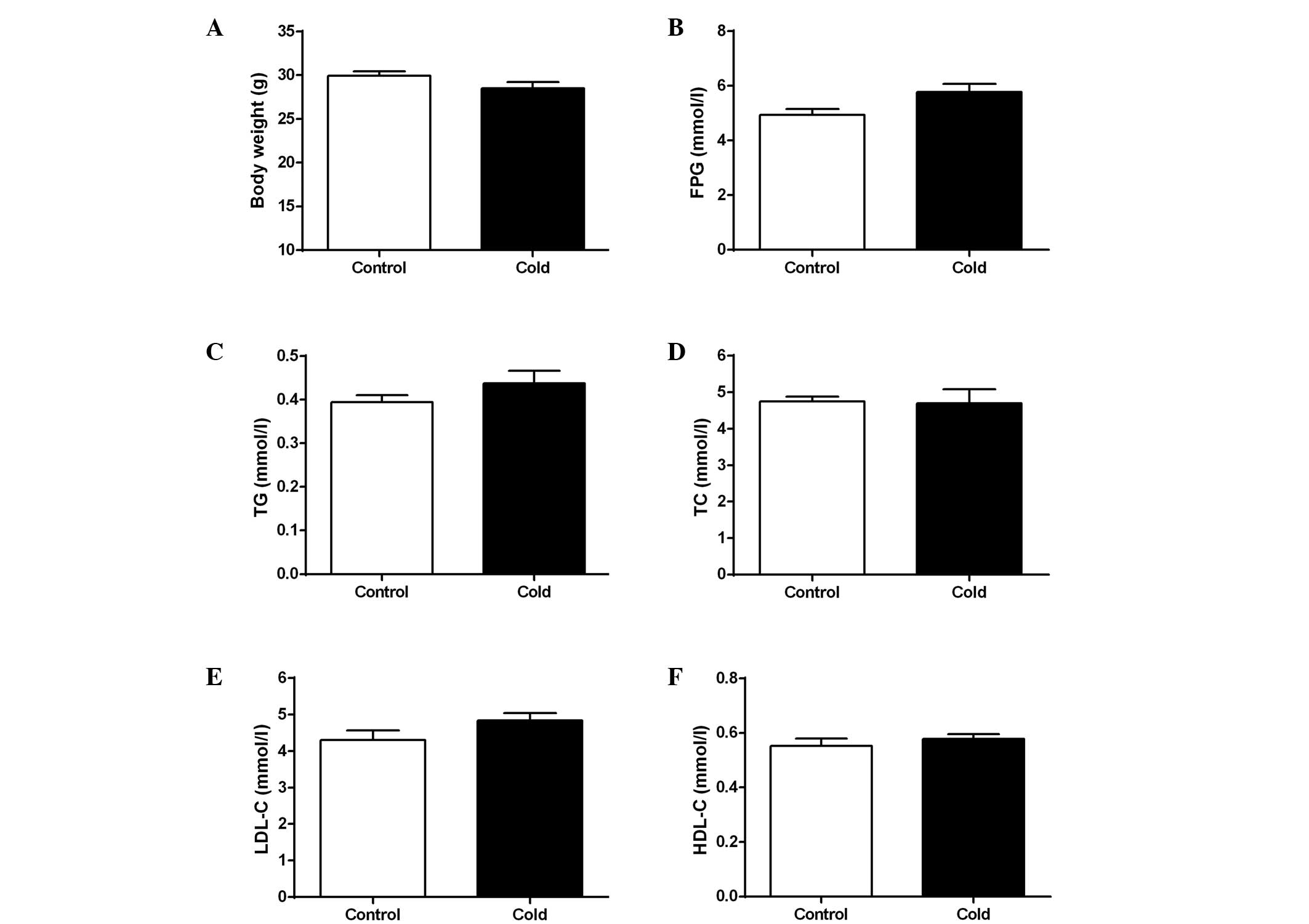
Whether cold stress affects glycolipid metabolism
was initially investigated. At the end of the experiment, mice
exposed to a cold environment had a similar body weight compared
with the controls (Fig. 1A).
Exposure to a cold environment did not affect the fasting plasma
glucose concentration (Fig. 1B)
and plasma lipid profiles, including TG, TC, LDL-C and HDL-C
(Fig. 1C–F; all P>0.05). These
findings suggest that cold stress may not affect the glycolipid
metabolism of ApoE−/− mice.
Atherosclerotic plaque size
Secondly, the present study investigated whether
cold stress affected the growth of atherosclerotic plaques. The
atherosclerotic plaque size in the aortic root from the cold group
was significantly greater than the control group (P<0.01;
Fig. 2). This finding suggested
that cold stress may promote plaque growth.
Plaque instability
Thirdly, the present study examined whether cold
stress affected the features of atherosclerotic plaque instability.
The contents of collagen and vascular smooth muscle cells (SMCs) in
atherosclerotic plaques were significantly decreased in the cold
group compared with the control group (P<0.01; Fig. 3A, C, E and G), while the contents
of macrophages and lymphocytes were significantly increased
following 8-week intermittent cold stress (P<0.05 or P<0.01;
Fig. 3B, D, F and H). These
findings suggest that cold stress may enhance plaque
instability.
Expression of MMPs/TIMPs
Finally, it was revealed which molecules may be
involved in cold-induced plaque instability. The protein expression
of MMP-2, MMP-3, MMP-9, MMP-14, TIMP-1 and TIMP-2 in the thoracic
aorta was measured by western blot analysis. Exposure to a cold
environment significantly increased the expression of MMP-2, MMP-9
and MMP-14 (P<0.01 or P<0.05), decreased the expression of
TIMP-1 (P<0.05) and had no significant effect on MMP-3 and
TIMP-2 in aortic tissues (Fig. 4).
These findings suggest that the imbalance between the protein
expression of MMPs and TIMPs in vessels may contribute to
cold-induced plaque instability.
Discussion
The present study demonstrated that chronic
intermittent cold stress promotes the growth of atherosclerotic
plaques and enhances the features of plaque instability.
Additionally, these effects are associated with the imbalance
between MMPs and TIMPs, however, are not associated with obesity,
hyperglycemia or hyperlipidemia. These findings suggest that
chronic intermittent cold stress may promote plaque instability
through enhancing the action of MMPs and subsequently weakening the
fibrous cap.
There is a growing body of evidence that exposure to
a cold environment is associated with a high risk of sudden cardiac
death. However, a causal link between a cold environment and sudden
cardiac death has not been established. The main finding of the
present study was that eight-week cold exposure resulted in plaque
instability, as indicated by decreased collagen and vascular SMCs
deposition and increased macrophage and lymphocyte contents. The
fibromuscular cap is a crucial factor for the stability of plaques.
Collagen and the vascular SMCs are important parts of the fibrous
cap, which are able to resist the mechanical strength and promote
the stability of atherosclerotic plaques. These results indicated
that a cold environment may induce acute coronary events and sudden
cardiac death by increasing the vulnerability of the
atherosclerotic plaque.
Metabolic disorders, including obesity,
hyperglycemia and hyperlipidemia may promote plaque growth and
instability. Chronic cold stress induces adaptive metabolic
alterations in several different types of animals. Kozyreva et
al (18) reported that cold
stress caused a marked decrease in plasma LDL and a significant
increase in HDL in hypertensive and normotensive rats. However, a
recent study demonstrated that cold stress significantly increased
the plasma total cholesterol and LDL in ApoE−/− and LDL
receptor−/− mice following persistent exposure to 4°C
for eight weeks (19). These
varied results may be attributed to the different temperatures,
exposure time, cooling speed or the genetic background of the
animals. In the present study, no effect on body weight, plasma
glucose and lipids was observed following eight-week intermittent
cold stress. These findings indicated that cold stress-induced
plaque growth and instability are not associated with the
alterations in glycolipid metabolism.
Hypertension is an important factor in the
development of atherosclerosis and the rupture of plaques. Previous
studies have demonstrated that chronic cold stress significantly
increased systolic and diastolic blood pressure (20,21).
However, the present study demonstrated that the blood pressure was
similar between the two groups (data not shown). Thus, it was
hypothesized that cold-induced plaque instability is not attributed
to the alteration in blood pressure.
Mechanisms by which a cold environment causes plaque
instability have not been fully elucidated. A previous study
demonstrated that chronic cold exposure promotes plaque instability
via uncoupling protein 1-dependent lipolysis and
hypercholesterolemia (19).
However, similar biochemical features were not observed in the
present study. Several studies have suggested that MMPs and TIMPs
are involved in plaque instability (22–27).
Furthermore, MMPs have become recognized as crucial regulators in
cold-induced biological processes (11,12).
The present study revealed that cold-induced plaque instability was
concomitant with the increased expression of MMP-2, MMP-9 and
MMP-14 and the decreased expression of TIMP-1. The results from the
present study indicated that the imbalance of MMPs and TIMPs may be
responsible for the association between intermittent cold stress
and plaque vulnerability. Therefore, restoring the balance between
MMPs and TIMPs may be a potent method to prevent cold-induced
plaque rupture.
In conclusion, the present study demonstrated that
chronic intermittent cold exposure increases atherosclerotic plaque
size and promotes plaque instability in ApoE−/− mice.
Furthermore, the cold stress-induced vulnerability to plaques is
associated with the imbalance between MMP-2/-9/-14 and TIMP-1. The
present findings may contribute to mechanistic insights into sudden
cardiac death in cold environments.
Acknowledgements
This study was supported by grants from the National
Natural Science Foundation of China (nos. 81070191 and
81100232).
References
|
1
|
Bi Y, Gao R, Patel A, et al:
Evidence-based medication use among Chinese patients with acute
coronary syndromes at the time of hospital discharge and 1 year
after hospitalization: results from the Clinical Pathways for Acute
Coronary Syndromes in China (CPACS) study. Am Heart J. 157:509–516.
2009.
|
|
2
|
Bhaskaran K, Hajat S, Haines A, Herrett E,
Wilkinson P and Smeeth L: Effects of ambient temperature on the
incidence of myocardial infarction. Heart. 95:1760–1769. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Barnett AG, Dobson AJ, McElduff P, et al:
Cold periods and coronary events: an analysis of populations
worldwide. J Epidemiol Community Health. 59:551–557. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
De Lorenzo F, Sharma V, Scully M and
Kakkar VV: Cold adaptation and the seasonal distribution of acute
myocardial infarction. QJM. 92:747–751. 1999.PubMed/NCBI
|
|
5
|
Dilaveris P, Synetos A, Giannopoulos G,
Gialafos E, Pantazis A and Stefanadis C: Climate impacts on
myocardial infarction deaths in the Athens territory: the CLIMATE
study. Heart. 92:1747–1751. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Guo Y, Li S, Zhang Y, et al: Extremely
cold and hot temperatures increase the risk of ischaemic heart
disease mortality: epidemiological evidence from China. Heart.
99:195–203. 2013. View Article : Google Scholar
|
|
7
|
Arbab-Zadeh A, Nakano M, Virmani R and
Fuster V: Acute coronary events. Circulation. 125:1147–1156. 2012.
View Article : Google Scholar
|
|
8
|
Li F, Gao Z, Xu D, Jing J and Wang J:
Effect of cold and dry environment on the expression of matrix
metalloproteinase 1 mRNA in bone tissues of different pathogenic
factors induced models. Zhongguo Zuzhi Gongcheng Yanjiu.
16:9414–9418. 2012.
|
|
9
|
Kawai N, Kawanishi M, Okada M, Matsumoto Y
and Nagao S: Treatment of cold injury-induced brain edema with a
nonspecific matrix metalloproteinase inhibitor MMI270 in rats. J
Neurotrauma. 20:649–657. 2003. View Article : Google Scholar
|
|
10
|
Hori T, Uemoto S, Hata T, et al: Matrix
metalloproteinase-9 after the cold ischemia/reperfusion injury
and/or shear stress with portal hypertension: an overview. Surg
Today. 44:201–203. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Upadhya AG, Harvey RP, Howard TK, Lowell
JA, Shenoy S and Strasberg SM: Evidence of a role for matrix
metalloproteinases in cold preservation injury of the liver in
humans and in the rat. Hepatology. 26:922–928. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Li C, Li X, Shen Q, et al: Critical role
of matrix metalloproteinase-9 in acute cold exposure-induced stroke
in renovascular hypertensive rats. J Stroke Cerebrovasc Dis.
28:e477–e485. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Jiang S, Jin F, Li D, et al: Intermittent
hypobaric hypoxia promotes atherosclerotic plaque instability in
ApoE-deficient mice. High Alt Med Biol. 14:175–180. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ma S, Yang D, Li D, Tang B and Yang Y:
Oleic acid induces smooth muscle foam cell formation and enhances
atherosclerotic lesion development via CD36. Lipids Health Dis.
10:532011. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Jin F, Jiang S, Yang D, et al: Acipimox
attenuates atherosclerosis and enhances plaque stability in
ApoE-deficient mice fed a palmitate-rich diet. Biochem Biophys Res
Commun. 428:86–92. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Li D, Ma S, Yang Y, et al: BTEB2 knockdown
suppresses neointimal hyperplasia in a rat artery balloon injury
model. Mol Med Rep. 4:413–417. 2011.PubMed/NCBI
|
|
17
|
Ma S, Yang D, Li D, Tang B, Sun M and Yang
Y: Cardiac extracellular matrix tenascin-C deposition during
fibronectin degradation. Biochem Biophys Res Commun. 409:321–327.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Kozyreva TV, Lomakina SV, Tuzikov FV and
Tuzikova NA: Plasma lipoproteins under the effect of cold exposure
in normotensive and hypertensive rats. J Therm Biol. 29:67–72.
2004. View Article : Google Scholar
|
|
19
|
Dong M, Yang X, Lim S, et al: Cold
exposure promotes atherosclerotic plaque growth and instability via
UCP1-dependent lipolysis. Cell Metab. 18:118–129. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Hintsala H, Kandelberg A, Herzig KH, et
al: Central aortic blood pressure of hypertensive men during
short-term cold exposure. Am J Hypertens. Aug 20–2013.(Epub ahead
of print).
|
|
21
|
Modesti PA: Season, temperature and blood
pressure: A complex interaction. Eur J Intern Med. 24:604–607.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Sapienza P, di Marzo L, Borrelli V, et al:
Metalloproteinases and their inhibitors are markers of plaque
instability. Surgery. 137:355–363. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shah PK and Galis ZS: Matrix
metalloproteinase hypothesis of plaque rupture: players keep piling
up but questions remain. Circulation. 104:1878–1880. 2001.
|
|
24
|
Johnson JL, Fritsche-Danielson R, Behrendt
M, et al: Effect of broad-spectrum matrix metalloproteinase
inhibition on atherosclerotic plaque stability. Cardiovasc Res.
71:586–595. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Xie S, Nie R and Wang J: Inhibiting
extracellular matrix metalloproteinase inducer maybe beneficial for
diminishing the atherosclerotic plaque instability. J Postgrad Med.
55:284–286. 2009. View Article : Google Scholar
|
|
26
|
Smeglin A and Frishman WH: Elastinolytic
matrix metalloproteinases and their inhibitors as therapeutic
targets in atherosclerotic plaque instability. Cardiol Rev.
12:141–150. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Loftus IM, Naylor AR, Bell PR and Thompson
MM: Matrix metalloproteinases and atherosclerotic plaque
instability. Br J Surg. 89:680–694. 2002. View Article : Google Scholar : PubMed/NCBI
|