Introduction
The auditory sense is regulated by a specialized
mechanosensory epithelium (the organ of Corti) in the cochlea,
consisting of hair cells and nonsensory supporting cells (1), which extends along the length of the
coiled cochlear duct in the ventral region of the inner ear
(2). During early development,
processes refine the afferent innervation of the sensory hair
cells, leading to a precise pattern of innervation (3). Cochlear hair cells release
neurotrophic factors, which participate in cochlear development,
and the survival and maintenance of spiral ganglion neurons depend
on neural activity and neurotrophic support (4). The cochlear spiral ganglion involves
primary afferent bipolar neurons that propagate auditory signals
from hair cells of the organ of Corti to the central auditory
system (4). However, the signaling
pathway that regulates cochlear prosensory specification requires
further investigation (5). Thus,
it is necessary to identify the signaling pathways that induce the
precise and unique responses in cochlear prosensory cells.
Chemotactic cytokines (chemokines) are a large
family of small protein ligands that are classified into four
subfamilies, CXC, CC, CX3C and C chemokines, based on the location
and organization of the first two cysteine residues (6). CXC chemokine ligand 12 (CXCL12), also
termed stromal cell-derived factor 1, and its cognate receptor, CXC
chemokine receptor 4 (CXCR4), are crucial for neurogenesis and
regulate essential processes during the establishment of neural
networks in neuronal systems, including neuronal migration, cell
positioning and axon guidance (6–8). A
previous study demonstrated that CXCL12 and CXCR4 are involved in
regulating cerebellar granule cell migration (9). Additionally, the CXCL12/CXCR4
signaling pathway is critical for the formation of appropriate
assemblies of γ-aminobutyric acid-ergic interneurons in the
cerebral cortex and the pontine nuclei in the pons (10,11).
Furthermore, in the spinal cord, the CXCL12/CXCR4 signaling pathway
regulates the precision of initial sensory and motor axon
trajectories of sensory and motor neurons (12). However, it is unclear whether the
CXCL12/CXCR4 signaling pathway participates in cochlear
development.
The present study analyzed the expression of CXCL12
and CXCR4 during cochlear development in neonatal mice. It was
demonstrated that CXCL12 and CXCR4 expression was significantly
increased during cochlear development. Treatment of spiral ganglion
neurons with CXCL12 in vitro significantly increased cell
survival and neurite growth of spiral ganglion neurons, whereas
blocking CXCL12/CXCR4 signaling impaired the above-mentioned
processes. Furthermore, inhibiting CXCL12/CXCR4 signaling reduced
the cell number and altered the morphology of spiral ganglion
neurons in vivo.
Materials and methods
Mice and tissue sample collection
A total of 24 female neonatal C57BL/6J mice (age, 3
weeks; weight, 25–30 g), provided by the Laboratory Animal Center
of the Fourth Military Medical University (Xi'an, China), were
housed under a 12-h light/dark cycle at 24±1°C and 45±5% humidity,
with ad libitum access to food and water. The animal
procedures were revised and approved by the Institutional Animal
Care and Use Committee of Fourth Military Medical University. The
mice were sacrificed by an intramuscular injection of ketamine (100
mg/kg; Yongnuo Pharmaceutical Co., Ltd., Yichang, China), after
which cochleae were quickly and carefully removed, and the round
window and vestibular membranes were opened then soaked in 4%
paraformaldehyde solution (Sangon Biotech Co., Ltd., Shanghai,
China) overnight at 4°C. Cochlear tissue slices were stored at
−20°C until analysis.
Cell culture
Cochlear cartilage was removed with fine forceps,
and the spiral ganglion tissue was separated and transferred to
ice-cold Hanks' balanced salt solution (Sangon Biotech Co., Ltd.).
The modiolus was cultured in Hibernate-A medium, containing 2% B27
and 0.1% penicillin (100 U/ml) and 0.1 mg/ml streptomycin (all
Thermo Fisher Scientific, Inc., Waltham, MA, USA) as described
previously (13). To assess the
effect of CXCL12 on the spiral ganglion, spiral ganglion neurons
were treated in medium containing 100 ng/ml CXCL12 (Sigma-Aldrich,
St. Louis, MO, USA) with or without 20 μg/ml AMD3100 (CXCR4
antagonist; Sigma-Aldrich). The cells were maintained in a
humidified 5% CO2 incubator at 37°C. The expression
level of CXCR4 in spiral ganglion neurons was detected following
incubation at 37°C for 7 days. Microtubule-associated protein 2
(MAP2) was also detected after the 7-day incubation. To evaluate
spiral ganglion neuron survival and neurite outgrowth in the stripe
assays, the maintenance medium was supplemented with 30 ng/ml
recombinant brain-derived neurotrophic factor (BDNF; Sigma-Aldrich)
for a further 4 days of incubation.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from cells using TRIzol
reagent (Invitrogen; Thermo Fisher Scientific, Inc.) according to
the manufacturer's instructions, and treated with DNase
(Sigma-Aldrich) to remove contaminating genomic DNA. cDNA was
synthesized from 5 μg total RNA using the PrimeScript RT
Reagent kit (Takara Biotechnology Co., Ltd., Dalian, China). The
primers used in the present study were as follows: Forward,
5′-GTTTGCTTTGGAGCTTCTCG-3′ and reverse, 5′-GCTCTGGTGGAAGGTTGCTA-3′
for CXCL12; forward, 5′-GCCATGGCTGACTGGTACTT-3′ and reverse,
5′-GATGAAGGCCAGGATGAGAA-3′ forCXCR4; and forward,
5′-ACCCATCACCATCTTCCAGGAG-3′ and reverse,
5′-GAAGGGGCGGAGATGATGAC-3′ for glycer-aldehyde-3-phosphate
dehydrogenase (GAPDH). qPCR was performed using the SsoFast
EvaGreen Supermix (Bio-Rad Laboratories, Inc., Hercules, CA, USA)
on the 7500 Fast Real-Time PCR System (Applied Biosystems; Thermo
Fisher Scientific, Inc.) under the following conditions: 94°C for 4
min, followed by 35 cycles at 94°C for 20 sec, 55°C for 30 sec and
72°C for 20 sec. The data were analyzed using the 2−ΔΔCq
method (14). GAPDH served as the
internal control for normalization of gene expression.
Western blot analysis
Proteins from the spiral ganglion neurons were
extracted by homogenization in lysis buffer containing protease
inhibitors (both Beyotime Institute of Biotechnology, Haimen,
China). Protein concentration was measured using a Bradford protein
assay kit. The proteins (40 μg) were separated by 10–15%
sodium dodecyl sulfate-polyacrylamide gel electrophoresis and
transferred to a nitrocellulose membrane (Bio-Rad Laboratories,
Inc.). Membranes were blocked with 3% skimmed milk for 1 h at room
temperature, then incubated with rabbit anti-CXCR4 (1:800; cat. no.
ab2074; Abcam, Cambridge, UK), anti-MAP2 (1:500; cat. no. M3696;
Sigma-Aldrich), anti-caspase-3 (1:500; cat. no. sc-98785; Santa
Cruz Biotechnology, Inc., Dallas, TX, USA), anti-cleaved caspase-3
(1:200; cat. no. ab2302; Abcam) and anti-β-actin (1:1,000; cat. no.
sc-130656; Santa Cruz Biotechnology, Inc.) polyclonal antibodies
overnight at 4°C. Subsequently, the membranes were washed three
times for 15 min each with Tris-buffered saline containing
Tween-20, followed by incubation with goat anti-rabbit horseradish
peroxidase-conjugated secondary polyclonal antibody (1:2,000; cat.
no. bs-0295G-HRP; BIOSS, Beijing, China) for ~1 h at 37°C. The
blots were visualized using ECL Prime Western Blotting Detection
Reagent (GE Healthcare Life Sciences, Little Chalfont, UK) and the
ChemiScope 5300 Pro gel imaging system (Clinx Science Instruments
Co., Ltd., Shanghai, China). Protein band intensities were
quantified using Image-Pro Plus software version 6.0 (Media
Cybernetics, Inc., Rockville, MD, USA) and data were obtained by
normalization to the control group.
ELISA
Freshly isolated spiral ganglion specimens were
obtained from the mice. The protein extract was centrifuged and the
concentration of CXCL12 in the supernatant was determined using a
commercially available CXCL12 ELISA kit (cat. no. CSB-E04723m;
Cusabio Biotech Co., Ltd., Wuhan, China) according to the
manufacturer's protocol.
Quantification of neurite outgrowth
Neurite outgrowth from the spiral ganglion neurons
was assessed according to the number and lengths of the processes.
Neurite outgrowth was observed from digital images of 7–10 randomly
selected fields (magnification, ×20), as previously described
(15). The entire length of the
longest process from each neuron was determined using a TE300 Nikon
semi-automatic microscope (Nikon Corporation, Tokyo, Japan). The
mean neurite length per cell (the maximal possible distance along a
neurite) and the mean number of neurites per cell were
measured.
Immunohistochemistry
To prepare samples for immunohistochemical staining,
mice were sacrificed by an intramuscular injection of ketamine (100
mg/kg) 48 h after intraperitoneal injection of AMD3100 (2.5 mg/kg),
and the spiral ganglion was isolated as described previously
(16). The spiral ganglion samples
were fixed with 4% paraformaldehyde, then embedded in paraffin
(Sigma-Aldrich) and cut into 5-μm serial sections using a
microtome (Leica RM2016; Leica Microsystems GmbH, Wetzlar,
Germany). Spiral ganglion sections were incubated overnight at 4°C
with a rabbit polyclonal neuron-specific enolase antibody (1:250;
cat. no. ab53025; Abcam). Sections were then incubated with
biotinylated goat anti-rabbit IgG (1:1,000; cat. no. bs-0295G-Bio;
BIOSS) for 40 min at 37°C, followed by incubation with pre-prepared
streptavidin-biotinylated alkaline phosphatase complex (Beyotime
Institute of Biotechnology) for 20 min. The sections were
counterstained with Mayer's hematoxylin solution (Sigma-Aldrich).
For immunofluorescence detection, the cochleae were sectioned at a
10-μm thickness across the modiolus. The sections were
permeabilized with 0.5% Triton X-100 (Sangon Biotech Co., Ltd.) for
5 min, blocked with 5% goat serum (Gibco; Thermo Fisher Scientific,
Inc.) for 1 h, and then incubated with polyclonal rabbit
anti-CXCL12 (1:100; ab2074; Abcam) and anti-CXCR4 antibodies
overnight at 4°C. Subsequently, the sections were washed three
times for 5 min each with phosphate-buffered saline and then
incubated for 1 h at room temperature with the appropriate
secondary antibodies (1:1,000), as follows: CXCR4, Alexa Fluor
488-conjugated goat anti-rabbit polyclonal antibody (cat. no.
ab150077; Abcam); and CXCL12, Alexa Fluor 647-conjugated donkey
anti-rabbit polyclonal antibody (cat. no. ab150075; Abcam).
Subsequently, the nuclei were stained with
4′-6-diamidino-2-phenylindole dihydrochloride (1 μg/ml;
BIOSS) for 10 min at room temperature. The fluorescence was
observed with an Olympus BX51 fluorescence microscope (Olympus
Corporation, Tokyo, Japan), and the fluorescence intensity was
quantified using ImageJ 1.48u software (https://imagej.nih.gov/ij/).
Statistical analysis
All results from the current study are presented as
means ± standard deviation. Statistical differences were assessed
using Student's t-test or one-way analysis of variance and
P<0.05 was considered to indicate a statistically significant
difference.
Results
CXCL12 and CXCR4 are upregulated in
spiral ganglion neurons during cochlear development
To investigate whether CXCL12/CXCR4 is important in
the development of cochleae, the expression levels of CXCL12 and
CXCR4 in the cochlea of neonatal C57BL/6J mice were assessed using
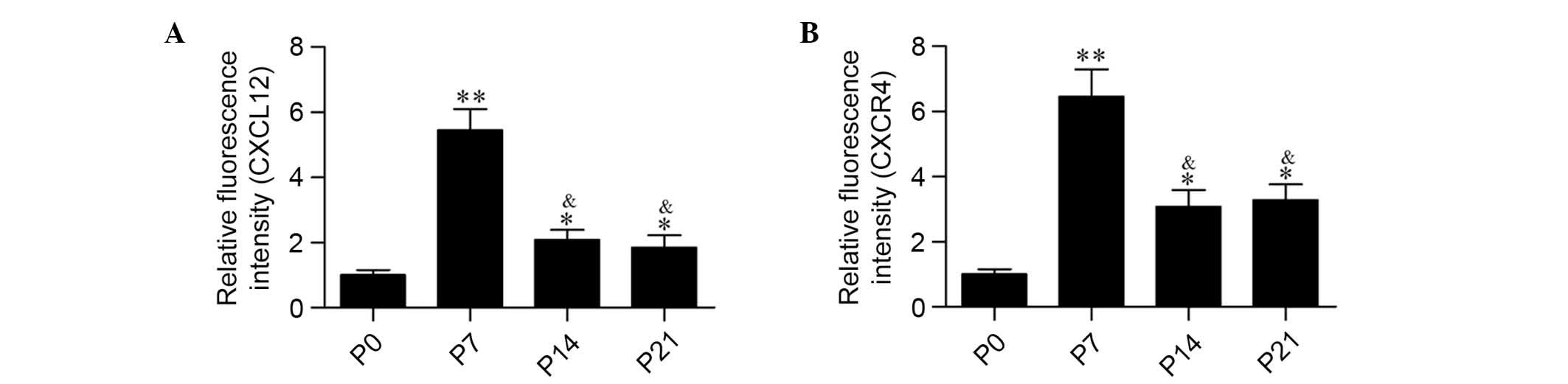
immunofluorescence (Fig. 1) and
the relative intensities were quantified (Fig. 2). Compared with postnatal day 0
(P0), the protein expression levels of CXCL12 and CXCR4 were
significantly increased in spiral ganglion neurons on P7
(P<0.01). Furthermore, the protein expression levels of CXCL12
and CXCR4 at P14 and P21 were decreased compared with P7
(P<0.05), but remained significantly increased compared with P0
(P<0.05; Fig. 2).
To further confirm the expression pattern of CXCL12
and CXCR4 in spiral ganglion neurons, the mRNA and protein
expression levels of CXCL12 and CXCR4 were analyzed by RT-qPCR, and
western blotting and ELISA, respectively. The results demonstrated
that the mRNA expression levels of CXCL12 and CXCR4 were
significantly increased on P7 (P<0.01), P14 and P21 (P<0.05)
compared with P0, reaching a peak on P7 (Fig. 3A). Consistent with these findings,
the protein concentration of CXCL12 (Fig. 3B) and levels of CXCR4 (Fig. 3C and D) were also significantly
increased on P7 (P<0.01), P14 and P21 (P<0.05). Overall, the
results of the present study indicated that CXCL12 and CXCR4 may
participate in the development of the cochlea.
CXCL12 enhances the expression of CXCR4
in spiral ganglion neurons
CXCL12 and its receptor, CXCR4 are vital for central
nervous system development by mediating cell migration, enhancing
precursor cell proliferation and inducing neuronal circuit
formation (17). Thus, the current
study measured the expression of CXCR4 in the presence of CXCL12 to
confirm that the CXCL12/CXCR4 signaling pathway may be involved in
the development of spiral ganglion neurons. AMD3100, a CXCR4
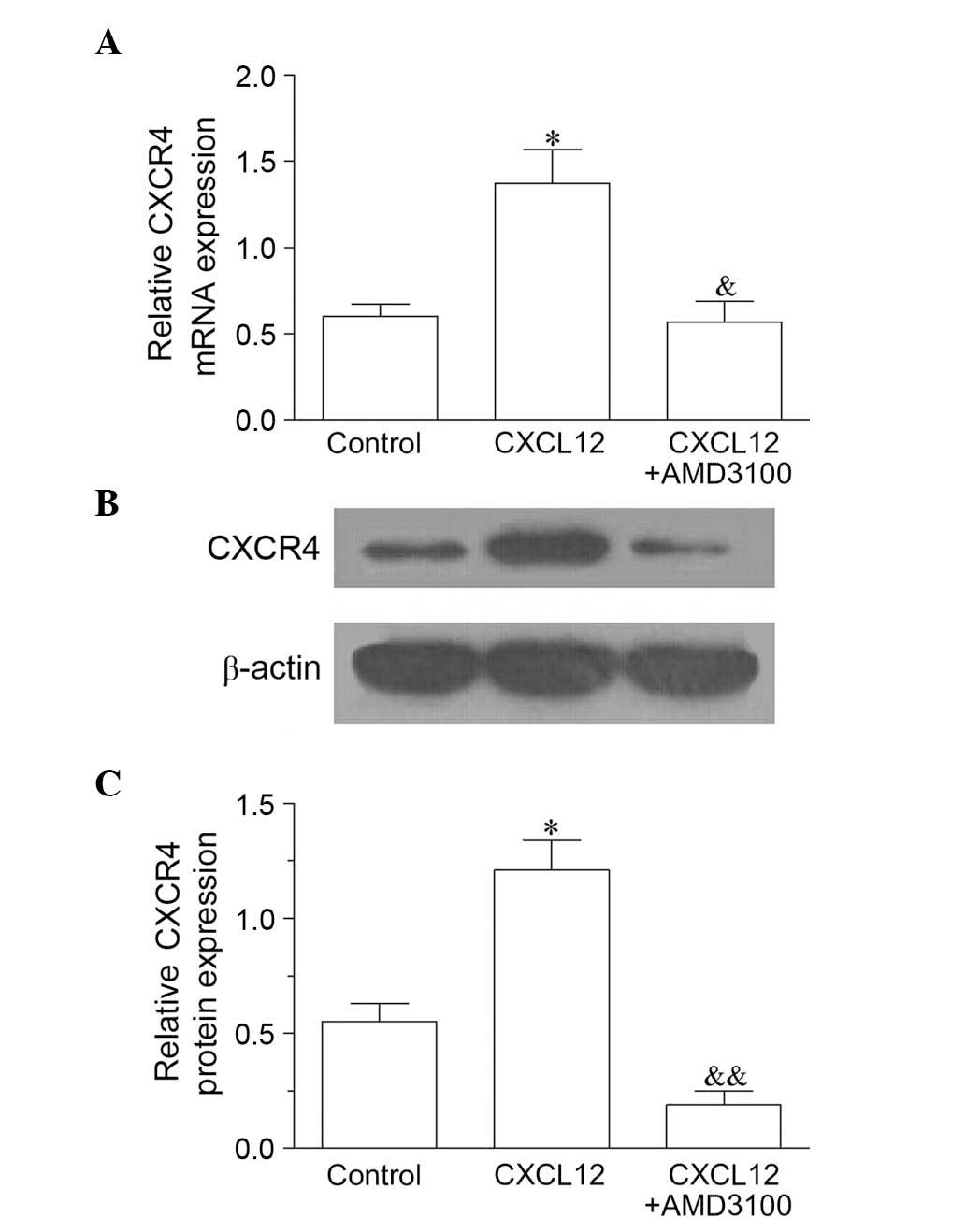
antagonist, inhibits the CXCL12/CXCR4 signaling pathway (18). As demonstrated in Fig. 4A–C, compared with the control, the
mRNA and protein expression levels of CXCR4 were significantly
increased following treatment with CXCL12. By contrast, AMD3100
treatment reversed the effect of CXCL12 as the CXCR4 mRNA
(P<0.05) and protein (P<0.01) levels were significantly
decreased by AMD3100 compared with CXCL12 treatment only (Fig. 4A–C).
Blocking CXCL12/CXCR4 signaling enhances
spiral ganglion neuron apoptosis
To verify that CXCL12/CXCR4 is important for the
regulation of cochlear development, CXCL12/CXCR4 signaling was
inhibited by AMD3100 and the effect on spiral ganglion neuron
apoptosis was evaluated. The present study demonstrated that
treatment with CXCL12 significantly decreased the protein
expression levels of caspase-3 and cleaved caspase-3 compared with
the control (P<0.05). However, compared with CXCL12 treatment,
the effect was significantly abrogated by AMD3100 (P<0.05;
Fig. 5). The results indicated
that CXCL12/CXCR4 signaling may inhibit cell apoptosis of spiral
ganglion neurons.
Blocking CXCL12/CXCR4 signaling impairs
the neurite growth of spiral ganglion neurons in vitro
To further confirm that CXCL12/CXCR4 signaling is
important for neuronal circuit formation in the spiral ganglion,
the current study analyzed its effect on neurite growth in
vitro. The protein expression levels of the neurite marker,
MAP2, were examined by western blot analysis. The results
demonstrated that the protein expression level of MAP2 in spiral
ganglion neurons treated with CXCL12 was significantly increased on
day 7 compared with the control group (P<0.05). However, AMD3100
treatment abolished this effect, as the level of MAP2 was
significantly reduced when compared with CXCL12 treatment (Fig. 6A and B). Furthermore, neurite
outgrowth from the spiral ganglion neurons was evaluated by
measuring the number and length of the neurites. When spiral
ganglion neurons were treated with CXCL12, the number of neurites
per cell increased compared with the control group (P<0.05;
Fig. 6C). Similarly, the average
neurite length in cells treated with CXCL12 was significantly
longer compared with the control group (P<0.05; Fig. 6D). By contrast, compared with
CXCL12 treatment only, the effects of CXCL12 on neurite number and
length were significantly abrogated by AMD3100 treatment
(P<0.05; Fig. 6C and D).
Blocking CXCL12/CXCR4 signaling impairs
the number and alters the morphology of spiral ganglion neurons in
vivo
To further confirm that the CXCL12/CXCR4 pathway is
involved in the development of spiral ganglion neurons, the present
study investigated the effect of CXCL12/CXCR4 inhibition on spiral
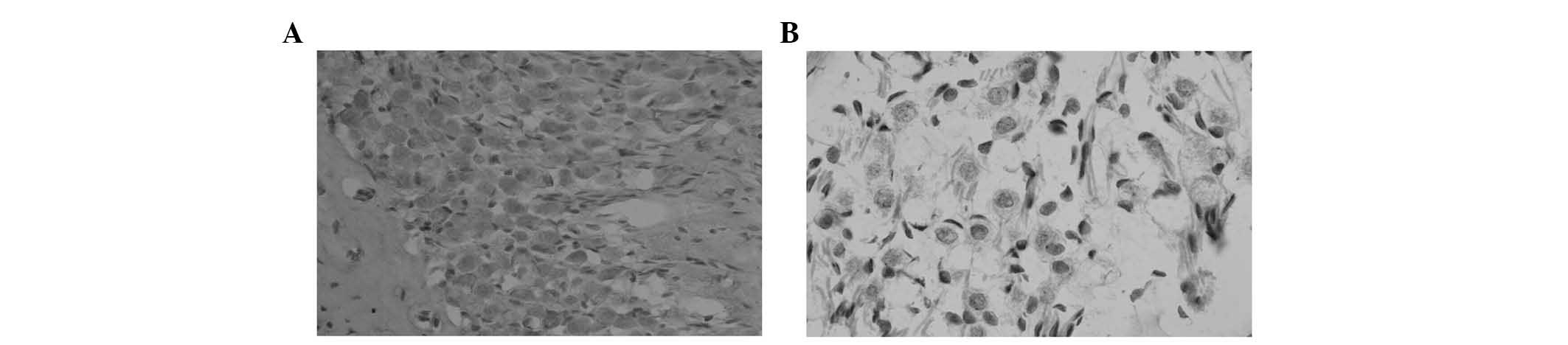
ganglion neurons in vivo. The results demonstrated that
spiral ganglion neurons were closely arranged with obviously
stained cell bodies in the control group (Fig. 7A). By contrast, the number of
spiral ganglion neurons was markedly reduced and the cells were
irregularly arranged with fuzzy cell morphology in mice treated
with AMD3100 (CXCR4 antagonist; Fig.
7B).
Discussion
In the present study, elevated CXCL12 and CXCR4
levels in the cochleae of neonatal C57BL/6J mice indicated that the
CXCL12/CXCR4 signaling pathway is vital in the process of auditory
sense formation. In addition, it was observed that treatment with
CXCL12 significantly increased spiral ganglion neuron survival and
neurite growth. This result was consistent with a previous
observation on dissociated spiral ganglion neurons (19).
Phosphatidylinositol-4,5-bisphosphate 3-kinase/Akt signaling
participates in mediating the effects of BDNF on spiral ganglion
neurons in vitro, including neuronal survival and neurite
extension (20). However, certain
neurotrophins exert opposing effects on spiral ganglion neurons and
alter the balance of competing signals to influence neurite
formation. For example, activation of Ras-related C3 botulinum
toxin substrate 1 (Rac)/cell division cycle 42 (cdc42)/c-Jun
N-terminal kinases (JNK) signaling by BDNF may reduce the
generation of neurites (20),
which conflicts with previous findings indicating that rat sarcoma
viral oncogene homolog or mitogen-activated protein kinase
kinase/extracellular signal-regulated kinase signaling was the
primary mediator of spiral ganglion neuron outgrowth in
vitro; the inhibition of which blocked the effects of
neurotrophin-3 on spiral ganglion neurons (21). Previous studies have demonstrated
that these effects on spiral ganglion neurons are mediated by
various signaling pathways, with neurite number being increased by
the p38 and Akt signaling pathway, but inhibited by the
Rac/cdc42/JNK signaling pathway. In the current study, neurite
number and length were increased when CXCL12/CXCR4 was activated,
indicating that the CXCL12/CXCR4 signaling pathway is involved in
the development of spiral ganglion neurons.
CXCL12 and CXCR4 are constitutively expressed in the
developing and mature central nervous system, stimulating a series
of downstream signaling pathways (22). During early developmental stages,
the expression of CXCR4 assists with the survival and proliferation
of cortical neuronal precursors. In the mature central nervous
system, the expression levels of CXCL12 and CXCR4 vary across a
wide range of differentiated cells (23,24).
In the hippocampus, neurogenesis and expression of CXCR4 persist
throughout life (25).
Additionally, following stroke, neuronal CXCR4 is upregulated in
areas of neurogenesis (25). These
observations indicate that CXCR4 possesses proliferative potential
in neuronal cells. The present study demonstrated that following
activation of the CXCL12/CXCR4 signaling pathway in spiral ganglion
neurons, cell apoptosis was significantly decreased. However,
inhibiting CXCL12/CXCR4 signaling significantly reversed the effect
of CXCL12. These results suggest that the CXCL12/CXCR4 signaling
pathway is important for the regulation of spiral ganglion neuron
survival.
Notably, CXCL12 and CXCR4 modulate synapse formation
and function. Normal neural function is required for connectivity,
neurites must find their targets and dendrites must achieve normal
morphology. A previous study demonstrated that activation of CXCR4
regulates the synaptic transmission of cerebellar and hippocampal
granule cells in vitro, and induces transmission at
nociceptive synapses (26).
Furthermore, activation of CXCR4 is involved in the formation of
parallel fiber synapses between cerebellar granule cells and
Purkinje cells, in which CXCL12 modulates calcium transients
(27–29). In the present study, the neurite
marker, MAP2 was detected by western blot analysis, confirming that
the CXCL12/CXCR4 signaling pathway was involved in neuronal circuit
formation of spiral ganglion neurons. In addition, the in
vivo morphology of spiral ganglion neurons was observed to
demonstrate that the CXCL12/CXCR4 signaling pathway was involved in
synapse formation.
A previous study demonstrated that the CXCL12
expression level is increased in the auditory nerve following
cochlear injury (30). Another
study provided further insight into the association between CXCL12
and transplanted stem cells following spiral ganglion neuron
degeneration (31). Upregulation
of CXCL12 in the host microenvironment and activation of CXCR4 in
donor stem cells improved the migration efficiency of transplanted
cells in the injured region of rat cochleae. However, the precise
function of the CXCL12/CXCR4 signaling pathway in the development
of the cochlea in neonatal mice remains to be determined.
In conclusion, the present study demonstrated that
the expression levels of CXCL12 and CXCR4 were increased in spiral
ganglion neurons during cochlear development. Treatment with CXCL12
increased cell survival and dendrite growth in spiral ganglion
neurons, an effect that was blocked by the CXCR4 antagonist,
AMD3100, implying that CXCL12/CXCR4 signaling is critical for
spiral ganglion neuron growth. Furthermore, inhibition of
CXCL12/CXCR4 signaling significantly reduced the number and altered
the morphology of spiral ganglion neurons in vivo.
Collectively, the current study suggests that the CXCL12/CXCR4
signaling pathway is important in cochlear development of neonatal
mice.
Abbreviations:
|
CXCL12
|
CXC chemokine ligand 12
|
|
CXCR4
|
CXC chemokine receptor 4
|
|
MAP2
|
microtubule-associated protein 2
|
|
BDNF
|
brain-derived neurotrophic factor
|
Acknowledgments
The present study was supported by the National
Natural Science Foundation of China (grant nos. 81100714, 81271070
and 81371099).
References
|
1
|
Bok J, Zenczak C, Hwang CH and Wu DK:
Auditory ganglion source of Sonic hedgehog regulates timing of cell
cycle exit and differentiation of mammalian cochlear hair cells.
Proc Natl Acad Sci USA. 110:13869–13874. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Tateya T, Imayoshi I, Tateya I, Hamaguchi
K, Torii H, Ito J and Kageyama R: Hedgehog signaling regulates
prosensory cell properties during the basal-to-apical wave of hair
cell differentiation in the mammalian cochlea. Development.
140:3848–3857. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Barclay M, Ryan AF and Housley GD: Type I
vs type II spiral ganglion neurons exhibit differential survival
and neuritogenesis during cochlear development. Neural Dev.
6:332011. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Leake PA, Hradek GT, Hetherington AM and
Stakhovskaya O: Brain-derived neurotrophic factor promotes cochlear
spiral ganglion cell survival and function in deafened, developing
cats. J Comp Neurol. 519:1526–1545. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Dabdoub A, Puligilla C, Jones JM, Fritzsch
B, Cheah KS, Pevny LH and Kelley MW: Sox2 signaling in prosensory
domain specification and subsequent hair cell differentiation in
the developing cochlea. Proc Natl Acad Sci USA. 105:18396–18401.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Yang S, Edman LC, Sánchez-Alcañiz JA,
Fritz N, Bonilla S, Hecht J, Uhlén P, Pleasure SJ, Villaescusa JC,
Marín O and Arenas E: Cxcl12/Cxcr4 signaling controls the migration
and process orientation of A9-A10 dopaminergic neurons.
Development. 140:4554–4564. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zou YR, Kottmann AH, Kuroda M, Taniuchi I
and Littman DR: Function of the chemokine receptor CXCR4 in
haematopoiesis and in cerebellar development. Nature. 393:595–599.
1998. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Nagasawa T: CXC chemokine ligand 12
(CXCL12) and its receptor CXCR4. J Mol Med Berl. 92:433–439. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Ma Q, Jones D, Borghesani PR, Segal RA,
Nagasawa T, Kishimoto T, Bronson RT and Springer TA: Impaired
B-lymphopoiesis, myelopoiesis, and derailed cerebellar neuron
migration in CXCR4- and SDF-1-deficient mice. Proc Natl Acad Sci
USA. 95:9448–9453. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhu Y, Matsumoto T, Mikami S, Nagasawa T
and Murakami F: SDF1/CXCR4 signalling regulates two distinct
processes of precerebellar neuronal migration and its depletion
leads to abnormal pontine nuclei formation. Development.
136:1919–1928. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Stumm RK, Zhou C, Ara T, Lazarini F,
Dubois-Dalcq M, Nagasawa T, Höllt V and Schulz S: CXCR4 regulates
interneuron migration in the developing neocortex. J Neurosci.
23:5123–5130. 2003.PubMed/NCBI
|
|
12
|
Lieberam I, Agalliu D, Nagasawa T, Ericson
J and Jessell TM: A Cxcl12-CXCR4 chemokine signaling pathway
defines the initial trajectory of mammalian motor axons. Neuron.
47:667–679. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
García-Hernández S, Potashner SJ and
Morest DK: Role of fibroblast growth factor 8 in neurite outgrowth
from spiral ganglion neurons in vitro. Brain Res. 1529:39–45. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Livak and Schmittgen: Analysis of relative
gene expression data using real-time quantitative PCR and the
2−ΔΔCt method. Methods. 25:402–408. 2001. View Article : Google Scholar
|
|
15
|
Xu N, Engbers J, Khaja S, Xu L, Clark JJ
and Hansen MR: Influence of cAMP and protein kinase A on neurite
length from spiral ganglion neurons. Hear Res. 283:33–44. 2012.
View Article : Google Scholar :
|
|
16
|
Wang F, Gao X, Chen J, Liu SL, Wang FY,
Hei RY, Chen Y and Qiu JH: Effect of early postnatal air-conduction
auditory deprivation on the development and function of the rat
spiral ganglion. J Laryngol Otol. 125:917–923. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Peng H, Kolb R, Kennedy JE and Zheng J:
Differential expression of CXCL12 and CXCR4 during human fetal
neural progenitor cell differentiation. J Neuroimmune Pharmacol.
2:251–258. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Schols D, Struyf S, Van Damme J, Esté JA,
Henson G and De Clercq E: Inhibition of T-tropic HIV strains by
selective antagonization of the chemokine receptor CXCR4. J Exp
Med. 186:1383–1388. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Hartnick CJ, Staecker H, Malgrange B,
Lefebvre PP, Liu W, Moonen G and Van de Water TR: Neurotrophic
effects of BDNF and CNTF, alone and in combination, on postnatal
day 5 rat acoustic ganglion neurons. J Neurobiol. 30:246–254. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Mullen LM, Pak KK, Chavez E, Kondo K,
Brand Y and Ryan AF: Ras/p38 and PI3K/Akt but not Mek/Erk signaling
mediate BDNF-induced neurite formation on neonatal cochlear spiral
ganglion explants. Brain Res. 1430:25–34. 2012. View Article : Google Scholar
|
|
21
|
Aletsee C, Beros A, Mullen L, Palacios S,
Pak K, Dazert S and Ryan AF: Ras/MEK but not p38 signaling mediates
NT-3-induced neurite extension from spiral ganglion neurons. J
Assoc Res Otolaryngol. 2:377–387. 2001. View Article : Google Scholar
|
|
22
|
Li M and Ransohoff RM: Multiple roles of
chemokine CXCL12 in the central nervous system: A migration from
immunology to neurobiology. Prog Neurobiol. 84:116–131. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Banisadr G, Fontanges P, Haour F, Kitabgi
P, Rostène W and Mélik Parsadaniantz S: Neuroanatomical
distribution of CXCR4 in adult rat brain and its localization in
cholinergic and dopaminergic neurons. Eur J Neurosci. 16:1661–1671.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Krumbholz M, Theil D, Cepok S, Hemmer B,
Kivisäkk P, Ransohoff RM, Hofbauer M, Farina C, Derfuss T, Hartle
C, et al: Chemokines in multiple sclerosis: CXCL12 and CXCL13
up-regulation is differentially linked to CNS immune cell
recruitment. Brain. 129:200–211. 2006. View Article : Google Scholar
|
|
25
|
Stumm RK, Rummel J, Junker V, Culmsee C,
Pfeiffer M, Krieglstein J, Höllt V and Schulz S: A dual role for
the SDF-1/CXCR4 chemokine receptor system in adult brain:
Isoform-selective regulation of SDF-1 expression modulates
CXCR4-dependent neuronal plasticity and cerebral leukocyte
recruitment after focal ischemia. J Neurosci. 22:5865–5878.
2002.PubMed/NCBI
|
|
26
|
Oh SB, Tran PB, Gillard SE, Hurley RW,
Hammond DL and Miller RJ: Chemokines and glycoprotein120 produce
pain hypersensitivity by directly exciting primary nociceptive
neurons. J Neurosci. 21:5027–5035. 2001.PubMed/NCBI
|
|
27
|
Limatola C, Giovannelli A, Maggi L,
Ragozzino D, Castellani L, Ciotti MT, Vacca F, Mercanti D, Santoni
A and Eusebi F: SDF-1α-mediated modulation of synaptic transmission
in rat cerebellum. Eur J Neurosci. 12:2497–2504. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gillard SE, Lu M, Mastracci RM and Miller
RJ: Expression of functional chemokine receptors by rat cerebellar
neurons. J Neuroimmunol. 124:16–28. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ragozzino D, Renzi M, Giovannelli A and
Eusebi F: Stimulation of chemokine CXC receptor 4 induces synaptic
depression of evoked parallel fibers inputs onto Purkinje neurons
in mouse cerebellum. J Neuroimmunol. 127:30–36. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Kilpatrick LA, Zhu J, Lee FS and Lang H:
Role of stromal cell-derived factor-1 expression in the injured
mouse auditory nerve. Otolaryngol Head Neck Surg. 145:1007–1015.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zhang PZ, He Y, Jiang XW, Chen FQ, Chen Y,
Xue T, Zhou K, Li X, Wang Y, Wu YX, et al: Up-regulation of stromal
cell-derived factor-1 enhances migration of transplanted neural
stem cells to injury region following degeneration of spiral
ganglion neurons in the adult rat inner ear. Neurosci Lett.
534:101–106. 2013. View Article : Google Scholar
|