Introduction
Patients with irritable bowel syndrome (IBS) often
suffer from multiple symptoms in the gastrointestinal tract and in
other organ systems (1). A
previous study by our laboratory demonstrated high proportions of
chronic fatigue and musculoskeletal pain in IBS patients referred
to Lovisenberg Diaconal Hospital (Oslo, Norway) for investigation
of perceived food hypersensitivity (2). Notably, these extra-intestinal or
systemic symptoms may be more debilitating than the intestinal
complaints (3). It has been
hypothesized that these common comorbid symptoms may share a common
pathogenesis (4). However, the
cause remains to be elucidated.
A lactulose challenge test to induce symptoms in
patients with IBS has been developed by our laboratory (5) and was used as a tool in our previous
study (6); other studies have used
a modified test (7,8). Ingestion of a small dose of lactulose
may elicit abdominal and systemic symptoms, including chills and
fatigue. This suggests that the intestines may be the primary site
of the ailment, and that blood borne substances may be involved in
symptom generation. These putative factors remain unidentified.
However, previous studies have indicated a low-grade intestinal
inflammation in patients with IBS (9), and consequent increased intestinal
permeability (10).
Therefore, the present study aimed to investigate
the association between symptoms induced during a lactulose
challenge test, and circulating levels of biomarkers indicating
enterocyte disintegrity [intestinal fatty acid binding protein
(iFABP)], endotoxemia [lipopolysaccharide (LPS) and the LPS
co-receptor soluble cluster of differentiation (sCD)14], and
inflammation [monocyte chemoattractant protein-1 (MCP-1) and
calprotectin]. Alterations in any one of these factors may indicate
disruption of the intestinal mucosal barrier and consequently,
immune activation, and thus potentially explain systemic symptoms
in response to lactulose ingestion.
Subjects and methods
Study participants
Patients with IBS were recruited consecutively from
the outpatient clinic of A.B. at Lovisenberg Diaconal Hospital
(Oslo, Norway) between November 2011 and January 2013. The patients
were diagnosed with IBS according to the Rome III criteria
(11). Patients with organic
diseases of the gastrointestinal tract were excluded. Healthy
controls were recruited from the staff at Lovisenberg Diaconal
Hospital. They were included if they considered themselves to be
healthy and were not otherwise medically examined. However, they
were excluded if they met the Rome III criteria for IBS, based on
questionnaires. Written informed consent was obtained from all
participants. The study was performed according to the Declaration
of Helsinki and approved by the regional Committee for Medical
Research Ethics (REK Sør-Øst; reference number 2011/2474).
Outline of study design
Participants were assessed following an overnight
fast. Questionnaires for grading of habitual symptoms were filled
in. Subsequently, the subjects underwent a lactulose challenge
test, in which a solution of 10 g lactulose dissolved in 200 ml tap
water was ingested. The participants filled in questionnaires
assessing habitual and lactulose-induced symptoms and were not
allowed to eat or drink during the assessment. Blood samples were
obtained prior to and 90 min following lactulose ingestion, and
were rapidly transferred into a −20°C freezer for storage until
analysis. To minimalize interassay variability, samples from
patients and controls were analyzed in the same run.
Assessment of symptoms
Habitual symptoms were assessed in all patients
using previously validated questionnaires. IBS severity was
measured according to the IBS Severity Scoring System (IBS-SSS)
(12). Grading of chronic fatigue
was estimated according to the Fatigue Impact Scale (FIS) (13). Severity of musculoskeletal pain was
graded using Visual Analogue Scales (VAS) (14). The increase in extra-intestinal
symptoms following lactulose ingestion was graded from 0 to 3 for
sickness and chills, giving a maximum achievable score of 6 points.
Increase in intestinal symptoms following lactulose ingestion was
graded from 0 to 3 for borborygmi, bloating and pain, giving a
maximum achievable score of 9 points.
Analysis of blood samples
iFABP was analyzed in the serum using a commercial
ELISA kit (catalog no. HK 406; Hycult Biotech, Uden, The
Netherlands), according to the manufacturer's protocol. LPS was
analyzed in plasma using a Limulus Amebocyte Lysate colorimetric
assay (Lonza, Walkersville, MD, USA), according to the
manufacturer's protocol, with the following modifications: Samples
were diluted 10-fold to avoid interference with background color
and were preheated to 68°C for 12 min prior to analysis to dissolve
immune complexes, as previously described by Trøseid et al
(15). sCD14 was analyzed in
plasma using a commercial ELISA kit (R&D Systems, Inc.,
Minneapolis, MN, USA), according to the manufacturer's protocol.
MCP-1 was analyzed in serum using a commercial
Quantikine® ELISA kit (R&D Systems Europe, Ltd.,
Abingdon, UK), according to the manufacturer's protocol.
Calprotectin was analyzed in serum using a commercial ELISA kit
(Bühlmann Laboratories AG, Schönenbuch, Switzerland), according to
the manufacturer's protocol.
Statistical analysis
The data were analyzed in GraphPad Prism (version 6;
GraphPad Software, Inc., La Jolla, CA, USA). In the text, data are
expressed as the mean ± standard deviation. In the figures, data
are expressed as mean ± standard error. Paired Student's t-tests
were performed to compare means within groups and unpaired
Student's t-tests were performed to compare means between groups.
Correlations were assessed using Pearson's correlation coefficient.
All tests were two-tailed. P<0.05 was considered to indicate a
statistically significant difference.
Results
Study participants
A total of 94 IBS patients (30 male and 64 female;
mean age, 40 years; range, 19–73 years) and 20 healthy controls (8
male and 12 female; mean age, 38 years; range, 25–65 years) were
recruited consecutively to participate in the present study. Of the
IBS patients, 41 were classified as diarrhea-predominant IBS
(IBS-D), 15 as constipation-predominant IBS (IBS-C) and 38 as IBS
with mixed stool pattern (IBS-M).
Symptoms
As determined by IBS-SSS, habitual IBS symptoms were
classified as mild in none of the patients, moderate in 42 patients
and severe in 52 patients. The mean score for musculoskeletal pain
according to the VAS was 30.3±25.2 cm, and the mean score for
fatigue according to the FIS was 83.4±48.3 points. The mean scores
for lactulose-induced symptoms were 4.0±2.4 and 2.0±1.8 points for
intestinal and extra-intestinal symptoms in patients. In healthy
controls, the scores were 0.4±2.8 and 0±0 points, respectively.
Biomarkers
The numbers of blood samples from analyzed
consecutive individuals (patients/healthy controls) were as
follows: iFABP (48/19), LPS (26/12), sCD14 (11/11), MCP-1 (10/10),
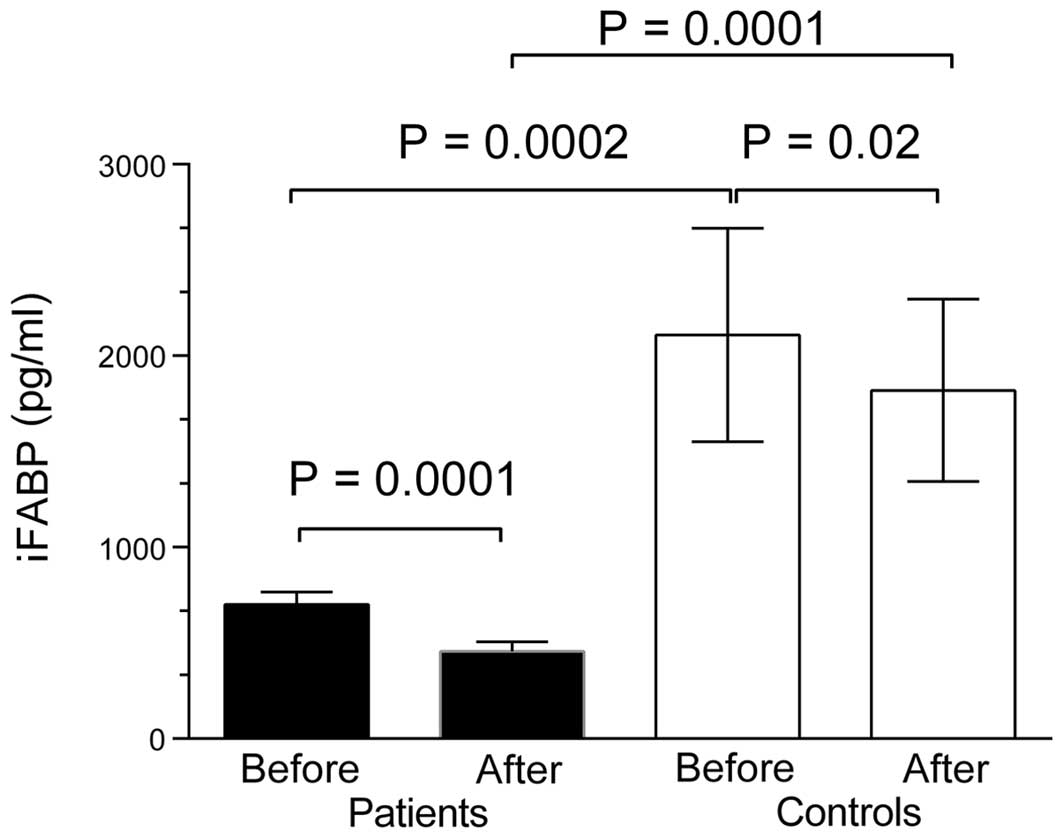
and calprotectin (10/10). Serum levels of iFABP were reduced in IBS
patients compared with healthy controls, prior to (702±442 vs.
2,110±2,429 pg/ml; P=0.0002) and 90 min following lactulose
ingestion (402±344 vs. 1,820±2,022 pg/ml; P=0.0001). Following
lactulose ingestion, iFABP levels decreased in healthy controls
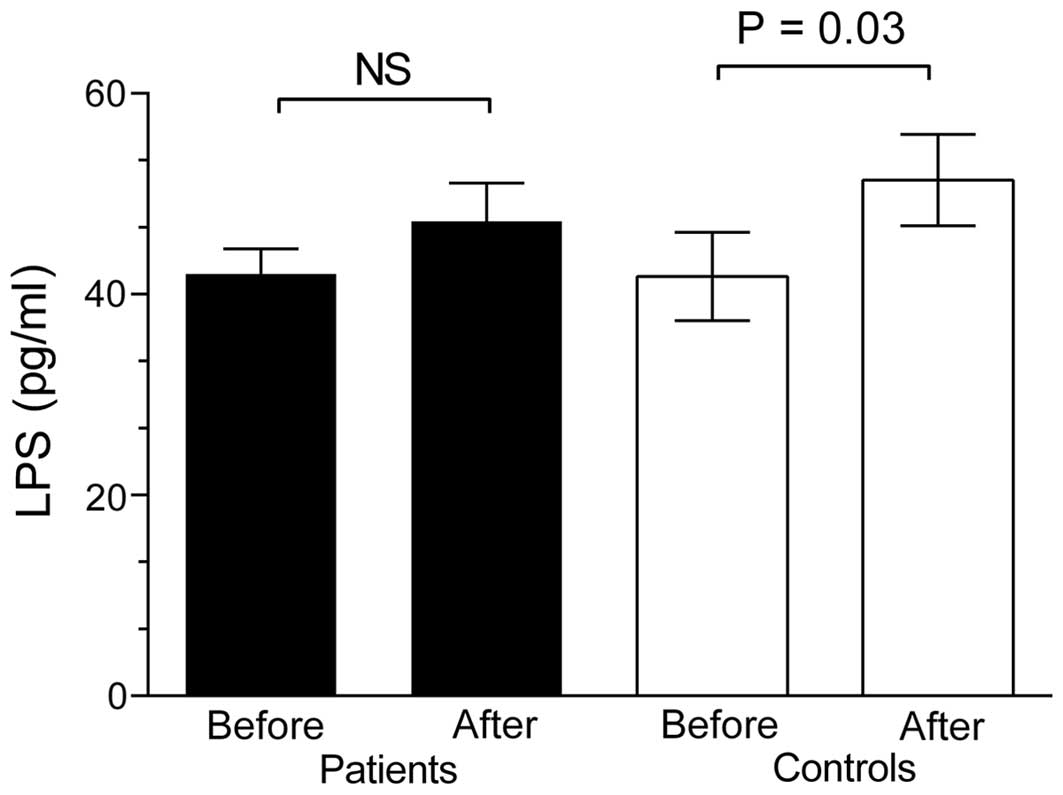
(P=0.02) and in IBS patients (P=0.0001; Fig. 1). Fasting levels of LPS were not
significantly different in IBS patients (42±15 pg/ml) compared with
healthy controls (42±15 pg/ml); however, following lactulose
ingestion, LPS levels increased in healthy controls (P=0.03),
whereas the levels did not increase significantly in IBS patients
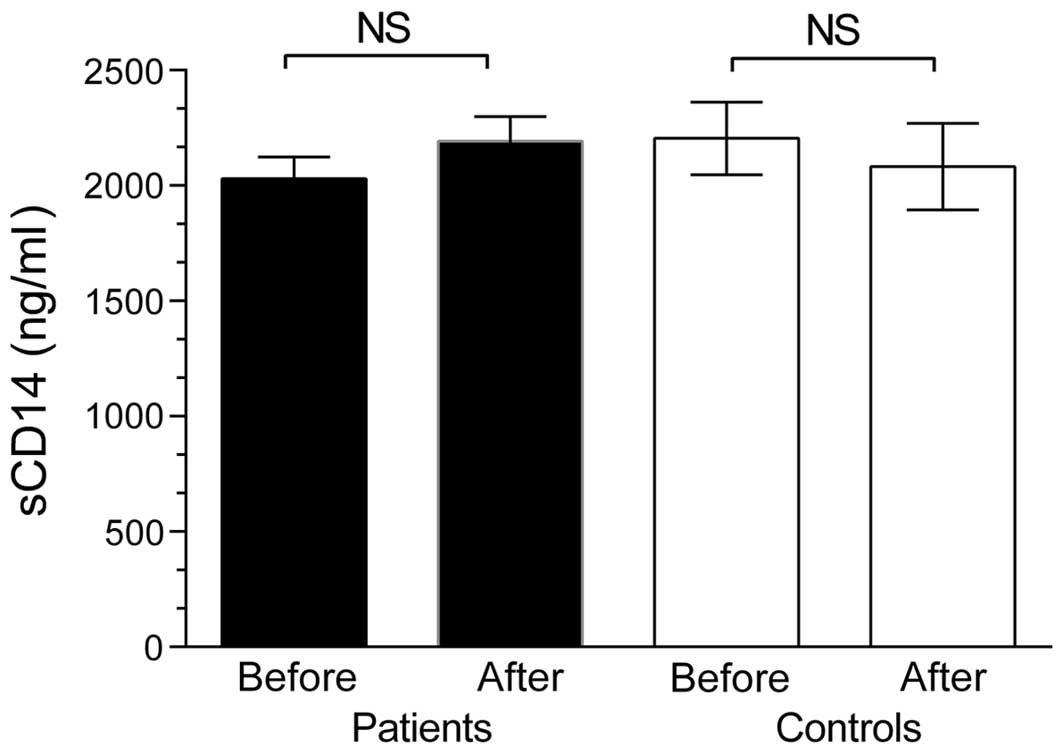
(Fig. 2). Fasting levels of sCD14
were not significantly different in IBS patients (2,030±560 ng/ml)
compared with healthy controls (2,204±544 ng/ml), and levels were
not altered by lactulose ingestion, in IBS patients or in healthy
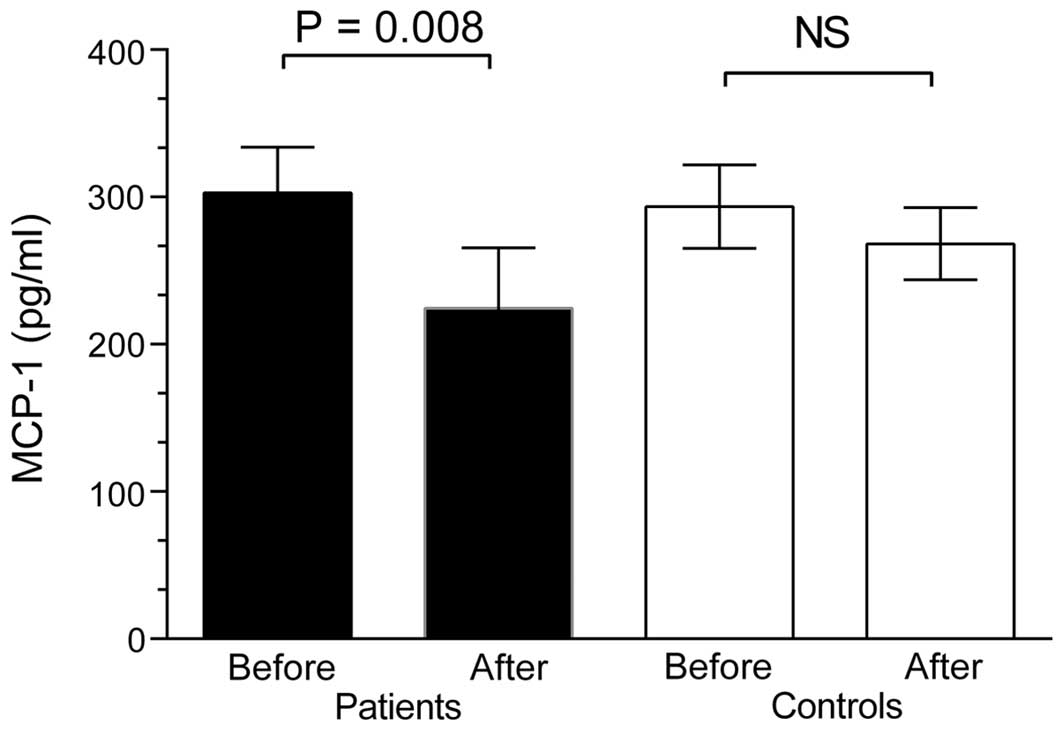
controls (Fig. 3). Fasting levels
of MCP-1 were not significantly different in IBS patients (303±98
pg/ml) compared with healthy controls (294±90 pg/ml). Following
lactulose ingestion, MCP-1 levels decreased in IBS patients
(P=0.008), whereas the levels did not alter significantly in the
healthy controls (Fig. 4). No
significant differences were observed in the serum levels of
calprotectin in patients compared with healthy controls prior to
(4.2±1.2 vs. 4.2±1.22 µg/ml; P=0.99) or following (P=0.37)
lactulose administration (Fig. 5).
The levels of biomarkers were similar in patients classified as
IBS-D, IBS-C and IBS-M. No correlations were observed between any
of the biomarkers and habitual or lactulose-induced symptoms.
Discussion
The present study revealed no evidence of enterocyte
disintegrity, endotoxemia or inflammation in patients with IBS,
prior to or following lactulose provocation. Therefore, the results
do not support the hypothesis that IBS-associated systemic symptoms
are due to mucosal damage, as measured by iFABP, leakage of
microbial endotoxin, as measured by LPS/sCD14 or inflammation, as
measured by MCP-1 or calprotectin in blood.
The FABP family is comprised of 14–15 kDa
intracellular proteins expressed in intestinal epithelial cells
(16). iFABP is present throughout
the intestine, with the greatest expression in the jejunum
(17). Following intestinal
mucosal damage, iFABP is released into the circulation and its
plasma concentration increases, for example in patients with
untreated celiac disease (18),
following surgical trauma, in severe sepsis or in intestinal
malignant disease (19). Although
various previous studies refer to low-grade inflammation and
impaired intestinal permeability in IBS patients (20–22),
the present study observed low levels of serum iFABP in patients,
and a further decrease following lactulose administration.
Therefore, no indication of mucosal damage was determined in
patients with IBS, prior to or following lactulose challenge. In
accordance with these results, a recent study reported that serum
iFABP is not a useful biomarker of intestinal barrier dysfunction
driven by gut microbiota changes in obesity (23). Therefore, marginally increased
intestinal permeability, as reported in IBS and obesity (21,22),
may not be a consequence of mucosal damage measurable by iFABP.
Passage of small molecules (<600 Da) through
tight junctions of the intestinal mucosa is regulated by
cell-to-cell adhesion proteins (20). The iFABP molecule is too large to
cross the mucosal barrier through these intercellular spaces. A
molecule that may pass through paracellular spaces without overt
epithelial damage, although which still requires impaired barrier
function, is LPS from Gram-negative bacteria. Following
intestinal-to-blood leakage, LPS activates immune cells via its
co-receptor CD14, a surface antigen expressed on monocytes and
macrophages. In patients with overt mucosal damage, as in
inflammatory bowel disease (IBD), plasma levels of LPS correlate
with disease severity (18). In
diseases with subtle mucosal damage, associated with insulin
resistance and low-grade intestinal inflammation, conditions often
referred to as ‘metabolic endotoxemia’, LPS may be a causal or
complicating factor (24). Whether
lactulose challenge may impair mucosal barrier function at a tight
junction level, without causing ‘mucosal damage’ as measured by
iFABP, remains unclear. Intravenous infusion of LPS in healthy
volunteers triggers visceral hypersensitivity with characteristics
similar to those observed in IBS patients (25), and elevated plasma levels of LPS
were associated with IBS (IBS-D) in a recent study (26). A separate study demonstrated
elevated serum immunoglobulin A against LPS in patients with
fatigue (27), a condition often
linked to IBS (28). In the
present study, similar blood levels of LPS in IBS patients and
healthy controls were observed, with a slight increase in the mean
LPS levels in the two groups following lactulose administration
(statistically significant only in the healthy controls). These
results suggested that lactulose ingestion may increase intestinal
leakage of LPS; however, the lactulose-induced leakage of LPS was
not specific for IBS patients, and thus may not explain their
systemic symptoms. It is possible that the increase in LPS is
associated with the decrease in iFABP. LPS contains fatty acids and
low levels of iFABP may be a consequence, or an indication, of a
defence reaction to subtle LPS stimulation. Obesity is
characterized by subtle leakage of LPS into the circulation
(24), and low levels of iFABP
have been reported in obese patients (23).
The proinflammatory cytokine, MCP-1, is critical for
monocyte recruitment in inflammatory conditions. In the present
study, similar levels of MCP-1 in patients and healthy controls
were observed, and these levels decreased significantly in patients
following lactulose challenge. In addition, calprotectin levels
were similar in patients and healthy controls and tended to
decrease (non-significantly) following lactulose challenge. A
previous study revealed elevated MCP-1 levels in IBS patients
(29). The reason for this
contradictory data remains to be elucidated.
In conclusion, the results of the present study
revealed no indication of mucosal damage, endotoxemia or
inflammation in the IBS patients assessed. Notably, patients had
abnormally low values of iFABP, a finding that requires further
investigation.
Acknowledgements
The authors sincerely thank Dr Marius Trøseid at the
Institute of Clinical Medicine, University of Oslo (Oslo, Norway)
for helpful academic advice and for performing the LPS and sCD14
analyses.
References
|
1
|
Sperber AD and Dekel R: Irritable bowel
syndrome and co-morbid gastrointestinal and extra-gastrointestinal
functional syndromes. J Neurogastroenterol Motil. 16:113–119. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Berstad A, Undseth R, Lind R and Valeur J:
Functional bowel symptoms, fibromyalgia and fatigue: A food-induced
triad? Scand J Gastroenterol. 47:914–919. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Vandvik PO, Lydersen S and Farup PG:
Prevalence, comorbidity and impact of irritable bowel syndrome in
Norway. Scand J Gastroenterol. 41:650–656. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kim SE and Chang L: Overlap between
functional GI disorders and other functional syndromes: What are
the underlying mechanisms? Neurogastroenterol Motil. 24:895–913.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Valeur J, Morken MH, Norin E, Midtvedt T
and Berstad A: Carbohydrate intolerance in patients with
self-reported food hypersensitivity: Comparison of lactulose and
glucose. Scand J Gastroenterol. 44:1416–1423. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Undseth R, Berstad A, Kløw NE, Arnljot K,
Moi KS and Valeur J: Abnormal accumulation of intestinal fluid
following ingestion of an unabsorbable carbohydrate in patients
with irritable bowel syndrome: An MRI study. Neurogastroenterol
Motil. 26:1686–1693. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Le Nevé B, Posserud I, Böhn L, Guyonnet D,
Rondeau P, Tillisch K, Naliboff B, Mayer EA and Simrén M: A
combined nutrient and lactulose challenge test allows symptom-based
clustering of patients with irritable bowel syndrome. Am J
Gastroenterol. 108:786–795. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Le Nevé B, Brazeilles R, Derrien M, Tap J,
Guyonnet D, Ohman L, Törnblom H and Simrén M: Lactulose challenge
determines visceral sensitivity and severity of symptoms in
patients with irritable bowel syndrome. Clin Gastroenterol Hepatol.
14:226-233.e1–e3. 2016.
|
|
9
|
Ohman L and Simrén M: Pathogenesis of IBS:
Role of inflammation, immunity and neuroimmune interactions. Nat
Rev Gastroenterol Hepatol. 7:163–173. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Wilcz-Villega E, McClean S and O'Sullivan
M: Reduced E-cadherin expression is associated with abdominal pain
and symptom duration in a study of alternating and diarrhea
predominant IBS. Neurogastroenterol Motil. 26:316–325. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Longstreth GF, Thompson WG, Chey WD,
Houghton LA, Mearin F and Spiller RC: Functional bowel disorders.
Gastroenterology. 130:1480–1491. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Francis CY, Morris J and Whorwell PJ: The
irritable bowel severity scoring system: A simple method of
monitoring irritable bowel syndrome and its progress. Aliment
Pharmacol Ther. 11:395–402. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fisk JD, Ritvo PG, Ross L, Haase DA,
Marrie TJ and Schlech WF: Measuring the functional impact of
fatigue: Initial validation of the fatigue impact scale. Clin
Infect Dis. 18:(Suppl 1). S79–S83. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Aaron LA, Burke MM and Buchwald D:
Overlapping conditions among patients with chronic fatigue
syndrome, fibromyalgia, and temporomandibular disorder. Arch Intern
Med. 160:221–227. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Trøseid M, Nowak P, Nyström J, Lindkvist
A, Abdurahman S and Sonnerborg A: Elevated plasma levels of
lipopolysaccharide and high mobility group box-1 protein are
associated with high viral load in HIV-1 infection: Reduction by
2-year antiretroviral therapy. AIDS. 24:1733–1737. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Gajda AM and Storch J: Enterocyte fatty
acid-binding proteins (FABPs): Different functions of liver and
intestinal FABPs in the intestine. Prostaglandins Leukot Essent
Fatty Acids. 93:9–16. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sacchettini JC, Hauft SM, Van Camp SL,
Cistola DP and Gordon JI: Developmental and structural studies of
an intracellular lipid binding protein expressed in the ileal
epithelium. J Biol Chem. 265:19199–19207. 1990.PubMed/NCBI
|
|
18
|
Bottasso Arias NM, García M, Bondar C,
Guzman L, Redondo A, Chopita N, Córsico B and Chirdo FG: Expression
pattern of fatty acid binding proteins in celiac disease
Enteropathy. Mediators Inflamm. 2015:7385632015.PubMed/NCBI
|
|
19
|
Bingold TM, Franck K, Holzer K,
Zacharowski K, Bechstein WO, Wissing H and Scheller B: Intestinal
fatty acid binding protein: A sensitive marker in abdominal surgery
and abdominal infection. Surg Infect (Larchmt). 16:247–253. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
González-Castro AM, Martínez C,
Salvo-Romero E, Fortea M, Pardo-Camacho C, Pérez-Berezo T,
Alonso-Cotoner C, Santos J and Vicario M: Mucosal pathobiology and
molecular signature of epithelial barrier dysfunction in the small
intestine in Irritable Bowel Syndrome. J Gastroenterol Hepatol.
18–Apr;2016.(Epub ahead of print). View Article : Google Scholar
|
|
21
|
Arslan G, Kahrs GE, Lind R, Frøyland L,
Florvaag E and Berstad A: Patients with subjective food
hypersensitivity: The value of analyzing intestinal permeability
and inflammation markers in gut lavage fluid. Digestion. 70:26–35.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Berstad A, Arslan G and Folvik G:
Relationship between intestinal permeability and calprotectin
concentration in gut lavage fluid. Scand J Gastroenterol. 35:64–69.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Lau E, Marques C, Pestana D, Santoalha M,
Carvalho D, Freitas P and Calhau C: The role of I-FABP as a
biomarker of intestinal barrier dysfunction driven by gut
microbiota changes in obesity. Nutr Metab (Lond). 13:312016.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Cani PD, Amar J, Iglesias MA, Poggi M,
Knauf C, Bastelica D, Neyrinck AM, Fava F, Tuohy KM, Chabo C, et
al: Metabolic endotoxemia initiates obesity and insulin resistance.
Diabetes. 56:1761–1772. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Layé S, Parnet P, Goujon E and Dantzer R:
Peripheral administration of lipopolysaccharide induces the
expression of cytokine transcripts in the brain and pituitary of
mice. Brain Res Mol Brain Res. 27:157–162. 1994. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Dlugosz A, Nowak P, D'Amato M, Mohammadian
KG, Nyström J, Abdurahman S and Lindberg G: Increased serum levels
of lipopolysaccharide and antiflagellin antibodies in patients with
diarrhea-predominant irritable bowel syndrome. Neurogastroenterol
Motil. 27:1747–1754. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Maes M, Mihaylova I and Leunis JC:
Increased serum IgA and IgM against LPS of enterobacteria in
chronic fatigue syndrome (CFS): Indication for the involvement of
gram-negative enterobacteria in the etiology of CFS and for the
presence of an increased gut-intestinal permeability. J Affect
Disord. 99:237–240. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Han CJ and Yang GS: Fatigue in irritable
bowel syndrome: A systematic review and meta-analysis of pooled
frequency and severity of Fatigue. Asian Nurs Res (Korean Soc Nurs
Sci). 10:1–10. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Tülübaş F, Oran M, Mete R, Turan F, Yilmaz
A, Yildiz ZD and Gürel A: Investigation of serum macrophage
migration inhibitor factor and monocyte chemotactic protein-1
levels in irritable bowel syndrome. Turk J Med Sci. 44:967–971.
2014. View Article : Google Scholar : PubMed/NCBI
|