Introduction
Some nutrients present in food possessed some
medicinal properties and may be used for the prevention and/or
treatment of different diseases. Generally, these nutrients are
called nutraceutic, a term derived from ‘nutrition’ and
‘pharmaceutics’ (1). Nutraceutics
received a lot of attention because of their potential therapeutic
effects, but also thanks to their safety. Indeed, given that they
are natural compounds, nutraceutics can be safer and without
adverse reactions. Among nutraceutical compounds, glucosinolates
(GLSs) and flavonoids are among the most investigated
categories.
In the last decades the consumption of members of
the Brassicaceae family increased, because they are able to exert
beneficial effects on human health, due to their content in
nutraceutics (2). In particular,
Brassicaceae are known to contain GLSs, that mediate some of the
health benefits, but the health promoting effects seem to be
associated also with the presence of phenolic compounds, including
flavonoids (3,4). In particular, Brassica seeds seemed
very rich in GLSs (5).
Brassicaceae family includes vegetables normally consumed in our
diet such as broccoli, Brussels sprouts, cabbage and collards.
Another member of this family is Eruca sativa, generally
known as rocket salad, that is an edible plant widespread in the
Mediterranean area, known for its medicinal properties. Indeed,
Eruca sativa extracts were reported to exert antioxidant,
antiplatelet, antithrombotic, anticancer and antimicrobial
properties (6–9). Eruca sativa, including its
seeds, contained different compounds known for their nutraceutic
potential, including GLSs and flavonoids (9,10).
Interestingly, these classes of nutraceutics have shown
neuroprotective effects, thank to their antioxidant and
anti-inflammatory activities (11–13),
suggesting their use for the prevention of neurodegenerative
diseases.
The anti-inflammatory action of these compounds is
particularly interesting considering that inflammation plays a main
role in different pathologies. Indeed, inflammation acts as a
defensive response when a tissue is damaged by toxins or infective
agents in order to repair the tissue and eliminate the causative
factors, but, when it is prolonged, it may be detrimental. In
particular, nervous system is more sensitive to inflammation
compared to other tissues, given its limited regeneration capacity.
Indeed, neuroinflammation has been associated with different
disorders, including neurodegenerative diseases (14,15).
During neuroinflammation, different mechanisms cause neuronal
degeneration and death, including the release of different
cytokines by activated microglia and the infiltration of the
central nervous system by immune cells from the periphery (14). Given this, the research is focused
on the discovery of new anti-inflammatory compounds that can be
helpful for the treatment and/or prevention of neurodegenerative
disorders.
The aim of this study was the evaluation of the
anti-inflammatory potential of Eruca sativa seed extract
(ESE). Specifically, we evaluated the anti-inflammatory and
neuroprotective effects exerted by ESE pre-treatment in NSC-34
motor neurons exposed to the culture medium of lipopolysaccharide
(LPS)-stimulated RAW 264.7 macrophages.
Materials and methods
ESE preparation and
characterization
Eruca sativa seeds were supplied by SAIS spa
(Cesena, Italy; license: no. 12, 6/7/1973; authorization from
Regione Emilia Romagna 43) and guaranteed by the producer for the
quality and the genetic homogeneity of the product. Seeds were
soaked for 15 min in 1% sodium hypochlorite and washed with tap
water. Subsequently, the seeds were air dried at a temperature of
30°C for approximately 8–12 min, were ground and again air dried
with forced ventilation. ESE was characterized by HPLC
analysis.
For cell culture experiments, ESE was dissolved in
dimethyl sulfoxide (DMSO) and diluted in the cell culture
medium.
RAW 264.7 macrophage cell culture and
treatment
RAW 264.7, a murine macrophage cell line obtained
from ‘Centro Substrati Cellulari, Istituto Zooprofilattico
Sperimentale della Lombardia e dell'Emilia’ (Brescia, Italy) were
cultured in monolayer at 37°C in a moisturized atmosphere of 5%
CO2 and 95% air using DMEM-high glucose medium
(Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) supplemented with
10% fetal bovine serum (FBS; Sigma-Aldrich; Merck KGaA). In order
to induce an inflammatory state, cells were grown until 70–80%
confluence was reached, and after they were incubated with 1 µg/ml
LPS from Escherichia coli 0111:B4 (Sigma-Aldrich; Merck KGaA) for
24 h. Untreated cells were used as control. At the end of the
treatment, cells were either fixed or harvested for other analyses,
while the culture medium was collected to carry out experiments
with NSC-34 cells. All the experiments were made in triplicates and
repeated three independent times.
NSC-34 motor neurons cell culture and
treatment with the culture medium of LPS-stimulated RAW 264.7
NSC-34 motor neurons, a murine cell line acquired
from Cedarlane/CELLutions Biosystems Inc. (Burlington, ON, Canada),
were cultured in DMEM-high glucose medium (Sigma-Aldrich; Merck
KGaA) with the addiction of 10% FBS (Sigma-Aldrich; Merck KGaA), at
37°C in a moisturized atmosphere of 5% CO2 and 95% air.
We evaluated the cytotoxicity of ESE, exposing NSC-34 cells for 24
h to different concentrations of ESE. In particular, we tested the
following concentrations of ESE: 0.1, 0.2, 0.3 and 0.4 µg/ml. To
examine anti-inflammatory effects of ESE against LPS-induced
inflammation, NSC-34 cells were pre-treated for 24 h with ESE. At
the end of pre-treatment, medium was replaced with cell-free
conditioned medium from LPS-stimulated RAW 264.7 macrophage, and
incubated for 24 h. As controls, NSC-34 were incubated with the
medium of unstimulated RAW 264.7. Cells treated with vehicle
(<0.1% DMSO) or with the ESE alone were also included as
controls. Then, motor neuronal cells were fixed for
eosin/hematoxylin staining or immunocytochemistry analysis. All the
experiments were done in triplicate and repeated for three
independent times.
Eosin and hematoxylin staining
RAW 264.7 and NSC-34 morphological changes were
evaluated by eosin and hematoxylin staining applying a standard
protocol. Cells, grown on coverslips (10 mm; Thermo Fisher
Scientific, Darmstadt, Germany), were fixed with 4%
paraformaldehyde (Santa Cruz Biotechnology, Inc., Dallas, TX, USA)
for 15 min at room temperature. Cells were washed with 1X
phosphate-buffered saline (PBS) and stained with hematoxylin Harris
(Bio-Optica, Milan, Italy) for 1 min, rinsed with tap water,
stained with eosin (Bio-Optica) for 5 min, and rinsed with
distilled water. After, coverslips were dehydrated with ethanol
series (Carlo Erba Reagents, Valde-Reuil, France; 50, 70, 80, 96,
and 100%) and xylene (J.T. Baker, Deventer, The Netherlands).
Subsequently, coverslips were mounted on microscope slides with
mounting medium (Eukitt, Carlo Erba Reagents, Valde-Reuil, France)
and allowed to dry. Microscopy was performed using a light
microscope (Leica DM 2000 combined with Leica ICC50 HD camera) with
the objective ×20 (for RAW 264.7 macrophages) or ×40 (for NSC-34
motoneurons). All images are representative of three independent
experiments.
Immunocytochemistry
RAW 264.7 macrophages and NSC-34 motor neurons were
grown on coverslips (10 mm; Thermo Fisher Scientific, Oberhausen,
Germany) and, at the end of the treatments, they were fixed with 4%
paraformaldehyde at room temperature for 15 min and after washed
with PBS (pH 7.5). Then, cells were incubated with 3% hydrogen
peroxide (H2O2) at room temperature for 15
min to suppress the endogenous peroxidase activity. After three
washes with PBS, cells were blocked with horse serum +0.1% Triton
X-100 for 20 min and incubated overnight at 4°C with primary
antibodies. In particular, RAW 264.7 cells were incubated with the
following antibodies against examined proteins: mouse monoclonal
antibody against interleukin (IL)-1β (cat. no. 12242; 1:100; Cell
Signaling Technology, Inc., Danvers, MA, USA), rabbit polyclonal
antibody against Fas Ligand (FasL; cat. no. ab15285; 1:100; Abcam,
Cambridge, UK), goat polyclonal antibody against P-selectin (cat.
no. sc-6943; 1:50; Santa Cruz Biotechnology, Inc.) and rat
monoclonal antibody against IL-10 (cat. no. sc-73309; 1:100; Santa
Cruz Biotechnology, Inc.).
NSC-34 were incubated with the following primary
antibodies against examined proteins: rabbit polyclonal antibody
against tumor necrosis factor-α (TNF-α; cat. no. 3707; 1:250; Cell
Signaling Technology, Inc.), rat monoclonal antibody against IL-10
(cat. no. sc-73309; 1:100; Santa Cruz Biotechnology, Inc.), rabbit
polyclonal antibody against FasL (cat. no. ab15285; 1:100; Abcam),
mouse monoclonal antibody against cyclooxygenase 2 (COX2; cat. no.
sc-166475; 1:100; Santa Cruz Biotechnology, Inc.), rabbit
monoclonal antibody against caspase 1 (cat. no. ab108362; 1:100;
Abcam), rat monoclonal antibody against NLR family pyrin domain
containing 3 (NLRP3; cat. no. MAB7578; 1:100; R&D Systems,
Minneapolis, MN, USA), rabbit polyclonal antibody against IL-18
(cat. no. ab191152; 1:100; Abcam), mouse monoclonal antibody
against IL-1β (cat. no. 12242; 1:100; Cell Signaling Technology,
Inc.) and mouse monoclonal antibody against Toll-like receptor 4
(TLR4; cat. no. ab22048; 1:100; Abcam).
After, cells were washed with PBS and incubated with
biotinylated secondary antibody (1:200; Vector Laboratories, Inc.,
Burlingame, CA, USA) and streptavidin AB Complex-HRP (ABC-kit from
Dako, Glostrup, Denmark). The immunostaining was developed with the
DAB peroxidase substrate kit (Vector Laboratories, DBA Italia
S.r.l., Milan, Italy; brown color; positive staining) and
counterstaining with nuclear fast red (Vector Laboratories, DBA
Italia S.r.l.; pink background; negative staining).
The immunocytochemical assays were repeated three
times and each experimental group was plated in duplicate, for a
total of 6 coverslips for each antibody. With the aim of
calculating the percentage of positive cells stained, the images
were captured using a light microscopy (LEICA DM 2000 combined with
LEICA ICC50 HD camera) with an objective ×40 and the densitometric
analysis was carried out using the software LEICA Application Suite
ver. 4.2.0. Quantitative analysis was performed on 6 coverslips by
covering approximately 90% of the total area.
Protein extraction and western blot
analysis
At the end of the treatment, RAW 264.7 cells were
harvested and lysed using buffer A [320 mM sucrose, 10 mM, 1 mM
EGTA, 2 mM EDTA, 5 mM NaN3, 50 mM NaF, β-mercaptoethanol, and
protease/phosphatase inhibitor cocktail (Roche Molecular
Diagnostics, Branchburg, NJ, USA)] in ice for 15 min, and
centrifuged at 1,000 × g for 10 min at 4°C. The supernatant was
collected as cytosolic extract. The obtained pellet was lysed using
buffer B [150 mM NaCl, 10 mM Tris-HCl (pH 7.4), 1 mM EGTA, 1 mM
EDTA, Triton X-100, and protease/phosphatase inhibitor cocktail
(Roche Molecular Diagnostics)] in ice for 15 min and centrifuged at
15,000 × g for 30 min at 4°C. The supernatant was harvested as
nuclear extract. Protein concentrations were analyzed using the
Bradford assay (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Twenty µg of proteins were heated for 5 min at 95°C and resolved by
SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and after
transferred onto a PVDF membrane (Immobilon-P, Millipore, DBA
Italia S.r.l., Milan, Italy).
After, membranes were blocked with 5% skim milk in
PBS for 1 h at room temperature and incubated overnight at 4°C with
the following primary antibodies: Rabbit polyclonal antibody
against TNF-α (cat. no. 3707; 1:500; Cell Signaling Technology,
Inc.) and rat monoclonal antibody against IL-10 (cat. no. sc-73309;
1:500; Santa Cruz Biotechnology, Inc.). After the membranes were
washed with PBS 1X and incubated with horse radish peroxidase
(HRP)-conjugated goat anti-rabbit or chicken anti-rat IgG secondary
antibody (cat. nos. sc-2004 and sc-2956, respectively; 1:2,000;
Santa Cruz Biotechnology, Inc.) for 1 h at room temperature. In
order to evaluate if blots were loaded with equal amounts of
proteins, membranes were incubated with antibody for glyceraldehyde
3-phosphate dehydrogenase (GAPDH) HRP Conjugated (cat. no. 3683;
1:1,000; Cell Signaling Technology, Inc.). The relative expression
of protein bands was analyzed using an enhanced chemiluminescence
system (Luminata Western HRP Substrates; Millipore, DBA Italia
S.r.l.) and protein bands were acquired with ChemiDoc™ MP System
(Bio-Rad Laboratories, Inc.) and quantified using the computer
program ImageJ software. All blots are representative of three
independent experiments.
Statistical data analysis
Statistical analysis was performed using GraphPad
Prism version 6.0 computer software program (GraphPad Software,
Inc., La Jolla, CA, USA). The data were statistically analyzed by
Student's t-test for the comparison between 2 groups or by one-way
ANOVA test and Bonferroni post hoc test for multiple comparisons.
P<0.05 was considered to indicate a statistically significant
difference. Results are reported as mean ± SEM of N
experiments.
Results
Characterization of Eruca sativa
seed
ESE was chemically characterized by HPLC. Analysis
showed that glucoerucin was the GLS present in higher quantity in
ESE, and in particular the extract contained 46.36 mg glucoerucin/g
of ESE. Moreover, the ESE contained 0.16 mg/g dry weight of
ascorbic acid and 1.99 mg/g dry weight of total flavonoids.
Evaluation of the presence of
inflammation in LPS-treated RAW 264.7 macrophages
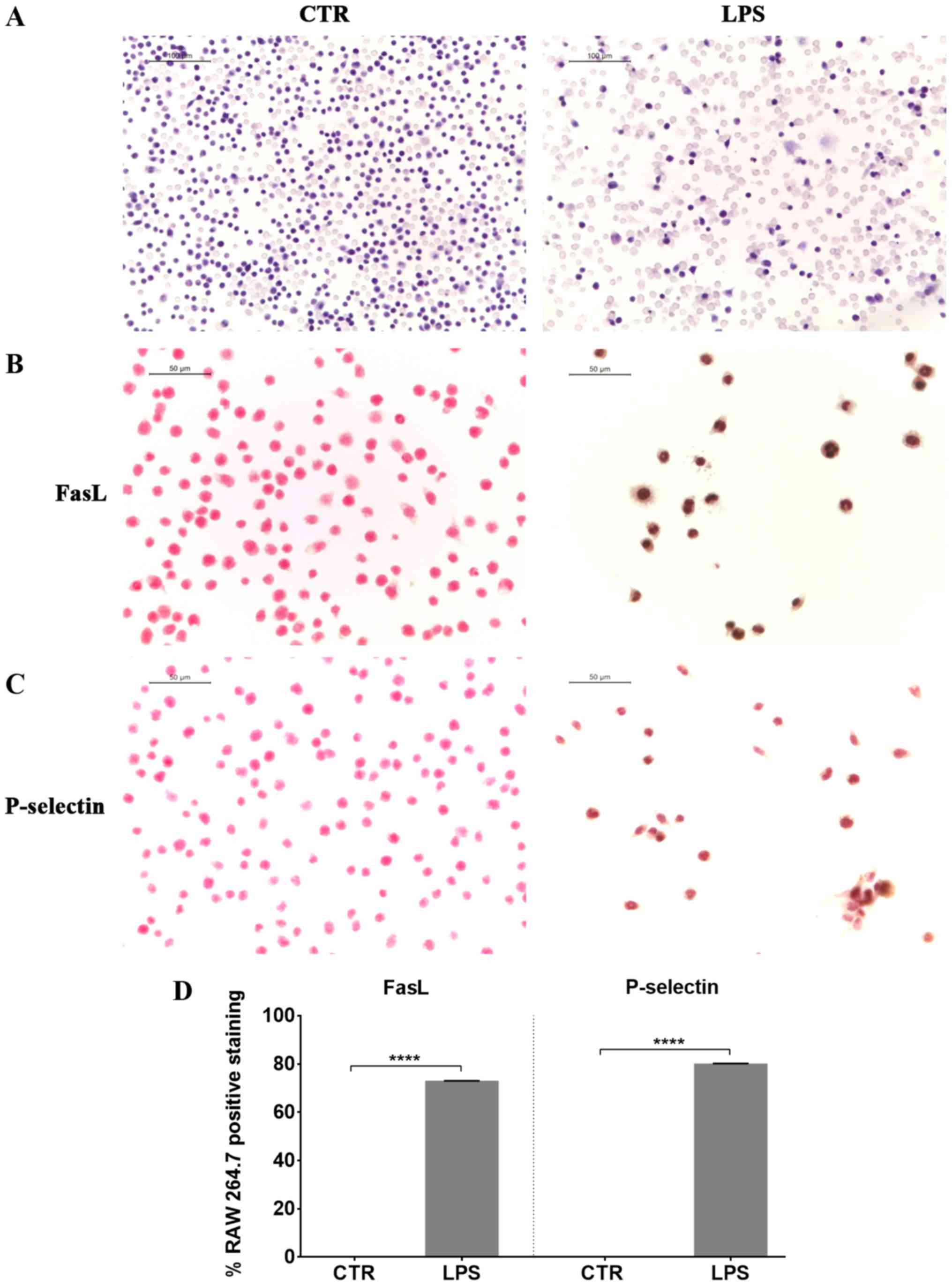
At first we induced inflammation in RAW 264.7 cells,
exposing them to LPS. Eosin and hematoxylin staining of LPS
stimulated RAW 264.7 showed morphological changes, such as an
increase in cell size and the extension of lamellipodia and
filopodia (Fig. 1). We observed a
positive staining for FasL in LPS treated cells compared to
controls (Fig. 1), indicating the
induction of apoptosis in LPS-treated macrophages. Furthermore, RAW
264.7 macrophages exposed to LPS presented a positive staining for
P-selectin (Fig. 1).
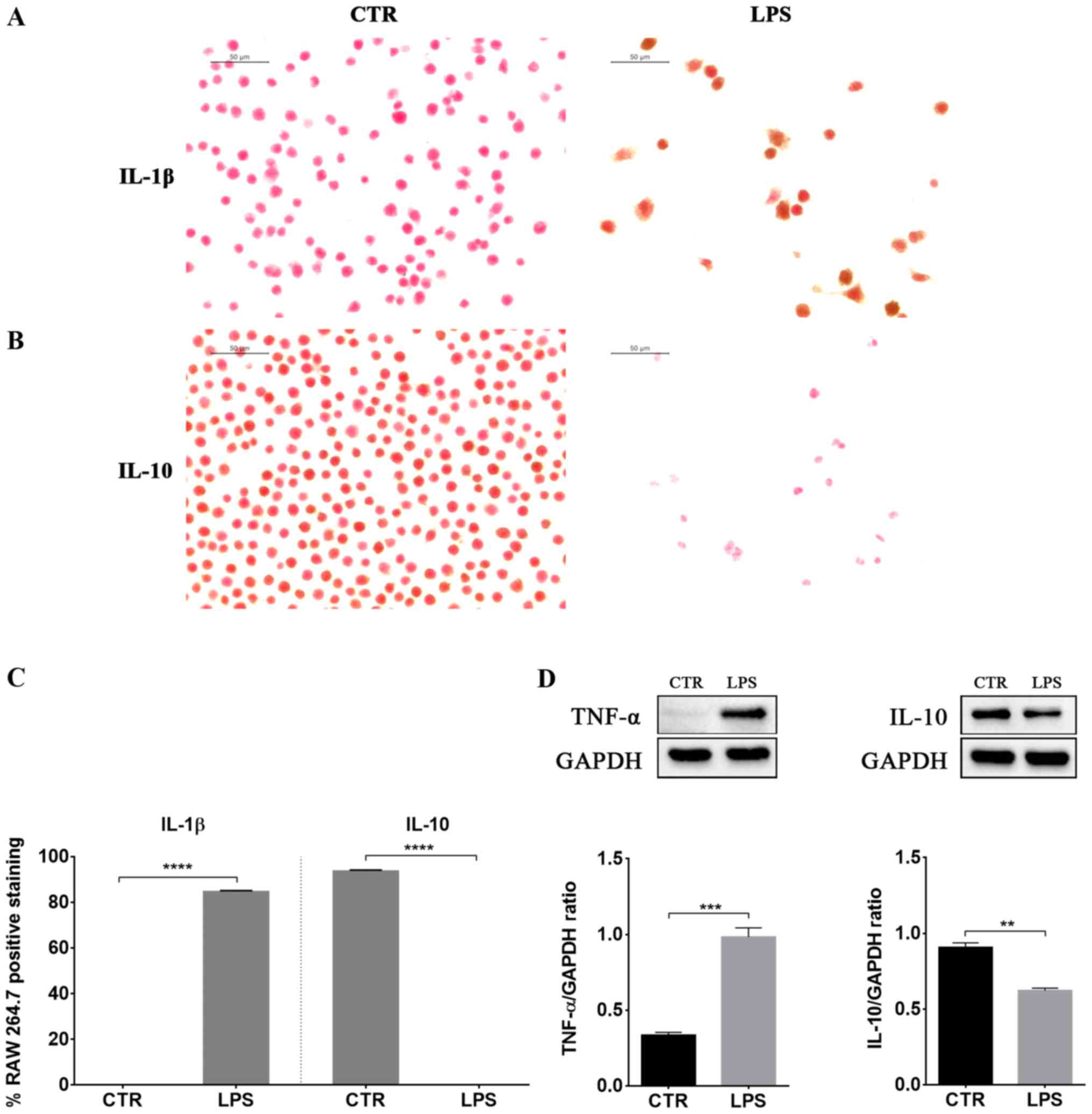
LPS-induced inflammation was associated with
increased protein levels of the pro-inflammatory cytokines TNF-α
and IL-1β (Fig. 2). Instead, IL-10
levels, a well known anti-inflammatory cytokine, decreased in LPS
stimulated macrophages compared to control cells (Fig. 2).
Evaluation of ESE anti-inflammatory
and neuroprotective effects in NSC-34 motor neurons exposed to the
medium of LPS-treated RAW 264.7
After confirming the presence of inflammation in
macrophages, we evaluated whether ESE was able to counteract the
inflammation induced in NSC-34 by the medium of LPS-treated RAW
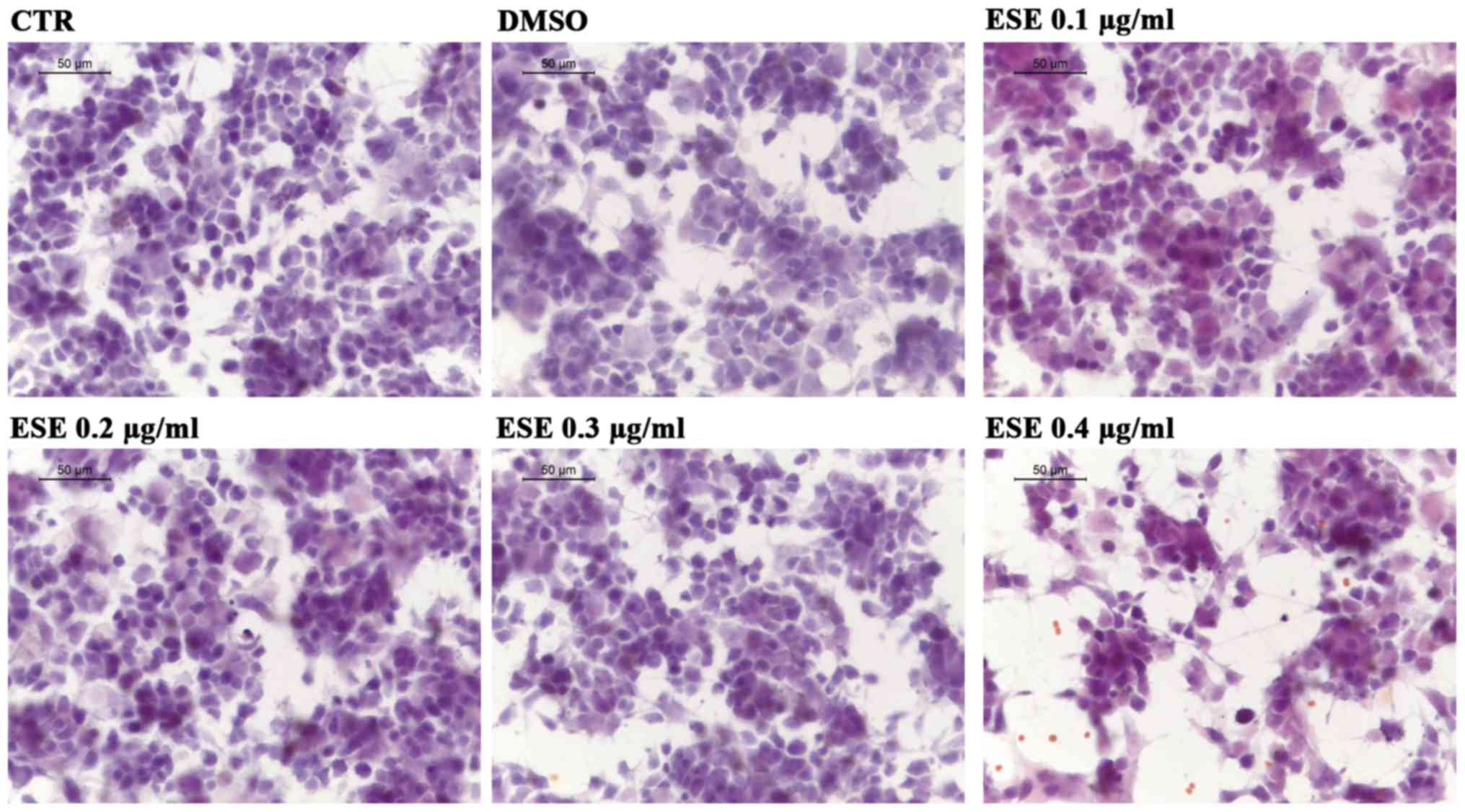
264.7. At first we assessed whether the ESE caused cytotoxicity or
morphological changes in NSC-34, using the concentrations of the
extract 0.1, 0.2, 0.3 and 0.4 µg/ml. Our results showed no
cytotoxicity for the concentrations 0.1, 0.2 and 0.3 µg/ml of ESE,
while the highest concentration showed a moderate cytotoxicity,
associated with the presence of cell debris (Fig. 3). Given that the extract was
dissolved in DMSO, we included in our analysis also NSC-34 motor
neurons incubated with a concentration of DMSO comparable to that
used for the highest concentration of ESE, but no cytotoxicity was
observed (Fig. 3).
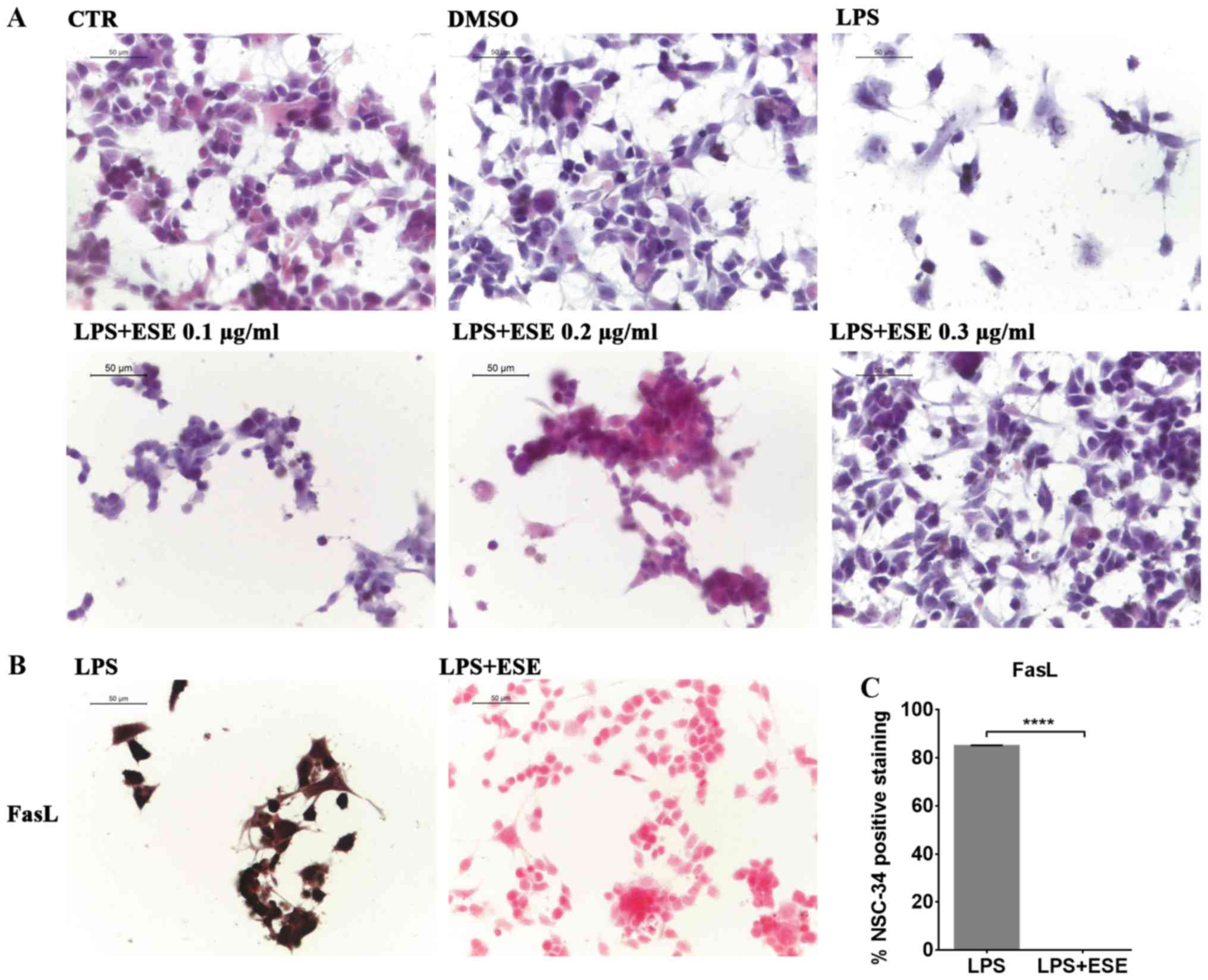
We evaluated the morphological changes induced in
NSC-34 by the medium of LPS-stimulated RAW 264.7. By
eosin/hematoxylin staining, we observed that NSC-34 motor neurons
exposed to the medium of LPS-treated RAW 264.7 showed cell death
and degeneration. However, only the pre-treatment with ESE 0.3
µg/ml was able to prevent LPS-induced morphological changes,
preserving the neural morphology that was similar to that of
control cells (Fig. 4A). Instead,
lower concentrations of the extract were not able to counteract LPS
induced effects (Fig. 4A), while
we did not evaluate the concentration 0.4 µg/ml of ESE, given its
moderate cytotoxicity. For this reason we decided to continue the
experiments with the concentration 0.3 µg/ml of ESE.
The treatment of NSC-34 motor neurons with the
medium of LPS-stimulated RAW 264.7, induced apoptosis associated
with FasL positive staining (Fig.
4B). However, our results showed that the pre-treatment with
ESE was able to counteract apoptosis, as demonstrated by FasL
negative staining (Fig. 4B and C).
NSC-34 control cells, and those incubated with DMSO or with ESE
alone did not express FasL (data not shown).
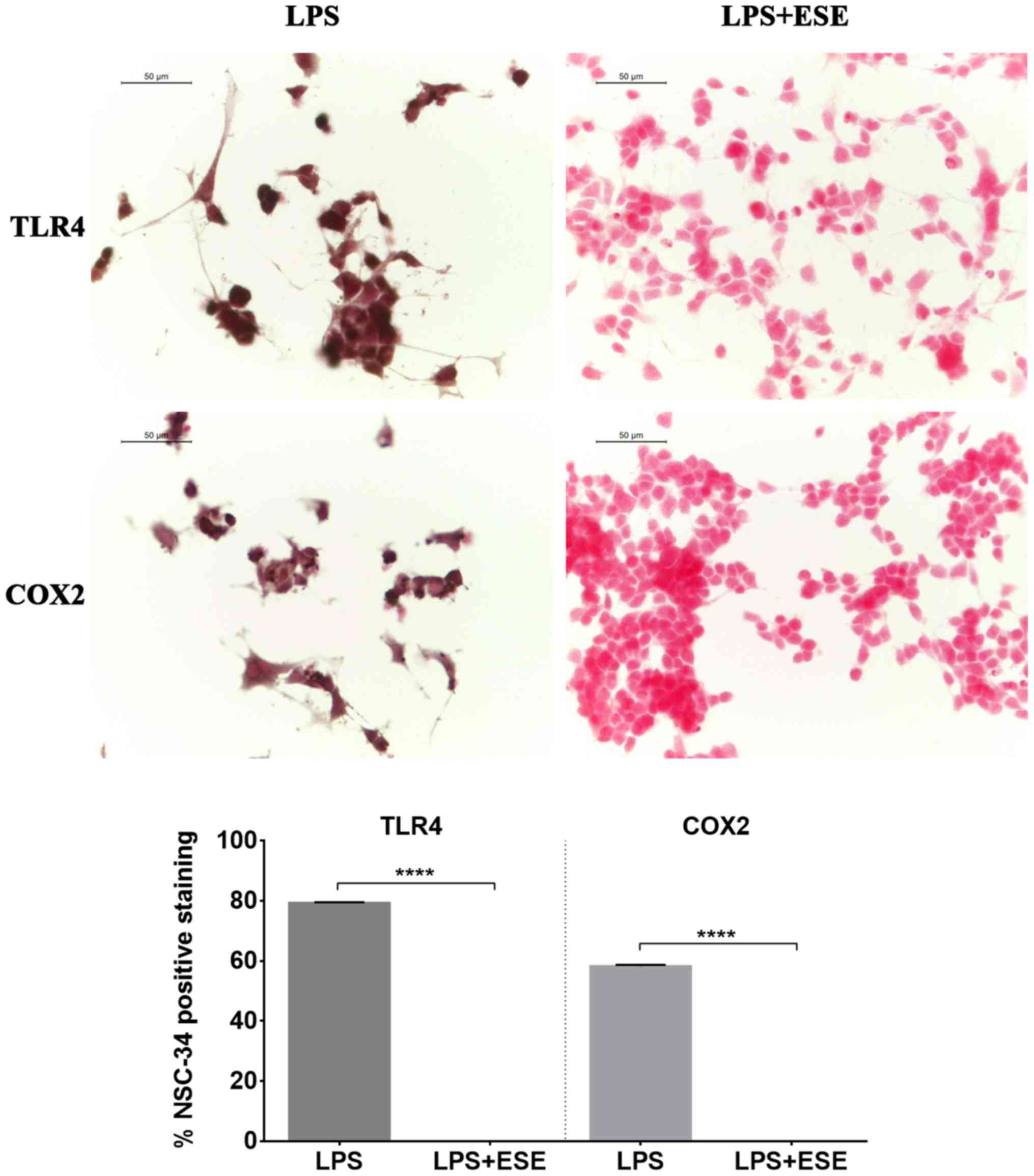
We observed TLR4 positive staining in NSC-34 motor
neurons exposed to the culture medium of LPS-stimulated macrophages
(Fig. 5). Moreover, we evaluated
also COX2 expression, and we found that NSC-34 motor neurons
stimulated with the medium of LPS-treated RAW 264.7 macrophages
showed a positive staining for COX2 (Fig. 5). These results suggested that
inflammation depends on both TLR4 and COX2 pathway activation.
Interestingly, NSC-34 pre-treated with ESE did not express TLR4 and
COX2 proteins. NSC-34 motor neurons used as controls or incubated
with DMSO, or with the extract alone did not show expression of
neither TLR4 nor COX2 (data not shown), indicating the absence of
inflammation.
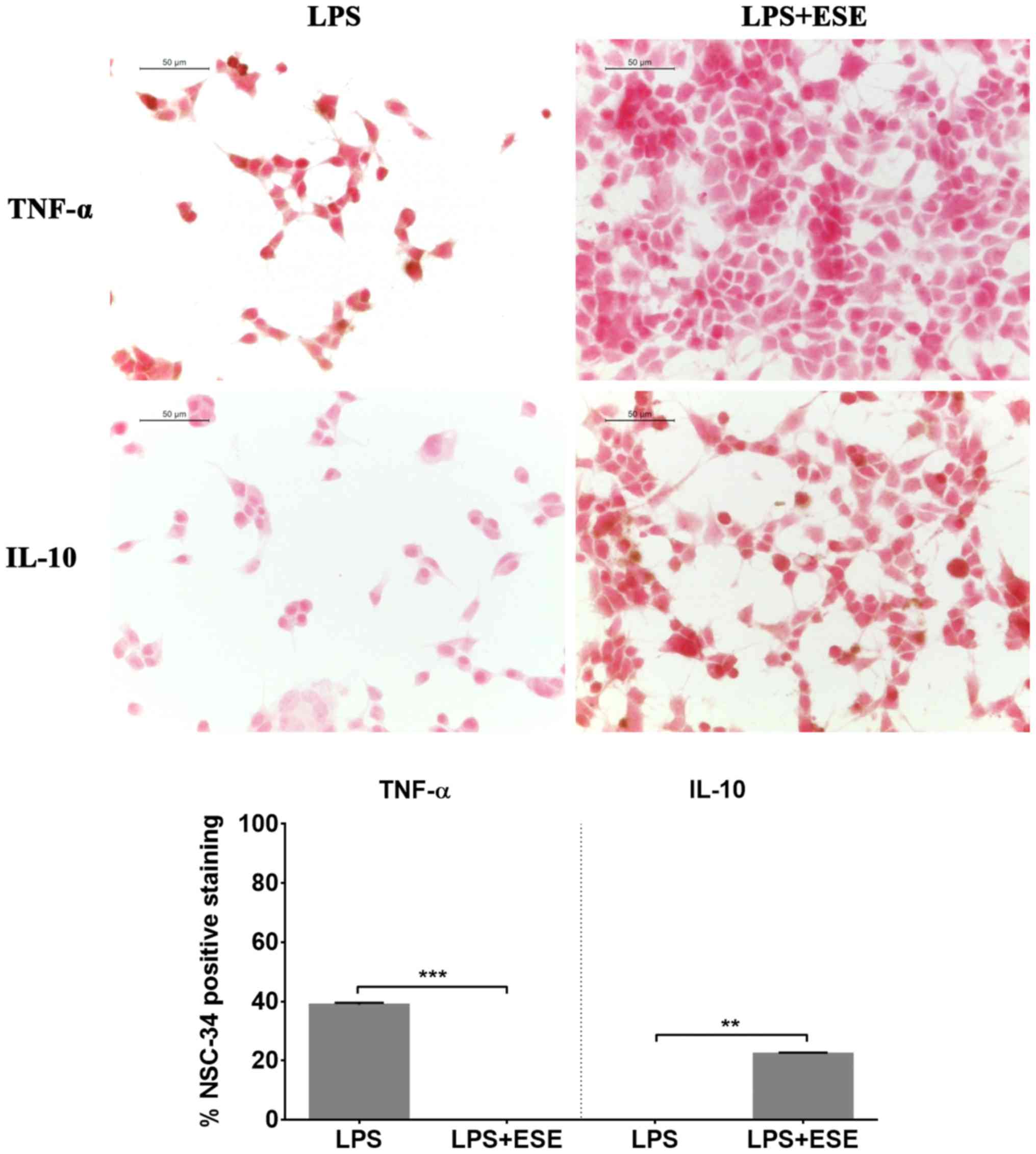
Moreover, we evaluated cytokine levels and observed
that NSC-34 cells exposed to the culture medium of LPS-stimulated
RAW 264.7 did not show the expression of IL-10, but they showed a
positive staining for the pro-inflammatory cytokine TNF-α (Fig. 6). Pre-treatment with ESE recovered
the expression of IL-10, while the expression of the
pro-inflammatory cytokine was abolished. Cells incubated with DMSO
or with the seed extract alone expressed IL-10, but not TNF-α (data
not shown).
Inflammation may be also mediated by a protein
complex known as inflammasome, a cytoplasmic protein complex that
mediates pro-inflammatory responses. It is formed by a pattern
recognition receptor, acting like sensor of damage, such as of
pathogen associated molecular patterns, that when activated,
induced the production of active caspase 1 (16,17).
The NLRP3 inflammasome, formed by NLRP3 that belongs to the
NOD-like receptor (NLR) family, is the most studied. When the
inflammasome is activated, the cleavage of pro-caspase 1 is induced
to form the active caspase 1, that in turn converts the inactive
IL-18 and IL-1β into the active forms, triggering inflammation
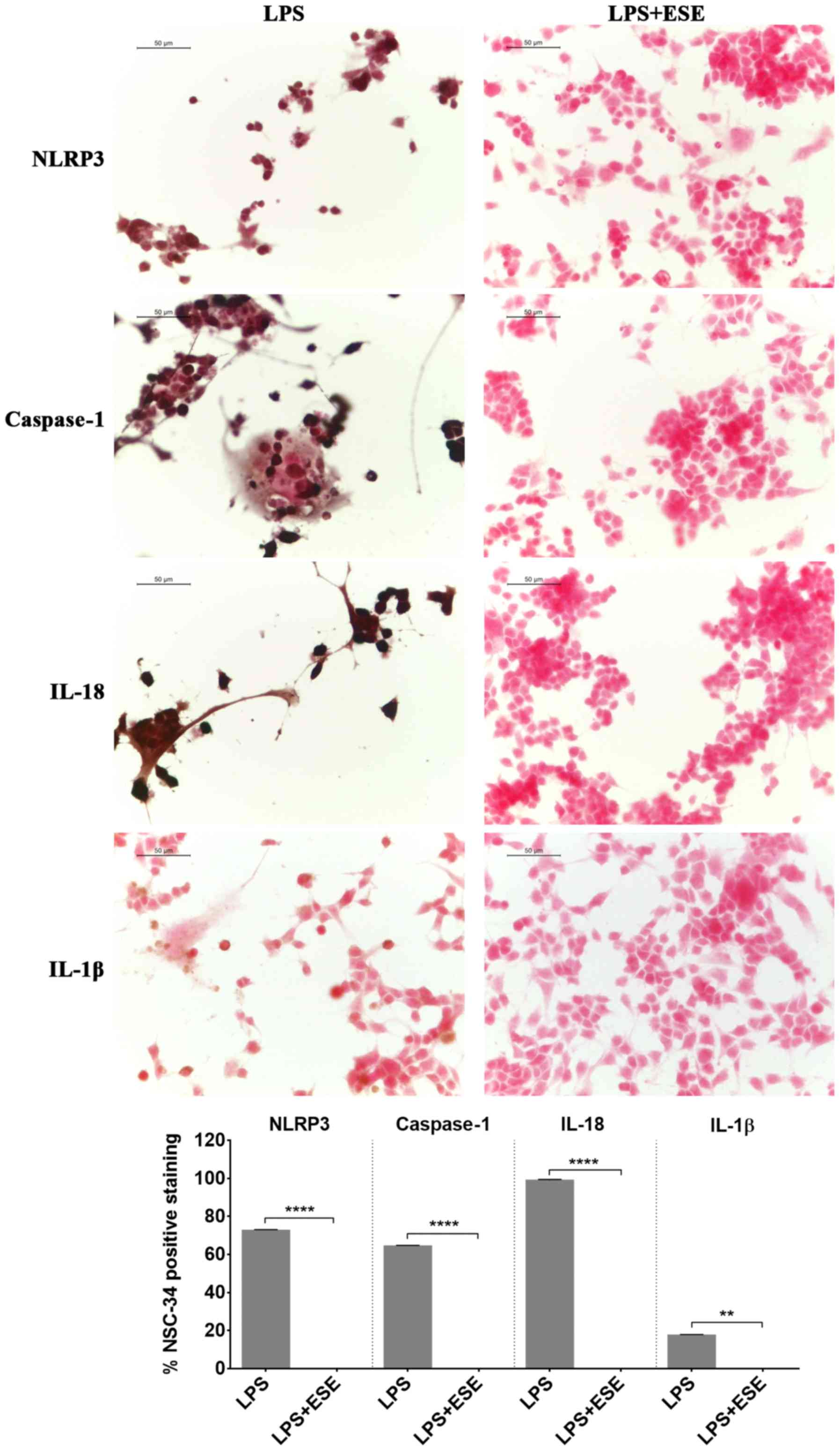
(16,17). We found NLRP3 and caspase 1
positive staining in NSC-34 stimulated with the LPS-conditioned
medium of RAW 264.7 macrophages indicating the activation of
inflammasome. According to the activation of caspase 1, we observed
the expression of the proinflammatory cytokines IL-18 and IL-1β
(Fig. 7). However, the
pre-treatment of NSC-34 with ESE was able to abolish the expression
of NLRP3 and caspase 1 and, consequently, the production of IL-18
and IL-1β (Fig. 7). NSC-34 control
cells, or incubated with DMSO or with the extract alone, did not
express inflammasome proteins (data not shown).
Discussion
Nutrition plays a main role in human health, and may
influence the development and progression of chronic diseases,
including neurodegenerative disorders (18). For this reason the use of
nutritional supplements and plant extracts in disease prevention is
of great importance, especially for neurodegenerative conditions,
where the therapeutic options are limited. In particular,
Brassicaceae vegetables consumption has demonstrated different
health benefits, thank to the presence of different phytochemicals
(2,19,20).
The aim of our work was to evaluate the
anti-inflammatory effects of ESE. We focalized our attention on
neuroinflammation, that plays a pivotal role in different
neurodegenerative disorders (14,15).
For this reason, we evaluated the anti-inflammatory properties of
the extract in NSC-34 motor neurons exposed to the cell culture
medium of LPS-stimulated RAW 264.7 macrophages. The ESE contained
the GLS glucoerucin, ascorbic acid and flavonoids. As reported in
the literature, the GLSs present in bigger amount in Eruca
sativa seeds is glucoerucin (21,22).
Our results confirmed the induction of inflammation
in LPS-stimulated RAW 264.7. Indeed, LPS-treated RAW 264.7 showed
morphological changes, including the extension of lamellipodia and
filopodia and an increased cell size, as reported in literature
(23). Moreover, it is known that
LPS was able to induce apoptosis in macrophages, through the
autocrine secretion of TNF-α and the production of nitric oxide
(NO) (24). Accordingly, in our
model we observed the induction of apoptosis as demonstrated by
FasL positive staining in LPS treated cells compared to controls.
FasL is a member of the TNF family of cytokines that binds the Fas
receptor, one of the TNF-receptor family of proteins. The Fas-FasL
system plays a main role in the induction of the extrinsic
apoptotic pathway (25). Moreover,
our results showed the expression of P-selectin in RAW 264.7
macrophages exposed to LPS. P-selectin is an adhesion molecule
expressed mainly in platelets and endothelial cells, but it was
reported that it may be also expressed in macrophages (26). Selectins mediate the leukocyte
extravasation and rolling on vascular surface in order to permit to
reach the inflammation site, and in particular some cytokines and
LPS are able to increase P-selectin transcription (27). LPS induced inflammation is mediated
by the release of pro-inflammatory cytokine (28). Indeed, also in our model we
observed that LPS treatment increased the protein levels of the
pro-inflammatory cytokines TNF-α and IL-1β in RAW 264.7, while the
level of the anti-inflammatory cytokine IL-10 decreased.
After having assessed the presence of inflammation
in RAW 264.7 macrophages exposed to LPS, we evaluated whether ESE
was able to counteract the inflammation induced in NSC-34 by the
medium of LPS-treated RAW 264.7. We observed that the pre-treatment
with 0.3 µg/ml ESE was able to prevent LPS-induced morphological
changes and apoptosis, as demonstrated by FasL negative staining.
Notably, the system Fas-FasL showed an increased expression in the
brain during inflammation, neurologic and neurodegenerative
diseases, associated with elevated apoptosis (29).
NSC-34 pre-treated with ESE did not express TLR4 and
COX2 proteins, indicating that the anti-inflammatory and
neuroprotective action of ESE may be mediated by the inhibition of
TLR4 and COX2 pro-inflammatory pathways. TLR4 was shown to be
involved in neuroinflammation in the brain, and has a role in
neurodegenerative diseases, such as Alzheimer's disease,
Parkinson's disease, amyotrophic lateral sclerosis and multiple
sclerosis (30). Moreover, it was
reported that COX2 expression may be triggered by pro-inflammatory
cytokines such as IL-1β and TNF-α (31,32).
Interestingly, the inhibition of TLR4 signaling has shown
neuroprotective effects (33,34).
Also COX2 inhibition has shown protective effects. Indeed, it was
observed that the use of celecoxib, a COX2 inhibitor, showed
neuroprotective effects in NSC-34 exposed to the supernatant of
LPS-stimulated macrophages, inhibiting the release of
pro-inflammatory mediators from activated macrophages (35). Furthermore, COX2 inhibition showed
also protective action on neurons exposed to excitotoxic stimuli
(36). ESE was shown to decrease
COX2 gene expression level in a model of rat mammary gland
carcinogenesis induced by 7,12 dimethylbenz(α)anthracene (DMBA),
and to exert antioxidant, anti-inflammatory and anticancer
activities (37). Moreover, other
Brassicaceae extracts were able to inhibit COX2 expression
(38).
Notably, the pre-treatment with ESE recovered the
expression of IL-10, while the expression of the pro-inflammatory
cytokine was abolished, indicating the capacity of ESE of
counteracting inflammation, inhibiting the release of TNF-α.
Furthermore, it was able to induce an anti-inflammatory and
prosurvival response associated with IL-10 production.
Interestingly, IL-10 is a potent anti-inflammatory cytokine that
regulate host immune response (39). In the brain, other than its
anti-inflammatory effect inhibiting the production of
pro-inflammatory cytokines, IL-10 is able to mediate neurogenesis
and exerts a prosurvival effect in neurons (40,41).
Zhou et al (42) reported
that IL-10 was able to exert prosurvival and neuroprotective
actions on spinal cord neurons, protecting them against
excitotoxicity in vitro.
Moreover, we observed the activation of the NLRP3
inflammasome in NSC-34 cells treated with the medium of LPS-treated
RAW 264.7, associated with the presence of IL-18 and IL-1β. The
NLRP3 inflammasome is the most studied. After inflammasome
activation, pro-caspase 1 is cleaved to produce the active caspase
1, leading to the production of IL-18 and IL-1β, inducing
pro-inflammatory responses, that have a role also in some nervous
system disorders (16,17). It is known that the signaling
mediated by TLR4, whose expression increased in LPS stimulated
NSC-34, is necessary for the priming of the inflammasome, inducing
the transcription of NLRP3 and pro-IL-1β, whose amounts during the
resting state are not sufficient for inflammasome activation
(16). However, ESE pre-treatment
abolished the expression of NLRP3 inflammasome proteins and the
production of IL-18 and IL-1β. Moreover, data reported that COX2,
that in our model was expressed in LPS stimulated NSC-34, may act
as a regulator of the NLRP3 inflammasome. Indeed, it was shown that
COX2 was able to promote NLRP3 inflammasome activation in
macrophages, regulating in this way the release of IL-1β, while the
genetic knockdown or COX2 inhibition reduced the priming and the
activation of NLRP3 inflammasome (43).
Interestingly, some natural compounds were reported
to be able to regulate NLRP3 inflammasome, including GLSs and
flavonoids (44,45). However, to our knowledge this is
the first study demonstrating the ability of ESE to inhibit NLRP3
inflammasome activation. Given the involvement of inflammasome also
in central nervous system disorders, it is possible to speculate
that the abolition of inflammasome activation could be a good
strategy to counteract neuroinflammation in nervous system
pathologies.
All together, our data indicated that ESE was able
to reduce inflammation, mediating in this way neuroprotective
effects, protecting NSC-34 motor neurons from cell death. Notably,
our results showing the anti-inflammatory properties of ESE are in
line with other evidence showing the anti-inflammatory properties
of other Brassicaceae extracts (38,46,47).
In conclusion, our results showed that ESE was able
to induce anti-inflammatory and neuroprotective effects in NSC-34
motor neurons exposed to the culture medium of LPS-stimulated RAW
264.7 macrophages. In particular, it exerted these actions
inhibiting COX2, TLR4, NLRP3 inflammasome pathways, and
consequently limiting pro-inflammatory cytokines production and
apoptosis. These results confirm the health promoting effects of
Brassicaceae extracts and encourage their use as dietary supplement
in the prevention/treatment of those pathological conditions where
inflammation plays a pivotal role.
Acknowledgements
The study was supported by current research funds
2017 of IRCCS ‘Centro Neurolesi Bonino-Pulejo’, Messina, Italy.
Glossary
Abbreviations
Abbreviations:
|
COX2
|
cyclooxygenase 2
|
|
DMSO
|
Dimethyl sulfoxide
|
|
ESE
|
Eruca sativa seed extract
|
|
FasL
|
Fas Ligand
|
|
FBS
|
fetal bovine serum
|
|
GAPDH
|
Glyceraldehyde 3-phosphate
dehydrogenase
|
|
GLS
|
glucosinolate
|
|
H2O2
|
hydrogen peroxide
|
|
HRP
|
horse radish peroxidase
|
|
IL
|
interleukin
|
|
LPS
|
lipopolysaccharide
|
|
NLR
|
NOD-like receptor
|
|
NLRP3
|
NLR Family Pyrin Domain Containing
3
|
|
NO
|
nitric oxide
|
|
PBS
|
phosphate buffered saline
|
|
TLR
|
Toll-like receptor
|
|
TNF-α
|
tumor necrosis factor-α
|
References
|
1
|
Kalra EK: Nutraceutical-definition and
introduction. AAPS Pharm Sci. 5:E252003. View Article : Google Scholar
|
|
2
|
Björkman M, Klingen I, Birch AN, Bones AM,
Bruce TJ, Johansen TJ, Meadow R, Mølmann J, Seljåsen R, Smart LE
and Stewart D: Phytochemicals of Brassicaceae in plant protection
and human health-influences of climate, environment and agronomic
practice. Phytochemistry. 72:538–556. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Cartea ME, Francisco M, Soengas P and
Velasco P: Phenolic compounds in Brassica vegetables. Molecules.
16:251–280. 2011. View Article : Google Scholar
|
|
4
|
Prakash D and Gupta C: Glucosinolates: The
phytochemicals of nutraceutical importance. J Complement Integr
Med. 9:132012. View Article : Google Scholar
|
|
5
|
Bhandari SR, Jo JS and Lee JG: Comparison
of glucosinolate profiles in different tissues of nine brassica
crops. Molecules. 20:15827–15841. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Khoobchandani M, Ganesh N, Gabbanini S,
Valgimigli L and Srivastava MM: Phytochemical potential of Eruca
sativa for inhibition of melanoma tumor growth. Fitoterapia.
82:647–653. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Khoobchandani M, Ojeswi BK, Ganesh N,
Srivastava MM, Gabbanini S, Matera R, Iori R and Valgimigl L:
Antimicrobial properties and analytical profile of traditional
Eruca sativa seed oil: Comparison with various aerial and
root plant extracts. Food Chem. 120:217–224. 2010. View Article : Google Scholar
|
|
8
|
Fuentes E, Alarcon M, Fuentes M, Carrasco
G and Palomo I: A novel role of Eruca sativa Mill. (rocket)
extract: Antiplatelet (NF-κB inhibition) and antithrombotic
activities. Nutrients. 6:5839–5852. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Sarwar Alam M, Kaur G, Jabbar Z, Javed K
and Athar M: Eruca sativa seeds possess antioxidant activity
and exert a protective effect on mercuric chloride induced renal
toxicity. Food Chem Toxicol. 45:910–920. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bell L, Oruna-Concha MJ and Wagstaff C:
Identification and quantification of glucosinolate and flavonol
compounds in rocket salad (Eruca sativa, Eruca vesicaria and
Diplotaxis tenuifolia) by LC-MS: Highlighting the potential
for improving nutritional value of rocket crops. Food Chem.
172:852–861. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Dinkova-Kostova AT and Kostov RV:
Glucosinolates and isothiocyanates in health and disease. Trends
Mol Med. 18:337–347. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Spencer JP, Vafeiadou K, Williams RJ and
Vauzour D: Neuroinflammation: Modulation by flavonoids and
mechanisms of action. Mol Aspects Med. 33:83–97. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Orhan IE, Daglia M, Nabavi SF, Loizzo MR,
Sobarzo-Sánchez E and Nabavi SM: Flavonoids and dementia: An
update. Curr Med Chem. 22:1004–1015. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Kempuraj D, Thangavel R, Natteru PA,
Selvakumar GP, Saeed D, Zahoor H, Zaheer S, Iyer SS and Zaheer A:
Neuroinflammation induces neurodegeneration. J Neurol Neurosurg
Spine. 1:pii: 1003. 2016.PubMed/NCBI
|
|
15
|
Chen WW, Zhang X and Huang WJ: Role of
neuroinflammation in neurodegenerative diseases (Review). Mol Med
Rep. 13:3391–3396. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
He Y, Hara H and Núñez G: Mechanism and
regulation of NLRP3 inflammasome activation. Trends Biochem Sci.
41:1012–1021. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Zhou K, Shi L, Wang Y, Chen S and Zhang J:
Recent advances of the NLRP3 inflammasome in central nervous system
disorders. J Immunol Res. 2016:92382902016. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Virmani A, Pinto L, Binienda Z and Ali S:
Food, nutrigenomics, and neurodegeneration-neuroprotection by what
you eat! Mol Neurobiol. 48:353–362. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wagner AE, Terschluesen AM and Rimbach G:
Health promoting effects of brassica-derived phytochemicals: From
chemopreventive and anti-inflammatory activities to epigenetic
regulation. Oxid Med Cell Longev. 2013:9645392013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Kapusta-Duch J, Kopeć A, Piatkowska E,
Borczak B and Leszczyńska T: The beneficial effects of Brassica
vegetables on human health. Rocz Panstw Zakl Hig. 63:389–395.
2012.PubMed/NCBI
|
|
21
|
Iori R, Bernardi R, Gueyrard D, Rollin P
and Palmieri S: Formation of glucoraphanin by chemoselective
oxidation of natural glucoerucin: A chemoenzymatic route to
sulforaphane. Bioorg Med Chem Lett. 9:1047–1048. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Barillari J, Canistro D, Paolini M,
Ferroni F, Pedulli GF, Iori R and Valgimigli L: Direct antioxidant
activity of purified glucoerucin, the dietary secondary metabolite
contained in rocket (Eruca sativa Mill.) seeds and sprouts.
J Agric Food Chem. 53:2475–2482. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Pi J, Li T, Liu J, Su X, Wang R, Yang F,
Bai H, Jin H and Cai J: Detection of lipopolysaccharide induced
inflammatory responses in RAW264.7 macrophages using atomic force
microscope. Micron. 65:1–9. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Xaus J, Comalada M, Valledor AF, Lloberas
J, López-Soriano F, Argilés JM, Bogdan C and Celada A: LPS induces
apoptosis in macrophages mostly through the autocrine production of
TNF-alpha. Blood. 95:3823–3831. 2000.PubMed/NCBI
|
|
25
|
Ashe PC and Berry MD: Apoptotic signaling
cascades. Prog Neuropsychopharmacol Biol Psychiatry. 27:199–214.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tchernychev B, Furie B and Furie BC:
Peritoneal macrophages express both P-selectin and PSGL-1. J Cell
Biol. 163:1145–1155. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
McEver RP: Selectins: Initiators of
leucocyte adhesion and signalling at the vascular wall. Cardiovasc
Res. 107:331–339. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Rossol M, Heine H, Meusch U, Quandt D,
Klein C, Sweet MJ and Hauschildt S: LPS-induced cytokine production
in human monocytes and macrophages. Crit Rev Immunol. 31:379–446.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Choi C and Benveniste EN: Fas ligand/Fas
system in the brain: Regulator of immune and apoptotic responses.
Brain Res Brain Res Rev. 44:65–81. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Trotta T, Porro C, Calvello R and Panaro
MA: Biological role of Toll-like receptor-4 in the brain. J
Neuroimmunol. 268:1–12. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Hoozemans JJ, Veerhuis R, Janssen I,
Rozemuller AJ and Eikelenboom P: Interleukin-1beta induced
cyclooxygenase 2 expression and prostaglandin E2 secretion by human
neuroblastoma cells: Implications for Alzheimer's disease. Exp
Gerontol. 36:559–570. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Mark KS, Trickler WJ and Miller DW: Tumor
necrosis factor-alpha induces cyclooxygenase-2 expression and
prostaglandin release in brain microvessel endothelial cells. J
Pharmacol Exp Ther. 297:1051–1058. 2001.PubMed/NCBI
|
|
33
|
Ulbrich F, Kaufmann K, Roesslein M,
Wellner F, Auwärter V, Kempf J, Loop T, Buerkle H and Goebel U:
Argon mediates anti-apoptotic signaling and neuroprotection via
inhibition of Toll-like receptor 2 and 4. PLoS One.
10:e01438872015. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
De Paola M, Sestito SE, Mariani A, Memo C,
Fanelli R, Freschi M, Bendotti C, Calabrese V and Peri F: Synthetic
and natural small molecule TLR4 antagonists inhibit motoneuron
death in cultures from ALS mouse model. Pharmacol Res. 103:180–187.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Huang Y, Liu J, Wang LZ, Zhang WY and Zhu
XZ: Neuroprotective effects of cyclooxygenase-2 inhibitor celecoxib
against toxicity of LPS-stimulated macrophages toward motor
neurons. Acta Pharmacol Sin. 26:952–958. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Strauss KI and Marini AM: Cyclooxygenase-2
inhibition protects cultured cerebellar granule neurons from
glutamate-mediated cell death. J Neurotrauma. 19:627–638. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Abdel-Rahman S, Shaban N, Haggag A, Awad
D, Bassiouny A and Talaat I: Inhibition of NF-κB, Bcl-2 and COX-2
Gene expression by an extract of Eruca sativa seeds during
rat mammary gland carcinogenesis. Asian Pac J Cancer Prev.
16:8411–8418. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Herz C, Tran HT, Márton MR, Maul R,
Baldermann S, Schreiner M and Lamy E: Evaluation of an aqueous
extract from horseradish root (Armoracia rusticana Radix)
against lipopolysaccharide-induced cellular inflammation reaction.
Evid Based Complement Alternat Med. 2017:19506922017. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sabat R, Grütz G, Warszawska K, Kirsch S,
Witte E, Wolk K and Geginat J: Biology of interleukin-10. Cytokine
Growth Factor Rev. 21:331–344. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Lobo-Silva D, Carriche GM, Castro AG,
Roque S and Saraiva M: Balancing the immune response in the brain:
IL-10 and its regulation. J Neuroinflammation. 13:2972016.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Pereira L, Font-Nieves M, Van den Haute C,
Baekelandt V, Planas AM and Pozas E: IL-10 regulates adult
neurogenesis by modulating ERK and STAT3 activity. Front Cell
Neurosci. 9:572015. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Zhou Z, Peng X, Insolera R, Fink DJ and
Mata M: Interleukin-10 provides direct trophic support to neurons.
J Neurochem. 110:1617–1627. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Hua KF, Chou JC, Ka SM, Tasi YL, Chen A,
Wu SH, Chiu HW, Wong WT, Wang YF, Tsai CL, et al: Cyclooxygenase-2
regulates NLRP3 inflammasome-derived IL-1β production. J Cell
Physiol. 230:863–874. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Lee HW, Lee CG, Rhee DK, Um SH and Pyo S:
Sinigrin inhibits production of inflammatory mediators by
suppressing NF-κB/MAPK pathways or NLRP3 inflammasome activation in
macrophages. Int Immunopharmacol. 45:163–173. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Tőzsér J and Benkő S: Natural compounds as
regulators of NLRP3 inflammasome-mediated IL-1β production.
Mediators Inflamm. 2016:54603022016. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Sadeghi H, Mostafazadeh M, Sadeghi H,
Naderian M, Barmak MJ, Talebianpoor MS and Mehraban F: In vivo
anti-inflammatory properties of aerial parts of Nasturtium
officinale. Pharm Biol. 52:169–174. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kook SH, Choi KC, Lee YH, Cho HK and Lee
JC: Raphanus sativus L. seeds prevent LPS-stimulated inflammatory
response through negative regulation of the p38 MAPK-NF-κB pathway.
Int Immunopharmacol. 23:726–734. 2014. View Article : Google Scholar : PubMed/NCBI
|