Introduction
Bladder cancer (BC) is the most common malignant
tumor of the urinary system in both sexes and occurs in the mucosa
of the bladder, with 74,400 new cases and 29,400 new deaths
occurring in 2013 in China as well as increasing incidence rate
with age (1,2). In spite of advancements in bladder
carcinoma therapeutic strategies, including radiotherapy, surgical
resection and chemotherapy, the survival rate remains low due to
recurrence and metastasis (3). The
highly metastatic and invasive characteristics of BC reduce the
effectiveness of treatments; therefore, these characteristics have
become a focus of research (4,5). In
addition, numerous clinicians emphasize that management, prevention
and diagnosis are also of importance to effective control of BC
(6–8). Therefore, it is necessary to
elucidate the underlying molecular mechanisms of BC.
MicroRNAs (miRNAs) are non-coding small RNAs,
encoded by genes of ~22 nucleotides in length, which regulate
expression of other genes on post-transcriptional levels by
promoting degradation of mRNAs or suppressing translation by
complementary biding to the 3′untranslated region (UTR) of the
target mRNA (9,10). Increasing amount of evidence
indicates that miRNAs serve a role in regulating tumorigenesis and
tumor development including growth, metastasis and apoptosis
(11,12). miRNAs have been reported to
modulate cancer progression and are considered as potential
biomarkers for diagnosis, pathogenesis and progression prediction,
as well as therapy in numerous cancers, including lung cancer,
breast cancer, thyroid carcinoma and colorectal cancer (13–16).
Particularly, miRNA-145 exerts a regulatory effect on multiple
types of human tumors, including colorectal cancer, non-small cell
lung cancer, pancreatic cancer and oral cancer (17–20).
Nevertheless, the underlying mechanism of miRNA-145-mediated
modulation of migration and invasion of BC cells remains to be
elucidated.
In the present study, miRNA-145 was downregulated in
BC tissues and cells. Furthermore, miRNA-145 inhibited
migration/invasion of BC cells, which may be mediated by targeting
N-cadherin and its downstream effector matrix metalloproteinase-9
(MMP9). Investigation of miRNA-145 and its targets may aid in
identification of novel therapeutic targets and diagnostic
biomarkers of BC.
Materials and methods
Reagents and antibodies
miRNA-145 mimics, inhibitor and negative controls
(NCs) were obtained from Guangzhou RiboBio Co., Ltd. (Guangzhou,
China). Lipofectamine® 2000, Dulbecco's modified Eagle's
medium (DMEM), fetal bovine serum (FBS) and a bicinchoninic acid
(BCA) protein assay kit were purchased from Thermo Fisher
Scientific Inc. (Waltham, MA, USA). Transwell chamber was purchased
from Corning Incorporated (Corning, NY, USA). RNAiso reagent, miRNA
cDNA Synthesis kit and SYBR Premix Ex Taq™ kit were purchased from
Takara Biotechnology Co., Ltd. (Dalian, China).
Radioimmunoprecipitation assay (RIPA) lysis buffer was purchased
from Beyotime Institute of Biotechnology (Shanghai, China). Primary
antibodies against N-cadherin, matrix metalloproteinase-9 (MMP9)
and GAPDH, and anti-rabbit horseradish peroxidase (HRP) conjugated
immunoglobulin G was purchased from Abcam (Cambridge, MA, USA).
Tissue specimens
Clinical tissue specimens were collected from
patients with BC (n=10; all stage III; mean age, 61.8 years; 7 male
and 3 female) who had undergone surgical resection of their bladder
tumors and matched adjacent non-tumor bladder tissues (ANT) at
Affiliated Hospital of BeiHua University from January 2014 to
January 2015. All tissue specimens were snap frozen in liquid
nitrogen and stored at −80°C. Written informed consent was obtained
from all patients and the present study was approved by the Ethics
Committee of Affiliated Hospital of BeiHua University (Jilin City,
China).
Cell culture
Human BC cell lines T24 and 5637 were obtained from
American Type Culture Collection (Manassas, VA, USA). Normal
bladder cells SV-HUC-1 were purchased from the Type Culture
Collection of the Chinese Academy of Sciences (Shanghai, China).
Cells were maintained in DMEM medium supplemented with 10% (v/v)
FBS, containing penicillin (100 U/ml) and streptomycin (100 µg/ml)
at 37°C in a humidified incubator containing 5% CO2.
Cells in exponential growth phase (~1×106 cells/ml) were
used for the subsequent experiments.
Transient transfection
T24 cells were seeded at a density of
1×105 cells/well into 6-well plates and cell confluence
was 30% at the time of transfection. A total of 1 µg miRNA-145
mimics (forward 5′-TCGGGGGAGTCTCTTGACCTATT-3′; reverse
5′-TAGGTCAAGACTCCCCCGATT-3′), miRNA-145 inhibitor
(5′-TAGGTCAAGACTCCCCCGA-3′), or negative control (NC:
5′-TAGGTCAAGAGACTCCCCCGA-3′) were transfected into BC cells using
Lipofectamine® 2000 following the manufacturer's
protocol. Subsequent experiments were performed 48 h
post-transfection.
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA of tissues and cells were extracted with
RNAiso reagent (Takara Biotechnology Co., Ltd.) following the
manufacturer's protocol. RNA purity and concentration were examined
spectrophotometrically and ribosomal ratios of 28S, 18S and 5S were
detected by agarose gel electrophoresis. Reverse transcription was
performed using miRNA cDNA Synthesis kit (Takara Biotechnology Co.,
Ltd.) according to the manufacturer's instructions with the
following temperature protocol: 1 h at 37°C and termination at 85°C
for 5 min. PCR was performed using the SYBR Premix Ex Taq™ kit
(Takara Biotechnology Co., Ltd.) in accordance with manufacturer's
protocols. The thermocycling conditions were as follows: 95°C for
10 sec, followed by 95°C for 5 sec and 60°C for 20 sec for 40
cycles, 95°C for 60 sec, 55°C for 30 sec and 95°C for 30 sec. U6
small nuclear RNA was used as internal control. Forward and reverse
primers sequences for miRNA-145 were 5′-GTCCAGTTTTCCCAGGAATC-3′ and
5′-AGAACAGTATTTCCAGGAAT-3′, and primers for U6 included
5′CTCAACTGGTGTCGTGGAGTCGGCAATTGACAAGTTGAAATATG-3′ and
5′-ACACTCCAAGGGCTGTAACGGGTGCCGGAA-3′. The qRT-PCR results of
miRNA-145 expression levels were analyzed using the
2−ΔΔCq calculation method (21) from three independent repeats.
Wound healing and transwell
assays
To determine alterations in cell mobility following
transfection with miRNA-145, wound healing and transwell assays
were performed. A total of 1×105 cells were seeded into
6-well plates with complete culture medium (serum free DMEM
supplemented with 100 U/ml penicillin and 100 µg/ml streptomycin)
and the confluent monolayer of cells was scratched using a 200 µl
pipette tip. Subsequently, cell migration into the wound closure
was visualized under a light microscope. For detection of cell
migration by transwell assay, 100 µl cell suspension containing
~1×105 cells in serum-free medium was plated in the
upper chamber while 500 µl DMEM containing 10% FBS was added into
the lower chamber. Following incubation for 24 h, non-migrating
cells on the top chamber were gently removed by a cotton-tipped
swab. The cells that migrated through the membrane into the lower
chamber were fixed with 4% paraformaldehyde for 30 min and stained
with 0.1% crystal violet for 20 min at 37°C. Subsequently, the
migrated cells were captured under an inverted light microscope
(Olympus Corporation, Tokyo, Japan) and counted. For detection of
cell invasion, the experimental procedures were the same as for
cell migration detection, but Matrigel was added into the bottom
center of the upper chamber.
Western blot analysis
Cells were lysed with RIPA cell lysis buffer
(Beyotime Institute of Biotechnology) and quantified using a BCA
kit (Thermo Fisher Scientific, Inc.). Equal amounts (20 µg/lane) of
protein were separated by 10% Tris-glycine gradient gels via
SDS-PAGE and transferred to polyvinylidene difluoride (PVDF)
membranes. Subsequently, the membranes were blocked with 5% skimmed
milk at room temperature in TBST (containing 0.1% Tween-20) for 1.5
h and incubated with antibodies (1:1,000 dilution) against
N-cadherin (cat. no. ab18203), MMP9 (cat. no. ab38898) and GAPDH
(cat. no. ab8245) at 4°C overnight, followed by incubation with
anti-rabbit HRP-conjugated secondary antibody (1:5,000) for 2 h at
room temperature. Western blot bands were visualized by Enhanced
Chemiluminescence kit (Pierce; Thermo Fisher Scientific, Inc.) and
measured with Quantity One 4.6 version software (version 4.6;
Bio-Rad Laboratories, Inc., Hercules, CA, USA).
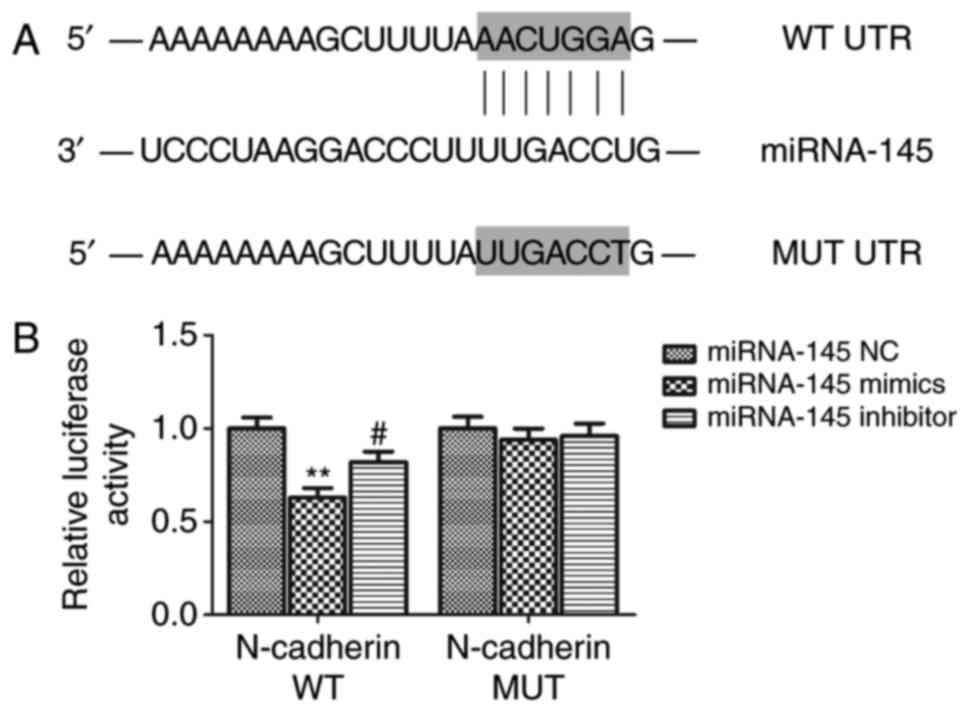
Luciferase-reporter assay
To predict the potential target genes of miRNA-145,
prediction software programs TargetScan 7.1 (http://www.targetscan.org/) and miRTarBase 7.0
(http://miRTarBase.mbc.nctu.edu.tw/)
(22) were used. The luciferase
reporter assay was carried out to determine whether miRNA-145
directly targets N-cadherin. The wild-type (WT) and mutant-type
(MUT) N-cadherin 3′-UTR were cloned into the pmirGLO-reporter
vector (Promega Corporation, Madison, WI, USA). For the luciferase
reporter assay, cells were seeded into 96-well plates for 24 h and
co-transfected with luciferase reporter vectors with miRNA-145
mimic, miRNA-145 inhibitor or NC using Lipofectamine®
2000. Following transfection for 48 h, cells were collected. The
relative luciferase activity was examined using a dual luciferase
reporter assay system (Promega Corporation). Ratio of Firefly
luciferase and Renilla luciferase activity was used for
normalization. Each assay was performed in triplicate.
Statistical analysis
Statistical analysis was performed using SPSS
software version 20.0 (IBM Corp., Armonk, NY, USA) and GraphPad
Prism 5.0 (GraphPad Software, Inc., La Jolla, CA, USA). All data
are presented as the mean ± standard deviation of three independent
experiments. One-way analysis of variance followed by Fisher's
Least Significant Difference post-hoc test or two-tailed Student's
t-test was used to determine the statistical significance of
differences. P<0.05 was considered to indicate a statistically
significant difference.
Results
Expression of miRNA-145 is
down-regulated in bladder cancer
In order to investigate the expression level of
miRNA-145 in BC, miRNA-145 expression levels were determined by
RT-qPCR in 10 cases of clinical BC tissues and corresponding ANT,
as well as BC cell lines (T24 and 5637). Expression levels of
miRNA-145 in tissues and cell lines were significantly reduced
compared with paired normal bladder tissues and SV-HUC-1 cells,
respectively (P<0.01; Fig. 1A and
B). The expression level of miRNA-145 was the lowest in T24
cells, and, therefore, the subsequent experiments were performed
using this cell line. Following transfection with miRNA-145 mimics,
inhibitor or NC, expression of miRNA-145 was measured in T24 cells
and the results indicated that miRNA-145 level in miRNA-145 mimics
group was significantly higher compared with the other groups (both
P<0.01; Fig. 1C) while
miRNA-145 expression in miRNA-145 inhibitor group was markedly
reduced compared with the miRNA-145 mimics group (P<0.01).
Inhibitory effect of miRNA-145 on
migration and invasion of BC cells
To further measure the effect of miRNA-145 on cell
migration and invasion, wound healing and transwell assays were
performed. The wound healing assay demonstrated that transfection
with miRNA-145 mimics significantly decreased wound-healing rate
compared with the NC mimics group (P<0.01; Fig. 2A and B). Furthermore, the
wound-healing rate of the miRNA-145 inhibitor group was
significantly lower than the miRNA-145 mimic group (P<0.01). The
results of transwell assay demonstrated that transfection with
miRNA-145 mimics markedly reduced the migrated cell number and
invaded cell number compared with cells transfected with NC mimics
(P<0.01; Fig. 2C and D).
Additionally, the migrated and invaded cell number in the miRNA-145
inhibitor group was significantly reduced compared with the
miRNA-145 mimic group (P<0.01). These results indicate that
miRNA-145 serves an inhibitory role in BC cell migration and
invasion.
miRNA-145 regulates the expression of
N-cadherin via binding to its 3′UTR
Using bioinformatics software, it was predicted that
N-cadherin may be a potential target gene of miRNA-145. Further, in
order to confirm this direct interaction between miRNA-145 and
N-cadherin, a luciferase reporter assay was carried out. The
results demonstrated that relative luciferase activity in the WT
group co-transfected with N-cadherin 3′UTR and miRNA-145 mimics was
lower compared with the miRNA-145 NC group (P<0.01; Fig. 3). Furthermore, the relative
luciferase activity in the WT group co-transfected with miRNA-145
inhibitor was significantly higher than the miRNA-145 mimic group
(P<0.01). Relative luciferase activity in the MUT group
exhibited no significant difference between co-transfection with
miRNA-145 mimics or miRNA-145 inhibitor and NC mimics. Furthermore,
N-cadherin expression level was detected via western blot assay and
it was determined that N-cadherin was significantly downregulated
in the miRNA-145 mimics group compared with the miRNA-124 NC mimics
group (P<0.01; Fig. 4A). The
expression of N-cadherin was increased in the miRNA-145 inhibitor
group compared with the miRNA-145 mimics group (P<0.01).
Furthermore, since MMP9 was previously demonstrated to be one of
N-cadherin downstream molecules (23), its expression level was determined
by western blotting. The results were similar to that of N-cadherin
described above (P<0.01; Fig.
4B).
Discussion
The results of the present study revealed the
regulatory mechanism of miRNA-145 and N-cadherin in the migration
and invasion of in BC cells. It was identified that miRNA-145 was
downregulated in BC tissues and cell lines. Furthermore, miRNA-145
suppressed BC cell migration and invasion. miRNA-145 directly
targeted N-cadherin and regulated its and MMP9 expression level,
suggesting that miRNA-145 may exert a potentially inhibitory role
on migration and invasion of BC cells by targeting N-cadherin and
regulating MMP9. These results provide novel insights into
molecular mechanism underlying BC metastasis and invasion and
regulation of miRNA-145 could become a potential biomarker or a
therapeutic target for treatment of BC.
Bioinformatics tools have predicted that high-energy
bonding exists in the interactions between miRNA and target genes,
and that miRNA overexpression results in target protein expression
downregulation (9,24). It is well-known that numerous genes
are responsible for carcinogenesis and cancer development.
Therefore, miRNAs can act as tumor suppressors or oncogenes, by
binding to regulators associated with cancer cell survival,
proliferation, apoptosis and metastasis (25,26).
A growing body of evidence has demonstrated that miRNA-145 serves a
suppressive role in various cancers (27–30).
It has been observed that miRNA-145 exerts tumor suppressive
effects, by modulating cell proliferation, metastasis, invasion,
apoptosis, chemo-resistance effect and epithelial-to-mesenchymal
transition of multiple tumors (31–34).
However, the effect and underlying mechanisms of miRNA-145 in BC
remain to be elucidated. In line with aforementioned studies, the
present study demonstrated that miRNA-145 acted as a
tumor-suppressor and was downregulated in patients with BC.
Furthermore, the present study revealed that miRNA-145 inhibited
migration and invasion in BC cells.
Metastasis and invasion are the main characteristics
of malignancy and are responsible for the majority of
tumor-associated mortality (35,36).
N-cadherin, a homophilic transmembrane adhesion glycoprotein, acts
as a transmembrane signal transduction receptor influencing
cell-cell adhesion and therefore serves a role in invasion and
migration during cancer progression (37–39).
Accumulating evidence suggests that N-cadherin is implicated in
promoting cancer cell motility (40), thyroid tumorigenesis (41), migration and invasion (42). One report suggests that N-cadherin
serves a prognostic role in patients with BC (43). Further, another research group has
demonstrated that miRNA-145 can target N-cadherin in lung
adenocarcinoma cells (44),
whereas, to the best of the authors' knowledge, the modulation of
cell metastasis via N-cadherin by miRNA-145 has not been previously
studied in the context of BC. Based on the aforementioned studies,
the authors of the present study hypothesize that N-cadherin may
serve as an oncogene mediating the effect of miRNA-145 on BC
metastasis. In the present study, luciferase-reporter assay and
western blot analysis results suggested that miRNA-145 contributed
to inhibition of BC cells invasion and migration by directly
targeting N-cadherin. MMPs, known as proteolytic enzymes, were
previously reported to be associated with tumor metastasis and
invasion (45,46). Additionally, MMP9, an extracellular
matrix-degrading enzyme, serves a role in metastasis and invasion
of carcinoma cells (47,48). MMP9 was identified as a modulatory
target of N-cadherin-dependent signaling (23). Furthermore, MMP9 expression level
was upregulated in the presence of N-cadherin (49). Nonetheless, MMP9 is not a direct
target of miRNA-145, as previously demonstrated by a luciferase
assay, but its expression was decreased in miRNA-145-transfected
gastric cell lines (50). In
agreement with this evidence, the results of the present study
indicated that levels of MMP9 were decreased in BC cells
transfected with miRNA-145 mimics while these levels increased in
BC cells transfected with miRNA-145 inhibitor. The above results
indicate that miRNA-145 exerts an anti-invasive and anti-migratory
roles in BC by directly targeting N-cadherin and regulating MMP9.
However, the limitations of the present study included lack of
verification of the results in an animal model of BC. Furthermore,
the modulatory roles of miRNA-145 and N-cadherin in BC are likely
through complex molecular mechanisms; thus, further investigation
is required.
In conclusion, the present study demonstrated that
miRNA-145 served as a tumor-inhibitor and was downregulated in BC
tissues and cell lines. Furthermore, miRNA-145 directly regulated
expression of N-cadherin in BC cells and its exogenous expression
inhibited migration and invasion of BC cells. It can be
hypothesized that miRNA-145 may inhibit migration and invasion of
BC cells possibly by directly suppressing the protein expression of
N-cadherin. Therefore, miRNA-145 and N-cadherin may be novel
candidates for development of efficient therapeutic strategies for
treatment of BC.
Acknowledgements
Not applicable.
Funding
Not applicable.
Availability of data and materials
All data generated or analyzed during this study are
included in this published article.
Authors' contributions
XQZ designed the study and revised the manuscript.
XFZ performed the experiments and wrote the manuscript. ZXC, CCW
and HG performed the experiments and analyzed the data. All authors
read and approved the final manuscript.
Ethics approval and consent to
participate
Written informed consent was obtained from all
patients and the present study was approved by the Ethics Committee
of Affiliated Hospital of BeiHua University (Jilin City,
China).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests.
References
|
1
|
Chen W, Zheng R, Zhang S, Zeng H, Xia C,
Zuo T, Yang Z, Zou X and He J: Cancer incidence and mortality in
China, 2013. Cancer Lett. 401:63–71. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Chen W, Zheng R, Baade PD, Zhang S, Zeng
H, Bray F, Jemal A, Yu XQ and He J: Cancer statistics in China,
2015. CA Cancer J Clin. 66:115–132. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Choe J, Braschi-Amirfarzan M, Tirumani SH,
Shinagare AB, Kim KW, Ramaiya NH and Krajewski KM: Updates for the
radiologist in non-muscle-invasive, muscle-invasive, and metastatic
bladder cancer. Abdom Radiol (NY). 42:2710–2724. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Lobo N, Mount C, Omar K, Nair R,
Thurairaja R and Khan MS: Landmarks in the treatment of
muscle-invasive bladder cancer. Nat Rev Urol. 14:565–574. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hermans TJN, Voskuilen CS, van der Heijden
MS, Schmitz-Dräger BJ, Kassouf W, Seiler R, Kamat AM, Grivas P,
Kiltie AE, Black PC and van Rhijn BWG: Neoadjuvant treatment for
muscle-invasive bladder cancer: The past, the present, and the
future. Urol Oncol. Nov 8–2017.(Epub ahead of print). View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Korpics MC, Block AM, Martin B, Hentz C,
Gaynor ER, Henry E, Harkenrider MM and Solanki AA: Concurrent
chemotherapy is associated with improved survival in elderly
patients with bladder cancer undergoing radiotherapy. Cancer.
123:3524–3531. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Kang Z, Li Y, Yu Y and Guo Z: Research
progress on bladder cancer molecular genetics. J Cancer Res Ther.
10 Suppl:C89–C94. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Na SW, Yu SH, Kim KH, Hwang EC, Jung SI,
Kwon DD and Kang TW: The prognosis of patients less than 40 years
with bladder cancer. J Cancer Res Ther. 10:710–714. 2014.PubMed/NCBI
|
|
9
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Bartel DP: MicroRNA Target recognition and
regulatory functions. Cell. 136:215–233. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zhang H, Liu A, Feng X, Tian L, Bo W, Wang
H and Hu Y: MiR-132 promotes the proliferation, invasion and
migration of human pancreatic carcinoma by inhibition of the tumor
suppressor gene PTEN. Prog Biophys Mol Biol. Sep 20–2017.(Epub
ahead of print). View Article : Google Scholar :
|
|
12
|
Slabakova E, Culig Z, Remsik J and Soucek
K: Alternative mechanisms of miR-34a regulation in cancer. Cell
Death Dis. 8:e31002017. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zhou Q, Huang SX, Zhang F, Li SJ, Liu C,
Xi YY, Wang L, Wang X, He QQ, Sun CC and Li DJ: MicroRNAs: A novel
potential biomarker for diagnosis and therapy in patients with
non-small cell lung cancer. Cell Prolif. 50:2017.doi:
10.1111/cpr.12394. View Article : Google Scholar
|
|
14
|
Jena MK: MicroRNAs in the development and
neoplasia of the mammary gland. Version 2. F1000Res. 6:10182017.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Celano M, Rosignolo F, Maggisano V, Pecce
V, Iannone M, Russo D and Bulotta S: MicroRNAs as biomarkers in
thyroid carcinoma. Int J Genomics. 2017:64965702017. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Masuda T, Hayashi N, Kuroda Y, Ito S,
Eguchi H and Mimori K: MicroRNAs as biomarkers in colorectal
cancer. Cancers (Basel). 9:1242017. View Article : Google Scholar :
|
|
17
|
Sathyanarayanan A, Chandrasekaran KS and
Karunagaran D: microRNA-145 downregulates SIP1-expression but
differentially regulates proliferation, migration, invasion and Wnt
signaling in SW480 and SW620 cells. J Cell Biochem. 119:2022–2035.
2018. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Chen GM, Zheng AJ, Cai J and Han P:
microRNA-145-3p inhibits non-small cell lung cancer cell migration
and invasion by targeting PDK1 via the mTOR signaling pathway. J
Cellular Biochem. 119:885–895. 2018. View Article : Google Scholar
|
|
19
|
Setua S, Khan S, Doxtater K, Yallapu MM,
Jaggi M and Chauhan SC: miR-145: Revival of a dragon in pancreatic
cancer. J Nat Sci. 3:pii:e3322017.
|
|
20
|
Moon S, Kim DK and Kim J:
Apoptosis-related microRNA-145-5p enhances the effects of
pheophorbide a-based photodynamic therapy in oral cancer.
Oncotarget. 8:35184–35192. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Chou CH, Chang NW, Shrestha S, Hsu SD, Lin
YL, Lee WH, Yang CD, Hong HC, Wei TY, Tu SJ, et al: miRTarBase
2016: Updates to the experimentally validated miRNA-target
interactions database. Nucleic Acids Res. 44:D239–D247. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Rieger-Christ KM, Lee P, Zagha R,
Kosakowski M, Moinzadeh A, Stoffel J, Ben-Ze'ev A, Libertino JA and
Summerhayes IC: Novel expression of N-cadherin elicits in vitro
bladder cell invasion via the Akt signaling pathway. Oncogene.
23:4745–4753. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bajan S and Hutvagner G: Regulation of
miRNA processing and miRNA mediated gene repression in cancer.
Microrna. 3:10–17. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wozniak M, Mielczarek A and Czyz M: miRNAs
in melanoma: Tumor suppressors and oncogenes with prognostic
potential. Curr Med Chem. 23:3136–3153. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Tutar Y, Özgur A, Tutar E, Tutar L,
Pulliero A and Izzotti A: Regulation of oncogenic genes by
MicroRNAs and pseudogenes in human lung cancer. Biomed
Pharmacother. 83:1182–1190. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zhang Z, Zhang M, Chen Q and Zhang Q:
Downregulation of microRNA-145 promotes epithelial-mesenchymal
transition via regulating Snail in osteosarcoma. Cancer Gene Ther.
24:83–88. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Sheng N, Tan G, You W, Chen H, Gong J,
Chen D, Zhang H and Wang Z: MiR-145 inhibits human colorectal
cancer cell migration and invasion via PAK4-dependent pathway.
Cancer Med. 6:1331–1340. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zhou X, Yue Y, Wang R, Gong B and Duan Z:
MicroRNA-145 inhibits tumorigenesis and invasion of cervical cancer
stem cells. Int J Oncol. 50:853–862. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Li Y, Liu J, Liu ZZ and Wei WB:
MicroRNA-145 inhibits tumour growth and metastasis in osteosarcoma
by targeting cyclin-dependent kinase, CDK6. Eur Rev Med Pharmacol
Sci. 20:5117–5125. 2016.PubMed/NCBI
|
|
31
|
Sathyanarayanan A, Chandrasekaran KS and
Karunagaran D: microRNA-145 modulates epithelial-mesenchymal
transition and suppresses proliferation, migration and invasion by
targeting SIP1 in human cervical cancer cells. Cell Oncol (Dordr).
40:119–131. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Zhang JH, Du AL, Wang L, Wang XY, Gao JH
and Wang TY: Episomal lentiviral Vector-mediated miR-145
overexpression inhibits proliferation and induces apoptosis of
human esophageal carcinomas cells. Recent Pat Anticancer Drug
Discov. 11:453–460. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xiang Y, Zhang Y, Tang Y and Li Q: MALAT1
modulates TGF-β1-induced Endothelial-to-mesenchymal transition
through downregulation of miR-145. Cell Physiol Biochem.
42:357–372. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zeng JF, Ma XQ, Wang LP and Wang W:
MicroRNA-145 exerts tumor-suppressive and chemo-resistance lowering
effects by targeting CD44 in gastric cancer. World J Gastroenterol.
23:2337–2345. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Hecht JR: Metastasis in colorectal cancer:
What makes the tumor aggressive? Eur J Surg Suppl. 104–110.
1998.PubMed/NCBI
|
|
36
|
Ellenrieder V, Adler G and Gress TM:
Invasion and metastasis in pancreatic cancer. Ann Oncol. 10 Suppl
4:S46–S50. 1999. View Article : Google Scholar
|
|
37
|
Singh M, Darcy KM, Brady WE, Clubwala R,
Weber Z, Rittenbach JV, Akalin A, Whitney CW, Zaino R, Ramirez NC
and Leslie KK: Cadherins, catenins and cell cycle regulators:
Impact on survival in a Gynecologic Oncology Group phase II
endometrial cancer trial. Gynecol Oncol. 123:320–328. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Derycke LD and Bracke ME: N-cadherin in
the spotlight of cell-cell adhesion, differentiation,
embryogenesis, invasion and signalling. Int J Dev Biol. 48:463–476.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Mariotti A, Perotti A, Sessa C and Ruegg
C: N-cadherin as a therapeutic target in cancer. Expert Opin
Investig Drugs. 16:451–465. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Wu HM, Huang HY, Schally AV, Chao A, Chou
HH, Leung PC and Wang HS: Growth hormone-releasing hormone
antagonist inhibits the invasiveness of human endometrial cancer
cells by down-regulating twist and N-cadherin expression.
Oncotarget. 8:4410–4421. 2017.PubMed/NCBI
|
|
41
|
Da C, Wu K, Yue C, Bai P, Wang R, Wang G,
Zhao M, Lv Y and Hou P: N-cadherin promotes thyroid tumorigenesis
through modulating major signaling pathways. Oncotarget.
8:8131–8142. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wu X, Wang H, Lian Y, Chen L, Gu L, Wang J
and Huang Y, Deng M, Gao Z and Huang Y: GTSE1 promotes cell
migration and invasion by regulating EMT in hepatocellular
carcinoma and is associated with poor prognosis. Sci Rep.
7:51292017. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Abufaraj M, Haitel A, Moschini M, Gust K,
Foerster B, Özsoy M, D'Andrea D, Karakiewicz PI, Rouprêt M,
Briganti A and Shariat SF: Prognostic role of N-cadherin expression
in patients with invasive bladder cancer. Clin Genitourin Cancer.
Aug 15–2017.(Epub ahead of print).
|
|
44
|
Mo D, Yang D, Xiao X, Sun R, Huang L and
Xu J: MiRNA-145 suppresses lung adenocarcinoma cell invasion and
migration by targeting N-cadherin. Biotechnol Lett. 39:701–710.
2017. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Merchant N, Nagaraju GP, Rajitha B,
Lammata S, Jella KK, Buchwald ZS, Lakka SS and Ali AN: Matrix
metalloproteinases: Their functional role in lung cancer.
Carcinogenesis. 38:766–780. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Han YH, Kee JY, Kim DS, Mun JG, Park SH,
Kim YJ, Um JY and Hong SH: Arctii fructus inhibits colorectal
cancer cell proliferation and MMPs mediated invasion via AMPK. Am J
Chin Med. 45:1309–1325. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Lyu ZK, Li CL, Jin Y, Liu YZ, Zhang X,
Zhang F, Ning LN, Liang ES, Ma M, Gao W, et al: Paeonol exerts
potential activities to inhibit the growth, migration and invasion
of human gastric cancer BGC823 cells via downregulating MMP-2 and
MMP-9. Mol Med Rep. 16:7513–7519. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Chen W, Zhu H, Yin L, Wang T, Wu J, Xu J,
Tao H, Liu J and He X: lncRNA-PVT1 facilitates invasion through
upregulation of MMP9 in nonsmall cell lung cancer cell. DNA Cell
Biol. 36:787–793. 2017.PubMed/NCBI
|
|
49
|
Suyama K, Shapiro I, Guttman M and Hazan
RB: A signaling pathway leading to metastasis is controlled by
N-cadherin and the FGF receptor. Cancer Cell. 2:301–314. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Gao P, Xing AY, Zhou GY, Zhang TG, Zhang
JP, Gao C, Li H and Shi DB: The molecular mechanism of microRNA-145
to suppress invasion-metastasis cascade in gastric cancer.
Oncogene. 32:491–501. 2013. View Article : Google Scholar : PubMed/NCBI
|