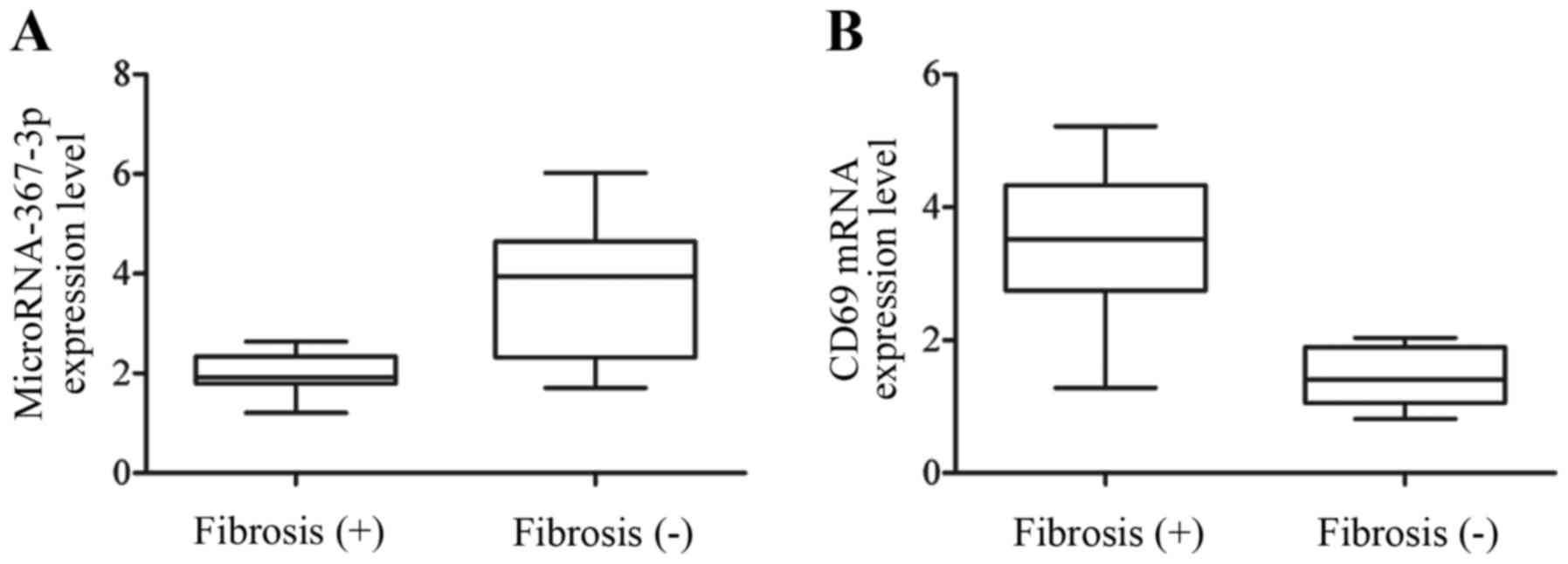
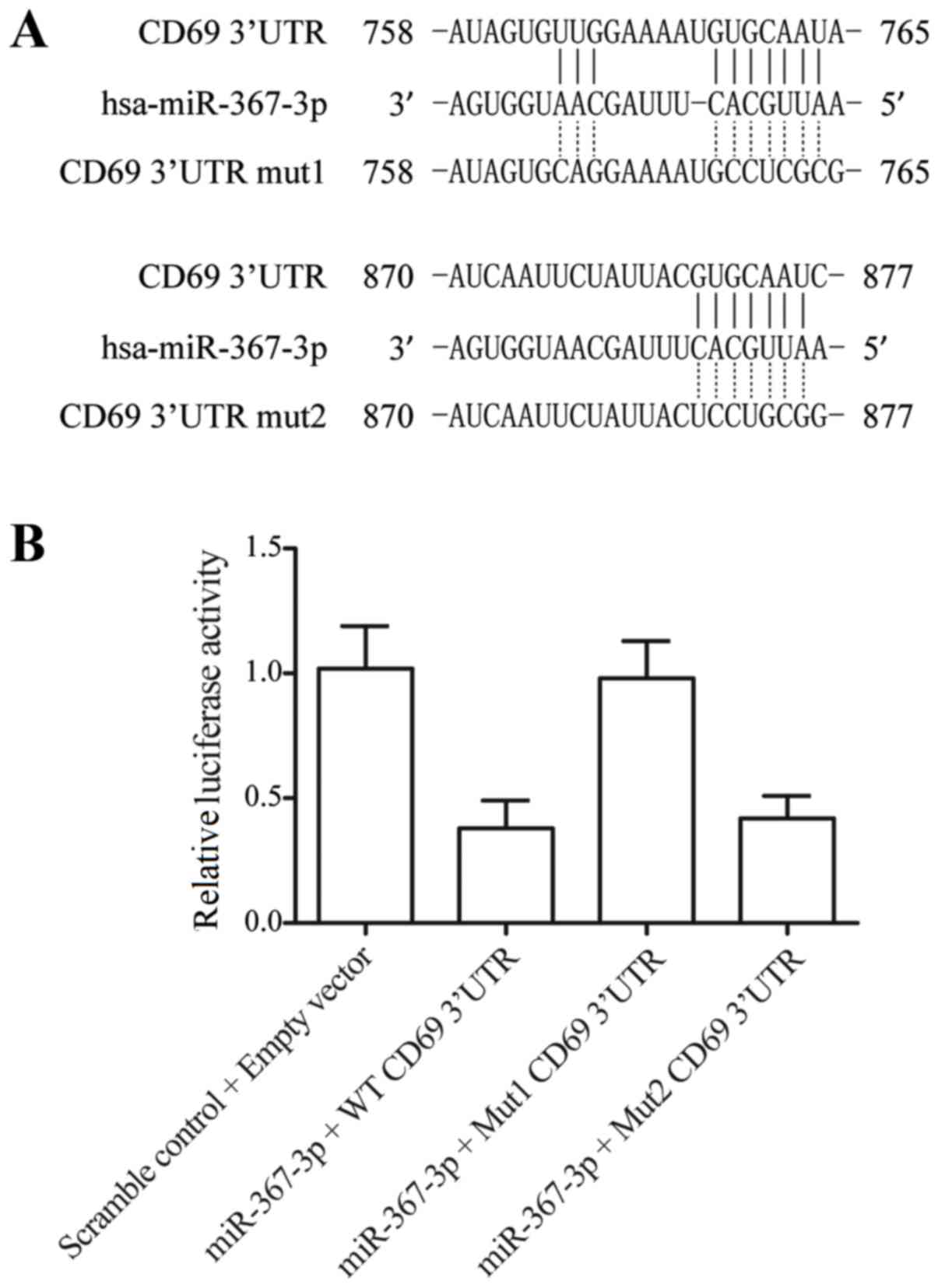
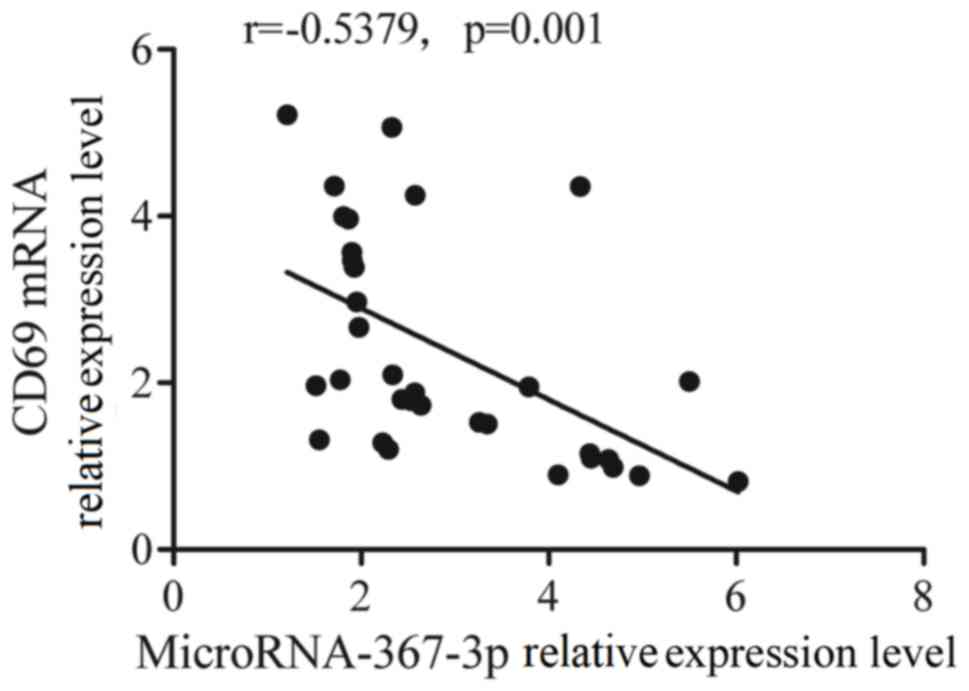
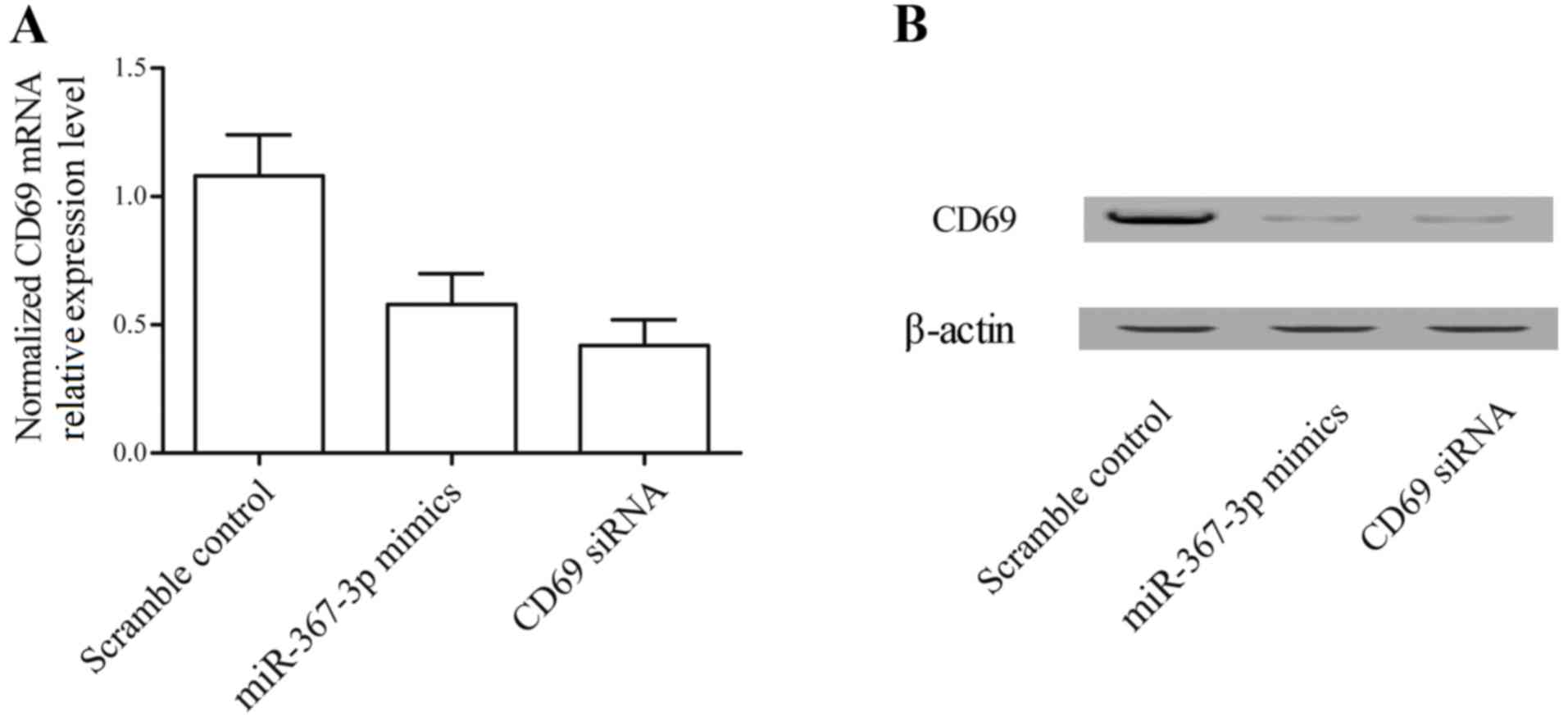
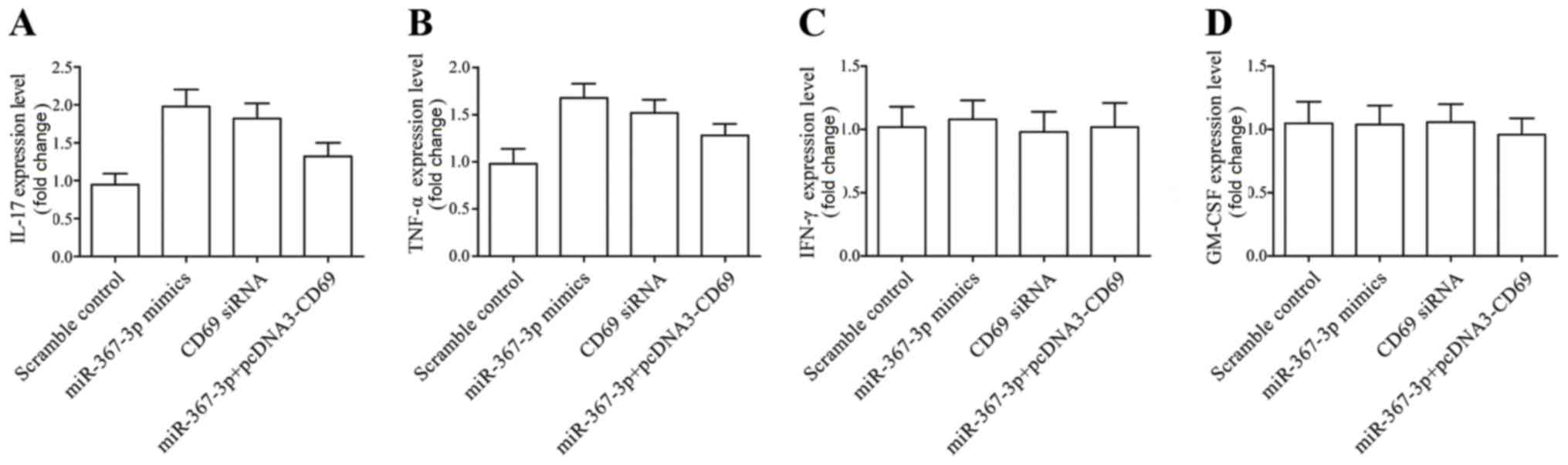
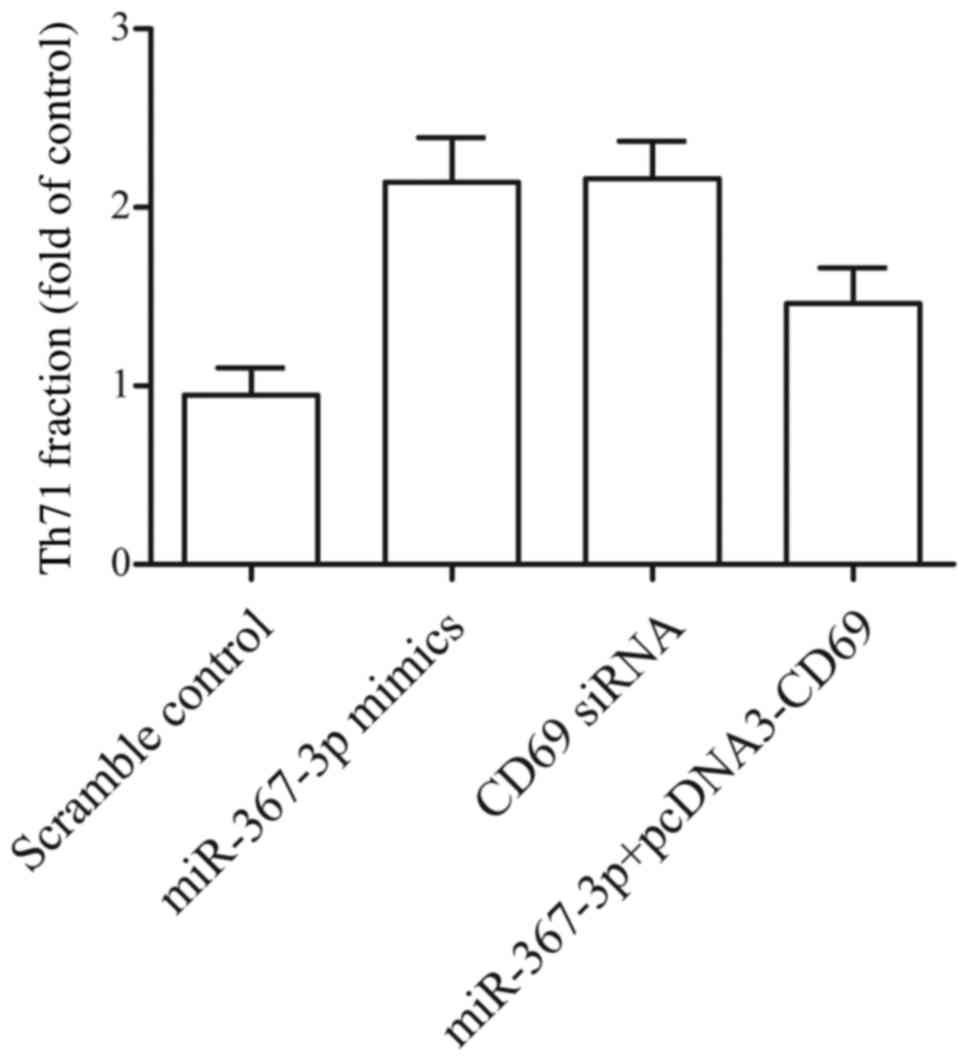
|
1
|
Berk BC, Fujiwara K and Lehoux S: ECM
remodeling in hypertensive heart disease. J Clin Invest.
117:568–575. 2007. View
Article : Google Scholar : PubMed/NCBI
|
|
2
|
Frangogiannis NG: Regulation of the
inflammatory response in cardiac repair. Circ Res. 110:159–173.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kong P, Christia P and Frangogiannis NG:
The pathogenesis of cardiac fibrosis. Cell Mol Life Sci.
71:549–574. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
van den Borne SW, Diez J, Blankesteijn WM,
Verjans J, Hofstra L and Narula J: Myocardial remodeling after
infarction: The role of myofibroblasts. Nat Rev Cardiol. 7:30–37.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Zamilpa R and Lindsey ML: Extracellular
matrix turnover and signaling during cardiac remodeling following
MI: Causes and consequences. J Mol Cell Cardiol. 48:558–563. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Tomasek JJ, Gabbiani G, Hinz B, Chaponnier
C and Brown RA: Myofibroblasts and mechano-regulation of connective
tissue remodelling. Nat Rev Mol Cell Biol. 3:349–363. 2002.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Bujak M and Frangogiannis NG: The role of
TGF-beta signaling in myocardial infarction and cardiac remodeling.
Cardiovasc Res. 74:184–195. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Bartel DP: MicroRNAs: Genomics,
biogenesis, mechanism, and function. Cell. 116:281–297. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Griffiths-Jones S: The microRNA registry.
Nucleic Acids Res. 32:(Database Issue). D109–D111. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chen JF, Mandel EM, Thomson JM, Wu Q,
Callis TE, Hammond SM, Conlon FL and Wang DZ: The role of
microRNA-1 and microRNA-133 in skeletal muscle proliferation and
differentiation. Nat Genet. 38:228–233. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Martín P, Gómez M, Lamana A, Cruz-Adalia
A, Ramírez-Huesca M, Ursa MA, Yáñez-Mo M and Sánchez-Madrid F: CD69
association with Jak3/Stat5 proteins regulates Th17 cell
differentiation. Mol Cell Biol. 30:4877–4889. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Shinoda K, Tokoyoda K, Hanazawa A,
Hayashizaki K, Zehentmeier S, Hosokawa H, Iwamura C, Koseki H,
Tumes DJ, Radbruch A and Nakayama T: Type II membrane protein CD69
regulates the formation of resting T-helper memory. Proc Natl Acad
Sci USA. 109:7409–7414. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Alari-Pahissa E, Notario L, Lorente E,
Vega-Ramos J, Justel A, López D, Villadangos JA and Lauzurica P:
CD69 does not affect the extent of T cell priming. PLoS One.
7:e485932012. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Lieberman SM, Kim JS, Corbo-Rodgers E,
Kambayashi T, Maltzman JS, Behrens EM and Turka LA: Site-specific
accumulation of recently activated CD4+ Foxp3+ regulatory T cells
following adoptive transfer. Eur J Immunol. 42:1429–1435. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Shiow LR, Rosen DB, Brdicková N, Xu Y, An
J, Lanier LL, Cyster JG and Matloubian M: CD69 acts downstream of
interferon-alpha/beta to inhibit S1P1 and lymphocyte egress from
lymphoid organs. Nature. 440:540–544. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Lamana A, Martin P, de la Fuente H,
Martinez-Muñoz L, Cruz-Adalia A, Ramirez-Huesca M, Escribano C,
Gollmer K, Mellado M, Stein JV, et al: CD69 modulates
sphingosine-1-phosphate-induced migration of skin dendritic cells.
J Invest Dermatol. 131:1503–1512. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Sancho D, Gómez M, Viedma F, Esplugues E,
Gordón-Alonso M, García-López MA, de la Fuente H, Martínez-A C,
Lauzurica P and Sánchez-Madrid F: CD69 downregulates autoimmune
reactivity through active transforming growth factor-beta
production in collagen-induced arthritis. J Clin Invest.
112:872–882. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Cruz-Adalia A, Jiménez-Borreguero LJ,
Ramírez-Huesca M, Chico-Calero I, Barreiro O, López-Conesa E,
Fresno M, Sánchez-Madrid F and Martín P: CD69 limits the severity
of cardiomyopathy after autoimmune myocarditis. Circulation.
122:1396–1404. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Radulovic K, Manta C, Rossini V, Holzmann
K, Kestler HA, Wegenka UM, Nakayama T and Niess JH: CD69 regulates
type I IFN-induced tolerogenic signals to mucosal CD4 T cells that
attenuate their colitogenic potential. J Immunol. 188:2001–2013.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gómez M, Sanz-González SM, Nabah Abu YN,
Lamana A, Sánchez-Madrid F and Andrés V: Atherosclerosis
development in apolipoprotein E-null mice deficient for CD69.
Cardiovasc Res. 81:197–205. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Khallou-Laschet J, Caligiuri G, Groyer E,
Tupin E, Gaston AT, Poirier B, Kronenberg M, Cohen JL, Klatzmann D,
Kaveri SV and Nicoletti A: The proatherogenic role of T cells
requires cell division and is dependent on the stage of the
disease. Arterioscler Thromb Vasc Biol. 26:353–358. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Laffón A, García-Vicuña R, Humbría A,
Postigo AA, Corbí AL, de Landázuri MO and Sánchez-Madrid F:
Upregulated expression and function of VLA-4 fibronectin receptors
on human activated T cells in rheumatoid arthritis. J Clin Invest.
88:546–552. 1991. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
García-Monzón C, Moreno-Otero R, Pajares
JM, García-Sánchez A, López-Botet M, de Landázuri MO and
Sánchez-Madrid F: Expression of a novel activation antigen on
intrahepatic CD8+ T lymphocytes in viral chronic active hepatitis.
Gastroenterology. 98:1029–1035. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Khan M, Nickoloff E, Abramova T, Johnson
J, Verma SK, Krishnamurthy P, Mackie AR, Vaughan E, Garikipati VN,
Benedict C, et al: Embryonic stem cell-derived exosomes promote
endogenous repair mechanisms and enhance cardiac function following
myocardial infarction. Circ Res. 117:52–64. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ong S, Ligons DL, Barin JG, Wu L, Talor
MV, Diny N, Fontes JA, Gebremariam E, Kass DA, Rose NR and Čiháková
D: Natural killer cells limit cardiac inflammation and fibrosis by
halting eosinophil infiltration. Am J Pathol. 185:847–861. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(-Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Pauley KM, Cha S and Chan EK: MicroRNA in
autoimmunity and autoimmune diseases. J Autoimmun. 32:189–194.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Miranda KC, Huynh T, Tay Y, Ang YS, Tam
WL, Thomson AM, Lim B and Rigoutsos I: A pattern-based method for
the identification of MicroRNA binding sites and their
corresponding heteroduplexes. Cell. 126:1203–1217. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Thum T, Gross C, Fiedler J, Fischer T,
Kissler S, Bussen M, Galuppo P, Just S, Rottbauer W, Frantz S, et
al: MicroRNA-21 contributes to myocardial disease by stimulating
MAP kinase signalling in fibroblasts. Nature. 456:980–984. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
van Rooij E, Sutherland LB, Thatcher JE,
DiMaio JM, Naseem RH, Marshall WS, Hill JA and Olson EN:
Dysregulation of microRNAs after myocardial infarction reveals a
role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci USA.
105:13027–13032. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Duisters RF, Tijsen AJ, Schroen B,
Leenders JJ, Lentink V, van der Made I, Herias V, van Leeuwen RE,
Schellings MW, Barenbrug P, et al: miR-133 and miR-30 regulate
connective tissue growth factor: Implications for a role of
microRNAs in myocardial matrix remodeling. Circ Res. 104:170–178,
6p following 178. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Nicoletti A, Heudes D, Mandet C, Hinglais
N, Bariety J and Michel JB: Inflammatory cells and myocardial
fibrosis: Spatial and temporal distribution in renovascular
hypertensive rats. Cardiovasc Res. 32:1096–1107. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Xia Y, Lee K, Li N, Corbett D, Mendoza L
and Frangogiannis NG: Characterization of the inflammatory and
fibrotic response in a mouse model of cardiac pressure overload.
Histochem Cell Biol. 131:471–481. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Schellings MW, Pinto YM and Heymans S:
Matricellular proteins in the heart: Possible role during stress
and remodeling. Cardiovasc Res. 64:24–31. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Frangogiannis NG: Matricellular proteins
in cardiac adaptation and disease. Physiol Rev. 92:635–688. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Testi R, D'Ambrosio D, De Maria R and
Santoni A: The CD69 receptor: A multipurpose cell-surface trigger
for hematopoietic cells. Immunol Today. 15:479–483. 1994.
View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Lu H, Crawford RB, North CM, Kaplan BL and
Kaminski NE: Establishment of an immunoglobulin m antibody-forming
cell response model for characterizing immunotoxicity in primary
human B cells. Toxicol Sci. 112:363–373. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Hamann J, Fiebig H and Strauss M:
Expression cloning of the early activation antigen CD69, a type II
integral membrane protein with a C-type lectin domain. J Immunol.
150:4920–4927. 1993.PubMed/NCBI
|
|
39
|
López-Cabrera M, Santis AG, Fernández-Ruiz
E, Blacher R, Esch F, Sánchez-Mateos P and Sánchez-Madrid F:
Molecular cloning, expression, and chromosomal localization of the
human earliest lymphocyte activation antigen AIM/CD69, a new member
of the C-type animal lectin superfamily of signal-transmitting
receptors. J Exp Med. 178:537–547. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Ziegler SF, Ramsdell F, Hjerrild KA,
Armitage RJ, Grabstein KH, Hennen KB, Farrah T, Fanslow WC, Shevach
EM and Alderson MR: Molecular characterization of the early
activation antigen CD69: A type II membrane glycoprotein related to
a family of natural killer cell activation antigens. Eur J Immunol.
23:1643–1648. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Ziegler SF, Levin SD, Johnson L, Copeland
NG, Gilbert DJ, Jenkins NA, Baker E, Sutherland GR, Feldhaus AL and
Ramsdell F: The mouse CD69 gene. Structure, expression, and mapping
to the NK gene complex. J Immunol. 152:1228–1236. 1994.PubMed/NCBI
|
|
42
|
Schnittger S, Hamann J, Dannenberg C,
Fiebig H, Strauss M and Fonatsch C: Regional sublocalization of the
human CD69 gene to chromosome bands 12p12.3-p13.2, the predicted
region of the human natural killer cell gene complex. Eur J
Immunol. 23:2711–2713. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Borrego F, Peña J and Solana R: Regulation
of CD69 expression on human natural killer cells: Differential
involvement of protein kinase C and protein tyrosine kinases. Eur J
Immunol. 23:1039–1043. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Bezouska K, Nepovím A, Horváth O, Pospísil
M, Hamann J and Feizi T: CD 69 antigen of human lymphocytes is a
calcium-dependent carbohydrate-binding protein. Biochem Biophys Res
Commun. 208:68–74. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Gerosa F, Tommasi M, Benati C, Gandini G,
Libonati M, Tridente G, Carra G and Trinchieri G: Differential
effects of tyrosine kinase inhibition in CD69 antigen expression
and lytic activity induced by rIL-2, rIL-12, and rIFN-alpha in
human NK cells. Cell Immunol. 150:382–390. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Felts JM, Itakura H and Crane RT: The
mechanism of assimilation of constituents of chylomicrons, very low
density lipoproteins and remnants-a new theory. Biochem Biophys Res
Commun. 66:1467–1475. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Karlhofer FM and Yokoyama WM: Stimulation
of murine natural killer (NK) cells by a monoclonal antibody
specific for the NK1.1 antigen. IL-2-activated NK cells possess
additional specific stimulation pathways. J Immunol. 146:3662–3673.
1991.PubMed/NCBI
|
|
48
|
Luan L, Han S, Wang H and Liu X:
Down-regulation of the Th1, Th17, and Th22 pathways due to
anti-TNF-α treatment in psoriasis. Int Immunopharmacol. 29:278–284.
2015. View Article : Google Scholar : PubMed/NCBI
|