Introduction
One of the exceptional features of the liver is its
remarkable regenerative capacity following resection or other
injuries (1). Liver
transplantation remains the only cure for many end-stage hepatic
diseases (2). However, one key
limiting factor in the application of liver surgery or
transplantation is defective regeneration of small-for-size and
partial grafts (3). Therefore,
there is impetus to investigate novel therapeutic strategies to
enhance liver regeneration after surgical injury (4).
Liver regeneration is a highly organized and complex
progress that occurs initially through the proliferation of
hepatocytes and then through the proliferation of nonparenchymal
cells, including bile duct epithelial cells, hepatic stellate cells
and sinusoidal endothelial cells (5–7).
After partial hepatectomy (PHx), the intrahepatic angiogenesis,
characterized by the increased sinusoidal cell proliferation and
microvascular architecture remodeling, is essential for liver
regeneration, as it serves a pivotal role in the supply of blood to
the newly replicating hepatocytes (8). Vascular endothelial growth factor A
(VEGF-A), a 45-kilodalton dimeric heparin-binding glycoprotein, is
the main growth factor in angiogenesis and vasculogenesis (9). The effects of VEGF-A are mediated
through three distinct high-affinity cell membrane receptors
tyrosine kinases VEGFR1, VEGFR2 and VEGFR3 (10), of which the VEGF-A/VEGFR2 pathway
contributes to the majority of VEGF-regulated proangiogenic effects
(11). Previous studies
demonstrate that VEGF-A and VEGFR2 expression is increased during
liver regeneration (12,13), whereas blockage of VEGF
significantly delays liver tissue repair, indicating VEGF as a
promising target for liver regeneration (14). Systemic administration of
exogenous VEGF or adenovirus-mediated gene transfer of VEGF promote
functional hepatic recovery following hepatectomy (15,16), as a result of neovascularization.
However, due to the short half-life and rapid diffusion, it is hard
to retain an effective local concentration of VEGF in the remnant
liver tissue (17,18). Furthermore, high doses treatment
may induce undesirable side effects (19,20). Therefore, developing an effective
local VEGF delivery system may offer an optimal therapy for liver
regeneration.
Our previous study constructed a fusion protein that
consisted of VEGF and collagen binding domain (CBD), a short
peptide with 7 amino acids (TKKTLRT) that could specifically bind
to type I collagen (21). Another
previous study had shown that CBD-VEGF can promote tissue
regeneration and improve neo-urethra function in a beagle extensive
urethral defect model (22). It
was shown that CBD-VEGF could attenuate left ventricular
remodeling, reduce infarct size and promote cardio-angiogenesis in
a porcine chronic myocardial infraction model (23). The present study investigated the
effect of CBD-VEGF on liver regeneration using a mice partial
hepatectomy model. The results demonstrated that CBD-VEGF could
significantly promote the proliferation of hepatocytes, enhance the
reconstruction of vascularization and promote liver
regeneration.
Materials and methods
Preparation of CBD-VEGF
CBD-VEGF was prepared as previously described
(22,23). Briefly, full length complementary
DNA of human VEGF165 was constructed and amplified linking a
sequence that encodes the collagen binding domain (TKKTLRT). Then,
the encoding gene of CBDVEGF was inserted into plasmid pET28a,
followed by transfer into BL21-strain Escherichia coli.
Selected single clones were inoculated into 4 ml Luria-Bertani (LB)
medium as primary culture, followed by inoculation 4 ml into 200 ml
fresh LB medium and culture at 37°C for ~4 h at 200 rpm. When A600
reached 0.6-0.8, 1 mM isopropyl β-D-thiogalactopyranoside was used
to induce protein expression for 5 h. After the inclusion body was
refolded, the protein containing 6X-His tag was purified by nickel
chelate chromatography and ion exchange chromatography on a
prepacked HiTrap heparin HP columns (Cytiva).
Animals
A total of 60 C57BL/6 male mice (22–24 g body
weight) 8–10 weeks, were purchased from Jinan Pengyue Experimental
Animal Breeding Co. Ltd. All animal experiments were approved by
The Ethics Committee Medical College of Qingdao University
(approval no. QDU-AEC-2021166) and were conducted according to the
Guide for the Care and Use of Laboratory Animals published by the
National Academy of Sciences and the National Institutes of Health
(24). The mice were housed in a
standard room with controlled humidity (55–60%) and temperature
(23–25°C) under a12-h light/dark cycle with free access to water
and food.
PHx model
Mice were subjected to two-thirds PHx as previously
described (25,26). Briefly, after anesthetization by
isoflurane (4% for induction, 2% for maintenance; RWD Life Science
Co., Ltd.), the median lobe plus gall bladder and the left lateral
lobe were removed. Immediately after PHx, native VEGF (500
ng/dose), or CBD-VEGF (500 ng/dose) was injected via portal vein.,
with PBS as a vehicle control (n=5 per group). The dose of
CBD-VEGF/VEGF was chosen based on our previous studies and
preliminary experiments (21,27). The sham operation group was
conducted midline laparotomy incision without resection of the
liver lobe. The mice were euthanized by cervical dislocation after
deep anesthesia with isoflurane (4% for induction) at various time
points after surgery. The serum and liver tissue were collected for
subsequent experiments. Levels of alanine transaminase (ALT),
aspartate transaminase (AST) in serum were measured by an automatic
biochemical analyzer (Chemray 800; Rayto Life and Analytical
Sciences) according to the manufacturer's instructions (Rayto Life
and Analytical Sciences).
ELISA assay
Frozen liver tissue from VEGF or CBD-VEGF group at
24 h were homogenized and the levels of VEGF in the liver tissue of
PHx mice were detected using an ELISA kit (mlBio; cat. no.
ml064255) according to manufacturer's instructions.
Immunofluorescence (IF) analysis
IF analysis was performed as previously described
(28). Briefly, The liver tissues
of experimental and control group mice were embedded in OCT and
stored at −80°C. Next, the liver tissue samples sections (8 µm
thick) were fixed with 4% paraformaldehyde for 30 min and blocked
with 5% bovine serum albumin (BSA; Wuhan Servicebio Technology Co.,
Ltd.; cat. no. G5001) both for 1 h at room temperature. For
bromodeoxyuracil (BrdU) staining, BrdU (50 mg/kg body weight) was
injected intraperitoneally 4 h before liver tissue were harvested.
Subsequently, the primary antibody including anti-VEGFR2 (ABclonal
Biotech Co., Ltd.; 1:200; cat. no. A1484), anti-BrdU (ABclonal
Biotech Co., Ltd.;, 1:200; cat. no. A1482) anti-Ki67 (Abcam; 1:300;
cat. no. ab16667), anti-CD31 (BD Pharmingen; BD Biosciences; 1:300;
cat. no. 553373) and β-catenin (CTNNB1) antibody (ABclonal Biotech
Co., Ltd.; 1:200; cat. no. A19657) were incubated overnight at 4°C,
followed by incubating with the corresponding
fluorescein-conjugated secondary antibodies.
4′,6-diamidino-2-phenylindole (DAPI; Wuhan Servicebio Technology
Co., Ltd.) was used to stain cell nuclei for 5 min at room
temperature. The sections were visualized using a fluorescence
microscope (Olympus BX50; Olympus Corporation). Then 5–8 images
were captured for each sample and the percentage of positive nuclei
was analyzed using ImageJ software (version 1.43; National
Institutes of Health).
Immunohistochemistry staining
The formalin-fixed liver tissue was dehydrated
through graded alcohols and embedded in paraffin wax (4%
phosphate-buffered and formalin-fixed for 24 h at room temperature;
5 µm thick). The sections were blocked with 5% BSA for 1 h and then
incubated with anti-proliferating cell nuclear antigen (PCNA;
Affinity Biosciences; 1:200; cat. no. AF0239) overnight at 4°C. The
slides were washed with PBS, then incubated with the HRP conjugated
secondary antibody (ABclonal Biotech Co., Ltd.; 1:1,000; cat. no.
AS014) for 1 h at room temperature. Staining was developed with
diaminobenzidine substrate solution (DAB; Wuhan Servicebio
Technology Co., Ltd.) and the sections were counterstained with
hematoxylin for 1 min at room temperature, then they were
dehydrated through graded alcohols, mounted and visualized under a
light microscope. Data were expressed as the percentage of PCNA
positive nuclei analyzed using ImageJ software (version 1.43;
National Institutes of Health).
Hematoxylin and eosin staining
The liver tissue was fixed with phosphate-buffered
and formalin (Wuhan Servicebio Technology Co., Ltd.) for 24 h at
room temperature. After dehydration through graded alcohols, the
tissue was embedded in paraffin wax and sectioned at 5 µm. The
sections were stained with hematoxylin-eosin for 5 min at room
temperature and observed under an Olympus light microscope
(magnification, ×200; Olympus Corporation), and three fields of
view were examined per section.
Protein-dye conjugation and in vivo
distribution
CBD-VEGF was labeled with 775-B2 NHS ester according
to the manufacturer's instructions. CBD-VEGF conjugated with the
775-B2 NHS ester was injected into the portal vein of PHx mice.
Bioluminescence images were acquired and processed using an IVIS
Lumina XRMS III in vivo imaging system (PerkinElmer, Inc.)
at 6, 24, 48 and 72 h after administration. Then, the livers were
harvested for frozen sections (8 µm). The sections were incubated
with DAPI for 5 min at room temperature and the distribution of
fluorescein labeled protein was examined by fluorescence microscopy
(magnification, ×200; Olympus Corporation), and three fields of
view were examined per section.
Western blot analysis
Total protein was extracted from liver tissues using
RIPA lysis buffer (Beyotime Institute of Biotechnology). The
protein concentration was confirmed using a BCA Protein Assay kit
(Epizyme; cat. no. ZJ102) and protein samples (40 µg) were
collected and subjected to SDSPAGE (6% separation gel and 10%
concentration gel) and then transferred to a polyvinylidene
fluoride membrane (MilliporeSigma). Next, the membranes were
blocked with skimmed milk (PPLYGEN; cat. no. P1622) for 1 h at room
temperature and followed by incubating with primary antibodies
overnight at 4°C: native VEGF (R & D Systems; 1:1,000; cat. no.
293-VE/CF), VEGFR2 (Abcam; 1:1,000; cat. no. AB39256), PCNA
(Affinity Biosciences; 1:1,000), β-actin (ABclonal Biotech Co.,
Ltd.; 1:1,000; cat. no. AC028) and GAPDH (Aksomics Inc.; 1:3,000;
cat. no. KC-5G5) and HRP conjugated secondary antibody (ABclonal
Biotech Co., Ltd.; 1:1,000; cat. no. AS014) at room temperature for
1 h. The protein band were detected ECL regents, imaged using Tanno
imaging system (Tanon 5200; Tanon Science & Technology) and the
protein bands analyzed with ImageJ (version 1.43; National
Institutes of Health).
Reverse transcription-quantitative PCR
(RT-qPCR)
Total RNA was extracted using the GeneJet RNA
Purification Kit (Thermo Fisher Scientific, Inc.) from the liver
tissue. Then, cDNA was synthesized from the purified RNA (200
ng/sample) using a transcription kit (Takara Biotechnology Co.,
Ltd.), qPCR were performed in triplicate using a SYBR Premix Ex Taq
(Vazyme Biotech Co., Ltd.). The following thermo cycling conditions
were used: Pre-incubation at 95°C for 30 sec; amplification at 95°C
for 15 sec, 57°C for 15 sec and 72°C for 1 min, for 55 cycles;
melting curve at 95°C for 15 sec, 60°C for 1 min and 95°C for 15
sec. The following primers were used in the current study: HGF
forward 5′-CACTCCCGAGAACTTCAAATGC-3′ reverse
5′-TGTCCACTTGACACGTCACACTT-3′. GAPDH forward
5′-CAGTTACTTCCCCAGCAA-3′ and reverse 5′-CACGACTCATACAGCACCT-3′.
GAPDH was regarded as the internal control. The target gene
expression was quantified using the 22−ΔΔCq method
(29).
Statistical analysis
All data were presented as mean ± standard deviation
and all experiments were performed at ≤3 times. Differences between
two groups were analyzed using unpaired Student's ttest and the
multiple groups were performed using oneway ANOVA followed by
Tukey's post hoc test. Data were analyzed using the GraphPad Prism
program version 8.0 (GraphPad Software, Inc.). P<0.05 was
considered to indicate a statistically significant difference.
Results
Bio-distribution of CBD-VEGF in PHx
mice
CBD-VEGF was used to modify VEGF to enhance its
binding affinity with extracellular matrix (Fig. 1A). Purified CBD-VEGF and native
VEGF were analyzed by SDS-PAGE (Fig.
1B). As described previously, the in vivo
biodistribution of CBD-VEGF was evaluated by injecting 775-B2 NHS
ester conjugated CBD-VEGF via the hepatic portal vein (30). The biodistribution of CBD-VEGF was
measured at 6, 24, 48 and 72 h after injection. As shown in
Fig. 1D, the CBD-VEGF infused via
the portal vein initially accumulated in the liver of PHx mice. At
24 and 48 h post injection, fluorescence signals were detected in
the liver, intestines and kidney, possibly due to the metabolism of
CBD-VEGF in the body. However, at 72 h after injection, no visible
fluorescence signal were detected in the liver. In addition, ELISA
results showed that the concentration of CBD-VEGF in liver was
significantly increased at 24 and 48 h after PHx compared with
native VEGF group (24 h: 36.39±2.03 pg/ml vs. 22.53±1.62 pg/ml,
P<0.01; 48 h: 26.20±1.22 pg/ml vs. 7.72±1.04 pg/ml, P<0.01)
(Fig. 1C), indicating that
CBD-VEGF could be retained at the injury site with less diffusion
than that of native VEGF.
CBD-VEGF can promote the
reconstruction of vascularization
Previous reports has identified CD31 as a cell
marker for liver sinusoidal endothelial cells (LSECs) and VEGFR2
and serving an important role in regulating LSECs biological
function during liver regeneration (31,32). The extent of vascular remodeling
in regenerated liver was measured by immunostaining. As shown in
Fig. 2A, the neovascularization
in CBD-VEGF group was markedly enhanced compared with native VEGF
group. In addition, the quantitative analysis of VEGFR2 and CD31
revealed a more positive area in the CBD-VEGF group (Fig. 2B and C; VEGFR2: 20.25±2.31% vs.
11.5±1.34%, P<0.01; CD31: 18.75±1.32% vs. 12.75±0.74%,
P<0.01). Moreover, western blotting further verified that VEGFR2
expression was significantly upregulated in CBD-VEGF group
(Fig. 2D and E). Immunostaining
results further demonstrated that CBD-VEGF could improve
proliferation of LSECs (Fig. 2F and
G). Taken together, these results indicated that CBD-VEGF
promoted angiogenesis in remnant liver during liver
regeneration.
CBD-VEGF alleviates PHx-induced liver
injury
The therapeutic effect of CBD-VEGF on liver
regeneration was further verified via PHx mice model. No
significant discrepancy was detected in hematoxylin and
eosin-stained liver sections (Fig.
3A and Fig. S1. However,
serum levels of ALT and AST (two liver injury markers), were
significantly decreased in CBD-VEGF treated group compared with
those of native VEGF group, at 2 days and 3 days after PHx,
respectively (Fig. 3B and C).
These results suggested that CBDVEGF application alleviate
PHx-induced liver injury.
CBD-VEGF promotes liver regeneration
following PHx
The present study further investigated the effect of
CBD-VEGF on hepatocyte proliferation and liver regeneration
following PHx. The results showed that the liver-to-body weight
ratio of CBD-VEGF group was significantly restored compared with
that of native VEGF group, at 2 and 3 days post-PHx, respectively.
Liver weight could be restored almost to the original level at 7
days following surgery and no significant difference was found
between groups at 7 days following surgery, indicating that the
CBD-VEGF treated group could regenerate faster at the early phase
of liver regeneration (Fig. 4A and
B). Furthermore, no significant difference in the body weight
loss and liver cell size was noted between groups following
CBD-VEGF treatment (Fig. 4C-E).
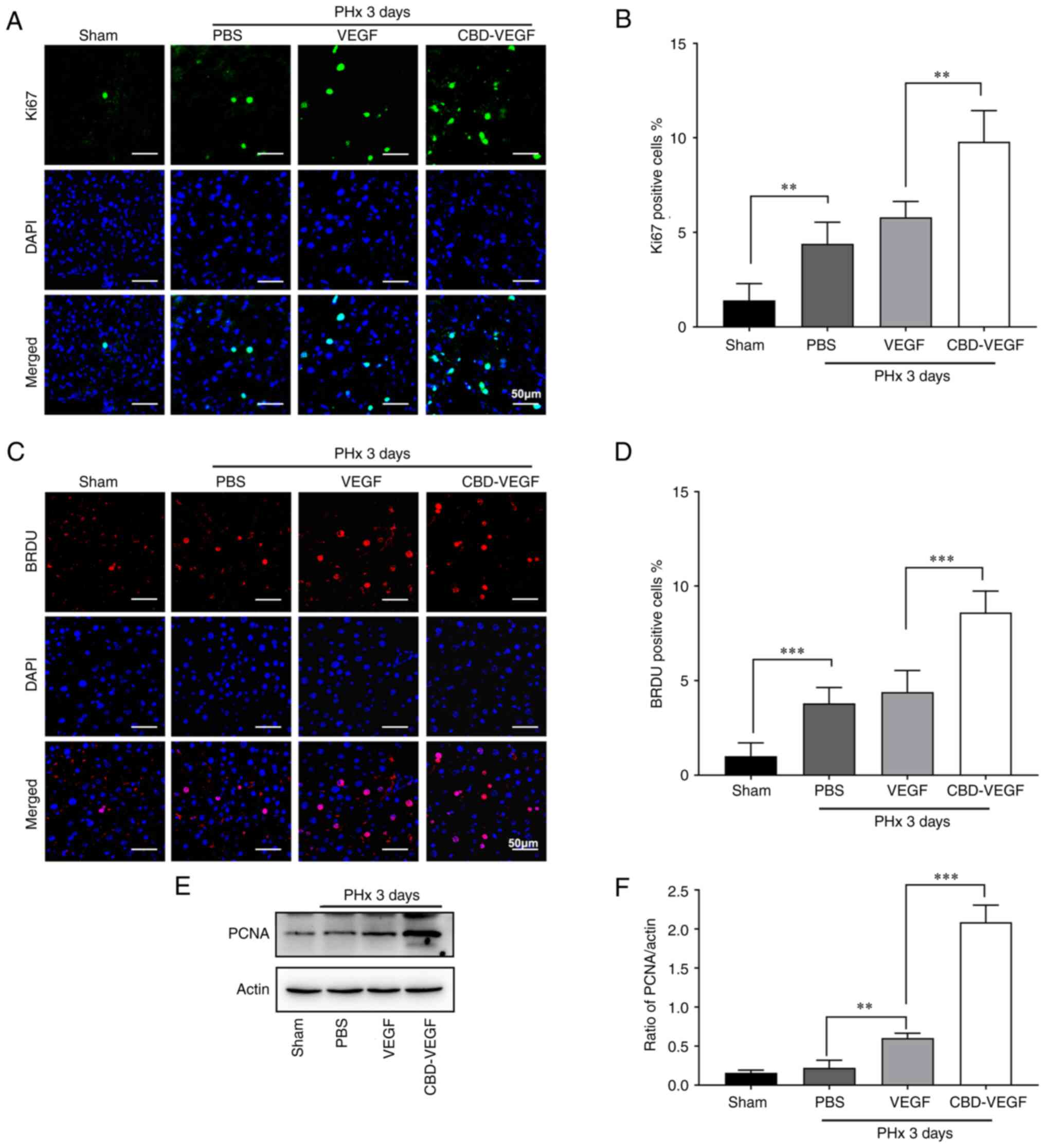
To further confirm the effect of CBD-VEGF on hepatocyte
proliferation, expression of Ki67 and BrdU were evaluated at 2 and
3 days after PHx (Figs. 5A and C
and 6A and C). CBDVEGF
administration could significantly increase the number of
Ki67+ cells when compared with the native VEGF (Figs. 5B and 6B). (day 2: 20.8±2.135% vs. 13.2±1.166%,
P<0.01; day 3 9.8±1.470% vs. 5.8±0.748%, P<0.01;)
Consistently, compared with native VEGF group, the ratio of BrdU
incorporated hepatocyte was significantly increased in CBD-VEGF
group (Figs. 5D and 6D; day 2: 16.4±1.85% vs. 10.4±1.85%,
P<0.01; day 3: 8.60±0.93% vs. 4.40±0.93%, P<0.01). The levels
of PCNA expression were also verified by IHC staining and western
blot analysis, which further confirmed that the ability of CBD-VEGF
on hepatocyte proliferation (Fig. 2F
and H; Fig. S2; Figs. 5E and F and 6E and F). Taken together, the results
indicated that CBD-VEGF treatment could significantly promote
hepatocyte proliferation and liver regeneration following PHx.
Discussion
Despite the robust regenerative capacity of liver,
liver failure remains one of the significant clinical challenge
with a high mortality rate (33).
The ability of liver to restore the cell mass loss is an important
component of hepatic functional recovery following resection
surgery (34). In the present
study, CBD-VEGF, which could specifically bind to the hepatic
extracellular matrix to retain VEGF in remnant liver mass, was used
in the regenerative therapy for 2/3 PHx. The findings demonstrated
that CBD-VEGF treatment significantly attenuate liver injury,
promote hepatocyte replication and enhance sinusoidal regeneration
after hepatectomy.
Research groups are working on localizing and
sustaining VEGF protein at the sites of injury tissue. Injectable
alginate and nanofiber have been used as vehicles to deliver VEGF
(17,35). A previous study by Yu et al
(35) reported a local delivery
of VEGF to regenerating liver tissue by using biodegradable
nanofiber meshes, which provided a sustained release of VEGF and
increased the proliferation of hepatocytes. As the main components
of hepatic extracellular matrix are type I and type III collagens,
collagen I could be a potential target for VEGF to be enriched and
control released in remnant liver tissue (36). In a previous study, de Souza et
al (37) reported that a
heptapeptide (TKKTLRT) could bind specifically to the type I
collagen. With this domain, growth factors showed a remarkable
collagen binding ability and remained excellent biological
activity. Indeed, several growth factors, such as basic fibroblast
growth factor, brain-derived neurotrophic factor and VEGF were
fused with CBD (38,39) Previous preclinical studies
demonstrated that the recombinant proteins could retain the
effective concentration at the injury site and exhibit enhanced
tissue regeneration. Consistently, the present study demonstrated
that CBD-VEGF injection through portal vein could collect and be
retained with a higher concentration in liver tissue compared with
native VEGF. Additionally, CBD-VEGF treatment promoted hepatocyte
replication and enhanced liver mass restoration.
Angiogenesis is essential for liver regeneration and
repair, as it serves a vital role in the supply of blood to the
newly replicating hepatocytes. LSEC is a specialized endothelial
cell that constituting ~10% of the liver cellular mass (40). LSECs have been noted to contribute
to liver regeneration following liver injury. During the early
phase of liver regeneration, the transition from quiescent to
replicative hepatocytes increase after 12 h and reaches a peak ~48
h after PHx, while the proliferation of LSECs is delayed in
comparison to hepatocytes (40–43). Previous studies have identified
VEGF as a powerful mitogen of LSECs via upregulating VEGF receptors
during liver regeneration (44,45). It has been demonstrated that
endogenous VEGF expression in the remnant liver is increased from
24 h after hepatectomy, with a peak at 72 h (46). Ding et al (32) revealed that inducible genetic
ablation of VEGFR2 in LSECs impairs the first wave of hepatocyte
proliferation and subsequent hepatovascular regeneration,
suggesting VEGFR2 as the main mediator of VEGF signaling in LSECs.
Thus, a synchronized proliferation of hepatocytes and LSECs is a
crucial requirement for proper liver regeneration. CD31, also known
as PECAM, is considered as golden standard marker of LSECs
(46). The present study showed
that CD31 and VEGFR2 expression in the remnant liver tissue were
significantly upregulated in the CBD-VEGF treatment group,
indicating that CBD-VEGF promoted liver regeneration possibly
through regulating sinusoidal regeneration and revascularization
following PHx. It is reported that hepatocyte growth factor (HGF),
one of the mitogenic growth factors, is involved in regulating
liver regeneration (47). The
expression of HGF was measured and the results of the present study
revealed that CBD-VEGF treatment could promote liver HGF expression
at 2 days after surgery (Fig.
S3).
In brief, the present study indicated that injection
of CBD-VEGF could significantly reduce the liver injury after
partial hepatectomy, enhance the reconstruction of
neovascularization and promote liver regeneration. These results
suggested that CBD-VEGF may be a potential clinical candidate for
liver regeneration.
Supplementary Material
Supporting Data
Acknowledgements
Not applicable.
Funding
The present study was supported by the National Key R&D
Program of China Grants (grant no. 2018YFA0109800) and China
Postdoctoral Science Foundation (grant no. 2019M652328).
Availability of data and materials
All data generated or analyzed during this study are
included in this published article
Authors' contributions
SW, GD and CS contributed to sample testing, data
analysis and study design; ZL contributed to sample testing and
data analysis; QS, XL, XY and YL contributed to sample preparation;
CG, XW contributed to study design; GD and CS directed the project,
contributed to the discussion, reviewed and edited the manuscript.
GD had full access to all the data in the study and had final
responsibility for the decision to submit for publication. GD and
SW confirm the authenticity of all the raw data. All authors have
read and approved the final manuscript.
Ethics approval and consent to
participate
The present study was approved by The Ethics
Committee Medical College of Qingdao University (approval no.
QDU-AEC-2021166).
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing
interests
References
|
1
|
Yagi S, Hirata M, Miyachi Y and Uemoto S:
Liver regeneration after hepatectomy and partial liver
transplantation. Int J Mol Sci. 21:84142020. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mathurin P: Early liver transplantation
for acute alcoholic hepatitis: We can't say no. J Hepatol.
75:718–722. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Song Z, Humar B, Gupta A, Maurizio E,
Borgeaud N, Graf R, Clavien PA and Tian Y: Exogenous melatonin
protects small-for-size liver grafts by promoting monocyte
infiltration and releases interleukin-6. J Pineal Res.
65:e124862018. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Zhu CH, Zhang DH, Zhu CW, Xu J, Guo CL, Wu
XG, Cao QL and Di GH: Adult stem cell transplantation combined with
conventional therapy for the treatment of end-stage liver disease:
A systematic review and meta-analysis. Stem Cell Res Ther.
12:5582021. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Michalopoulos GK: Liver regeneration. J
Cell Physiol. 213:286–300. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Fausto N, Campbell JS and Riehle KJ: Liver
regeneration. Hepatology. 43:S45–S53. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Taub R: Liver regeneration: From myth to
mechanism. Nat Rev Mol Cell Biol. 5:836–847. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Kraizer Y, Mawasi N, Seagal J, Paizi M,
Assy N and Spira G: Vascular endothelial growth factor and
angiopoietin in liver regeneration. Biochem Biophys Res Commun.
287:209–215. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Vento SI, Wolff CH, Salven PJ, Hytönen ML,
Ertama LO and Malmberg CH: Vascular permeability factor/vascular
endothelial growth factor in nasal polyps. Acta Otolaryngol Suppl.
543:170–174. 2000.PubMed/NCBI
|
|
10
|
Alizai PH, Bertram L, Kroy D, Kummer J,
Andert A, Neumann UP, Ulmer TF and Fragoulis A: Expression of
VEGFR-2 during liver regeneration after partial hepatectomy in a
bioluminescence mouse model. Eur Surg Res. 58:330–340. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Smith GA, Fearnley GW, Tomlinson DC,
Harrison MA and Ponnambalam S: The cellular response to vascular
endothelial growth factors requires co-ordinated signal
transduction, trafficking and proteolysis. Biosci Rep.
35:e002532015. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Ross MA, Sander CM, Kleeb TB, Watkins SC
and Stolz DB: Spatiotemporal expression of angiogenesis growth
factor receptors during the revascularization of regenerating rat
liver. Hepatology. 34:1135–1148. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Sato T, El-Assal ON, Ono T, Yamanoi A,
Dhar DK and Nagasue N: Sinusoidal endothelial cell proliferation
and expression of angiopoietin/Tie family in regenerating rat
liver. J Hepatol. 34:690–698. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Ronco MT, Francés D, de Luján Alvarez M,
Quiroga A, Monti J, Parody JP, Pisani G, Carrillo MC and Carnovale
CE: Vascular endothelial growth factor and nitric oxide in rat
liver regeneration. Life Sci. 81:750–755. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Yu Q, Que LG and Rockey DC:
Adenovirus-mediated gene transfer to nonparenchymal cells in normal
and injured liver. Am J Physiol Gastrointest Liver Physiol.
282:G565–G572. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Atta HM, Al-Hendy A, Salama SA, Shaker OG
and Hammam OA: Low-dose simultaneous delivery of adenovirus
encoding hepatocyte growth factor and vascular endothelial growth
factor in dogs enhances liver proliferation without systemic growth
factor elevation. Liver Int. 29:1022–1030. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Silva EA and Mooney DJ: Spatiotemporal
control of vascular endothelial growth factor delivery from
injectable hydrogels enhances angiogenesis. J Thromb Haemost.
5:590–598. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Wu K, Huang R, Wu H, Liu Y, Yang C, Cao S,
Hou X, Chen B, DaI J and Wu C: Collagen-binding vascular
endothelial growth factor attenuates CCl4-induced liver fibrosis in
mice. Mol Med Rep. 14:4680–4686. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lee RJ, Springer ML, Blanco-Bose WE, Shaw
R, Ursell PC and Blau HM: VEGF gene delivery to myocardium:
Deleterious effects of unregulated expression. Circulation.
102:898–901. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Matsuno H, Kozawa O, Yoshimi N, Akamatsu
S, Hara A, Mori H, Okada K, Ueshima S, Matsuo O and Uematsu T: Lack
of alpha2-antiplasmin promotes pulmonary heart failure via
overrelease of VEGF after acute myocardial infarction. Blood.
100:2487–2493. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Zhang J, Ding L, Zhao Y, Sun W, Chen B,
Lin H, Wang X, Zhang L, Xu B and Dai J: Collagen-targeting vascular
endothelial growth factor improves cardiac performance after
myocardial infarction. Circulation. 119:1776–1784. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Jia W, Tang H, Wu J, Hou X, Chen B, Chen
W, Zhao Y, Shi C, Zhou F, Yu W, et al: Urethral tissue regeneration
using collagen scaffold modified with collagen binding VEGF in a
beagle model. Biomaterials. 69:45–55. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Shi C, Zhao Y, Yang Y, Chen C, Hou X, Shao
J, Yao H, Li Q, Xia Y and Dai J: Collagen-binding VEGF targeting
the cardiac extracellular matrix promotes recovery in porcine
chronic myocardial infarction. Biomater Sci. 6:356–363. 2018.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bayne K, Ramachandra GS, Rivera EA and
Wang J: The evolution of animal welfare and the 3Rs in Brazil,
China, and India. J Am Assoc Lab Anim Sci. 54:181–191.
2015.PubMed/NCBI
|
|
25
|
Ito H, Ando K, Nakayama T, Taniguchi M,
Ezaki T, Saito K, Takemura M, Sekikawa K, Imawari M, Seishima M and
Moriwaki H: Role of Valpha 14 NKT cells in the development of
impaired liver regeneration in vivo. Hepatology. 38:1116–1124.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mitchell C and Willenbring H: A
reproducible and well-tolerated method for 2/3 partial hepatectomy
in mice. Nat Protoc. 3:1167–1170. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Lin N, Li X, Song T, Wang J, Meng K, Yang
J, Hou X, Dai J and Hu Y: The effect of collagen-binding vascular
endothelial growth factor on the remodeling of scarred rat uterus
following full-thickness injury. Biomaterials. 33:1801–1807. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Shi Q, Zhao G, Wei S, Guo C, Wu X, Zhao RC
and Di G: Pterostilbene alleviates liver ischemia/reperfusion
injury via PINK1-mediated mitophagy. J Pharmacol Sci. 148:19–30.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2(−Delta Delta C(T)) method. Methods. 25:402–408. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liang H, Li X, Wang B, Chen B, Zhao Y, Sun
J, Zhuang Y, Shi J, Shen H, Zhang Z and Dai J: A collagen-binding
EGFR antibody fragment targeting tumors with a collagen-rich
extracellular matrix. Sci Rep. 6:182052016. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Wang L, Wang X, Xie G, Wang L, Hill CK and
DeLeve LD: Liver sinusoidal endothelial cell progenitor cells
promote liver regeneration in rats. J Clin Invest. 122:1567–1573.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Ding BS, Nolan DJ, Butler JM, James D,
Babazadeh AO, Rosenwaks Z, Mittal V, Kobayashi H, Shido K, Lyden D,
et al: Inductive angiocrine signals from sinusoidal endothelium are
required for liver regeneration. Nature. 468:310–315. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rahnemai-Azar AA, Cloyd JM, Weber SM,
Dillhoff M, Schmidt C, Winslow ER and Pawlik TM: Update on liver
failure following hepatic resection: Strategies for prediction and
avoidance of post-operative liver insufficiency. J Clin Transl
Hepatol. 6:97–104. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Fausto N: Liver regeneration. J Hepatol.
32:19–31. 2000. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Yu YQ, Jiang XS, Gao S, Ma R, Jin Y, Jin
X, Peng SY, Mao HQ and Li JT: Local delivery of vascular
endothelial growth factor via nanofiber matrix improves liver
regeneration after extensive hepatectomy in rats. J Biomed
Nanotechnol. 10:3407–3415. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Hermert D, Martin IV, Reiss LK, Liu X,
Breitkopf DM, Reimer KC, Alidousty C, Rauen T, Floege J, Ostendorf
T, et al: The nucleic acid binding protein YB-1-controlled
expression of CXCL-1 modulates kidney damage in liver fibrosis.
Kidney Int. 97:741–752. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
de Souza SJ and Brentani R: Collagen
binding site in collagenase can be determined using the concept of
sense-antisense peptide interactions. J Biol Chem. 267:13763–13767.
1992. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Yin R, Zhao S and Qiu C: Brain-derived
neurotrophic factor fused with a collagen-binding domain inhibits
neuroinflammation and promotes neurological recovery of traumatic
brain injury mice via TrkB signalling. J Pharm Pharmacol.
72:539–550. 2020. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hao W, Han J, Chu Y, Huang L, Zhuang Y,
Sun J, Li X, Zhao Y, Chen Y and Dai J: Collagen/Heparin Bi-affinity
multilayer modified collagen scaffolds for controlled bFGF release
to improve angiogenesis in vivo. Macromol Biosci. 18:e18000862018.
View Article : Google Scholar : PubMed/NCBI
|
|
40
|
Gamble J, Vadas M and McCaughan G:
Sinusoidal endothelium is essential for liver regeneration.
Hepatology. 54:731–733. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Michalopoulos GK and DeFrances MC: Liver
regeneration. Science. 276:60–66. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Schiffer E, Frossard JL, Rubbia-Brandt L,
Mentha G and Pastor CM: Hepatic regeneration is decreased in a rat
model of sinusoidal obstruction syndrome. J Surg Oncol. 99:439–446.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Uda Y, Hirano T, Son G, Iimuro Y, Uyama N,
Yamanaka J, Mori A, Arii S and Fujimoto J: Angiogenesis is crucial
for liver regeneration after partial hepatectomy. Surgery.
153:70–77. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Shimizu H, Mitsuhashi N, Ohtsuka M, Ito H,
Kimura F, Ambiru S, Togawa A, Yoshidome H, Kato A and Miyazaki M:
Vascular endothelial growth factor and angiopoietins regulate
sinusoidal regeneration and remodeling after partial hepatectomy in
rats. World J Gastroenterol. 11:7254–7260. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Donahower BC, McCullough SS, Hennings L,
Simpson PM, Stowe CD, Saad AG, Kurten RC, Hinson JA and James LP:
Human recombinant vascular endothelial growth factor reduces
necrosis and enhances hepatocyte regeneration in a mouse model of
acetaminophen toxicity. J Pharmacol Exp Ther. 334:33–43. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Shimizu H, Miyazaki M, Wakabayashi Y,
Mitsuhashi N, Kato A, Ito H, Nakagawa K, Yoshidome H, Kataoka M and
Nakajima N: Vascular endothelial growth factor secreted by
replicating hepatocytes induces sinusoidal endothelial cell
proliferation during regeneration after partial hepatectomy in
rats. J Hepatol. 34:683–689. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Kang LI, Mars WM and Michalopoulos GK:
Signals and cells involved in regulating liver regeneration. Cells.
1:1261–1292. 2012. View Article : Google Scholar : PubMed/NCBI
|