Introduction
Chemokines are a large family of small (7–15 kDa),
structurally related heparin-binding proteins that bind to and
activate a family of chemokine receptors. More than 50 chemokines
have been identified and they are classified into 4 families (CXC,
CX3C, CC and C) based on the position of the first two conserved
cysteine residues (1). Chemokine
receptors are 7-transmembrane G protein-coupled receptors. Most are
promiscuous and can bind with high affinity to multiple chemokine
ligands (CXCR, CX3CR, CCR and XCR). At present, 20 chemokine
receptors have been identified.
In normal physiology, chemokines are involved in
proinflammatory and non-inflammatory cell homing by binding to
their cognate receptors (2).
However, increasing evidence implicates these small cytokine-like
proteins and their receptors in tumor biology (3–5).
CXCL12 and its cognate receptor CXCR4 have been shown to regulate
site-specific distant metastasis of many cancer types (6–10).
These studies demonstrated that tumor cells express a high level of
CXCR4 and that tumor metastasis target tissues (lung, liver and
bone) express high levels of the ligand CXCL12, allowing tumor
cells to directionally migrate to target organs via a CXCL12-CXCR4
chemotactic gradient. These studies have led to the current
CXCL12/CXCR4 ‘endocrine axis’ model; CXCR4 expression by metastatic
cells enables those cells to navigate towards organs abundantly
expressing CXCL12. Currently, despite compelling evidence for the
pro-metastatic function of the CXCL12/CXCR4 endocrine axis, little
attention has been devoted to the precise role of the CXCL12/CXCR4
autocrine loop in tumor metastasis.
A previous study reported a pro-metastatic effect of
the CXCL12/CXCR4 autocrine loop when CXCL12 was transfected into
the mammary carcinoma cell line MDA-MB-231 (11). This study also demonstrated that
CXCL12 expression was inversely correlated with disease-free and
overall survival in breast cancer patients. In another study,
increased proliferation was in contrast to reduced metastasis in
vivo, which has been demonstrated as responsible for forced
CXCL12-expressing MDA-MB-231 cells (12). These contradictory results suggest
that the function of the CXCL12/CXCR4 autocrine loop in tumor
growth and metastasis requires further elucidation.
Matrix metalloproteinases (MMPs), which are
multidomain zinc-dependent endopeptidases, are pivotal in cancer
invasion and metastasis (13,14).
Among these MMPs, MMP-2 and MMP-9 have been of particular interest
due to their pathogenic roles in non-small cell lung cancer (NSCLC)
(15–17). Degradation of ECM and basement
membranes by MMP-2 and/or MMP-9 is required for tumor cell invasion
and metastasis (16,18,19).
The correlation between the CXCL12/CXCR4 autocrine loop and the
expression of MMP-2 and MMP-9 remains unknown.
We performed this study to investigate the
previously unknown role of the CXCL12/CXCR4 autocrine loop in cell
growth and distant metastasis of NSCLC. This was achieved by using
a gene transfection technique. Human NSCLC cell line A549, which
does not express endogenous CXCL12, was transfected with
pIRES2-ZsGreen1-CXCL12 or the control vector pIRES2-ZsGreen1 to
establish stable CXCL12 (A549-CXCL12) and control vector
(A549-ZsGreen1) transfectants. In the stable CXCL12-expressing cell
line (A549-CXCL12), a functional CXCL12/CXCR4 autocrine loop was
constructed. The contribution of the CXCL12/CXCR4 autocrine loop
signaling pathway to the migration, proliferation and invasiveness
of A549-CXCL12 cells was evaluated and compared with A549-ZsGreen1
and wild-type A549 cells. We hypothesized that the CXCL12/CXCR4
autocrine loop may stimulate production of MMP-2 and MMP-9.
Therefore, we also investigated whether the CXCL12/CXCR4 autocrine
loop affected MMP-2 and MMP-9 expression in NSCLC cells.
Materials and methods
Materials
Anti-mouse and anti-rabbit IgG-conjugated
horseradish peroxidase and rabbit polyclonal antibodies specific
for extracellular signal-regulated kinase (ERK), phosphorylated
(p-) ERK, MMP-9 and MMP-2 were purchased from Santa Cruz
Biotechnology (Santa Cruz, CA, USA). Rabbit polyclonal antibodies
specific for GAPDH, CXCR4 and CXCL12 were purchased from R&D
Systems (Minneapolis, MN, USA). Lipofectamine® 2000
reagent was purchased from Invitrogen Ltd. (Paisley, UK). The human
lung cancer cell lines A549 and H1975 were purchased from American
Type Culture Collection (Rockville, MD, USA), and 95C and 95D cell
lines were obtained from China Center for Type Culture Collection
(Wuhan, China). Human full-length CXCL12 was purchased from
Biofavor Ltd. (Wuhan, China) and the pIRES2-ZsGreen1 vector was
purchased from Takara Biotechnology (Dalian, China). PCR primers
were designed using Beacon Designer (Palo Alto, CA, USA) and
synthesized by Yingjun Biotechnology (Shanghai, China). Molecular
biology-grade agarose was obtained from Invitrogen Ltd. The RNA
extraction and reverse transcription kits were obtained from AbGene
Ltd. (Epsom, UK). Matrigel was purchased from BD Biosciences (San
Jose, CA, USA). A Transwell plate equipped with a porous insert
(pore size, 8 μm) was obtained from Becton Dickinson Labware
(Franklin Lakes, NJ, USA).
Cell culture
Human NSCLC cell lines A549, H1975, 95C and 95D were
maintained in RPMI-1640 medium supplemented with 10% fetal calf
serum, 100 U/ml penicillin and 100 μg/ml streptomycin in a
humid atmosphere of 5% CO2 and 95% air, at 37°C. This
study was approved by the Ethics Committee of Renmin Hospital of
Wuhan University, Wuhan, China.
Stable transfection
The human full-length CXCL12 cDNA fragment was
ligated to the cloning site of pIRES2-ZsGreen1, followed by
transformation using One-Shot® E.coli (Invitrogen
Ltd.), verification and amplification. A549 cells were transfected
with either purified or control plasmid using Lipofectamine 2000
reagent (according to the manufacturer’s instructions) followed by
selection with G418. Stable CXCL12 (A549-CXCL12) and control
plasmid (A549-ZsGreen1) transfectants were subsequently established
and verified.
Western blot analyses and reverse
transcription-PCR (RT-PCR) analyses
Proteins in the total cell lysates were resolved by
SDS-PAGE and electrotransferred to a polyvinylidene difluoride
membrane (Millipore, Bedford, MA, USA). After the blot was blocked
in a solution of 4% bovine serum albumin, membrane-bound proteins
were probed overnight with primary antibodies against GAPDH,
CXCL12, CXCR4, MMP-2, MMP-9 or p-ERK. They were then incubated with
horseradish peroxidase-conjugated secondary antibodies for 1 h.
Antibody-bound protein bands were detected with enhanced
chemiluminescence reagents and photographed with Kodak X-OMAT LS
film (Eastman Kodak, Rochester, NY, USA). Quantitative data were
obtained using a computing densitometer and ImageQuant software
(Molecular Dynamics, Sunnyvale, CA, USA).
For RT-PCR analysis, RNA was extracted from total
cell lysates using a TRIzol kit (MDBio, Piscataway, NJ, USA). The
RNA concentration was determined using an ultraviolet
spectrophotometer. The reverse transcription reaction was performed
using 2 μg total RNA that was reverse-transcribed into cDNA
using oligo(dT) primer, then amplified for 30 cycles using 2
oligodeoxynucleotide primers: β-actin sense,
5′-CACGATGGAGGGGCCGGACTCATC-3′ and anti-sense,
5′-TAAAGACCTCTATGCCAACACAGT-3′; CXCL12 sense,
5′-GTCAGCCTGAGCTACAGATGC-3′ and anti-sense,
5′-CTTTAGCTTCGGGTCAATGC-3′; CXCR4 sense, 5′-CCGTGGCAAACTGGTACTTT-3′
and anti-sense, 5′-GACGCCAACATAGACCACCT-3′; MMP-2 sense,
5′-GTGCTGAAGGACACACTAAAGAAGA-3′ and anti-sense,
5′-TTGCCATCCTTCTCAAAGTTGTAGC-3′; or MMP-9 sense,
5′-CACTGTCCACCCCTCAGAGC-3′ and anti-sense,
5′-GCCACTTGTCGGCGATAAGC-3′. PCR was carried out as follows: 94°C
for 4 min, followed by 30 cycles of 94°C for 30 sec, 52°C for 30
sec, 72°C for 25 sec and a final extension for 4 min at 72°C. The
products were visualized on 2% agarose gel after staining with
ethidium bromide.
Wound assay
Cells were grown to confluence in 6-well plates and
starved with serum-free RPMI-1640 medium for 24 h. An injury line
was created with a pipette tip in the center of the dishes.
Following rinsing with phosphate-buffered saline (PBS), cells were
allowed to migrate for 12 h and then photographed (×100). Each
clone was plated and wounded in triplicate and each experiment was
repeated at least 3 times.
Invasion assay
The invasion assay was performed using Transwells
with filter inserts (pore size, 8 μm) coated with 50
μl Matrigel diluted 1:3. Approximately 2.5×104
cells in 100 μl serum-free RPMI-1640 medium were placed in
the upper chamber and 1 ml of the same medium was placed in the
lower chamber. Following 18 h incubation at 37°C in 5%
CO2, non-invading cells were removed from the upper
surface of the filter. Cells that had migrated through the filter
were fixed with 4% paraformaldehyde (Sigma, St. Louis, MO, USA),
stained with crystal violet for Matrigel and counted under a
microscope (×400). Each clone was plated in triplicate in each
experiment, and each experiment performed in triplicate.
MTT assay
Briefly, 1×104 cells were seeded onto
96-well plates and allowed to adhere for 8 h. Cell proliferation
was assessed at various time points by adding 10 μl
filter-sterilized MTT (5 mg/ml in PBS) to a single row of 6 wells.
Following 4 h incubation with MTT, the media was removed and the
blue formazan crystals trapped in the cells were dissolved in 100
μl sterile DMSO, by incubating at 37°C for 30 min.
Absorbance at 568 nm was measured in each well with a plate reader.
The growth curve was constructed by plotting absorbance against
time.
Statistics
Statistical differences between the means of the
different groups were evaluated with Prism 5.01 (GraphPad Software
Inc., La Jolla, CA, USA) using one-way ANOVA. P<0.05 was
considered to indicate a statistically significant difference.
Results
Expression pattern of CXCL12 and CXCR4 in
NSCLC cell lines
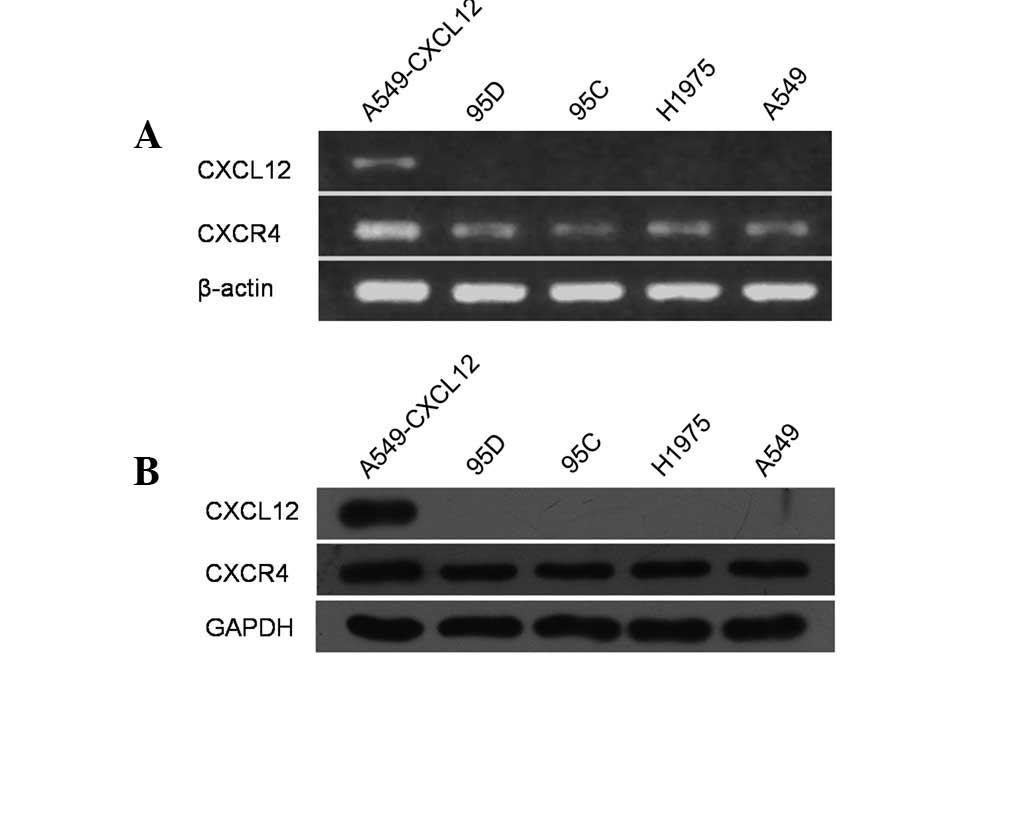
Several studies have demonstrated CXCR4 expression
in NSCLC cell lines, but its ligand CXCL12 has not been observed
(10,15,20).
We examined mRNA expression of CXCL12 and CXCR4 in a panel of human
NSCLC cell lines, including 95C, 95D, H1975 and A549, using RT-PCR.
In accordance with previous studies, all 4 cell lines
constitutively expressed CXCR4 mRNA, whereas CXCL12 mRNA expression
was not observed (Fig. 1A).
Following this, total cell lysates of these cell lines were
prepared and examined for CXCL12 and CXCR4 protein expression by
western blot analysis using CXCL12- and CXCR4-specific antibodies.
As demonstrated in Fig. 1B, all 4
cell lines constitutively expressed CXCR4 protein but not its
cognate ligand CXCL12.
Construction of functional CXCL12/CXCR4
autocrine loop in NSCLC cell line A549
The human NSCLC cell line A549 was transfected with
pIRES2-ZsGreen1 plasmid encoding human CXCL12 or empty vector
pIRES2-ZsGreen1 as a control, followed by selection with G418 to
yield a stable CXCL12-expressing cell line (A549-CXCL12) and a
stable control plasmid transfectant (A549-ZsGreen1). Using western
blot analysis, CXCL12 protein expression was observed in
A549-CXCL12 cells, but not in A549-ZsGreen1 or wild-type A549 cells
(Fig. 2A). Furthermore, p-ERK1/2
was detected in A549-CXCL12 cells but not A549-ZsGreen1 cells
(Fig. 2B), indicating autonomous
ERK1/2 activation through CXCL12 binding to its cognate receptor
CXCR4 in A549-CXCL12 cells. Taken together, these data indicated
that a functional CXCL12/CXCR4 autocrine loop was successfully
constructed in the A549-CXCL12 cell line.
CXCL12/CXCR4 autocrine loop promotes cell
migration, invasiveness and proliferation
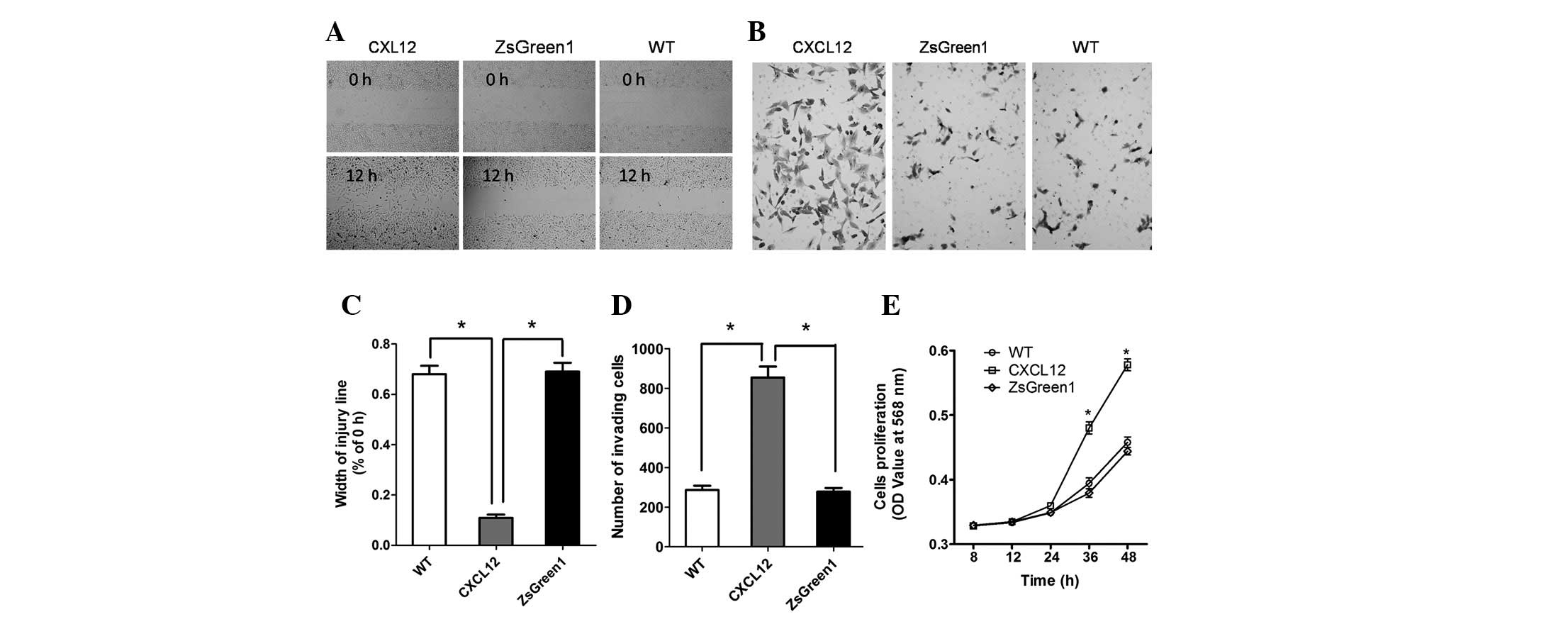
To investigate the influence of the CXCL12/CXCR4
autocrine loop on the malignant phenotype of NSCLC cells, we
measured the migration, invasiveness and proliferation of
A549-CXCL12, A549-ZsGreen1 and wild-type A549 cells. In wound
migration analyses, A549-CXCL12 cells demonstrated significantly
increased mobility compared with A549-ZsGreen1 and wild-type A549
cells (P<0.01; Fig. 3A and C).
The invasive potential of A549-CXCL12, A549-ZsGreen1 and wild-type
A549 cells was determined using a Matrigel chamber assay. The
number of invading A549-CXCL12 cells was significantly higher than
(triple) that of A549-ZsGreen1 or wild-type A549 cells (P<0.01;
Fig. 3B and D). The MTT
(3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) cell
proliferation assay was performed to measure cell proliferation
rate. As shown by the proliferation curve (Fig. 3E), the cell proliferation rate of
A549-CXCL12 cells was markedly higher than that of A549-ZsGreen1 or
wild-type A549 cells at 24 and 48 h (P<0.01), indicating that
the CXCL12/CXCR4 autocrine loop induced increased cellular
proliferation.
CXCL12/CXCR4 autocrine loop upregulates
MMP-2 and MMP-9 expression
Tumor cell invasion and metastasis involve ECM
degradation and remodeling in addition to cell locomotion. MMP-2
and MMP-9 are capable of degrading several ECM proteins, including
type IV and V collagens, laminin and elastin. To investigate
whether the CXCL12/CXCR4 autocrine loop influences MMP-2 and MMP-9
expression, we performed RT-PCR to detect mRNA expression in
A549-CXCL12, A549-ZsGreen1 and wild-type A549 cells. Markedly
increased MMP-2 and MMP-9 expression was observed in A549-CXCL12
cells compared with control cells (Fig.
4A–C). Western blot analysis also revealed significantly higher
expression levels of MMP-2 and MMP-9 in A549-CXCL12 cells.
Discussion
Primary non-small cell lung cancer is the leading
cause of cancer mortality worldwide. Most patients present with
locally advanced (37%) or metastatic (38%) disease at the time of
diagnosis (21). As with most
cancers, early-stage NSCLC is often be controlled with locally
directed therapy including radiation and surgery; it is the
development of metastatic disease that leads to the high mortality
rate of NSCLC. Therefore, possible mechanisms of metastasis, as
well as the early detection and screening of lung cancer, have been
the subject of growing interest.
Tumor metastasis is an organized process that occurs
in a stepwise fashion: i) uncontrolled proliferation and local
invasion; ii) intravasation into the vascular system and survival
in the circulation; iii) escape of cancer cells from the lumina of
blood vessels into the parenchyma of distant tissues
(extravasation), and iv) the formation of secondary tumors
(colonization) (22–24). Tumor metastasis is also a
non-random, highly organ-specific pathophysiological process
(23,25,26). A
growing body of evidence has indicated that the chemokine CXCL12
and its cognate receptor CXCR4 are critical in this process
(6,10,27–30).
These studies have led to the current CXCL12/CXCR4 ‘endocrine axis’
model; CXCR4 expression by metastatic cells enables these cells to
navigate towards organs abundantly expressing CXCL12. In the case
of NSCLC, the role of the CXCL12/CXCR4 endocrine axis has been well
established (10), but the precise
effect of the CXCL12/CXCR4 autocrine loop on NSCLC cells has not
yet been demonstrated. Several studies have been conducted to
evaluate the functionality of the CXCL12/CXCR4 autocrine loop in
human mammary carcinoma (11,12),
oral squamous cell carcinoma (31)
and colorectal carcinoma cells (32). However, the evidence arising from
these studies has been controversial and even contradictory.
In this study, CXCR4 was constitutively expressed in
all 4 human NSCLC cell lines evaluated, but CXC12 was not observed
consistently. To investigate the effect of the CXCL12/CXCR4
autocrine loop on the NSCLC cells, the full-length CXCL12
gene was transfected into human NSCLC cell line A549 to generate
the stable CXCL12-expressing transfectant, A549-CXCL12. A549-CXCL12
cells overexpressed CXCL12 and autonomous ERK1/2 activation was
also observed, indicating that this transfectant had acquired a
functional CXCL12/CXCR4 autocrine loop. The ability to migrate was
evaluated first, as this is a significant characteristic of the
aggressive phenotype of malignant tumor cells. Our results
demonstrated that the CXCL12/CXCR4 autocrine loop signaling pathway
significantly enhanced A549-CXCL12 cell migration compared with
control A549-ZsGreen1 and wild-type A549 cells (P<0.01). These
results are consistent with those revealed by Kang et
al(11) and Uchida et
al(31), who performed similar
experiments using human mammary carcinoma cells and oral squamous
cell carcinoma cells, respectively. Metastasis fundamentally
involves the movement of cells from one site to another. These
results indicated that the CXCL12/CXCR4 autocrine loop may promote
the metastatic potential of NSCLC cells.
The most fundamental trait of malignant tumor cells
is their ability to sustain chronic proliferation. Thus, another
significant characteristic of the aggressive phenotype of malignant
tumor cells is uncontrolled cellular proliferation. As a primary
tumor grows, its blood supply cannot support its metabolic needs;
lack of oxygen causes tumor cells to move away from the site of
hypoxia and spread to new locations through activation of genes
such as c-Met and CXCR4(33). Our results revealed that the
CXCL12/CXCR4 autocrine loop induced a significant increase in
A549-CXCL12 cell proliferation compared with the vector control
cell line A549-ZsGreen1 and wild-type parent cell line A549,
indicating that the CXCL12/CXCR4 autocrine loop may play a critical
role in tumor growth so as to facilitate tumor metastasis. These
results are in accordance with studies demonstrating that the
CXCL12/CXCR4 autocrine loop increased mammary carcinoma cell
proliferation (13). However, they
are in disagreement with previous results by Wendt et
al(33), who revealed increased
apoptosis in forced CXCL12-expressing colorectal cancer cells
compared to control eGFP clones. These data highlight differences
in the pathophysiological impact of the CXCL12/CXCR4 autocrine loop
among different types of cancer cells.
In the present study, the CXCL12/CXCR4 autocrine
loop also markedly enhanced the invasiveness of NSCLC cells. Kang
et al and Uchida et al(11,31)
reported that the CXCL12/CXCR4 autocrine loop significantly
increased breast cancer and oral squamous cell carcinoma cell
invasiveness, respectively. By contrast, Wendt et al
reported decreased invasiveness and metastasis in breast cancer
cells and colorectal cancer cells in vivo(12,32),
suggesting that the role of the CXCL12/CXCR4 autocrine loop in
modulating the capacity for invasiveness may depend on the type of
cancer. Enzymatic degradation of ECM and basement membranes is a
key step in cancer invasion and metastasis. In human lung cancer,
MMP-2 and MMP-9 have been demonstrated to be correlated with
malignancy grade and metastasis (13,34).
Therefore, we hypothesized that the CXCL12/CXCR4 autocrine loop may
stimulate MMP-2 and MMP-9 production to degrade the ECM and
basement membranes. Both mRNA and protein expression of MMP-2 and
MMP-9 were markedly increased in A549-CXCL12 cells compared with
controls, further supporting the notion that the CXCL12/CXCR4
autocrine loop indirectly enhanced the capacity for NSCLC cell
invasiveness.
Overall, these results indicate that the
CXCL12/CXCR4 autocrine loop increases the metastatic potential of
NSCLC cells, confirming that the CXCL12/CXCR4 autocrine loop may
contribute to the motility, growth and invasiveness of NSCLC. To
our knowledge, this in vitro study is the first to reveal
that the CXCL12/CXCR4 autocrine loop increases the metastatic
potential of NSCLC. Furthermore, our findings suggest that targeted
therapies against CXCR4 (35)
should consider the CXCL12 expression status of the NSCLC to be
treated, since tumors with autocrine overexpression of CXCL12 may
be more sensitive to CXCR4 antagonists that compete with CXCL12 for
receptor binding and more suitable for the application of
chemokine-based anti-cancer therapies. Differences in the impact of
the CXCL12/CXCR4 autocrine loop on cancer cells may depend on the
type of cancer, and this possibility requires further
investigation.
References
|
1
|
Vindrieux D, Escobar P and Lazennec G:
Emerging roles of chemokines in prostate cancer. Endocr Relat
Cancer. 16:663–673. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Slettenaar VI and Wilson JL: The chemokine
network: a target in cancer biology? Adv Drug Deliv Rev.
58:962–974. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Balkwill F: Cancer and the chemokine
network. Nat Rev Cancer. 4:540–550. 2004. View Article : Google Scholar
|
|
4
|
Ben-Baruch A: The multifaceted roles of
chemokines in malignancy. Cancer Metastasis Rev. 25:357–371. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Keeley EC, Mehrad B and Strieter RM: CXC
chemokines in cancer angiogenesis and metastases. Adv Cancer Res.
106:91–111. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Müller A, Homey B, Soto H, et al:
Involvement of chemokine receptors in breast cancer metastasis.
Nature. 410:50–56. 2001.PubMed/NCBI
|
|
7
|
Rubie C, Frick VO, Ghadjar P, et al: CXC
receptor-4 mRNA silencing abrogates CXCL12-induced migration of
colorectal cancer cells. J Transl Med. 9:222011. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Liang JJ, Zhu S, Bruggeman R, et al: High
levels of expression of human stromal cell-derived factor-1 are
associated with worse prognosis in patients with stage II
pancreatic ductal adenocarcinoma. Cancer Epidemiol Biomarkers Prev.
19:2598–2604. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Castellone MD, Guarino V, De Falco V, et
al: Functional expression of the CXCR4 chemokine receptor is
induced by RET/PTC oncogenes and is a common event in human
papillary thyroid carcinomas. Oncogene. 23:5958–5967. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Phillips RJ, Burdick MD, Lutz M, Belperio
JA, Keane MP and Strieter RM: The stromal derived
factor-1/CXCL12-CXC chemokine receptor 4 biological axis in
non-small cell lung cancer metastases. Am J Respir Crit Care Med.
167:1676–1686. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Kang H, Watkins G, Parr C, Douglas-Jones
A, Mansel RE and Jiang WG: Stromal cell derived factor-1: its
influence on invasiveness and migration of breast cancer cells in
vitro, and its association with prognosis and survival in human
breast cancer. Breast Cancer Res. 7:R402–410. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wendt MK, Cooper AN and Dwinell MB:
Epigenetic silencing of CXCL12 increases the metastatic potential
of mammary carcinoma cells. Oncogene. 27:1461–1471. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Deryugina EI and Quigley JP: Matrix
metalloproteinases and tumor metastasis. Cancer Metastasis Rev.
25:9–34. 2006. View Article : Google Scholar
|
|
14
|
Ii M, Yamamoto H, Adachi Y, Maruyama Y and
Shinomura Y: Role of matrix metalloproteinase-7 (matrilysin) in
human cancer invasion, apoptosis, growth, and angiogenesis. Exp
Biol Med (Maywood). 231:20–27. 2006.PubMed/NCBI
|
|
15
|
Tang CH, Tan TW, Fu WM and Yang RS:
Involvement of matrix metalloproteinase-9 in stromal cell-derived
factor-1/CXCR4 pathway of lung cancer metastasis. Carcinogenesis.
29:35–43. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chakrabarti S and Patel KD: Matrix
metalloproteinase-2 (MMP-2) and MMP-9 in pulmonary pathology. Exp
Lung Res. 31:599–621. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Iijima T, Minami Y, Nakamura N, et al:
MMP-2 activation and stepwise progression of pulmonary
adenocarcinoma: analysis of MMP-2 and MMP-9 with gelatin
zymography. Pathol Int. 54:295–301. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Okada N, Ishida H, Murata N, Hashimoto D,
Seyama Y and Kubota S: Matrix metalloproteinase-2 and -9 in bile as
a marker of liver metastasis in colorectal cancer. Biochem Biophys
Res Commun. 288:212–216. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Rao JS, Gondi C, Chetty C, Chittivelu S,
Joseph PA and Lakka SS: Inhibition of invasion, angiogenesis, tumor
growth, and metastasis by adenovirus-mediated transfer of antisense
uPAR and MMP-9 in non-small cell lung cancer cells. Mol Cancer
Ther. 4:1399–1408. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Su L, Zhang J, Xu H, et al: Differential
expression of CXCR4 is associated with the metastatic potential of
human non-small cell lung cancer cells. Clin Cancer Res.
11:8273–8280. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Jemal A, Murray T, Ward E, et al: Cancer
statistics, 2005. CA Cancer J Clin. 55:10–30. 2005. View Article : Google Scholar
|
|
22
|
Talmadge JE and Fidler IJ: AACR centennial
series: the biology of cancer metastasis: historical perspective.
Cancer Res. 70:5649–5669. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Fidler IJ: The pathogenesis of cancer
metastasis: the ‘seed and soil’ hypothesis revisited. Nat Rev
Cancer. 3:453–458. 2003.
|
|
24
|
Geiger TR and Peeper DS: Metastasis
mechanisms. Biochim Biophys Acta. 1796:293–308. 2009.PubMed/NCBI
|
|
25
|
Hynes RO: Metastatic potential: generic
predisposition of the primary tumor or rare, metastatic variants-or
both? Cell. 113:821–823. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Liotta LA: An attractive force in
metastasis. Nature. 410:24–25. 2001. View
Article : Google Scholar : PubMed/NCBI
|
|
27
|
Liang Z, Yoon Y, Votaw J, Goodman MM,
Williams L and Shim H: Silencing of CXCR4 blocks breast cancer
metastasis. Cancer Res. 65:967–971. 2005.PubMed/NCBI
|
|
28
|
do Carmo A, Patricio I, Cruz MT,
Carvalheiro H, Oliveira CR and Lopes MC: CXCL12/CXCR4 promotes
motility and proliferation of glioma cells. Cancer Biol Ther.
9:56–65. 2010.
|
|
29
|
Zlotnik A: Involvement of chemokine
receptors in organ-specific metastasis. Contrib Microbiol.
13:191–199. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Wagner PL, Hyjek E, Vazquez MF, et al:
CXCL12 and CXCR4 in adenocarcinoma of the lung: association with
metastasis and survival. J Thorac Cardiovasc Surg. 137:615–621.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Uchida D, Onoue T, Tomizuka Y, et al:
Involvement of an autocrine stromal cell derived factor-1/CXCR4
system on the distant metastasis of human oral squamous cell
carcinoma. Mol Cancer Res. 5:685–694. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Wendt MK, Johanesen PA, Kang-Decker N,
Binion DG, Shah V and Dwinell MB: Silencing of epithelial CXCL12
expression by DNA hypermethylation promotes colonic carcinoma
metastasis. Oncogene. 25:4986–4997. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Staller P, Sulitkova J, Lisztwan J, Moch
H, Oakeley EJ and Krek W: Chemokine receptor CXCR4 downregulated by
von Hippel-Lindau tumour suppressor pVHL. Nature. 425:307–311.
2003. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Hsu CP, Shen GH and Ko JL: Matrix
metalloproteinase-13 expression is associated with bone marrow
microinvolvement and prognosis in non-small cell lung cancer. Lung
Cancer. 52:349–357. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Gangadhar T, Nandi S and Salgia R: The
role of chemokine receptor CXCR4 in lung cancer. Cancer Biol Ther.
9:409–416. 2010. View Article : Google Scholar : PubMed/NCBI
|