Introduction
Non-small-cell lung cancer (NSCLC) is the leading
cause of cancer mortality worldwide (1). Recently, molecular-targeting therapies
such as gefitinib and erlotinib have gained attention due to their
potential to improve survival and reduce toxic side effects in
patients with NSCLC (2–4). Four phase III trials with gefitinib or
erlotinib in patients with epidermal growth factor receptor (EGFR)
mutation-positive NSCLC have demonstrated higher response rates and
longer progression-free survival (PFS) times than those of patients
who received platinum doublets as first-line chemotherapy (5–8). These
results indicate that treatment with EGFR-tyrosine kinase
inhibitors (TKIs) may now be the standard treatment for EGFR
mutation-positive NSCLC patients. However, the clinical role of
EGFR-TKI treatment in EGFR mutation-negative patients has not yet
been elucidated. A number of researchers have reported that
erlotinib may also have efficacy against EGFR-negative NSCLC
(9–11).
The factors predicting the efficacy of erlotinib
treatment in patients with EGFR mutation-negative NSCLC have not
been well studied. In order to improve the survival of patients
with EGFR mutation-negative NSCLC receiving EGFR-TKIs including
erlotinib, a biomarker that can predict the efficacy of EGFR-TKIs
is required.
The presence of pulmonary metastasis and malignant
pleural effusion in patients with NSCLC has also been reported to
be a predictive factor of EGFR mutations (12,13).
However, the association between these characteristics and the
efficacy of erlotinib treatment in patients with EGFR
mutation-negative NSCLC remains uncertain. These findings prompted
the investigation of the correlation between the efficacy of
erlotinib treatment and sites of metastasis in patients with EGFR
mutation-negative NSCLC in the current study. It was investigated
whether metastasis to specific organs, including pulmonary
metastasis and malignant pleural effusion, may predict the efficacy
and outcome of erlotinib treatment in patients with EGFR
mutation-negative NSCLC.
Patients and methods
Patient characteristics
This retrospective study included cases of
histologically or cytologically diagnosed NSCLC, which were
advanced stage IIIB or IV, according to the International
Association for the Study of Lung Cancer staging system (14) or recurrent at initial diagnosis. In
total, 206 NSCLC patients were treated with EGFR-TKIs at Kurume
University Hospital (Kurume, Japan) between April 2008 and
September 2012. Of these patients, 53 were identified as EGFR
mutation-negative and thus, were enrolled in this study. The
clinical characteristics of the patients, including age, gender,
smoking history, tumor histology, Eastern Cooperative Oncology
Group (ECOG) performance status (PS) (15), onset of skin rash following
treatment and metastatic sites, were recorded. Tumor nodules in the
primary (T3) and in other ipsilateral lobes (T4) were included as
pulmonary metastases. Tumor response was examined by computed
tomography and evaluated using the Response Evaluation Criteria for
Solid Tumors, version 1.0 (RECIST, v 1.0) (16). The present study was conducted in
accordance with the Declaration of Helsinki and was approved by the
Institutional Review Board of Kurume University Hospital (Kurume,
Japan).
DNA extraction and peptic nucleic
acid-locked nucleic acid (PNA-LNA) polymerase chain reaction (PCR)
clamp assay
For EGFR mutation analysis, the PNA-LNA PCR
clamp method was adopted, using protocols described previously
(17). Specific PNA-LNA probe sets
for two mutation sites, exon 19 (delE746-A750) and exon 21 (L858R),
were developed and these covered >90% of EGFR mutations
reported previously in Japan. In brief, the genomic DNA was
purified from paraffin-embedded tissues using a QIAamp DNA Micro
kit (Qiagen, Valencia, CA, USA). The PCR primers employed were
synthesized by Invitrogen Life Technologies (Carlsbad, CA, USA),
PNA clamp primers and LNA mutant probes were purchased from FASMEC
(Kanagawa, Japan) and Integrated DNA Technologies, Inc.,
(Coralville, IA, USA), respectively. The PNA-LNA PCR clamp assay
was performed using a SDS-7500 System (Applied Biosystems Life
Technologies, Foster City, CA, USA).
Statistical analysis
Fisher’s exact test was used to analyze the
significance of associations between patient characteristics and
overall response [complete response (CR) and partial response (PR)
by RECIST]. The objective response rate (RR) was defined as the
proportion of CR or PR. PFS was defined as the period from the date
of initiation of erlotinib treatment to the onset of disease
progression or mortality from any cause. Overall survival (OS) was
measured from the administration of the initial dose of erlotinib
until the date of mortality or loss to follow-up. The Kaplan-Meier
method was used to assess the survival curves and the log-rank test
was used to evaluate the significance of differences between the
two groups. The univariate survival analyses were conducted by
means of log-rank test, and the multivariate regression was
performed using the Cox proportional-hazards regression model. All
variables that had P-values of <0.05 were included in the Cox
regression model. All tests were two-sided, and P<0.05 was
considered to indicate a statistically significant difference. All
statistical analyses were conducted using JMP, version 10 (SAS
Institute Inc., Cary, NC, USA).
Results
Patient characteristics
The clinical characteristics of the 53 patients are
shown in Table I. Overall, 13
patients were female and 12 were never-smokers; the age range was
35–80 years (median, 64.2 years). In total, 36 patients had
adenocarcinoma and 11 had squamous cell carcinoma. The PS was good
(ECOG, 0–1) in 44 patients, and poor (ECOG, 2–3) in the remaining
nine patients. Erlotinib was used as the first-line therapy in one
patient, as a second-line therapy in 13 patients, as a third-line
therapy in 29 patients, and as a fourth-line therapy or thereafter
in 10 patients. Among the 53 patients who exhibited distant
metastasis, 27 (50.9%), 13 (24.5%), 11 (20.8%), 10 (18.9%), 5
(9.4%), 6 (11.3%) and 14 (26.4%) also had pulmonary, brain, bone,
extrathoracic lymph node, adrenal gland and liver metastasis, and
malignant pleural effusion, respectively.
 | Table ICharacteristics of the 53 non-small
cell lung cancer patients. |
Table I
Characteristics of the 53 non-small
cell lung cancer patients.
| Characteristics | Patients, n | % |
|---|
| Age, years |
| Median | 64 |
| Range | 35–80 |
| Gender, n |
| Male | 40 | 75.5 |
| Female | 13 | 24.5 |
| Smoking history,
n |
| Never | 12 | 22.6 |
| Former/current | 41 | 77.4 |
| Histology, n |
| Adenocarcinoma | 36 | 67.9 |
| Squamous | 13 | 24.5 |
|
Adeno-squamous/unidentified | 1/3 | 1.9/5.7 |
| Performance status,
n |
| 0–1 | 44 | 83.0 |
| 2–3 | 9 | 17.0 |
| Metastatic site,
n |
| Lung | 27 | 50.9 |
| Brain | 13 | 24.5 |
| Bone | 11 | 20.8 |
| Extrathoracic lymph
node | 10 | 18.9 |
| Adrenal grand | 5 | 9.4 |
| Liver | 6 | 11.3 |
| Malignant pleural
effusion | 14 | 26.4 |
| Othersa | 6 | 11.3 |
Survival analysis
In total, four patients responded to erlotinib
therapy, exhibiting a response rate of 7.5%. All four of these
patients also had pulmonary metastasis and malignant pleural
effusion with adenocarcinoma. At the time of analysis, the median
duration of follow-up was 9.8 months (range, 1.2–31.3 months). The
median PFS time for the patients overall was 2.2 months and the
median OS time was 6.2 months. Table
II shows the patient demographics, excluding metastatic sites,
associated with RR, PFS and OS. The patients with improved PS and
skin rash following treatment, exhibited longer PFS and OS times
than those with poor PS and without skin rash, as indicated in
previous studies (PFS, P=0.0002 and P=0.0077; OS, P<0.0001 and
P=0.0026, respectively) (18,19).
However, other factors were demonstrated to be unrelated to PFS and
OS. The median PFS and median OS times for patients according to
metastatic sites are shown in Table
III. The PFS and OS did not depend on the presence or absence
of extrathoracic lymph node and adrenal gland metastasis. In
patients with brain, bone and liver metastasis, the median PFS
times were shorter than for those patients without these
metastases. Furthermore, patients with liver metastasis exhibited a
shorter OS time than patients without liver metastasis. The median
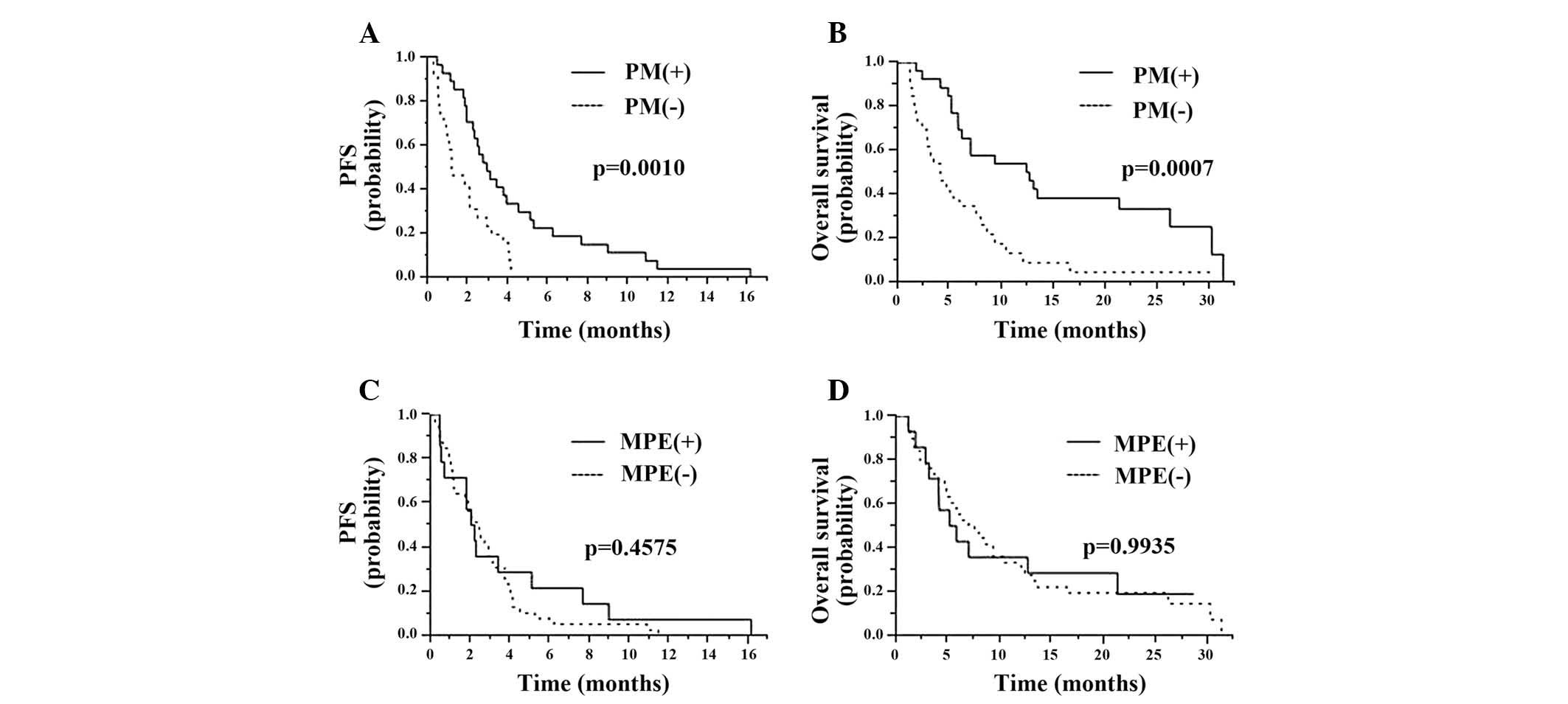
PFS times in the two groups of patients with and without pulmonary
metastasis were 2.9 months (95% CI, 1.9–4.5 months) and 1.2 months
(95% CI, 0.8–2.1 months), respectively (P=0.001; Fig. 1A). Although no significant
differences were identified between the response rate in patients
with and without pulmonary metastasis, the response rate tended to
be higher in patients with pulmonary metastasis (response rate,
14.8 vs. 0.0%; P=0.1110). The median duration of OS in the two
groups of patients with and without pulmonary metastasis was 12.4
months (95% CI, 5.8–26.2 months) and 4.1 months (95% CI, 2.3–7.6
months), respectively (P=0.0007; Fig.
1B). The response rate in patients with malignant pleural
effusion was significantly higher than that of patients without
malignant pleural effusion (response rate, 28.6% vs. 0.0%;
P=0.0034). However, as shown in Fig.
1C, the median PFS times in the patients with and without
malignant pleural effusion were 2.1 and 2.5 months, respectively
(P=0.4575). Furthermore, no significant differences were identified
in OS between the patients with and without malignant pleural
effusion (median OS time, 5.5 months vs. 7.3 months; P=0.9935;
Fig. 1D). Of the 13 variables
assessed, six were observed to be significantly associated with PFS
in univariate analysis: Pulmonary, brain, bone and liver
metastasis, plus the onset of skin rash and PS. The multivariate
analyses of PFS demonstrated that pulmonary metastasis was an
independent and significant predictive factor for PFS (P=0.0055)
(Table IV). By contrast, liver
metastasis and poor PS were risk factors for an unfavorable PFS
following erlotinib therapy (P=0.0279 and P=0.0214, respectively).
Additionally, four factors were observed to be significantly
associated with OS in the univariate analysis: Pulmonary and liver
metastasis plus the onset of skin rash and PS. The presence of
pulmonary metastasis was also an independent and significant
prognostic factor in the multivariate analysis (P=0.0022).
 | Table IIRR, PFS and OS for the patients
according to characteristics. |
Table II
RR, PFS and OS for the patients
according to characteristics.
| Factor | n | RR, % | Pa | mPFS, mo | P-valueb | mOS, mo | P-valueb |
|---|
| Age, years |
| >70 | 18 | 11.1 | 0.2493 | 1.9 | 0.1876 | 5.8 | 0.1151 |
| <71 | 35 | 5.7 | | 3.7 | | 13.1 | |
| Gender, n |
| Male | 40 | 5.0 | 0.2493 | 2.1 | 0.1235 | 5.8 | 0.1788 |
| Female | 13 | 15.4 | | 3.9 | | 16.6 | |
| Smoking history,
n |
| Never | 12 | 8.3 | 1.0000 | 2.3 | 0.2893 | 16.6 | 0.0975 |
| Former/current | 41 | 7.3 | | 2.2 | | 6.0 | |
| Histology, n |
| Adenocarcinoma | 35 | 11.4 | 0.5619 | 1.8 | 0.2847 | 5.8 | 0.8179 |
| Squamous | 13 | 0.0 | | 3.7 | | 9.3 | |
| Performance status,
n |
| 0–1 | 44 | 9.1 | 1.0000 | 2.9 | 0.0002 | 8.6 | <0.0001 |
| 2–3 | 9 | 0.0 | | 0.5 | | 1.9 | |
| Skin rash, n |
| Present | 35 | 11.4 | 0.5619 | 2.9 | 0.0077 | 8.6 | 0.0026 |
| Not present | 18 | 0.0 | | 1.0 | | 2.8 | |
 | Table IIIRR, PFS and OS for the 53 patients
according to the presence of metastatic sites. |
Table III
RR, PFS and OS for the 53 patients
according to the presence of metastatic sites.
| Metastatic site | n | RR, % | P-valuea | mPFS, mo | P-valueb | mOS, mo | P-valueb |
|---|
| Pulmonary metastasis,
n |
| Yes | 27 | 14.8 | 0.1110 | 2.9 | 0.0010 | 12.4 | 0.0007 |
| No | 26 | 0.0 | | 1.2 | | 4.1 | |
| Brain metastasis,
n |
| Yes | 13 | 7.7 | 1.0000 | 1.7 | 0.0440 | 5.0 | 0.0929 |
| No | 40 | 7.5 | | 2.7 | | 7.0 | |
| Bone metastasis,
n |
| Yes | 11 | 0.0 | 0.5688 | 1.2 | 0.0153 | 4.9 | 0.4427 |
| No | 42 | 9.5 | | 2.7 | | 7.0 | |
| Extrathoracic lymph
node metastasis, n |
| Yes | 10 | 0.0 | 1.0000 | 1.9 | 0.5291 | 7.0 | 0.3850 |
| No | 43 | 9.3 | | 2.3 | | 6.2 | |
| Adrenal grand
metastasis, n |
| Yes | 5 | 20.0 | 0.3355 | 1.7 | 0.3993 | 5.8 | 0.3109 |
| No | 48 | 6.3 | | 2.5 | | 7.0 | |
| Liver metastasis,
n |
| Yes | 6 | 0.0 | 1.0000 | 0.7 | <0.0001 | 2.9 | 0.0004 |
| No | 47 | 8.5 | | 2.5 | | 7.6 | |
| Malignant pleural
effusion, n |
| Yes | 14 | 26.4 | 0.0034 | 2.1 | 0.4575 | 5.5 | 0.9935 |
| No | 39 | 0.0 | | 2.5 | | 7.3 | |
 | Table IVMultivariate analysis of
progression-free survival. |
Table IV
Multivariate analysis of
progression-free survival.
| Independent
factor | Hazard ratio | 95% CI | P-value |
|---|
| Pulmonary
metastasis | 0.39 | 0.20–0.76 | 0.0055 |
| Brain
metastasis | 0.94 | 0.40–2.06 | 0.8721 |
| Bone
metastasis | 2.24 | 0.96–4.95 | 0.0616 |
| Liver
metastasis | 3.82 | 1.17–11.65 | 0.0279 |
| Onset of skin
rash | 0.49 | 0.25–1.01 | 0.0522 |
| PS (2–3 vs.
0–1) | 3.12 | 1.20–7.51 | 0.0214 |
Discussion
This study demonstrated that the presence of
pulmonary metastasis was a predictive marker of the outcome in
patients with EGFR-negative NSCLC, receiving erlotinib treatment.
Previously, a randomized controlled trial (BR21) investigating the
effects of erlotinib versus placebo demonstrated that erlotinib
significantly prolonged the median OS, PFS and improved the RR in
comparison with the placebo (9).
Furthermore, subset analysis in this trial demonstrated that
erlotinib treatment was effective in patients with EGFR
mutation-negative NSCLC. Several studies have reported that skin
rashes following erlotinib treatment tend to correlate with the
therapeutic efficacy in patients with NSCLC (18,19).
Therefore, the requirement for biomarkers that can predict the
efficacy of erlotinib therapy prior to initiation is evident. A
number of authors have examined the association between the
efficacy of EGFR-TKIs and patient demographics, including gender,
tumor histology, smoking history and ECOG-PS. However, few studies
have evaluated the efficacy of EGFR-TKIs focusing on metastatic
sites as a tumor property. The present study investigated the
association between patient characteristics, including metastatic
sites and the efficacy of erlotinib treatment in EGFR-mutation
negative NSCLC, and demonstrated that pulmonary metastasis was a
significant and independent factor associated with PFS and OS.
Together, these findings suggest that the presence of pulmonary
metastasis may be useful for predicting the efficacy of erlotinib
in patients with EGFR mutation-negative NSCLC.
Somatic mutations in the EGFR gene have been
identified as a major determinant of the clinical response to
treatment with EGFR-TKIs, such as gefitinib and erlotinib, in
individuals with NSCLC (2,3). In the current study, the four patients
who responded to erlotinib treatment had pulmonary metastasis and
malignant pleural effusion with adenocarcinoma. Recent studies have
suggested that the presence of pulmonary metastasis and malignant
pleural effusion is predictive of EGFR mutations, as is the case in
adenocarcinoma (12,13). In the current study, a number of
cases were reanalyzed for EGFR mutations, including minor
mutations, such as exon 20 insertions and G719X in exon 18;
however, no EGFR mutations were identified in the reanalyzed
samples (results not shown), suggesting that erlotinib may be
effective in certain patients with EGFR mutation-negative NSCLC.
Erlotinib inhibits the activity of EGFR mutation-negative NSCLC
tumor cells at a 50% inhibitory concentration of 2–20 nmol/l. By
contrast, three-fold higher concentrations of gefitinib are
required in order to block mutation-negative EGFR signaling
(20,21). In EGFR mutation-negative NSCLC, it
is postulated that erlotinib may bind to the EGFR more readily than
gefitinib. These results suggest that erlotinib treatment may be
effective in patients with EGFR mutation-negative NSCLC. Patients
who responded to erlotinib treatment in the current study exhibited
a rapid reduction of tumor size, as was the case for EGFR
mutation-positive NSCLC. The mean PFS of the patients in this study
was 9.5 months, which was equivalent to that observed in patients
with EGFR mutation-positive NSCLC (6–8). These
results suggested that erlotinib may inhibit an unknown survival
pathway or may act on tumors that have an unknown EGFR mutation
status.
A number of limitations were present in the current
study: i) The number of patients included was relatively small and,
therefore, assessing the significance of differences was
challenging and not necessarily representative of a larger
population; ii) the retrospective nature of this study did not
allow for a standardized measurement of PFS.
In conclusion, the findings suggest that the
presence of pulmonary metastasis may be a predictive marker of the
response to erlotinib in patients with EGFR mutation-negative
NSCLC. Currently, EGFR mutation-negative NSCLC patients have been
identified for whom treatment is terminated without receiving
erlotinib. However, EGFR mutation-negative NSCLC patients with
pulmonary metastasis may benefit from erlotinib treatment. A
prospective clinical trial is required to confirm the efficacy of
erlotinib treatment in EGFR mutation-negative NSCLC patients with
pulmonary metastasis.
References
|
1
|
Siegel R, Naishadham D and Jemal A: Cancer
statistics, 2012. CA Cancer J Clin. 62:10–29. 2012.
|
|
2
|
Lynch TJ, Bell DW, Sordella R, et al:
Activating mutations in the epidermal growth factor receptor
underlying responsiveness of non-small-cell lung cancer to
gefitinib. N Engl J Med. 350:2129–2139. 2004.
|
|
3
|
Peaz JG, Jänne PA, Lee JC, et al: EGFR
mutations in lung cancer: correlation with clinical response to
gefitinib therapy. Science. 304:1497–1500. 2004.
|
|
4
|
Mok TS, Wu YL, Thongprasert S, et al:
Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N
Engl J Med. 361:947–957. 2009.
|
|
5
|
Maemondo M, Inoue A, Kobayashi K, et al:
North-East Japan Study Group: Gefitinib or chemotherapy for
non-small-cell lung cancer with mutated EGFR. N Engl J Med.
362:2380–2388. 2010.
|
|
6
|
Mitsudomi T, Morita S, Yatabe Y, et al:
West Japan Oncology Group: Gefitinib versus cisplatin plus
docetaxel in patients with non-small-cell lung cancer harboring
mutations of the epidermal growth factor receptor (WJTOG3405): an
open label, randomized phase 3 trial. Lancet Oncol. 11:121–128.
2010.
|
|
7
|
Zhou C, Wu YL, Chen G, et al: Erlotinib
versus chemotherapy as first-line treatment for patients with
advanced EGFR mutation-positive non-small-cell lung cancer
(OPTIMAL, CTONG-0802): a multicenter, open-label, randomized, phase
3 study. Lancet Oncol. 12:735–742. 2011.
|
|
8
|
Rosell R, Carcereny E, Gervais R, et al:
Spanish Lung Cancer Group in collaboration with Groupe Français de
Pneumo-Cancérologie and Associazione Italiana Oncologia Toracica:
Erlotinib versus chemotherapy as first-line treatment for patients
with advanced EGFR mutation-positive non-small-cell lung cancer
(EURTAC): a multicenter, open-label, randomized, phase 3 trial.
Lancet Oncol. 13:239–246. 2012.
|
|
9
|
Shepherd FA, Rodrigues Pereira J, Ciuleanu
T, et al: National Cancer Institute of Canada Clinical Trials
Group: Erlotinib in previously treated non-small-cell lung cancer.
N Engl J Med. 353:123–132. 2005.
|
|
10
|
Ciuleanu T, Stelmakh L, Cicenas S, et al:
Efficacy and safety of erlotinib versus chemotherapy in second-line
treatment of patients with advanced, non-small-cell lung cancer
with poor prognosis (TITAN): a randomised multicentre, open-label,
phase 3 study. Lancet Oncol. 13:300–308. 2012.
|
|
11
|
Walleser S, Ray J, Bischoff H, et al:
Maintenance erlotinib in advanced nonsmall cell lung cancer:
cost-effectiveness in EGFR wild-type across Europe. Clinicoecon
Outcomes Res. 4:269–275. 2012.
|
|
12
|
Wu SG, Hu FC, Chang YL, et al: Frequent
EGFR mutations in nonsmall cell lung cancer presenting with miliary
intrapulmonary carcinomatosis. Eur Respir J. 41:417–424. 2013.
|
|
13
|
Wu SG, Yu CJ, Tsai MF, et al: Survival of
lung adenocarcinoma patients with malignant pleural effusion. Eur
Respir J. 41:1409–1418. 2013.
|
|
14
|
Detterbeck FC, Boffa DJ and Tanoue LT: The
new lung cancer staging system. Chest. 136:260–271. 2009.
|
|
15
|
Oken MM, Creech RH, Tormey DC, et al:
Toxicity and response criteria of the Eastern Cooperative Oncology
Group. Am J Clin Oncol. 5:649–655. 1982.
|
|
16
|
Therasse P, Arbuck SG, Eisenhauer EA, et
al: New guidelines to evaluate the response to treatment in solid
tumors. European Organization for Research and Treatment of Cancer,
National Cancer Institute of the United States, National Cancer
Institute of Canada. J Natl Cancer Inst. 92:205–216. 2000.
|
|
17
|
Nagai Y, Miyazawa H, Huqun, et al: Genetic
heterogeneity of the epidermal growth factor receptor in non-small
cell lung cancer cell lines revealed by a rapid and sensitive
detection system, the peptide nucleic acid-locked nucleic acid PCR
clamp. Cancer Res. 65:7276–7282. 2005.
|
|
18
|
Wacker B, Nagrani T, Weinberg J, et al:
Correlation between development of rash and efficacy in patients
treated with the epidermal growth factor receptor tyrosine kinase
inhibitor erlotinib in two large phase III studies. Clin Cancer
Res. 13:3913–3921. 2007.
|
|
19
|
Liu HB, Wu Y, Lv TF, et al: Skin Rash
could predict the response to EGFR tyrosine kinase inhibitor and
the prognosis for patients with non-small cell lung cancer: a
systematic review and meta-analysis. PLoS One. 8:e551282013.
|
|
20
|
Moyer JD, Barbacci EG, Iwata KK, et al:
Induction of apoptosis and cell cycle arrest by CP-358,774, an
inhibitor of epidermal growth factor receptor tyrosine kinase.
Cancer Res. 57:4838–4848. 1997.
|
|
21
|
Pollack VA, Savage DM, Baker DA, et al:
Inhibition of epidermal growth factor receptor-associated tyrosine
phosphorylation in human carcinoma with CP-358, 774: dynamics of
receptor inhibition in situ and antitumor effects in athymic mice.
J Pharmacol Exp Ther. 291:739–748. 1999.
|