Introduction
Antiviral drugs are key to the treatment of chronic
hepatitis B virus (HBV) infection (1). Standard antiviral treatment should be
administered as long as the indications and conditions allow
(2). Nucleoside analogs ar e one of
the two major classes of antiviral drugs (3). Compared with other nucleoside analogs,
adefovir dipivoxil is characterized by low cost, low drug
resistance and absence of cross-resistance, and has been applied
widely in developing countries (4,5). However,
renal toxicity is the most common adverse reaction associated with
adefovir dipivoxil, and it has been reported to cause Fanconi
syndrome (6–8), which is often misdiagnosed due to a
dearth of knowledge among the majority of clinicians (9). In addition, some common manifestations
of Fanconi syndrome, including systemic bone pain, osteoporosis and
spontaneous fracture, are easily misdiagnosed as osteoarthrosis,
osteoporosis and bone tumors in the early stages (9,10).
In the present study, an analysis of the possible
predictive factors for kidney damage in 28 patients with Fanconi
syndrome caused by long-term administration of adefovir dipivoxil
is reported, alongside possible predictive factors for kidney
damage in the clinic, and monitoring and prevention measures for
early detection of renal toxicity and Fanconi syndrome are
proposed.
Patients and methods
Patients
Patients at Mengchao Hepatobiliary Hospital of
Fujian Medical University (Fuzhou, China) between May 2014 and
February 2015 who were suffering from kidney damage and
manifestations of Fanconi syndrome caused by long-term
administration of adefovir dipivoxil (10 mg/day) for the treatment
of chronic HBV infection were included in the present study. Of
these patients (27 men and 1 woman), 19 cases were from rural areas
and 9 cases were from urban areas. The age of patients at the start
of adefovir dipivoxil administration ranged from 28 to 69
years.
The hepatitis B surface antigen (HBsAg)-positive
status history ranged from 1 to 46 years, and 20 cases had a family
history of HBsAg-positive status. There were 14 cases of chronic
hepatitis B, 10 cases of compensatory hepatitis B-associated
cirrhosis, 3 cases of decompensatory hepatitis B-associated
cirrhosis and 1 case of hepatic failure. The HBV-DNA levels ranged
from 2.0×103 to 8.6×109 IU/ml. In addition,
19 cases were positive for hepatitis B envelope antigen
(HBeAg).
In total, 6 cases were accompanied by type 2
diabetes, 6 cases were accompanied by hypertension and 3 cases were
accompanied by both diseases. Furthermore, 7 cases had previously
received lamivudine and presented resistance; among these, 4 cases
were changed to antiviral treatment with a combination of
lamivudine (GlaxoSmithkline Pharmaceuticals, Co., Ltd., Suzhou,
China) and adefovir dipivoxil (Chia Tai Tianqing Pharmaceutical
Group Co., Ltd., Nanjing, China), while 3 cases were changed to
antiviral treatment with a combination of entecavir (Sino-American
Shanghai Squibb Pharmaceuticals Ltd., Shanghai, China) and adefovir
dipivoxil. One case presented resistance against entecavir and was
changed to antiviral treatment with a combination of entecavir and
adefovir dipivoxil. In total, 2 cases did not respond to adefovir
dipivoxil treatment and were changed to antiviral treatment with a
combination of lamivudine and adefovir dipivoxil. Patients had
normal renal function, blood electrolytes and routine urine
examinations prior to administration of adefovir dipivoxil
(Table I).
 | Table I.Baseline characterization of 28
Fanconi syndrome cases. |
Table I.
Baseline characterization of 28
Fanconi syndrome cases.
| Variables | Baseline
characterization (n=28) | Reference
value |
|---|
| Age, years | 45.5 (17.0) |
|
| Gender, % |
|
|
|
Male | 96.4 |
|
Female | 3.6 |
| HbsAg+
family history, % |
|
|
| With | 71.4 |
|
| Without | 28.6 |
|
| HbsAg+
duration, years | 14.7±12.2 |
| Biochemical
level |
|
|
| ALB,
g/l | 4.25±4.16 | (34.00–54.00) |
| GLB,
g/l | 32.5 (6.0) | (20.0–40.0) |
| TBIL,
µmol/l | 21.23±8.40 | (3.00–25.00) |
| DBIL,
µmol/l | 6.0±5.9 | (0.0–8.6) |
| ALT,
U/l | 98.3±98.6 | (9.0–50.0) |
| AST,
U/l | 87±99 | (15–40) |
| GGT,
U/l | 54.9±36.6 | (10.0–60.0) |
| log10
HBV-DNA, IU/ml | 5.9 (1.6) |
|
| HBeAg, % |
|
|
|
Positive | 58.3 |
|
|
Negative | 41.7 |
|
The present study was approved by the Medical Ethics
Committee of Fuzhou Infectious Disease Hospital (Fuzhou, China).
Informed consent was obtained from the 28 patients. All data were
used with permission of the patients and published anonymously.
Histopathological and
immunohistochemical assessment
Fresh renal biopsy samples were fixed in 4%
paraformaldehyde at room temperature for 24 h, and then gradually
dehydrated using ethanol and embedded in paraffin. The paraffin
blocks were subsequently sectioned (5-µm) and stained with Mason's
Trichrome and periodic acid-Schiff staining kit (Beijing Solarbio
Science & Technology Co., Ltd., Beijing, China) or hematoxylin
and eosin (Nanjing Jiancheng Bioengineering Institute, Nanjing,
China), according to the manufacturer's protocol. To further assess
the degree of renal tubular damage, the accumulation of complement
component 3 (C3) in the renal tubule was detected using an anti-C3
monoclonal antibody (1:100 dilution; cat. no. ab11871; Abcam,
Cambridge, MA, USA) at 4°C overnight. After several rinses with
PBS, the tissue sections were incubated with horseradish
peroxidase-conjugated secondary antibody (1/1 dilution; cat. no.
PV9003; ZSGB-BIO, Beijing, China) at room temperature for 1 h.
Finally, the renal sections were stained with diaminobenzidine
(Beijing Solarbio Science & Technology Co., Ltd.) and
hematoxylin. Subsequently, double-blind evaluation of hepatic
steatosis was performed by two expert pathologists. The
histopathological examination was performed using an inverted
phase-contrast microscope.
Results
Clinical manifestations
Of the 28 cases, 15 presented clinically with
progressive fatigue and multiple joint pain; 11 cases manifested
differing degrees of difficulty in walking; and 3 cases had
pathological fractures. The time from medication to presentation of
the symptoms described ranged from 16 to 100 months [mean ±
standard deviation (SD), 54.80±25.64 months]. Pain was reported
predominantly in the ankle, knee, hip, and lumbosacral and
hypochondriac areas. Initially, pain was reported in one to two
areas, and gradually progressed to systemic multiple joint pain
with prolonged medication time. The pain was aggravated by adefovir
dipivoxil loading, and progressed to disability in walking and
pathological fractures. A total of 2 cases presented
acroanesthesia, while 1 case presented a 2-cm decrease in height
and an 8-kg reduction in body weight. All the patients had normal
sensation, muscular strength and muscular tension (Table II).
 | Table II.Main symptoms or serum/urine
examinations of 28 Fanconi syndrome cases. |
Table II.
Main symptoms or serum/urine
examinations of 28 Fanconi syndrome cases.
| Main symptoms or
serum/urine examinations | No. of positive
cases | Percentage | Total no. of
cases |
|---|
| Joint pain | 15 |
53.6 | 28 |
| Disability in
walking | 11 |
39.3 | 28 |
| Pathological
fractures | 3 |
10.7 | 28 |
| Osteoporosis | 7 | 100.0 | 7 |
| Hypokalemia | 11 |
39.3 | 28 |
| Hypocalcemia | 2 |
7.1 | 28 |
|
Hypophosphatemia | 23 |
82.1 | 28 |
| Blood urea nitrogen
upregulated | 2 |
7.1 | 28 |
| Serum creatinine
upregulated | 15 |
53.6 | 28 |
| Cystatin C
upregulated | 15 |
93.8 | 16 |
| Alkaline
phosphatase upregulated | 14 |
50.0 | 28 |
| Urinary protein
positive | 19 |
73.1 | 26 |
| Urinary glucose
positive | 13 |
50.0 | 26 |
| Urinary
erythrocytes positive | 18 |
69.2 | 26 |
Laboratory examinations
All 28 cases exhibited changes in the results of
routine urine examinations, serum phosphorus and renal function to
varying degrees, and the time from the start of treatment to the
presence of the described chemical abnormalities ranged from 16 to
106 months (mean ± SD, 53.64±25.09 months). In total, 22 cases
presented with hypophosphatemia, with serum phosphorus levels in
the range of 0.36–0.82 µmol/l (normal range, 0.90–1.62 µmol/l); 11
cases presented with hypokalemia, with serum potassium levels in
the range of 2.92–3.44 mmol/l (normal range, 3.50–5.30 mmol/l); 14
cases presented with increased serum alkaline phosphatase in the
range of 127–481 U/l (normal range, 45–125 U/l); and only 2 cases
presented with hypocalcemia, with calcium levels of 1.66 and 2.07
mmol/l, respectively (normal range, 2.08–2.60 mmol/l). Among the 16
cases tested for cystatin C (Cys-C), the levels of Cys-C were
increased in 15 cases, with values ranging from 1.06–1.49 mg/l
(normal range, 0.00–1.03 mg/l). In addition, 2 cases presented with
elevated blood urea nitrogen (BUN), with values of 8.9 and 9.2
mmol/l, respectively (normal range, 2.9–8.2 mmol/l), while 15 cases
presented increased serum creatinine (SCr), with values ranging
from 119–188 µmol/l (normal range, 53–115 µmol/l).
Urine routine examinations were performed in 26
cases, and urine pH in 1, 7 and 18 cases exhibited values that were
<5.5, >6.5 and normal, respectively (normal pH range,
5.5–6.5). Specific gravity examinations revealed that 7, 2 and 17
cases presented with values that were <1.015, >1.025 and
normal, respectively (normal range, 1.015–1.025). Urinary protein
examinations revealed that 2, 17 and 7 cases presented with urinary
protein levels that were ≥2 g/l, ≥1 g/l and normal (normal range,
0.04–0.99 g/l), respectively. Microalbuminuria was positive (≥0.56
g/l) in 19 cases, while urinary erythrocytes counts of ≥200
cells/µl, ≥80 cells/µl and ≥25 cells/µl were detected in 1, 7 and
10 cases, respectively. Urinary glucose levels of ≥28 mmol/l, ≥14
mmol/l and ≥5.6 mmol/l were detected in 7, 1 and 5 cases,
respectively. A total of 7 cases underwent bone mineral density
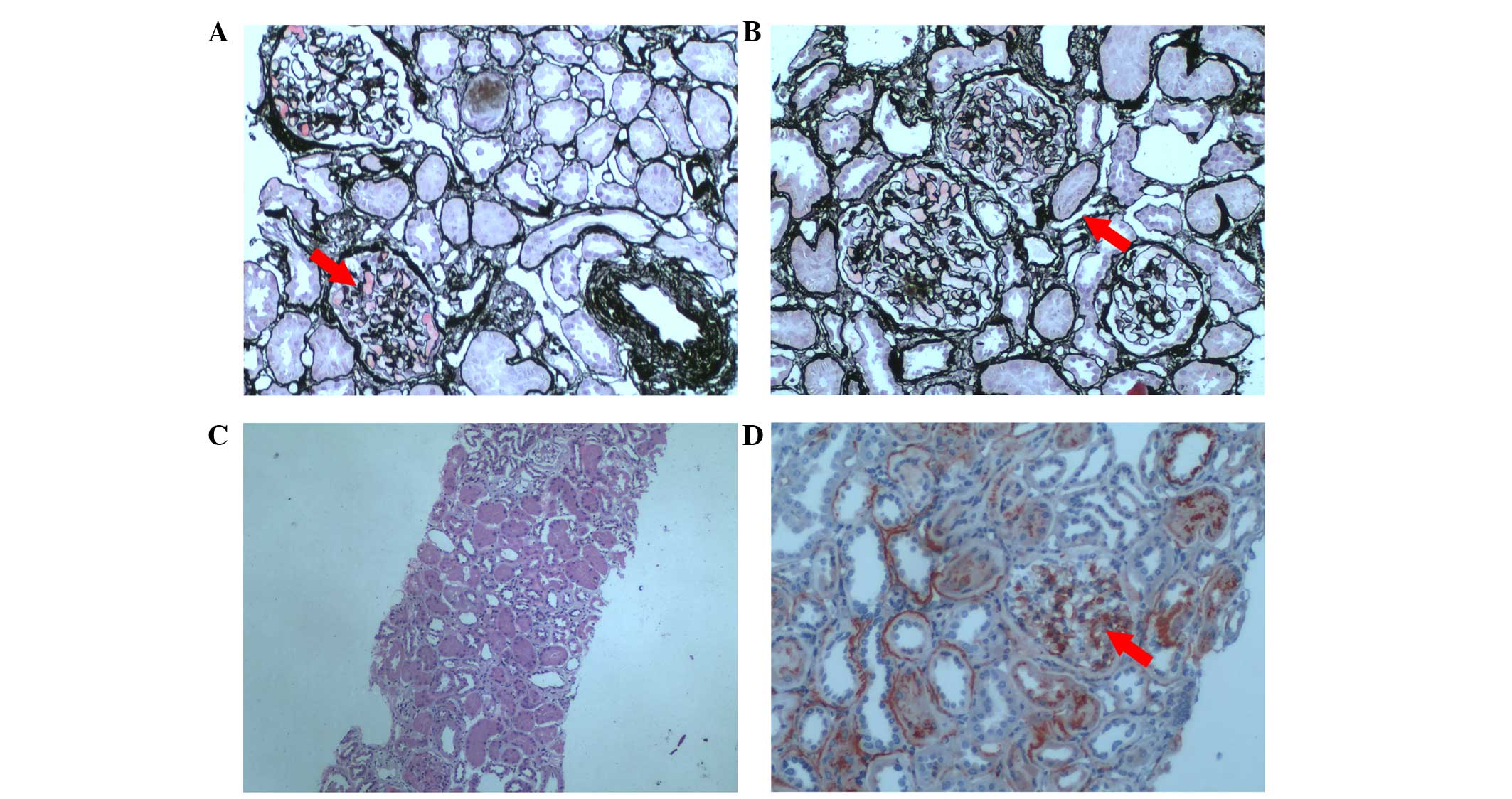
evaluation, and all returned osteoporosis. A renal pathological
examination was conducted for 1 patient, and the results revealed
diffuse mild mesangial cell proliferation accompanied by focal and
segmental glomerulosclerosis (ischemic sclerosis), as well as mild
renal tubular atrophy, interstitial fibrosis and atherosclerosis at
two points (Table II). The
microphotographs of a renal biopsy from one case are shown in
Fig. 1.
Age distribution
The age of the patients ranged from 34–76 years,
with the majority of patients aged between 40 and 59 years,
accounting for 57.1% of the total cases (Table III).
 | Table III.Age distribution of patients with
Fanconi syndrome. |
Table III.
Age distribution of patients with
Fanconi syndrome.
| Age, years | No. of cases | Percentage |
|---|
| 30–39 | 5 | 17.9 |
| 40–49 | 7 | 25.0 |
| 50–59 | 9 | 32.1 |
| 60–69 | 3 | 10.7 |
| 70–79 | 4 | 14.3 |
Duration of adefovir dipivoxil
treatment at first onset of symptoms or abnormal laboratory
examinations and total administration duration
In the 28 cases, the time of the first onset of
symptoms or laboratory examination abnormalities following
administration of adefovir dipivoxil ranged from 16 to 106 months
(mean ± SD, 53.64±25.09 months). Table
IV reveals that only 5 cases had a duration of <24 months,
accounting for 17.9% of all cases, while cases with a duration
exceeding 24 months accounted for 82.1% of all cases, presenting a
significant difference (P<0.05). The total time of
administration ranged from 18–118 months, which was longer than the
time from administration to the presence of abnormal laboratory
examinations.
 | Table IV.Duration of adefovir dipivoxil
treatment at first onset of symptoms or abnormal laboratory
examinations. |
Table IV.
Duration of adefovir dipivoxil
treatment at first onset of symptoms or abnormal laboratory
examinations.
| Time range,
months | No. of abnormal
cases | Percentage |
|---|
| 12–24 | 5 | 17.9 |
| 25–48 | 9 | 32.1 |
| 49–72 | 9 | 32.1 |
| 73–96 | 3 | 10.7 |
| >97 | 2 |
7.1 |
Estimated glomerular filtration rate
(eGFR) at pre- and post-administration of adefovir dipivoxil in the
28 cases
The eGFR values at pre-and post-administration of
adefovir dipivoxil were estimated in all patients using the Chronic
Kidney Disease (CKD)-Epidemiology Collaboration formula (11,12). The
eGFR values prior to the administration of adefovir dipivoxil
ranged from 48.4–111.8 ml/min/1.73 m2, among which, 9,
11 and 1 cases had eGFR values of >90 ml/min/1.73 m2
[stage 1 CKD (CKD1)], 60–90 ml/min/1.73 m2 [stage 2 CKD
(CKD2)] and 30–60 ml/min/1.73 m2 [stage 3 CKD (CKD3)],
respectively. The baseline eGFR was not obtained in 7 cases. The
eGFR values at diagnosis of Fanconi syndrome ranged from 30–94
ml/min/1.73 m2, and there were 2, 14 and 12 cases of
CKD1, CKD2 and CKD3, respectively. Among the 12 cases of CKD3, the
eGFR values preceding adefovir dipivoxil administration were
determined in 9 cases, of which, 6 cases had an eGFR of <90
ml/min/1.73 m2. The eGFR was reduced following adefovir
dipivoxil administration in 17 patients, while it was elevated in 4
cases (Table V).
 | Table V.eGFR values of patients at pre- and
post-treatment with adefovir dipivoxil. |
Table V.
eGFR values of patients at pre- and
post-treatment with adefovir dipivoxil.
| Case | Pre-treatment
(ml/min/1.73 m2) | Duration
(months) | Post-treatment
(ml/min/1.73 m2) |
|---|
| 1 |
79.0 | 48 | 30.0 |
| 2 |
76.0 | 108 | 36.0 |
| 3 |
75.0 | 78 | 42.0 |
| 4 |
48.4 | 60 | 43.0 |
| 5 | NR | 36 | 45.0 |
| 6 |
73.0 | 52 | 46.0 |
| 7 | NR | 100 | 46.0 |
| 8 |
69.0 | 73 | 53.0 |
| 9 |
95.0 | 106 | 53.0 |
| 10 | NR | 31 | 55.0 |
| 11 | 109.8 | 72 | 57.0 |
| 12 | 111.8 | 84 | 59.0 |
| 13 |
96.0 | 50 | 60.0 |
| 14 | 110.0 | 96 | 62.0 |
| 15 | NR | 96 | 63.0 |
| 16 |
89.0 | 78 | 63.0 |
| 17 | NR | 60 | 65.0 |
| 18 |
90.0 | 36 | 67.0 |
| 19 |
98.0 | 63 | 68.0 |
| 20 |
93.0 | 18 | 69.0 |
| 21 |
95.0 | 71 | 73.0 |
| 22 |
67.0 | 94 | 75.0 |
| 23 | NR | 27 | 78.1 |
| 24 | 107.3 | 70 | 80.0 |
| 25 | NR | 69 | 82.0 |
| 26 |
70.7 | 118 | 88.0 |
| 27 |
85.0 | 100 | 93.0 |
| 28 |
88.0 | 46 | 94.0 |
Outcome
Among the 28 patients, 1 case was changed to 0.5
mg/day entecavir, 1 case was changed to antiviral treatment with a
combination of 0.5 mg/day entecavir and 300 mg/day tenofovir; 1
case was changed to 300 mg/day tenofovir, according to the Cr
clearance rate; 1 case stopped antiviral treatment and received
phosphate and calcium supplements; and patients with hypokalemia
received potassium supplements. The follow-up duration ranged from
2–20 months. Patients with fatigue, joint pain and difficulty in
walking experienced significant remission after 2–6 months, while
patients with hypokalemia, hypophosphatemia and abnormalities in
routine urine examinations and SCr were restored to normal levels
after 1–4 months. A total of 5 patients with increased Cys-C were
retested 5 months later, and the abnormalities remained, but with a
mild reduction. Two months later, the eGFR values remained
unchanged in 2 cases, while they were reduced in 4 cases and
slightly elevated in 22 cases.
Discussion
Adefovir dipivoxil, which is a nucleotide analog
with a single adenosine phosphate, exerts its antiviral effects by
competing with the natural deoxyadenosine triphosphate substrate to
terminate the extension of viral DNA chains (13). This drug has been used to treat human
immunodeficiency virus infections in the early stages, with large
doses associated with significant renal toxicity, including acute
tubular necrosis and Fanconi syndrome (14), as well as mitochondrial abnormalities
in the proximal renal tubular ultrastructure (15). Prospective studies have shown that the
5-year renal toxicity of adefovir dipivoxil ranges from 3–8%
(16). Furthermore, adefovir
dipivoxil-induced Fanconi syndrome has been reported (7,8).
Fanconi syndrome is characterized by proximal tubule
transport dysfunction due to congenital or acquired factors, and is
manifested as renal glucosuria, aminoaciduria, phosphaturia and
renal tubular acidosis, thereby inducing a group of syndromes,
including hypokalemic paralysis, hypophosphatemia and hypocalcemia
osteopathy (osteoporosis and bone deformity) (10). This syndrome can be divided into
hereditary, idiopathic and acquired categories (17). Acquired Fanconi syndrome occurs mainly
in adults, and one of its main causes is treatment with various
drugs, including aristolochic acid, analgesics, contrast agents,
expired tetracycline, aminoglycoside antibiotics, azathioprine,
6-mercaptopurine, streptozotocin, cisplatin, ranitidine, valproate
anticonvulsants, and antiviral drugs such as cidofovir, tenofovir
and adefovir esters (18–26). Tenofovir-induced Fanconi syndrome is
often accompanied by acute renal failure (27–35).
It is generally accepted that the mechanism of
adefovir dipivoxil-induced liver damage is associated with the
aggregation effects caused by the human renal organic anion
transporter 1 (HOAT-1) on the drugs and by the toxicity of the
drugs on the mitochondria (36).
Several studies have reported that HOAT-1 has a strong affinity for
adefovir dipivoxil, and that individuals who show high expression
levels of HOAT-1 show a higher uptake of adefovir dipivoxil in the
renal proximal tubule compared with individuals who show low
expression of HOAT-1 or who are deficient (37,38); this
was observed in cases with tenofovir-related kidney damage
(39,40). Adefovir bisphosphonate, which is the
effective metabolite of adefovir dipivoxil, tends to suppress the
duplication of mitochondrial DNA, leading to mitochondrial DNA loss
in the renal proximal tubule, thus affecting renal tubular
reabsorption and secretion (36).
Previous studies demonstrated that adefovir dipivoxil-induced
proximal tubule damage tends to lead to Fanconi syndrome, which
presents different degrees of hypophosphatemia, hypouricemia and
combined dysfunction of the proximal renal tubule (41).
Patients with adefovir dipivoxil-induced Fanconi
syndrome in the present study were characterized by a number of
features. The male:female ratio was 27:1, with a significantly
larger number of men affected than women. This may be due to the
incidence of chronic hepatitis B, and consequently the number of
cases treated with adefovir dipivoxil is significantly higher among
males than females. Further studies are required to identify a
correlation between kidney damage and Fanconi syndrome caused by
adefovir dipivoxil with gender.
Furthermore, 57% of the patients in this group were
aged between 40 and 59 years. Among the patients younger than 60
years, the incidence gradually increased with age. It is speculated
that kidney damage caused by adefovir dipivoxil may be associated
with age. Age >40 years may be a risk factor for kidney damage
caused by adefovir dipivoxil; however, in the present study, the
incidence decreased in the population older than 60 years. This may
be associated with the fact that adefovir dipivoxil is suitable for
the population aged between 18 and 65 years (1,42);
consequently, a small number of patients aged >65 years are
treated with adefovir dipivoxil. Among the patients in the current
study, 19 cases came from rural areas and 9 cases came from urban
areas. We speculate that this difference is associated with poor
health consciousness, low follow-up probability and inferior
medical conditions in rural areas compared with those in urban
areas.
Among the 28 cases, 14 suffered from chronic
hepatitis B and 14 suffered from hepatitis B-associated cirrhosis.
Among the patients receiving adefovir dipivoxil, the proportion of
patients with chronic hepatitis B was significantly greater than
that among patients with hepatitis B-associated cirrhosis,
suggesting that kidney damage and Fanconi syndrome are more likely
to occur in patients with hepatitis B-associated cirrhosis
following adefovir dipivoxil administration. Liver function
remained normal upon receiving adefovir dipivoxil in 28 patients.
Although it was mildly elevated in 2 cases, the HBV-DNA levels were
normal in the remaining cases. The HBeAg-positive rate (58.30%) was
not significantly different from the HBeAg-negative rate
(P>0.05). It is not possible to determine the effects of liver
function, HBeAg-positive status or HBV-DNA levels on adefovir
dipivoxil-induced kidney damage with only 28 cases; therefore,
these effects require further investigations.
Among the 28 cases, 9 (32.1%) were accompanied by
hypertension or type 2 diabetes, exhibiting a significantly higher
prevalence than that in the population receiving adefovir
dipivoxil. This is due to the fact that hypertension and type 2
diabetes may induce secondary liver damage, particularly among
patients with poor control of blood pressure and glucose (43). Furthermore, antihypertensive and
hypoglycemic drugs may increase liver burden and aggravate liver
damage (44,45). In addition, liver damage is also a
risk factor for hypertension, exhibiting an interaction effect
(46). Therefore, patients with
Fanconi syndrome accompanied by hypertension and type 2 diabetes
may be subjected to a higher risk of liver damage when treated with
adefovir dipivoxil compared with patients with Fanconi syndrome
alone.
In total, 23 of the 28 patients had a total duration
of adefovir dipivoxil treatment exceeding 24 months. Among 11 cases
presenting progression to difficulty in walking, 9 had a total
duration of adefovir dipivoxil treatment >24 months, and the
symptoms were aggravated by prolonged duration of drug
administration. Therefore, we speculate that duration of adefovir
dipivoxil treatment >24 months is a predictive factor of liver
damage and Fanconi syndrome. In certain patients, the total
duration of adefovir dipivoxil treatment was longer than the period
in which abnormalities were present, suggesting that these patients
were not aware of having Fanconi syndrome and therefore continued
receiving adefovir dipivoxil treatment. Among the 28 patients, 14
continued to receive adefovir dipivoxil following the appearance of
joint pain, with a mean time of continued adefovir dipivoxil
administration of 21.3 months. Prior to being diagnosed as Fanconi
syndrome, these patients had visited a number of hospitals and
medical departments and undergone multiple examinations, but had
not obtained a distinct diagnosis or effective treatment;
consequently, the joint pain was progressively aggravated. Of these
patients, 11 had progressed to difficulty in walking when they
visited the Mengchao Hepatobiliary Hospital of Fujian Medical
University. Therefore, early detection of abnormalities in chemical
examinations and early diagnosis are particularly important for
timely alleviation of patients' pain. Since Fanconi syndrome is
rare, numerous physicians are unfamiliar with its symptoms and
signs, thus leading to delayed diagnosis and treatment (9). Therefore, improved knowledge of Fanconi
syndrome is important, particularly for hepatologists.
Of the 28 cases,9 received combined antiviral drug
treatment. Since nucleotide analogs, such as adefovir dipivoxil,
are excreted by the kidney in their original form, combined
antiviral drug treatment tends to increase the burden of the kidney
(47,48). Further studies are required to
investigate whether combined antiviral drug treatment increases the
risk of renal dysfunction.
In the current group of patients, there were 12
cases of CKD3 at the point at which Fanconi syndrome was diagnosed.
The eGFR was measured prior to adefovir dipivoxil administration in
9 cases, and was <90 ml/min/1.73 m2 in 6 cases,
suggesting that such patients are subjected to a greater risk of
renal damage than patients with and eGFR of >90 ml/min/1.73
m2. Thus, it is necessary to assess the renal function
in patients prior to adefovir dipivoxil administration.
The National Kidney Foundation-Kidney Disease
Outcomes Quality Initiative guidelines of chronic renal disease
state that, while Cr alone is insufficient, GFR is widely accepted
as the best index to assess renal function, wherein the GFR
decrease is parallel to renal function damage in chronic renal
disease (49). Among the 28 cases
included in the present study, Cys-C detection was performed in 19
cases at diagnosis. The levels of Cys-C were elevated in 18 cases
(range, 1.05–1.59 mg/l), among which, only 8 cases presented
elevated SCr. In total, 26 of 28 cases had eGFR <90 ml/min/1.73
m2, while 13 cases had eGFR <60 ml/min/1.73
m2 at the time of Fanconi syndrome diagnosis, and only
15 cases presented increased SCr, suggesting that Cys-C and GFR are
more sensitive measures of renal function than SCr (50), and that eGFR is the best index
(51). Therefore, Cys-C and GFR
accurately represent renal function.
All 28 patients discontinued adefovir dipivoxil when
they were diagnosed as Fanconi syndrome, and subsequently received
phosphorus and calcium supplementation. Significant remission in
fatigue, joint pain, difficulty in walking and abnormalities in
laboratory examinations were achieved after 2–6 months, indicating
that the key to improvement is the timely termination of adefovir
dipivoxil treatment.
A cross-sectional study involving 13,925 cases of
kidney disease in China revealed that age, previous administration
of nephrotoxic drugs, living in rural areas, history of
cardiovascular disease, a high-density lipoprotein level <40
mg/dl and hypertension are independent risk factors for kidney
function decline, while diabetes and abdominal obesity are
associated risk factors for eGFR decline (52–55); this
is consistent with the clinical characteristics of the patients in
the present study. The population with chronic HBV infection
presents a higher prevalence of, and susceptibility to, kidney
disease than the general population (56). The prevalence of kidney disease
increases gradually with the progression of liver disease,
particularly in patients with decompensated hepatic cirrhosis
(57). The eGFR significantly
decreases with age in patients with hepatic cirrhosis (54,58), with
an inverse correlation between baseline eGFR and risk of mortality
in patients with hepatic cirrhosis (59). Additionally, renal impairment is a
risk factor for mortality in patients with end-stage chronic
hepatitis B (60,61). Therefore, it is of particular
relevance to monitor renal function and to detect and correct renal
dysfunction in patients with chronic HBV infection in a timely
manner.
Adefovir dipivoxil has been used as an antiviral
agent for ~10 years, and is extensively used in primary hospitals
(62). Since the majority of
physicians are not yet fully aware of the adverse reactions of
adefovir dipivoxil, its use may seriously affect the quality of
life of patients, even leading to bone fractures and disability, if
treatment is not discontinued in a timely manner (16,63).
Patients with chronic HBV infection may incur proximal renal tubule
damage and acquired Fanconi syndrome upon receiving adefovir
dipivoxil, and tenofovir may cause similar effects via the same
mechanism (16,43,64).
Therefore, prior to administering adefovir dipivoxil, it is
necessary to assess renal function in patients. In addition to
monitoring BUN and SCr, blood potassium, serum phosphorus, Cys-C,
eGFR, urinary glucose, urinary protein, urine pH and other indexes
of renal tubular injury should be determined, with eGFR widely
accepted as the best index for assessing renal function. Timely
termination of adefovir dipivoxil treatment and replacement with
other antiviral agents is critical once renal impairment appears.
Furthermore, it is necessary to change to other antiviral agents
and to prolong the interval of administration according to the eGFR
level. Improvements in symptoms and chemical examination
abnormalities were achieved in the majority of patients in the
present study after 2–6 months.
In conclusion, the results of the present study
indicated that being aged >40 years, living in rural areas, a
history of nephrotoxic drug use, eGFR of <90 ml/min/1.73
m2, hypertension, diabetes, cirrhosis and duration of
adefovir dipivoxil treatment exceeding 24 months were predictive of
kidney damage and Fanconi syndrome.
Acknowledgements
The present study was supported by the National
Science and Technology Major Projects (grant no. 2008ZX10002-005),
the National Natural Science Foundation of China (grant no.
81602102), the Natural Science Foundation of Fujian Province (grant
nos. 2015J05174 and 2016J01592), the Project of Nanjing Military
Region (grant no. 15MS136), the Scientific Research Project of
Health and Family Planning Commission of Fujian province (grant
nos. 2014-2-43 and 2015-1-94) and the Scientific Foundation of
Fuzhou Health Department (grant no. 2014-S-139-3).
References
|
1
|
Segovia MC, Chacra W and Gordon SC:
Adefovir dipivoxil in chronic hepatitis B: history and current
uses. Expert Opin Pharmacother. 13:245–254. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Shepherd J, Jones J, Takeda A, Davidson P
and Price A: Adefovir dipivoxil and pegylated interferon alfa-2a
for the treatment of chronic hepatitis B: A systematic review and
economic evaluation. Health Technol Assess. 10(iii-iv): xi–xiv,
1–183. 2006.
|
|
3
|
Stein LL and Loomba R: Drug targets in
hepatitis B virus infection. Infect Disord Drug Targets. 9:105–116.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Chen Y and Ju T: Comparative meta-analysis
of adefovir dipivoxil monotherapy and combination therapy of
adefovir dipivoxil and lamivudine for lamivudine-resistant chronic
hepatitis B. Int J Infect Dis. 16:e152–158. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Han SH: Natural course, therapeutic
options and economic evaluation of therapies for chronic hepatitis
B. Drugs. 66:1831–1851. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Girgis CM, Wong T, Ngu MC, Emmett L,
Archer KA, Chen RC and Seibel MJ: Hypophosphataemic osteomalacia in
patients on adefovir dipivoxil. J Clin Gastroenterol. 45:468–473.
2011. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Law ST, Li KK and Ho YY: Nephrotoxicity,
including acquired Fanconi's syndrome, caused by adefovir
dipivoxil-is there a safe dose? J Clin Pharm Ther. 37:128–131.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Li L, Dong GF, Zhang X and Xie YS:
Adefovir dipivoxil-induced Fanconi syndrome and hypophosphatemic
osteomalacia associated with muscular weakness in a patient with
chronic hepatitis B. Nan Fang Yi Ke Da Xue Xue Bao. 31:19562011.(In
Chinese). PubMed/NCBI
|
|
9
|
Li X, Shen M, Sun WJ, Huang ZX, An N and
Zhang JJ: Misdiagnosis of Bone Metastasis Cancer After Using
Adefovir Dipivoxi in a Hepatitis B Patient with Fanconi Syndrome.
Indian J Hematol Blood Transfus. 32:329–331. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lee YS, Kim BK, Lee HJ and Dan J:
Pathologic Femoral Neck Fracture Due to Fanconi Syndrome Induced by
Adefovir Dipivoxil Therapy for Hepatitis B. Clin Orthop Surg.
8:232–236. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Stevens LA, Schmid CH, Greene T, Zhang YL,
Beck GJ, Froissart M, Hamm LL, Lewis JB, Mauer M, Navis GJ, et al:
Comparative performance of the CKD epidemiology collaboration
(CKD-EPI) and the modification of diet in renal disease (MDRD)
study equations for estimating GFR levels above 60 ml/min/1.73 m2.
Am J Kidney Dis. 56:486–495. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Levey AS, Stevens LA, Schmid CH, Zhang YL,
Castro AF III, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene
T, et al: A new equation to estimate glomerular filtration rate.
Ann Intern Med. 150:604–612. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zeng AZ, Lu P, Deng H, Cai SF, Yang C, Xin
XJ, Guo JJ, Li QL, Deng XH and Huang AL: Dissection of mechanism
for the adefovir dipivoxil resistance in chronic hepatitis B
patients. Zhonghua ganzangbing zazhi. 17:730–734. 2009.PubMed/NCBI
|
|
14
|
Kahn J, Lagakos S, Wulfsohn M, Cherng D,
Miller M, Cherrington J, Hardy D, Beall G, Cooper R, Murphy R, et
al: Efficacy and safety of adefovir dipivoxil with antiretroviral
therapy: A randomized controlled trial. JAMA. 282:2305–2312. 1999.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Brinkman K, ter Hofstede HJ, Burger DM,
Smeitink JA and Koopmans PP: Adverse effects of reverse
transcriptase inhibitors: Mitochondrial toxicity as common pathway.
AIDS. 12:1735–1744. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Fontana RJ: Side effects of long-term oral
antiviral therapy for hepatitis B. Hepatology. 49:(Suppl 5).
S185–S195. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Tsilchorozidou T and Yovos JG:
Hypophosphataemic osteomalacia due to de Toni-Debre-Fanconi
syndrome in a 19-year old girl. Hormones. 4:171–176. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Shaikh A, Wiisanen ME, Gunderson HD and
Leung N: Acquired Fanconi syndrome after treatment with
capecitabine, irinotecan, and bevacizumab. Ann Pharmacother.
43:1370–1373. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Lemire J and Kaplan BS: The various renal
manifestations of the nephropathic form of cystinosis. Am J
Nephrol. 4:81–85. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Cleveland WW, Adams WC, Mann JB and Nyhan
WL: Acquired Fanconi Syndrome Following Degraded Tetracycline. J
Pediatr. 66:333–342. 1965. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Schwartz JH and Schein P: Fanconi syndrome
associated with cephalothin and gentamicin therapy. Cancer.
41:769–772. 1978. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rossi R, Rath B, Ullrich K and Ehrich JH:
Ifosfamide-induced nephrotoxicity. Monatsschr Kinderheilkd.
141:594–601. 1993.(In German). PubMed/NCBI
|
|
23
|
Knorr M, Schaper J, Harjes M, Mayatepek E
and Rosenbaum T: Fanconi syndrome caused by antiepileptic therapy
with valproic Acid. Epilepsia. 45:868–871. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Venkatesan EP, Pranesh MB, Gnanashanmugam
G and Balasubramaniam J: Tenofovir induced Fanconi syndrome: A rare
cause of hypokalemic paralysis. Indian J Nephrol. 24:108–109. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Zhu LN, Mou LJ, Hu Y and Chen JX: Fanconi
syndrome caused by low-dose adefovir dipivoxil. Saudi Med J.
35:309–313. 2014.PubMed/NCBI
|
|
26
|
Yoshinami T, Yagi T, Sakai D, Sugimoto N
and Imamura F: A case of acquired Fanconi syndrome induced by
zoledronic acid. Intern Med. 50:1075–1079. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Zheng F, Zhao S and Li X: The clinical and
biochemical manifestations of Fanconi syndrome: A report of 42
cases. Zhonghua Nei Ke Za Zhi. 39:735–738. 2000.(In Chinese).
PubMed/NCBI
|
|
28
|
Belmouaz S, Sechet A, Fernandez B, Ayache
RA, Desport E, Bauwens M, Goujon JM, Gombert JM, Bridoux F and
Touchard G: Tubulo-interstitial nephritis with Fanconi syndrome in
Behcet disease. Nephrol Dial Transplant. 22:2079–2083. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Ghiculescu RA and Kubler PA:
Aminoglycoside-associated Fanconi syndrome. Am J Kidney Dis.
48:e89–e93. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Gupta SK: Tenofovir-associated Fanconi
syndrome: Review of the FDA adverse event reporting system. AIDS
Patient Care STDS. 22:99–103. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Kazory A, Singapuri S, Wadhwa A and Ejaz
AA: Simultaneous development of Fanconi syndrome and acute renal
failure associated with cidofovir. J Antimicrob Chemother.
60:193–194. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Francois H, Coppo P, Hayman JP, Fouqueray
B, Mougenot B and Ronco P: Partial fanconi syndrome induced by
imatinib therapy: A novel cause of urinary phosphate loss. Am J
Kidney Dis. 51:298–301. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Aslam N, Pesacreta M, Bastacky SI, McHale
T, Huston A, Palevsky PM and Weisbord SD: Light chain-associated
Fanconi syndrome with nephrotic-range proteinuria. Am J Kidney Dis.
47(A57): e43–e45. 2006.
|
|
34
|
Koike K, Lida S, Usui M, Matsumoto Y,
Fukami K, Ueda S, Tamaki K, Kato S and Okuda S: Adult-onset acute
tubulointerstitial nephritis and uveitis with Fanconi syndrome.
Case report and review of the literature. Clin Nephrol. 67:255–259.
2007. View
Article : Google Scholar : PubMed/NCBI
|
|
35
|
D'Ythurbide G, Goujard C, Méchaï F, Blanc
A, Charpentier B and Snanoudj R: Fanconi syndrome and nephrogenic
diabetes insipidus associated with didanosine therapy in HIV
infection: A case report and literature review. Nephrol Dial
Transplant. 22:3656–3659. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Izzedine H, Launay-Vacher V and Deray G:
Antiviral drug-induced nephrotoxicity. Am J Kidney Dis. 45:804–817.
2005. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Cihlar T, Lin DC, Pritchard JB, Fuller MD,
Mendel DB and Sweet DH: The antiviral nucleotide analogs cidofovir
and adefovir are novel substrates for human and rat renal organic
anion transporter 1. Mol Pharmacol. 56:570–580. 1999.PubMed/NCBI
|
|
38
|
Ho ES, Lin DC, Mendel DB and Cihlar T:
Cytotoxicity of antiviral nucleotides adefovir and cidofovir is
induced by the expression of human renal organic anion transporter
1. J Am Soc Nephrol. 11:383–393. 2000.PubMed/NCBI
|
|
39
|
Shimizu M, Furusyo N, Ikezaki H, Ogawa E,
Hayashi T, Ihara T, Harada Y, Toyoda K, Murata M and Hayashi J:
Predictors of kidney tubular dysfunction induced by adefovir
treatment for chronic hepatitis B. World J Gastroenterol.
21:2116–2123. 2015.PubMed/NCBI
|
|
40
|
Kearney BP, Ramanathan S, Cheng AK,
Ebrahimi R and Shah J: Systemic and renal pharmacokinetics of
adefovir and tenofovir upon coadministration. J Clin Pharmacol.
45:935–940. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Jung YK, Yeon JE, Choi JH, Kim CH, Jung
ES, Kim JH, Park JJ, Kim JS, Bak YT and Byun KS: Fanconi's syndrome
associated with prolonged adefovir dipivoxil therapy in a hepatitis
B virus patient. Gut Liver. 4:389–393. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ning H, Li K, Li W, Ding G, Xiao E, Mao Z,
Kang Y and Shang J: Adefovir dipivoxil effects on and related
factors of blood phosphorus metabolism in patients with chronic
hepatitis B. Zhonghua ganzangbing zazhi. 23:590–593.
2015.PubMed/NCBI
|
|
43
|
Ha NB, Ha NB, Garcia RT, Trinh HN, Vu AA,
Nguyen HA, Nguyen KK, Levitt BS and Nguyen MH: Renal dysfunction in
chronic hepatitis B patients treated with adefovir dipivoxil.
Hepatology. 50:727–734. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Krag A, Wiest R, Albillos A and Gluud LL:
The window hypothesis: Haemodynamic and non-haemodynamic effects of
beta-blockers improve survival of patients with cirrhosis during a
window in the disease. Gut. 61:967–969. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
García-Compeán D, González-González JA,
Lavalle-González FJ, González-Moreno EI, Maldonado-Garza HJ and
Villarreal-Pérez JZ: The treatment of diabetes mellitus of patients
with chronic liver disease. Ann Hepatol. 14:780–788. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Ge PS and Runyon BA: Treatment of Patients
with Cirrhosis. N Engl J Med. 375:767–777. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Cundy KC: Clinical pharmacokinetics of the
antiviral nucleotide analogues cidofovir and adefovir. Clin
Pharmacokinet. 36:127–143. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Jafari A, Khalili H and Dashti-Khavidaki
S: Tenofovir-induced nephrotoxicity: Incidence, mechanism, risk
factors, prognosis and proposed agents for prevention. Eur J Clin
Pharmacol. 70:1029–1040. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Vassalotti JA, Centor R, Turner BJ, Greer
RC, Choi M and Sequist TD: National Kidney Foundation Kidney
Disease Outcomes Quality Initiative: Practical Approach to
Detection and Management of Chronic Kidney Disease for the Primary
Care Clinician. Am J Med. 129:153–162.e7. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Safaei-Asl A, Enshaei M, Heydarzadeh A and
Maleknejad S: Correlation between cystatin C-based formulas,
Schwartz formula and urinary creatinine clearance for glomerular
filtration rate estimation in children with kidney disease. J Renal
Inj Prev. 5:157–161. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Stevens LA, Coresh J, Greene T and Levey
AS: Assessing kidney function-measured and estimated glomerular
filtration rate. N Engl J Med. 354:2473–2483. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zhang L, Zhang P, Wang F, Zuo L, Zhou Y,
Shi Y, Li G, Jiao S, Liu Z, Liang W and Wang H: Prevalence and
factors associated with CKD: A population study from Beijing. Am J
Kidney Dis. 51:373–384. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Weiner DE, Tighiouart H, Elsayed EF,
Griffith JL, Salem DN and Levey AS: Uric acid and incident kidney
disease in the community. J Am Soc Nephrol. 19:1204–1211. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Zhang L, Wang F, Wang L, Wang W, Liu B,
Liu J, Chen M, He Q, Liao Y, Yu X, et al: Prevalence of chronic
kidney disease in China: A cross-sectional survey. Lancet.
379:815–822. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Chen N, Wang W, Huang Y, Shen P, Pei D, Yu
H, Shi H, Zhang Q, Xu J, Lv Y and Fan Q: Community-based study on
CKD subjects and the associated risk factors. Nephrol Dial
Transplant. 24:2117–2123. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Deng CL, Song XW, Liang HJ, Feng C, Sheng
YJ and Wang MY: Chronic hepatitis B serum promotes apoptotic damage
in human renal tubular cells. World J Gastroenterol. 12:1752–1756.
2006. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Jaryal A, Kumar V and Sharma V: Renal
disease in patients infected with hepatitis B virus. Trop
Gastroenterol. 36:220–228. 2015. View
Article : Google Scholar : PubMed/NCBI
|
|
58
|
Amet S, Bronowicki JP, Thabut D, Zoulim F,
Bourliere M, Mathurin P, de Ledinghen V, Benhamou Y, Larrey DG,
Janus N, et al: Prevalence of renal abnormalities in chronic HBV
infection: The HARPE study. Liver Int. 35:148–155. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
59
|
De Silva ST, Perera RP, Niriella MA,
Pathmeswaran A and de Silva HJ: Frequency, pattern and short-term
outcome of chronic renal dysfunction in patients with cirrhosis: A
prospective study using HRS and ADQI-IAC criteria. Eur J
Gastroenterol Hepatol. 26:1296–1299. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Lim YS, Larson TS, Benson JT, Kamath PS,
Kremers WK, Therneau TM and Kim WR: Serum sodium, renal function,
and survival of patients with end-stage liver disease. J Hepatol.
52:523–528. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Kim SU, Han KH, Nam CM, Park JY, Kim do Y,
Chon CY and Ahn SH: Natural history of hepatitis B virus-related
cirrhotic patients hospitalized to control ascites. J Gastroenterol
Hepatol. 23:1722–1727. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Minde Z, Yimin M, Guangbi Y, JinLin H, Hao
W, Hong R, Yuming W, Xiaqiu Z, Daozhen X, Yagang C, et al: Five
years of treatment with adefovir dipivoxil in Chinese patients with
HBeAg-positive chronic hepatitis B. Liver Int. 32:137–146. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
63
|
George N, Basu G, Mohapatra A, Zachariah
U, Abraham P, Korula A, Varughese S, Jacob CK and Tamilarasi V:
Adefovir nephrotoxicity in a renal allograft recipient. Indian J
Nephrol. 25:180–183. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
64
|
Ho ES, Lin DC, Mendel DB and Cihlar T:
Cytotoxicity of antiviral nucleotides adefovir and cidofovir is
induced by the expression of human renal organic anion transporter
1. J Am Soc Nephrol. 11:383–393. 2000.PubMed/NCBI
|