Introduction
Epileptic seizures are a frequent and limiting
complication in patients with brain cancer that can also occur in
patients with systemic cancer (1–5). In total,
≤50% of patients with brain tumors are expected to have an
epileptic seizure during the course of their disease (4). Seizure management is crucial in primary
and metastatic brain tumors, but may be complicated (6). The main treatments available are
antiepileptic drugs (AEDs). In patients with brain tumors who are
experiencing seizures, the adverse events (AEs) and drug
interactions associated with traditional AEDs are observed more
frequently, compared with in the general population of patients
with epilepsy (6,7). A number of the traditional AEDs interact
with corticosteroids, a universal treatment for brain tumors,
reducing their efficacy (8–10). Certain AEDs also interact with
cytochrome P450, causing the accelerated metabolism of the majority
of chemotherapeutic agents (11,12).
Furthermore, the metabolism of AEDs can be altered by
corticosteroids and chemotherapies, leading to under- and
overdosing (13,14). Therefore, it is essential to identify
an AED that has proven efficacy, low and/or manageable toxicity
with low drug-drug interactions and is easy to titrate.
Lacosamide is a third-generation AED that
selectively enhances the slow inactivation of voltage-gated sodium
channels (15). The drug is approved
in the USA and EU for use as an adjunctive therapy, and in the USA
for use as monotherapy in the treatment of focal epilepsies. The
efficacy and tolerability of lacosamide is well established
(16–18). Lacosamide has a favorable
pharmacokinetic profile with low protein binding and a low
potential for drug-drug interactions (15), and is available as an intravenous
formulation.
To date, few studies have investigated the use of
lacosamide in patients with epileptic seizures due to brain tumors.
However, there is data from routine clinical practice that suggests
promising efficacy and tolerability in this patient population: A
retrospective chart review of 70 patients with brain tumors in the
USA found that lacosamide was well tolerated and demonstrated
effectiveness when used as an add-on AED treatment (19). The aim of the present study was to
investigate the efficacy and tolerability of lacosamide in
controlling epileptic seizures in patients with brain tumors in
Spain.
Materials and methods
Study design and patients
This observational retrospective study was conducted
at six centers in Spain: Hospital Provincial de Castellón
(Castellón), Hospital 12 de Octubre (Madrid), Hospital General de
Castellón (Castellón), Hospital Universitario Rey Juan Carlos
(Madrid), University Hospital La Fe (Valencia) and Hospital
Universitario Fundación Alcorcón (Madrid). Data from patients ≥18
years of age who had received at least one dose of lacosamide for
seizure management were collected. Criteria for study eligibility
depended upon the attending neurologist's criteria, and included at
least one convulsive seizure episode due to a brain tumor and
subsequent placement on lacosamide as an anticonvulsant treatment.
Resistance to previous AEDs was established following routine
clinical practice procedures at each study center. Epilepsy was
diagnosed based on the physician's clinical experience and symptoms
indicative of epilepsy. All patients (or their guardian) provided
written informed consent prior to inclusion in the present
analysis. Patients were excluded if they were receiving any
experimental drug other than lacosamide. The present study was
conducted according to the principles of the Declaration of
Helsinki and the study protocol was reviewed and approved by the
Ethics Committee of Hospital Provincial de Castellón (Castellón,
Spain).
Data collection
Data was collected by reviewing the medical records
of patients and included patient demographics (age, gender,
comorbidities, smoking status), tumor histology, functional status
[as per the Eastern Cooperative Oncology Group criteria (20)], cancer treatment, seizure type, mean
number of seizures and the number of previous and concomitant
AEDs.
Treatment outcomes
The primary objective was to assess the
effectiveness of lacosamide in patients with secondary epileptic
seizures by comparing the number of seizures experienced by the
patient prior to and subsequent to lacosamide treatment (at 3 and 6
months). An additional measure of efficacy was the need to progress
to combination AED therapy [lacosamide + other AED(s)] to achieve
seizure control. The secondary objective was to assess the
tolerability of lacosamide (via reported AEs). The efficacy
population included all patients who were still receiving
lacosamide treatment at 3 months. All patients who received at
least one dose of lacosamide during the study were included in the
safety analysis. A subgroup analysis was performed to determine the
efficacy and safety of lacosamide in patients who experienced a
lack of efficacy from previous AEDs.
Statistical analysis
Statistical analyses for the present study were
performed using the software package SAS version 9.3 (SAS Institute
Inc., Cary, NC, USA). All variables were analyzed by summary
statistical methods. For continuous variables, descriptive
statistics including the arithmetic mean, standard deviation (SD),
median, interquartile range and the minimum and maximum values were
used. Continuous variable data were collected at the baseline, and
at 3 and 6 months. For qualitative variables, absolute and relative
frequency tables were generated. Continuous variables were assessed
using the Student's t-test or the Wilcoxon signed-rank test when
variables did not meet the normality criteria. For categorical
variables, P-values were calculated using the χ2 test,
or the Fisher's exact test when the criteria for the χ2
test were not fulfilled. P<0.05 was considered to indicate a
statistically significant difference.
Considering the patient characteristics, it was
assumed that, following 3 months of lacosamide treatment, 16% of
patients would exhibit a ≥50% decrease in the number of seizures.
Based on this, a total of 47 evaluable patients were required to
achieve an accuracy of 10%, assuming a confidence level of 95%.
Another hypothesis examined in the present study was that
lacosamide would produce a decrease of ~30% in the number of
seizures with an estimated standard deviation of 60%. A sample of
46 patients was required to detect these differences as
statistically significant using a Wilcoxon signed-rank test paired
with a power of 90%. However, only 39 patients were found eligible
for the present study.
Results
Patient characteristics
Between July 2013 and November 2013, data from 39
patients with a brain tumor-associated epileptic seizure who had
received lacosamide as an anticonvulsant were collected and
included in the present analysis. The patients were distributed
across the six medical centers as follows: Hospital Provincial de
Castellón, n=8; Hospital 12 de Octubre, n=11; Hospital General de
Castellón, n=2; Hospital Universitario Rey Juan Carlos, n=6;
Hospital Universitari i Politècnic La Fe, n=4; Hospital
Universitario Fundación Alcorcón, n=8. Patients had a mean age of
54.1±13.8 years and 66.7% were male (Table I). The majority of patients (n=30;
76.9%) presented with a single brain lesion, and multiple brain
lesions were reported in 9 (23.1%) patients. Prior to the
initiation of lacosamide treatment, patients had experienced a mean
of 21.8 seizures (range, 0–200; SD 43.4) in the previous 3 months.
The most common type of seizure experienced prior to treatment was
a simple partial seizure. Of the 39 patients included in the study,
3- and 6-months of data were available for 33 (84.6%) and 26
(66.7%) patients, respectively (Table
II). The reasons for the unavailable 6-month data in 13
patients were: Cancer-associated mortality (n=7; 53.8%); change of
address (n=2); data not recorded (n=2); withdrawal from the study
prior to completion (n=1); and lacosamide treatment withdrawn due
to a lack of effectiveness (n=1). A total of 30 patients were
eligible for the subgroup analysis (Table
I), from which 19 patients (63.3%) completed the study. The
reasons for the unavailability of 6-month data in the subgroup
were: Cancer-associated mortality (n=7), withdrawal from the study
prior to completion (n=3), and lost to follow-up (n=1).
 | Table I.Baseline characteristics and
demographics. |
Table I.
Baseline characteristics and
demographics.
|
Characteristicsa | Main study
(n=39) | Subgroup analysis
(n=30) |
|---|
| Mean ± SD age,
years | 54.1±13.8 | 52.9±13.9 |
| Male, n (%) | 26 (66.7) | 22 (73.3) |
| Smoking status, n
(%) |
|
|
|
Smoker | 8 (21.1)b | 5 (17.2)b |
|
Ex-smoker | 9 (23.7)b | 9 (31.0)b |
| Type of brain lesion,
n (%) |
|
|
|
Single | 30 (76.9) | 22 (73.3) |
|
Multiple | 9 (23.1) | 8 (26.7) |
| Location of brain
tumor, n (%) |
|
|
|
Right | 16
(42.1)b | 13
(44.8)b |
| Left | 16
(42.1)b | 11
(37.9)b |
|
Bilateral | 6 (15.8)b | 5 (17.2)b |
| Specific location of
brain tumor, n (%) |
|
|
|
Lobar | 32
(84.2)b | 25
(86.2)b |
| Callosum
lobar + body | 2 (5.3)b | – |
| Lobar +
ganglia basal | 2 (5.3) | 2 (6.9)b |
|
Other | 1 (2.6) | 1 (3.5)b |
| Lobar +
other | 1 (2.6) | 1
(3.5)b |
| Tumor type, n
(%) |
| – |
|
Primary | 31 (81.6) | 22
(75.9)b |
|
Metastases | 7 (18.4) | 7
(24.1)b |
| Type of primary
tumor in case of metastases, n (%) |
|
|
|
Breast | 1 (2.6) | 1 (14.29) |
|
Lungc | 4 (10.3) | 4 (57.1) |
|
Colon | 1 (2.6) | 1 (14.29) |
|
Melanoma | 1 (2.6) | 1 (14.29) |
| Histological
diagnosis, n (%) |
|
|
|
Astrocytoma | 17 (43.6) | 2 (10.0) |
|
Oligodendroglioma | 8 (20.5) | 5 (25) |
|
Oligoastrocytoma | 1 (2.6) | 1 (5.0) |
|
Ependymoma | 1 (2.6) | 1 (5.0) |
|
Meningioma | 1 (2.6) | 1 (5.0) |
| Type of secondary
seizure, n (%) |
|
|
|
Post-traumatic | 1 (2.6) | 1 (3.3) |
|
Neoplastic | 38 (97.4) | 29 (96.7) |
| Status
epilepticus, n (%) | 10 (25.6) | 7 (23.3) |
|
Convulsive | 7 (17.9) | 5 (71.4) |
|
Non-convulsive | 3 (7.7) | 2 (28.6) |
| Type of seizure, n
(%) |
|
|
|
Generalized | 5 (13.5) | 2
(7.1)d |
| Simple
partial | 22 (59.5) | 18
(64.3)d |
| Complex
partial | 3 (8.1) | 2
(7.1)d |
|
Generalized + simple
partial | 3 (8.1) | 3
(10.7)d |
|
Generalized + complex
partial | 1 (2.7) | 1
(3.6)d |
|
Generalized + simple partial +
complex partial | 3 (8.1) | 2
(7.1)d |
| Previous AED, n
(%) |
|
|
|
Phenytoin | 7e (18.0) | 5 (16.7) |
|
Valproic acid | 11 (28.2) | 8 (26.7) |
|
Carbamazepine | 5 (12.8) | 3 (10.0) |
|
Oxcarbazepine | 2 (5.1) | 2 (6.7) |
|
Levetiracetam | 34 (87.2) | 27 (90.0) |
|
Gabapentin | 1 (2.6) | 1 (3.3) |
|
Topiramate | 1 (2.6) | 1 (3.3) |
|
Eslicarbazepine acetate | 2 (5.1) | 2 (6.7) |
| Comorbidities, n
(%) |
|
|
|
Hypertension | 11 (29.7) | 9 (32.1) |
|
Dyslipidemia | 7 (18.4) | 6 (20.7) |
|
Diabetes | 3 (8.3) | 3 (11.1) |
 | Table II.Histological diagnosis, oncology
treatment and the number of seizures reported in the study patients
prior to and subsequent to lacosamide treatment. |
Table II.
Histological diagnosis, oncology
treatment and the number of seizures reported in the study patients
prior to and subsequent to lacosamide treatment.
| Case no. | Histological
diagnosis | Oncology
treatment | Seizures of the 3
months prior to treatment, n | Seizures during
first 3 months of treatment, n | Seizures during
months 3–6 of treatment, n |
|---|
| 1 | Glioblastoma | – | 2 | 0 | – |
| 2 | Glioblastoma | Other | 0 | 0 | 0 |
| 3 | Pulmonary
neuroendocrine tumor | Radiotherapy | 2 | 0 | – |
| 4 | Non-small cell lung
cancer | – | 3 | – | – |
| 5 | Pulmonary
adenocarcinoma | Other | 3 | 5 | – |
| 6 | Melanoma | – | 3 | – | – |
| 7 | Glioblastoma | – | 3 | 0 | – |
| 8 | Glioblastoma | – | 3 | 0 | – |
| 9 | Oligodendroglioma
grade II | Chemotherapy | 7 | 60 | 58 |
| 10 | Glioblastoma | – | 12 | 4 | 8 |
| 11 | Oligodendroglioma
grade III | Chemotherapy | 1 | 0 | 1 |
| 12 | – | – | 2 | 0 | 0 |
| 13 | Glioblastoma | Other | 27 | 3 | – |
| 14 | Astrocytoma grade
II | Radiotherapy | 93 | 0 | 6 |
| 15 | Glioblastoma | – | 0 | 0 | 0 |
| 16 | Astrocytoma grade
II | Other | 6 | 0 | – |
| 17 | Glioblastoma | Other | 34 | 5 | – |
| 18 | Oligodendroglioma
grade III | Chemotherapy | 0 | 0 | 1 |
| 19 | Glioblastoma | – | 0 | 0 | 0 |
| 20 | High grade
glioma | – | 90 | 0 | 0 |
| 21 | – | – | 1 | 5 | 2 |
| 22 |
Oligodendroglioma | Radiotherapy,
Chemotherapy | 1 | 1 | 4 |
| 23 | Frontal parietal
oligodendroglioma | Chemotherapy | – | – | 6 |
| 24 | Oligodendroglioma
IL 1P 19Q | Chemotherapy | 3 | 0 | 0 |
| 25 | Infiltrating ductal
breast cancer | Radiotherapy |
| 3 | 0 |
| 26 | Non-small cell lung
cancer | Radiotherapy,
Palliative care | 2 | – | – |
| 27 | Right frontal
glioblastoma | – | – | – | – |
| 28 | Ependimoma | Other | 12 | 5 | – |
| 29 | Glioma grade
II | Radiotherapy | 200 | 12 | 12 |
| 30 | – | Radiotherapy,
Chemotherapy | – | – | – |
| 31 | – | – | 4 | 0 | 0 |
| 32 | Astrocytoma grade
II | Other | 63 | 0 | 12 |
| 33 | Glioblastoma | – | 3 | 2 | 0 |
| 34 | Oligodendroglioma
grade II | Other | 130 | 100 | 100 |
| 35 | Oligodendroglioma
grade II | Other | 15 | 1 | 0 |
| 36 | Oligoastrocytoma
grade II | – | 4 | 0 | 0 |
| 37 | Colon | Palliative
care | 3 | 0 | 0 |
| 38 | Glioblastoma | – | 20 | 13 | – |
| 39 | Meningioma | Other | 10 | 4 | 15 |
Lacosamide treatment
The main reason for the initiation of lacosamide
treatment was the inefficacy of other prior AEDs (76.9% of
patients), followed by a lack of tolerance to previous AEDs
(12.8%). At the initiation of treatment, patients received a mean
dose of 138.5±68.3 mg/day of lacosamide. The majority (66.7%) of
patients initiated treatment at a dose of 100 mg/day. The most
commonly used AEDs at the initiation of lacosamide treatment were
levetiracetam (84.6% of patients) and valproic acid (20.5%).
While receiving lacosamide, nine patients modified
their lacosamide treatment; seven due to a lack of efficacy and two
due to AEs. Of these, lacosamide was discontinued in one patient
due to dermal toxicity, which improved subsequent to the
discontinuation of lacosamide. The remaining lacosamide
modifications were an increase in dose. Two patients reported a
lack of efficacy with lacosamide and one other patient reported
unacceptable AEs; however, the dose of lacosamide those patients
received remained unchanged.
In the subgroup analysis, the mean dose of
lacosamide was 123.3±55.29 mg/day, with the majority of patients
(70%) receiving an initial dose of 100 mg/day. The most frequently
used AEDs with lacosamide in the subgroup were levetiracetam
(93.33%) and valproic acid (23.33%).
Concomitant medications
Table III depicts
the concomitant treatments received by the patients during the
study period. The most common concomitant AED administered during
the present study with lacosamide treatment was levetiracetam
(94.3% of patients). A total of 8 patients modified their
concomitant AEDs over the study period. A total of 4 patients
reduced their levetiracetam dose; 1 patient discontinued
carbamazepine and reduced their dose of valproic acid; 1 patient
initiated treatment with eslicarbazepine acetate and carbamazepine
(which was later discontinued); 1 patient initiated treatment with
valproic acid; 1 patient reduced the dose of valproic acid.
 | Table III.Concomitant treatments received
during the 6 months of lacosamide treatment. |
Table III.
Concomitant treatments received
during the 6 months of lacosamide treatment.
| Treatment | Main study, n
(%) | Subgroup analysis,
n (%) |
|---|
| Surgery only | 7 (17.9) | – |
| Radiotherapy +
chemotherapya | 6 (15.4) | 5 (16.7) |
| Chemotherapy
onlya | 11 (28.2) | 13 (43.3) |
| Radiotherapy
onlya | 6 (15.4) | 3 (10.0) |
| None | 16 (41.0) | 9 (30.0) |
| Chemotherapies
received during the study |
|
|
|
Temozolomide | 19 (48.7) | – |
|
Bevacizumab | 5 (12.8) | – |
|
Dacomitinib | 3 (7.7) | – |
|
Carboplatin | 1 (2.6) | – |
|
Carmustine | 1 (2.6) | – |
|
Fotemustine | 1 (2.6) | – |
|
Irinotecan | 1 (2.6) | – |
|
Lomustine | 1 (2.6) | – |
|
Paclitaxel | 1 (2.6) | – |
|
Pamidronic acid | 1 (2.6) | – |
|
Procarbazine | 1 (2.6) | – |
|
Vincristine | 1 (2.6) | – |
| Corticosteroids
received during the study |
| – |
|
Dexamethasone | 16 (41.0) | – |
|
Prednisolone | 1 (2.6) | – |
|
Fluticasone | 1 (2.6) | – |
| Lacosamide
monotherapy | 4 (10.3) | – |
| Lacosamide + one
AED | 20 (51.3) | – |
|
Lacosamide +
levetiracetam | 19 (48.7) | – |
| Lacosamide +
carbamazepine | 1 (2.6) | – |
|
Lacosamide + two AEDs | 13 (33.3) | – |
|
Lacosamide +
levetiracetam | 12 (30.8) | – |
|
Lacosamide + valproic
acid | 8 (20.5) | – |
|
Lacosamide + phenytoin | 3 (7.7) | – |
|
Lacosamide +
carbamazepine | 2 (5.1) | – |
|
Lacosamide + eslicarbazepine
acetate | 1 (2.6) | – |
| Lacosamide + four
AEDs | 2 (5.1) | – |
A total of 18 patients also received concomitant
treatment with a corticosteroid during the study period (Table III). Patients who received
dexamethasone modified their concomitant treatment: 5 patients
reduced their dose and 1 patient increased the dose. No additional
modifications to the concomitant corticosteroid treatment were made
during the study period. No drug-interaction between lacosamide and
radiotherapy, or lacosamide and chemotherapy was observed.
In the subgroup analysis, all patients received
lacosamide in combination with another drug. The numbers of
concomitant treatments used were 1, 2, 4 and 6 in 60, 33.33, 3.33
and 3.33% of patients, respectively. A total of 29 patients (96.7%)
received levetiracetam and lacosamide. Table III details the anticancer therapy
received by each of the patients included in the subgroup analysis.
No interaction between lacosamide and chemotherapy or radiotherapy
was reported.
Treatment effectiveness
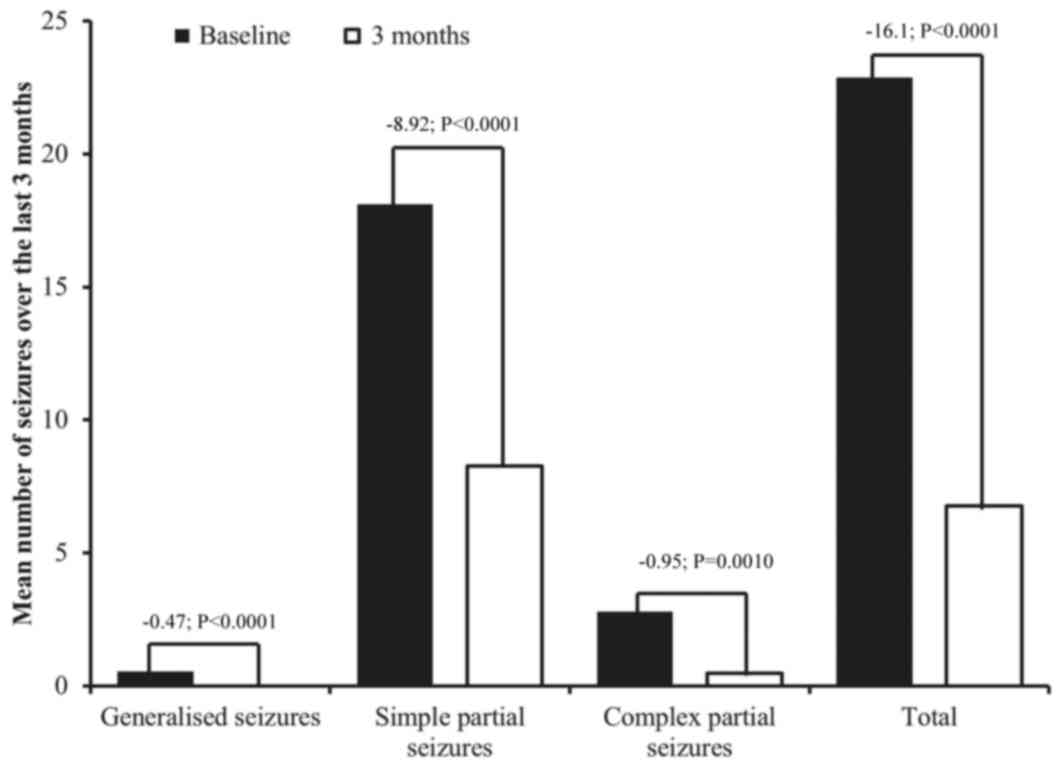
The number of seizures from the initiation of
treatment to 3 months [mean 6.8, SD 19.8; median 0.0 (range
0.0–100.0)] was significantly lower compared with the number of
seizures in the 3 months prior to lacosamide initiation [mean 22.9,
SD 44.5; P<0.0001; Fig. 1; median
3.0 (range 0.0–200.0)], for the 33 patients with data available
after 3 months of treatment. The majority of seizures experienced
in the first 3 months of lacosamide treatment were simple partial
seizures followed by complex partial seizures. No patient
experienced generalized seizures in the first 3 months of
lacosamide treatment (Fig. 1).
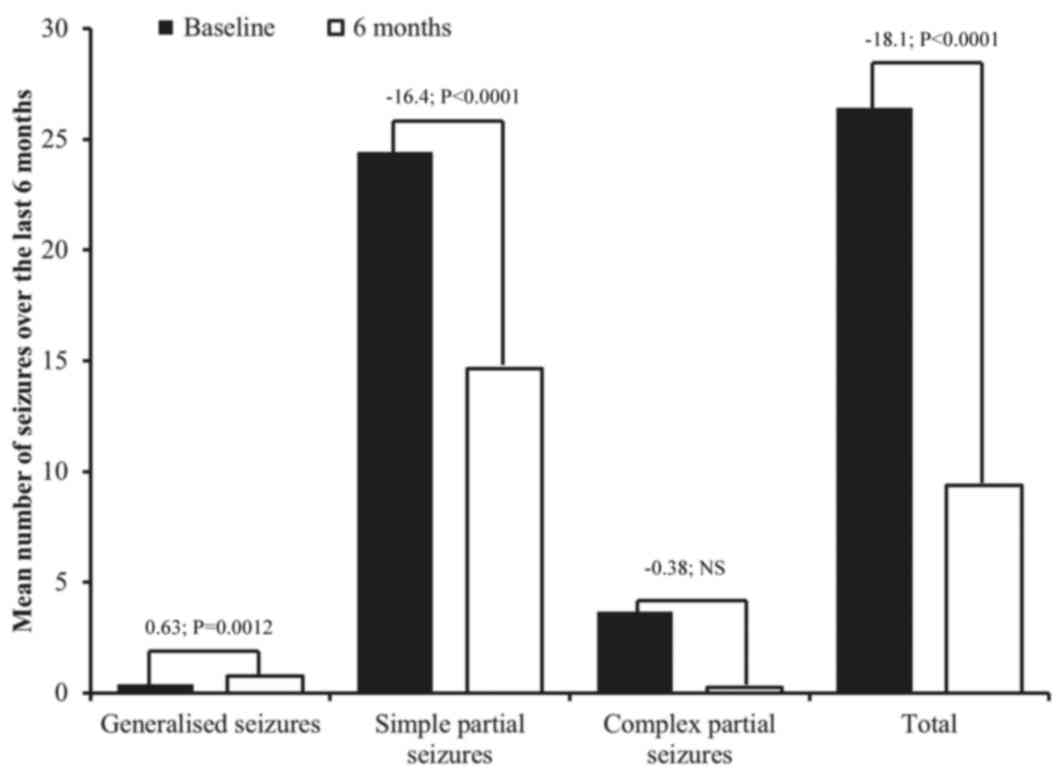
Similar results were observed after 6 months. Compared with the 6
months prior to lacosamide initiation, there was a significant
reduction in total seizures at 6 months [prior to treatment, mean
26.4, SD 50.4, median 4.0 (range 0–200); at 6 months, mean 9.4, SD
22.8, median 0.5 (range 0–100); difference in means P<0.0001;
Fig. 2].
Subsequent to 3 and 6 months of lacosamide
treatment, the majority of patients exhibited a reduction in the
number of seizures they experienced (72.7 and 56.5% of patients,
respectively), with 87.5 and 84.6% of patients experiencing a
reduction in seizure frequency of ≥50%. Furthermore, at the end of
the study period, 14 patients (53.8%) reported having no seizures
during treatment.
At 3 months, only 5 patients (15.2%) demonstrated no
reduction in their seizure frequency following lacosamide
initiation, and 4 patients (12.1%) had an increase in seizure
frequency. Of these 9 patients who did not respond to lacosamide
therapy, 3 were revealed to have cancer progression. Overall, 10
patients were considered to have disease progression during the
study, the 7 patients who succumbed to the cancer and
aforementioned 3 patients.
Finally, during the study, 10 patients experienced
status epilepticus (7 convulsive and 3 non-convulsive) and were
treated with levetiracetam (n=6), lacosamide (n=4), valproic acid
(n=6) phenytoin (n=3) and clonazepam (n=1).
In the subgroup analysis, the mean number of
seizures following 3 months of lacosamide treatment (mean 8.9, SD
22.5) was significantly lower compared with the mean number of
seizures at the baseline (mean 27.4, SD 49.1; P<0001). The
median number of seizures at 3 months was 2.0 (range 0.0–100.0) vs.
6.0 (range 0.0–200.0) at baseline. The majority of seizures
experienced subsequent to 3 months of treatment were partial
seizures followed by partial complex seizures. No generalized
seizures were reported at 3 months subsequent to lacosamide
treatment. After 3 months of lacosamide treatment, 80% of patients
exhibited a decrease in the number of seizures, with >50% of
reduction observed in 85% of these patients. Similarly, after 6
months of lacosamide treatment, the number of seizures (mean 12.2,
SD 26.6) was lower compared with those reported at baseline (mean
32.9, SD 56.8; P=0.0004). The median number of seizures at 6 months
was 0.0 (range 0.0–100.0) vs. 6.5 (range 0.0–200.0) at baseline. In
addition, after 6 months of treatment, 68.75% of patients exhibited
a reduction in the number of seizures, with 81.82% of these
patients reporting a >50% reduction.
Safety
Lacosamide was generally well tolerated. Of the 25
patients who had complete safety data available at the 6-month
follow-up, 3 patients (12%) reported an AE, including dizziness
(n=1), asthenia (n=2), instability (n=1), irritability (n=1) and
leg edema (n=1). None of these events were considered severe. No
neurocognitive deficits, cardiac adverse effects or liver function
test abnormalities were reported with the lacosamide treatment.
Furthermore, no AEs were reported that were considered to be
associated with anticancer-treatment. The subgroup analysis also
demonstrated that lacosamide treatment was well tolerated over the
6-month treatment period.
Discussion
This non-interventional, observational retrospective
analysis investigated the effectiveness and safety of lacosamide
for the treatment of epileptic seizures due to brain tumors. The
findings demonstrated that lacosamide significantly reduced the
total number of seizures and was well tolerated in this patient
population.
In the present study, lacosamide significantly
reduced the mean number of seizures experienced over a 6-month
period by a mean of 18.1 events. A large majority of patients in
the current study had a reduction in the number of seizures they
experienced, with 84.6% of patients experiencing a reduction in
seizure frequency of ≥50% after 6 months of lacosamide treatment.
Furthermore, at the end of the study period, 14 patients (40.0%)
reported having no seizures whilst receiving lacosamide treatment.
These efficacy results are consistent with those of clinical trials
of lacosamide in patients with epilepsy, which previously
demonstrated that lacosamide effectively reduces seizure frequency
compared with a placebo (16–18,21). It is
noteworthy that the 50% responder rate observed in this study was
larger compared with those reported in the major phase III trials
of lacosamide (33–41.2% of patients), despite larger doses of
lacosamide administered in the phase III trials. However, it is
important to note that the patients included in the phase III
trials were more refractory to treatment (with a mean duration of
epilepsy of 23 years) compared with the patients included in the
present study.
In the present study, lacosamide was generally well
tolerated, with only 3 patients (12%) reporting an AE over the
6-month treatment period. The AEs reported in this study included
dizziness, asthenia, instability, irritability and leg edema. These
results are consistent with clinical trials of lacosamide in
patients with epilepsy, which indicated that the most common AEs
associated with lacosamide involve the nervous or gastrointestinal
systems, and include dizziness, headache, fatigue, nausea and
diplopia (16–18,22,23). The
present study had a lower frequency of AEs compared with that
reported in clinical trials (range, 70–80% of patients who received
200 or 400 mg of lacosamide/day) (22,23);
however, the smaller number of patients may explain this. Notably,
none of the neurocognitive deficits typically associated with the
use of AEDs in patients with brain tumors were reported (24). Furthermore, no cardiac adverse effects
or liver function test abnormalities were reported following
lacosamide treatment.
To the best of our knowledge, only a limited number
of other studies have investigated the efficacy and safety of
lacosamide in a similar patient population (19,25). The
findings of the present study are in accordance with the results of
the retrospective analysis conducted by Saria et al
(19), which investigated the
efficacy and safety of lacosamide as an add-on AED in 70 patients
with primary brain tumors who received lacosamide for seizure
activity. Similar to the present study, Saria et al
(19) demonstrated that lacosamide
was effective and reduced the frequency of seizures in ~66% of
patients; lacosamide was additionally demonstrated to be well
tolerated, with 77% of patients in the study reporting no toxicity
(19). Maschio et al (25) published a preliminary report of the
efficacy and tolerability of lacosamide as an add-on therapy in 14
patients with brain tumor-associated epilepsy. The results from the
present study were consistent with the findings of Maschio et
al, which indicated that lacosamide was effective and well
tolerated (25). Therefore, in the
present patient population, lacosamide may provide a valid
alternative to other AEDs as an add-on therapy.
In patients with epilepsy due to brain tumors, the
ideal AED would provide complete seizure control while avoiding
significant AEs and drug-drug interactions, particularly as
patients with brain tumors are often receiving treatments for their
cancer and experiencing chemotherapy-associated AEs (2). However, there is a high risk of
drug-drug interactions between anticancer agents and AEDs,
particularly when using traditional enzyme-inducing AEDs (26). However, since the advent of second-
and third-generation AEDs, a number of newer AEDs have demonstrated
reduced drug-drug interactions, including lacosamide, gabapentin,
levetiracetam and pregabalin (27).
Based on the reported interactions of lacosamide with AEDs and
other drugs, and its pharmacokinetic profile, we hypothesize that
lacosamide may be an ideal anticonvulsant for patients with brain
tumors. The results of the present study support this hypothesis,
particularly as no serious drug interactions were observed during
the follow-up period.
There are a number of restrictions to the present
study, including the inherent limitations of the retrospective,
non-controlled study design. Due to the small sample size, the
confidence intervals around the point estimates obtained were
correspondingly large. Also, the effect of tumor status and
concomitant tumor treatment on the reduction in the number of
seizures during 6 months of lacosamide treatment cannot be ruled
out. As aforementioned, the efficacy and tolerability of lacosamide
has been well established in a large group of patients with
partial-onset seizures enrolled in three, randomized, multicenter
placebo-controlled clinical trials (16–18).
However, little information on the use of lacosamide in patients
with brain cancer is available; thus, the present study was
designed to clarify the efficacy and safety of lacosamide in this
group of patients. While this is a retrospective study without a
control group, data obtained in this analysis provides useful
information on the efficacy and tolerability of lacosamide in
patients with a high level of morbidity that are receiving multiple
drugs and are thus susceptible to drug-interaction
complications.
In conclusion, epilepsy is an important risk factor
for long-term disability in patients with brain tumors. The present
retrospective analysis suggests that lacosamide is an effective and
well-tolerated treatment for patients with brain tumors who
experience seizures, and additional prospective studies with a
larger patient population and randomized trial design are
warranted.
Acknowledgements
The authors would like to thank Ms. Ana Gentil
(Pertica, Castellón, Spain) for performing the statistical analysis
and Ms. Simone Boniface (Springer Healthcare Communications,
Auckland, New Zealand), for assistance with medical writing. The
present study was developed with the support of the Foundation of
the Provincial Hospital of Castellón.
Glossary
Abbreviations
Abbreviations:
|
AEs
|
adverse events
|
|
AEDs
|
antiepileptic drugs
|
|
SD
|
standard deviation
|
References
|
1
|
Hildebrand J, Lecaille C, Perennes J and
Delattre JY: Epileptic seizures during follow-up of patients
treated for primary brain tumors. Neurology. 65:212–215. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rossetti AO and Stupp R: Epilepsy in brain
tumor patients. Curr Opin Neurol. 23:603–609. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Singh G, Rees JH and Sander JW: Seizures
and epilepsy in oncological practice: Causes, course, mechanisms
and treatment. J Neurol Neurosurg Psychiatry. 78:342–349. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
4
|
van Breemen MS, Wilms EB and Vecht CJ:
Epilepsy in patients with brain tumours: Epidemiology, mechanisms,
and management. Lancet Neurol. 6:421–430. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rudà R and Soffietti R: What is new in the
management of epilepsy in gliomas? Curr Treat Options Neurol.
17:3512015. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Schiff D, Lee EQ, Nayak L, Norden AD,
Reardon DA and Wen PY: Medical management of brain tumors and the
sequelae of treatment. Neuro Oncol. 17:488–504. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Smith KC: The management of seizures in
brain tumor patients. J Neurosci Nurs. 42:28–39. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Haque N, Thrasher K, Knowles EE Jr, Werk
HC Jr and Sholiton LJ: Studies on dexamethasone metabolism in man:
Effect of diphenylhydantoin. J Clin Endocrinol Metab. 34:44–50.
1972. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Gambertoglio JG, Holford NH, Kapusnik JE,
Nishikawa R, Saltiel M, Stanik-Lizak P, Birnbaum JL, Hau T and
Amend WJ Jr: Disposition of total and unbound prednisolone in renal
transplant patients receiving anticonvulsants. Kidney Int.
25:119–123. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Chalk JB, Ridgeway K, Brophy T, Yelland JD
and Eadie MJ: Phenytoin impairs the bioavailability of
dexamethasone in neurological and neurosurgical patients. J Neurol
Neurosurg Psychiatry. 47:1087–1090. 1984. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Warren RD and Bender RA: Drug interactions
with antineoplastic agents. Cancer Treat Rep. 61:1231–1241.
1977.PubMed/NCBI
|
|
12
|
Rudà R, Bello L, Duffau H and Soffietti R:
Seizures in low-grade gliomas: Natural history, pathogenesis, and
outcome after treatments. Neuro Oncol. 14 Suppl 4:iv55–iv64. 2012.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Fincham RW and Schottelius DD: Decreased
phenytoin levels in antineoplastic therapy. Ther Drug Monit.
1:277–283. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Perucca E and Pisani F: Pharmacokinetics
and interactions of the new antiepileptic drugs. Epilepsy Res
Suppl. 3:79–88. 1991.PubMed/NCBI
|
|
15
|
Beydoun A, D'Souza J, Hebert D and Doty P:
Lacosamide: Pharmacology, mechanisms of action and pooled efficacy
and safety data in partial-onset seizures. Expert Rev Neurother.
9:33–42. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Ben-Menachem E, Biton V, Jatuzis D,
Abou-Khalil B, Doty P and Rudd GD: Efficacy and safety of oral
lacosamide as adjunctive therapy in adults with partial-onset
seizures. Epilepsia. 48:1308–1317. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Chung S, Sperling MR, Biton V, Krauss G,
Hebert D, Rudd GD and Doty P; SP754 Study Group, : Lacosamide as
adjunctive therapy for partial-onset seizures: A randomized
controlled trial. Epilepsia. 51:958–967. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Halász P, Kälviäinen R,
Mazurkiewicz-Beldzińska M, Rosenow F, Doty P, Hebert D and Sullivan
T; SP755 Study Group, : Adjunctive lacosamide for partial-onset
seizures: Efficacy and safety results from a randomized controlled
trial. Epilepsia. 50:443–453. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Saria MG, Corle C, Hu J, Rudnick JD,
Phuphanich S, Mrugala MM, Crew LK, Bota DA, Dan Fu B, Kim RY, et
al: Retrospective analysis of the tolerability and activity of
lacosamide in patients with brain tumors: Clinical article. J
Neurosurg. 118:1183–1187. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Oken MM, Creech RH, Tormey DC, Horton J,
Davis TE, McFadden ET and Carbone PP: Toxicity and response
criteria of the eastern cooperative oncology group. Am J Clin
Oncol. 5:649–655. 1982. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Pellerino A, Magistrello M, Franchino F,
Nicolotto E, Soffietti R and Rudà R: Efficacy and tolerability of
lacosamide in patients with glioma: A prospective study. Eur J
Neurol. 22 Suppl 1:732015.
|
|
22
|
European Medicines Agency, . Lacosamide
(Vimpat 50 mg tablets): Summary of product characteristics.
http://www.ema.europa.eu/docs/en_GB/document_library/EPAR_-_Product_Information/human/000863/WC500050338.pdf[Accessed
2016 May 20].
|
|
23
|
US Food and Drug Administration, . Vimpat
(lacosamide) tablets and injection: US prescribing information.
http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/022253lbl.pdf2016
May 20;
|
|
24
|
Klein M, Engelberts NH, van der Ploeg HM,
Kasteleijn-Nolst Trenité DG, Aaronson NK, Taphoorn MJ, Baaijen H,
Vandertop WP, Muller M, Postma TJ and Heimans JJ: Epilepsy in
low-grade gliomas: The impact on cognitive function and quality of
life. Ann Neurol. 54:514–520. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Maschio M, Dinapoli L, Mingoia M, Sperati
F, Pace A, Pompili A, Carapella CM, Vidiri A and Muti P: Lacosamide
as add-on in brain tumor-related epilepsy: Preliminary report on
efficacy and tolerability. J Neurol. 258:2100–2104. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Yap KY, Chui WK and Chan A: Drug
interactions between chemotherapeutic regimens and antiepileptics.
Clin Ther. 30:1385–1407. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Landmark C Johannessen and Patsalos PN:
Drug interactions involving the new second- and third-generation
antiepileptic drugs. Expert Rev Neurother. 10:119–140. 2010.
View Article : Google Scholar : PubMed/NCBI
|