Introduction
Khat is an herb that is used by millions of people
worldwide for its psychostimulatory effects, primarily in Africa
and the Middle East. The alkaloid in khat is the major ingredient
that can be efficiently extracted and absorbed orally (1). Khat contains numerous pharmacologically
active compounds (2). Cathinone is a
major alkaloid compound in fresh khat leaves, which is relatively
unstable and rapidly metabolized into cathine (norpseudoephedrine)
and norephedrine (1). Furthermore,
other alkaloids, including phenylpentenylamines and cathedulines
may exert pharmacological effects (3,4). The
majority of their pharmacological effects have been suggested to be
mediated by releasing biogenic amines through preferential binding
with the norepinephrine receptor, partially through binding with
dopamine and 5-hydroxytryptamine receptors (5).
Khat chewing has been reported to impact the sexual
behavior of males and females. Studies have shown that khat
possesses aphrodisiac activities (6–8) and may be
used to cure premature ejaculation (9). In addition, khat has been reported to be
a psychostimulant (10). Medicinal
uses of Khat for the treatment of depression, hunger, fatigue,
obesity and gastric ulcers have also been previously reported
(11). Khat can affect the
cardiovascular, digestive, endocrine, hepatobiliary, respiratory
and genitourinary systems (12).
Previous studies have indicated that khat is associated with oral
cancer development (13) and hepatic
cell apoptosis (14). To the best of
our knowledge, no studies have investigated the effects of khat
consumption on breast cancer.
Programmed cell death may occur via apoptosis,
necrosis or excessive autophagy, of which, mitochondria serve an
essential role in its regulation (15,16). The
B-cell lymphoma 2 (Bcl-2) family is involved in
mitochondria-mediated cell death by affecting the stability of the
outer mitochondrial membrane (17).
Anti-apoptotic Bcl-2 and Bax have been demonstrated to be
associated with spontaneous apoptosis in acute myeloid leukemia
cells in vitro (18). A
previous study has suggested the use of the Bax to Bcl-2 ratio in
patient cells to predict clinical response and outcome (19). Mitochondria participate in cell death
mechanisms through the release of apoptogenic proteins into the
cytosol and generation of excess reactive oxidative species (ROS).
The mitochondrial respiratory chain is a major source of cellular
ROS and therefore, represents a target for the effects of ROS
production (20).
Khat-induced hepatocyte apoptosis is primarily
regulated through the sustained activation of the c-Jun
NH2-terminal kinase (JNK) signaling pathway and partially through
the extracellular signal-regulated kinase (ERK) signaling cascade
(14). JNK and ERK are part of the
mitogen-activated protein kinase (MAPK) signaling pathways. The aim
of the present study was to investigate the effects of khat on the
viability and apoptosis of breast cancer cell MDA-MB-231.
Materials and methods
Khat extraction
Khat plants were purchased from a local Yemeni
market, Sana'a, Yemen. Fresh khat leaves with soft stems were
collected in the summer, weighed, washed three times with distilled
water and dried for three days in a clean, and dry room away from
sunlight. Subsequently, the leaves were weighed, packed in a tight
foil packet and stored at 4°C. Dried khat leaves (100 g) were
chopped into sections (5 mm) and dissolved in 100 ml 95% ethanol,
centrifuged at 5,031 × g for 5 min at room temperature. The
supernatant was filtered using filter paper. Ethanol (100 ml) was
added to the remaining leaves, and the procedure was repeated. The
extracted ethanol khat suspension was concentrated using a rotary
evaporator (LabTech, Inc., Hopkinton, MA, USA) at 30°C at a speed
of 0.44 × g until 70% of the ethanol solvent had evaporated. The
viscous solution was diluted with 100 ml distilled water and
stirred at 201 × g for 60 min at room temperature. The filtered
liquid was stored at −70°C for 24 h and subsequently dried using a
lyophilization apparatus (Lyophilization Technology, Inc.,
Warminster, PA, USA). Generally, 100 g dried leaves yielded 8 g
khat extract powder. The major alkaloids in khat leaves are
cathine, cathinone and norephedrine. The mean concentrations of
cathinone, cathine and norephedrine in fresh khat leaves are 0.95,
1.98, and 0.54 mg/g, respectively (21–23). The
lyophilized khat extracts were dissolved in Hank's balanced salt
solution (HyClone; GE Healthcare Life Sciences, Logan, UT, USA
without Ca2+ or Mg2+ to achieve a final
concentration of 200 µg/ml and sterilized using a filter with a
pore size of 0.2 µm.
Cell culture and treatment
MDA-MB-231 cells were cultured in Dulbecco's
modified Eagle's medium (HyClone; GE Healthcare Life Sciences) with
10% fetal bovine serum (Zhejiang Tianhang Biotechnology Co., Ltd.,
Hangzhou, Zhejiang, China) and maintained in a humidified incubator
at 37°C with 5% CO2. When 60–70% confluence was
achieved, the cells were treated with 400 µg/ml khat and left to
culture for 4, 8, 16, or 24 h at 37°C. The cells in the control
group were not treated with khat.
Analysis of cell viability
The cells were seeded into 6-well plates at 60–70%
confluence, overnight at 37°C. The cells were subsequently exposed
to various concentrations of Khat (20, 200, 300 and 400 µg/ml) for
4, 8, 16 and 24 h. Cells were digested using 0.25% pancreatic
enzyme liquid (without EDTA) and the resultant suspension was
collected and centrifuged at 168 × g for 5 min at room temperature.
Then the cells were stained with 0.2% trypan blue or 5 min at room
temperature prior to evaluation under an inverted light microscope.
The cells with blue-labeled nuclei were considered dead.
Annexin V/propidium iodide (PI)
staining assay
Apoptosis was assessed using an Annexin
V-fluorescein isothiocyanate (FITC) apoptosis detection kit
(Jiangsu KEYGEN BIOTECH Co., Ltd., Nanjing, Jiangsu, China)
according to the manufacturer's protocol. Following drug treatment,
cells in the 6-well plates were digested using 0.25% pancreatic
enzyme liquid (without EDTA), suspension was collected and
centrifuged at 168 × g for 5 min at room temperature, then washed
with phosphate-buffered saline (PBS) twice and resuspended in 500
µl of staining buffer with 5 µl FITC-conjugated Annexin V and 5 µl
PI staining solution. The cells were incubated on ice for 30 min
and analyzed using a FACSCalibur flow cytometer (BD Biosciences,
Franklin Lakes, NJ, USA) within 1 h. ModFit V3.2 software (BD
Biosciences) was used to analyze the cell apoptosis rate. Rates of
basal cell apoptosis and necrosis were determined in the untreated
control group. The rates of cell apoptosis were determined
following ≥3 independent experiments and each experiment was
performed in triplicate.
Staining of apoptotic cells with
Hoechst 33258
The cells were treated with khat and fixed in 4%
formaldehyde containing Hoechst 33258 (10 µg/ml). Following
incubation with 400 µg/ml khat for different times (0, 4, 8, 16 and
24 h) at 37°C, nuclear morphology was examined at ×200
magnification with an incident light fluorescent microscope to
assess apoptotic features. The apoptotic cells were determined as
the fraction of intensely stained, condensed and fragmented
nuclei.
Transmission electron microscopy
Cells underwent a 16-h incubation with 400 µg/ml
khat and were observed by transmission electron microscopy. The
cells were fixed in 0.1 M sodium cacodylate buffer (pH 7.4) with 2%
glutaraldehyde. The cells were rinsed with sodium cacodylate buffer
and post-fixed in 1% osmium tetroxide. The cells were dehydrated
with graded ethanol (50, 70 and 90% for 10 min each) and embedded
in epoxy resin. The sections were double-stained with 3% uranyl
acetate and lead citrate. The cells were examined under a JEOL 1230
transmission electron microscope (JEOL, Ltd., Tokyo, Japan), and
micrographs were produced using an Agfa Arcus II scanner (Agfa
Corporation, Shanghai, China) and Adobe Photoshop software (version
7.0.1; Adobe Systems Europe, Ltd., Maidenhead, UK).
Western blot analysis
The cells were harvested and lysed on ice for 30 min
in radioimmunoprecipitation buffer (50 mM Tris-HCl, pH 7.4, 150 mM
NaCl, 1% Nonidet P-40, 0.25% sodium deoxycholate, 50 mM NaF, 1 mM
Na3VO4, 5 mM sodium pyrophosphate and a protease inhibitor tablet
[Jiangsu Keygen Biotech Co., Ltd.]). The cell lysates were
centrifuged at 39,514 × g for 10 min at 4°C, and the supernatants
were used for western blotting. The total protein concentration was
determined using the BCA Protein assay reagent (Pierce; Thermo
Fisher Scientific, Inc., Waltham, MA, USA). The lysates were
denatured at 100°C following the addition of SDS loading buffer.
The proteins were separated on 4–20% SDS-PAGE gels and subsequently
transferred to nitrocellulose blotting membranes. Following
blocking of the membranes with 5% non-fat dry milk in 1X TBST for
>1 h at room temperature, the membranes were incubated with
1:1,000 diluted primary antibodies [p-JNK (cat. no. sc-135642), JNK
(cat. no. sc-1648), p-ERK (cat. no. sc-81492), ERK (cat. no.
sc-271270), Bcl-2 (cat. no. sc-7382), Bax (cat. no. sc-7480) and
Caspase-9 (cat. no. sc-73548); all Santa Cruz Biotechnology, Inc.,
Santa Cruz, California, USA] diluted in 5% non-fat dry milk in 1X
TBST for >12 h at 4°C. Membrane-bound primary antibodies were
detected with 1:3,000 diluted secondary antibodies (affinity
purified goat anti-rabbit IgG (cat. no. GB23303), goat anti-mouse
IgG (cat. no. GB23301), Servicebio Biotechnology, Ltd., Wuhan,
Huber, China) conjugated to horseradish peroxidase for 1 h at room
temperature. The blots were visualized using the ECL
chemiluminescence reagent kit (Servicebio Biotechnology, Ltd.,
Wuhan, Hubei, China), and analyzed using the ChemiDoc XRS gel
imaging device (Bio-Rad Laboratories, Inc., Hercules, CA, USA).
Measurement of ROS production
The production of ROS was measured using flow
cytometry with dichlorofluorescein-diacetate (DCFH-DA) as
previously described (24). The cells
were treated with khat (400 µg/ml) for 4, 8, 16 and 24 h,
dissociated with trypsin-EDTA, washed twice with cold PBS, and
suspended in PBS (1×106 cells/ml). The cell suspension
(500 µl) was transferred into a tube and incubated with DCFH-DA at
a final concentration of 5 µM for 30 min at 37°C. ROS production
was assessed by determining the DCF fluorescence intensity from
1×104 cells by flow cytometry.
Statistical analysis
Data are presented as the mean ± standard deviation
and analyzed using the SPSS software (version 17.0; SPSS Inc.,
Chicago, IL, USA). One-way analysis of variance (followed by
Student-Newman-Keuls post-hoc test) was used to analyze the
significance of differences among groups. P<0.05 was considered
to indicate a statistically significant difference.
Results
Effect of Khat treatment on viability
of MDA-MB-231 cells
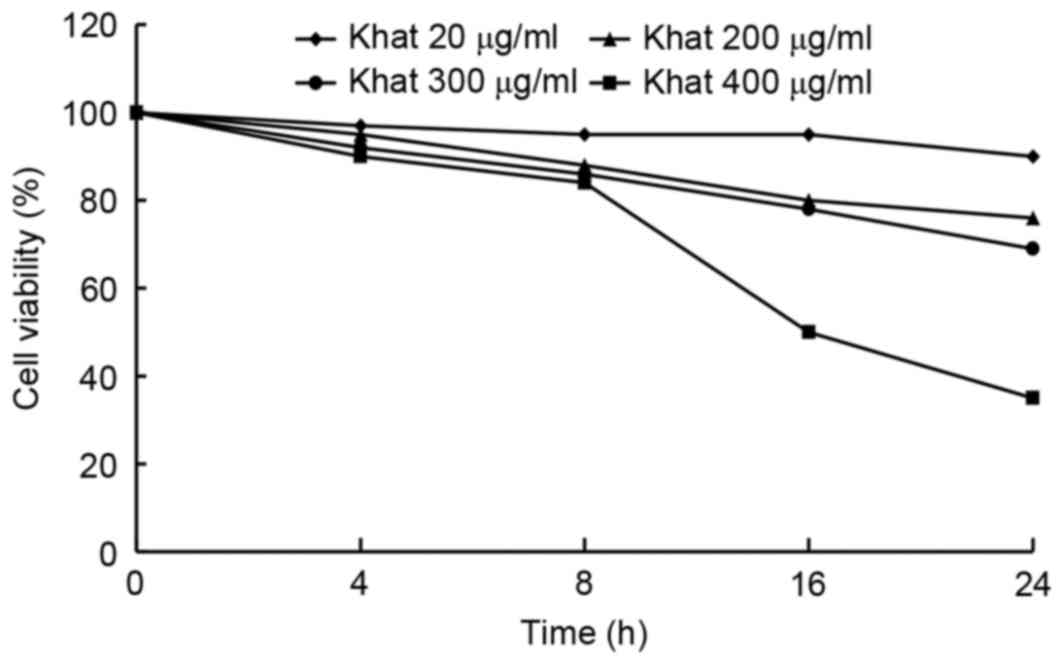
The inhibitory effect of Khat was assessed by
detecting the viability of MDA-MB-231 cells. As shown in Fig. 1, following Khat treatment at
concentrations of 20, 200, 300 and 400 µg/ml, the viability of
MDA-MB-231 cells was inhibited in a dose- and time-dependent
manner, with the greatest effect seen at the concentration of 400
µg/ml following a treatment duration of 8 h. Therefore, a
concentration of 400 µg/ml Khat was selected for use in subsequent
experiments.
Effect of khat treatment on apoptosis
of MDA-MB-231 cells
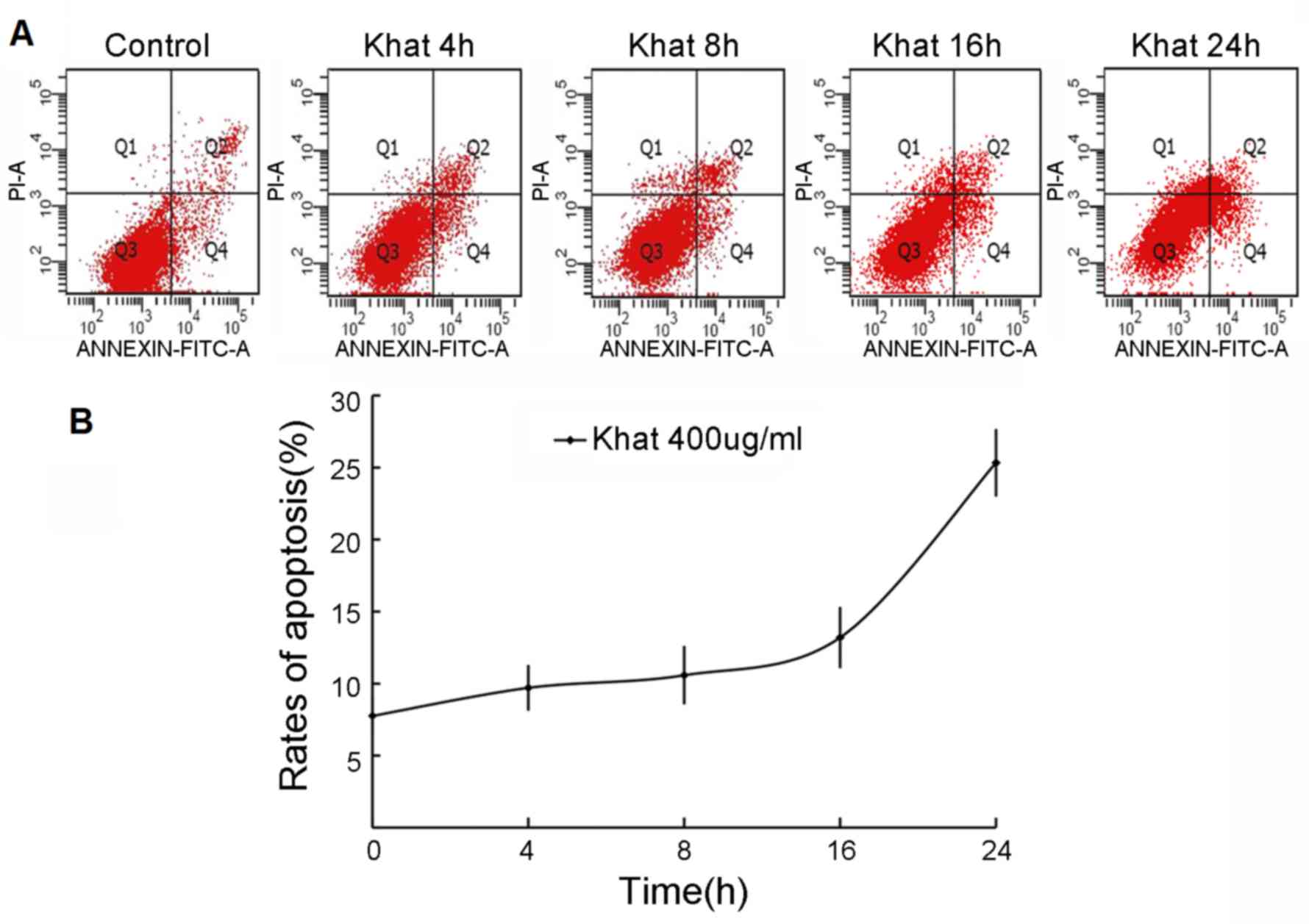
To investigate the effect of 400 µg/ml khat on
apoptosis of MDA-MB-231 cells, the cells were double-stained with
Annexin V and PI, prior to detection using flow cytometry.
Following serum-free culture for 24 h, MDA-MB-231 cells were
exposed to 400 µg/ml khat for 4, 8, 16 and 24 h prior to the
apoptosis assay. As shown in Fig. 2,
the apoptotic rates of the control, and cells exposed to khat for
4, 8, 16 and 24 h were 7.76±2.86, 9.7±1.59, 10.58±2.03, 13.2±4.14,
and 25.3±5.09%, respectively. At each of the time point indicated,
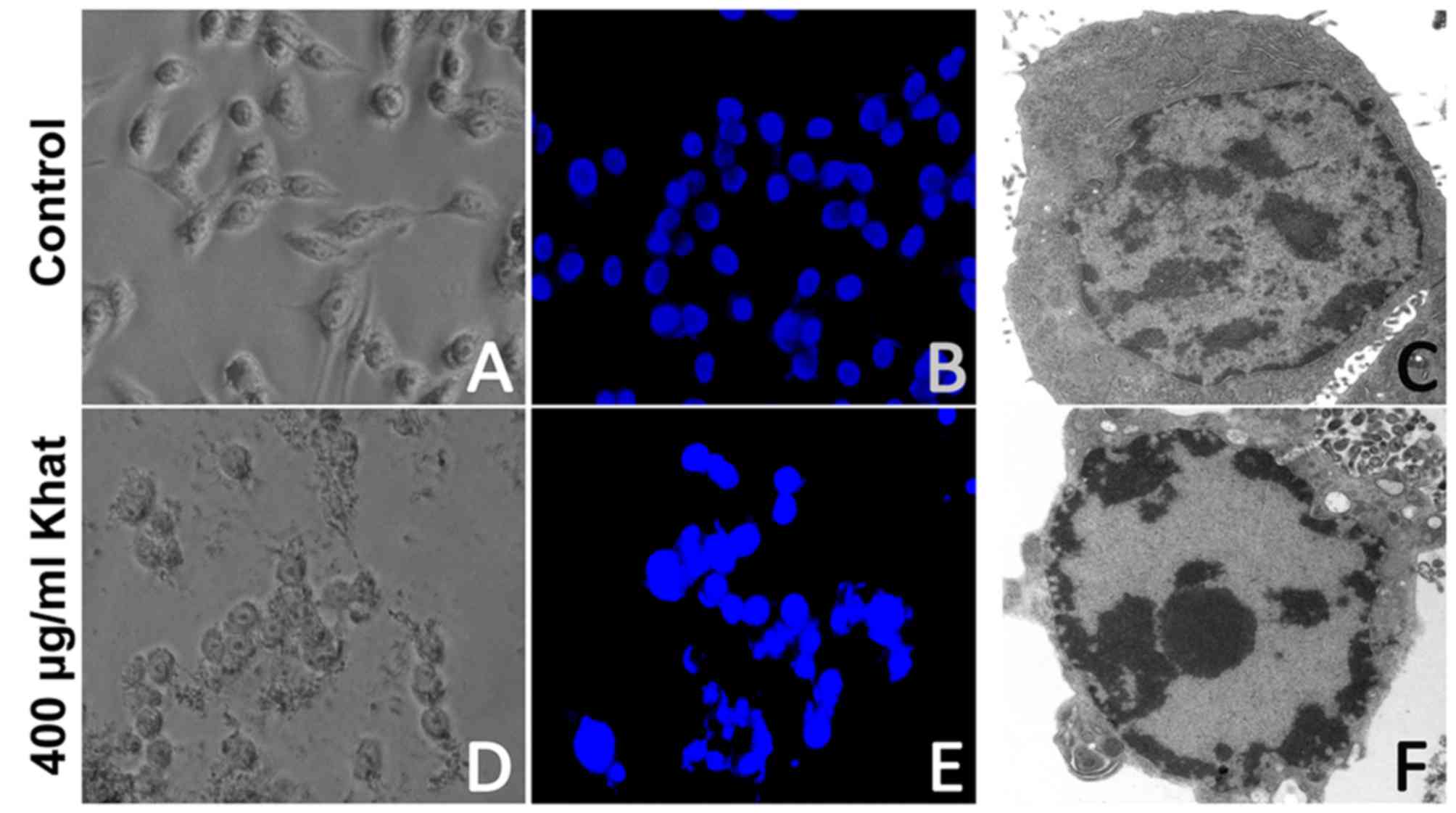
the treated cells were stained with Hoechst 33258. The nuclei of
the cells in the control group were regular and round-shaped
(Fig. 3A-C). By contrast, the nuclei
of the majority of khat-treated (400 µg/ml; 16 h) cells were
condensed and fragmented, which are characteristic of apoptotic
cells (Fig. 3D and E). When observed
under the electron microscope, the khat-treated cells exhibited
chromatin condensation and nuclei shrinkage, which further confirms
apoptosis (Fig. 3F).
Effect of Khat treatment on the
expression of apoptosis-associated proteins and the activation of
MAPKs
The expression of caspase-9 was determined in
MDA-MB-231 cells prior to and following khat treatment using
western blot analysis. The expression of caspase-9 was not detected
following Khat treatment (Fig. 4).
Following incubation with 400 µg/ml khat for different times (0, 4,
8, 16 and 24 h), the expression of pro-apoptotic protein Bax
markedly increased compared with untreated cells (i.e., cells at 0
h). Conversely, the expression of anti-apoptotic protein Bcl-2
markedly decreased compared with expression levels at 0 h.
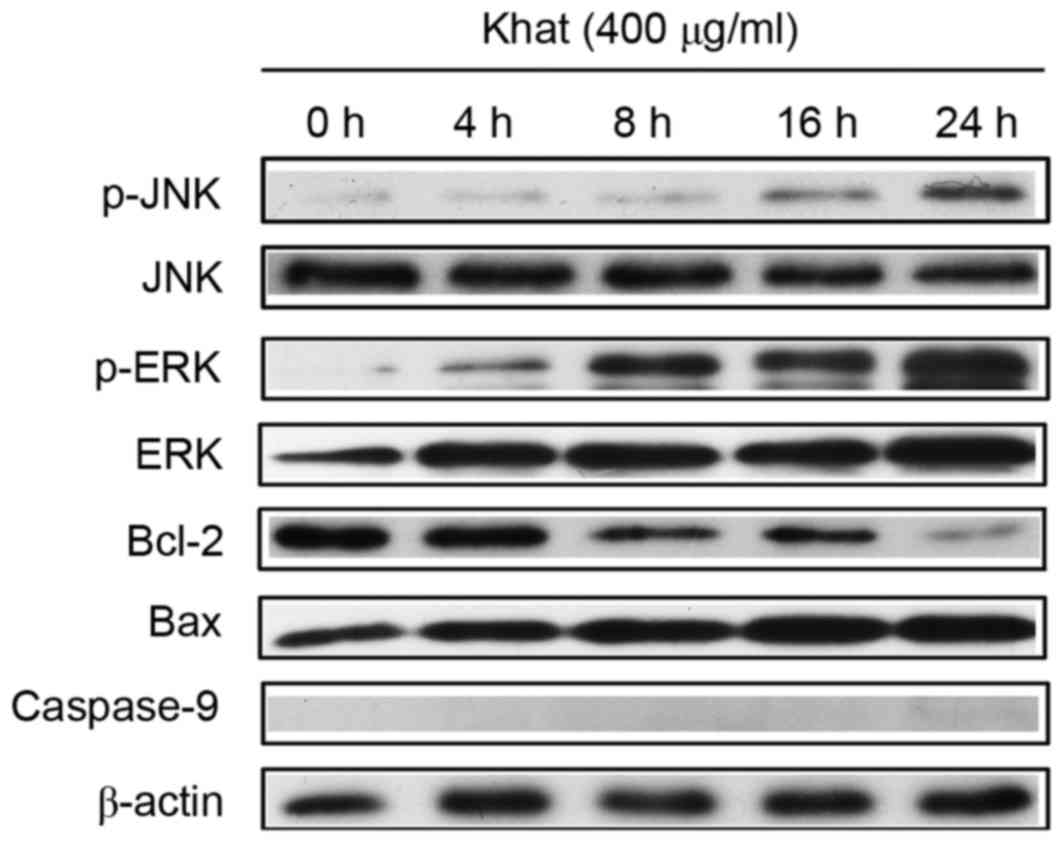 | Figure 4.Expression of apoptosis-associated
proteins and MAPKs (p-JNK, JNK, p-ERK, ERK). MDA-MB-231 cells were
incubated with 400 µg/ml khat for 4, 8, 16 and 24 h; apoptosis- and
MAPK-associated protein expression was detected by western
blotting. MAPK, mitogen-activated protein kinase; p,
phosphorylated; ERK, extracellular signal-regulated kinase; JNK,
janus kinase; Bax, apoptosis regulator Bax; Bcl-2, apoptosis
regulator Bcl-2. |
The involvement of MAPKs in khat-induced apoptosis
of MDA-MB-231 cells was subsequently investigated by measuring the
expression of the activated form of JNK and ERK. As shown in
Fig. 4, the size of the bands for
p-JNK seem to be less intense at 16 and 24 h than those at 0, 4 and
8 h. The band for p-ERK at 4 h is higher-intesity than that at 0 h.
Similarly, the band at 24 h is higher intensity than that at 16 h,
while the total JNK and ERK expression were not changed. Therefore,
the expression of p-JNK and p-ERK were increased when cells were
treated with khat compared with untreated cells (i.e., those at 0
h). It is likely that activation of ERK and JNK signal in
MDA-MB-231 cells may mediate the cellular apoptotic response to
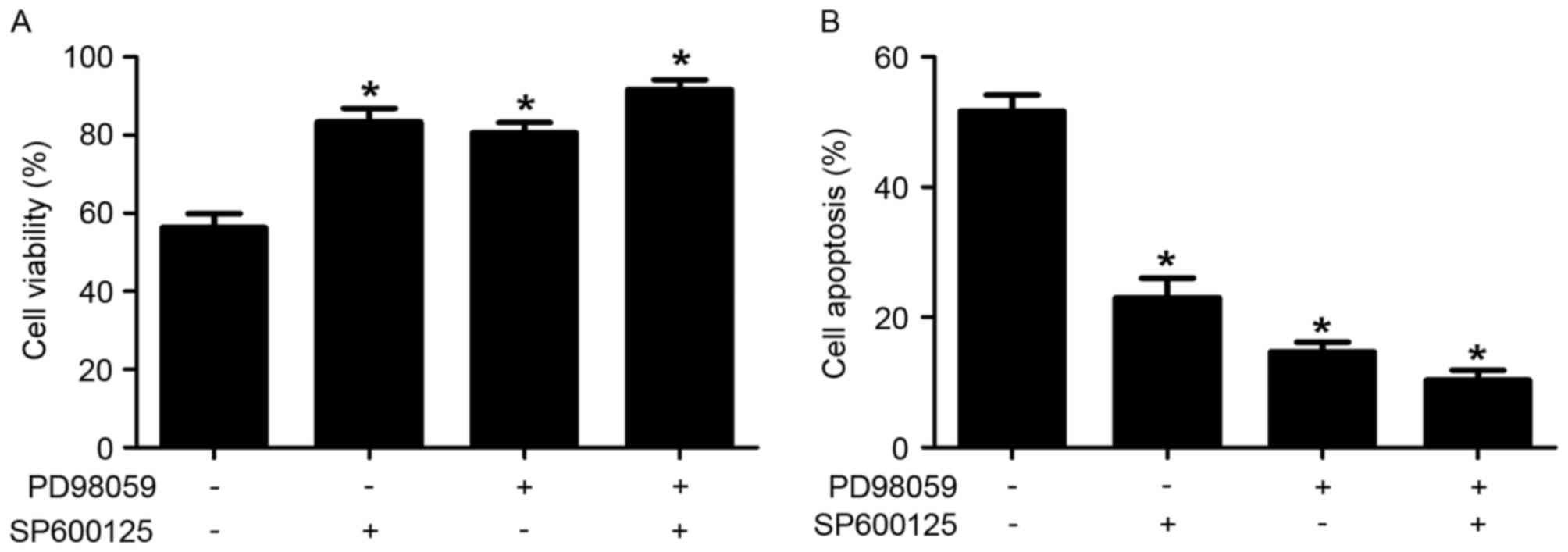
khat treatment. To confirm the possible roles of JNK and ERK in
khat-induced apoptosis, cell viability and apoptotic rates of
MDA-MB-231 cells were determined in response to inhibitors of JNK
(SP600125) and ERK (PD98059). As shown in Fig. 5, treatment with SP600125 and PD98059
significantly reversed khat-induced cell death compared with
untreated cells. These results indicated that JNK and ERK are
involved in khat-induced apoptosis in MDA-MB-231 cells.
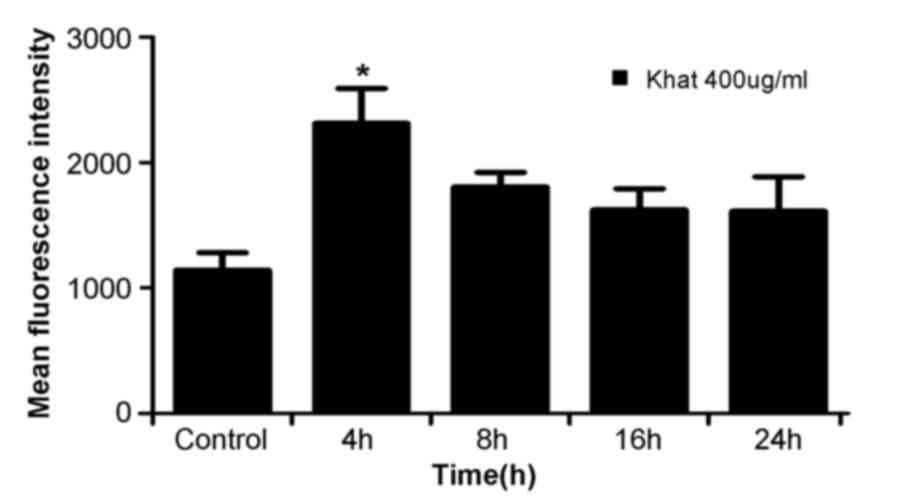
Effect of khat treatment on ROS
production
ROS has been considered as a potential regulator of
apoptosis (25). Therefore, in the
present study, the level of ROS was determined in khat-treated
cells. MDA-MB-231 cells were treated with 400 µg/ml khat for 4, 8,
16 and 24 h and subjected to flow cytometry for detection of ROS.
As shown in Fig. 6, ROS production in
cells treated with 400 µg/ml khat for 4 h was higher than control
cells (0 h cells). Then the ROS production decreased in 8–24 h
groups. khat induced a time-dependent decrease in ROS
production.
Discussion
In the present study, the cell viability assay
indicated that khat treatment inhibited the viability of MDA-MB-231
cells in a time- and dose-dependent manner. Flow cytometric
analysis demonstrated that 400 µg/ml khat induced apoptosis in
MDA-MB-231 cells in a time-dependent manner, which was consistent
with the cell viability assay.
Apoptosis is a major mechanism of cancer suppression
and is characterized by morphological and ultrastructural changes
associated with caspase-regulated biochemical processes (26,27). Khat
contains a complex phytochemisty, with fractions, including
flavonoids that are able to induce apoptosis and act against cancer
cells (28). In the present study,
Hoechst 33258 staining and transmission electron microscopy results
indicated that khat induced apoptosis in MDA-MB-231 cells, and
caused morphological changes, including microvilli loss, cell
membrane bubbling, nuclear chromatin condensation, cytosolic
compartment vacuolization and apoptotic body formation.
The MAPK signaling pathway includes the ERK, JNK and
p38 pathways, which are involved in cell survival, proliferation
and apoptosis. A previous study by the present authors demonstrated
no significant difference in p38 expression in hepatic cells
treated with khat and untreated cells (29). Therefore in the present study, the
expression levels of ERK and JNK were examined in khat-treated
breast cancer cells. The activation of ERK has been previously
demonstrated to be involved in cell cycle progression and
proliferation (30), whereas JNK is
generally activated in response to stress and toxicants that induce
cell apoptosis (31). Prior reports
demonstrated that sustained JNK activation results in apoptosis
(32,33). In the present study, western blotting
demonstrated that the levels of p-JNK and p-ERK were increased
following treatment with khat in a time-dependent manner, whilst
treatment with JNK and ERK inhibitors significantly reversed the
khat-induced cell death. These results suggest that khat-induced
breast cancer cell apoptosis is primarily mediated by the
activation of the JNK signaling pathway.
The Bcl-2 protein is known to be a suppressor of
apoptosis (34), and Bax is a
promoter of apoptosis (35). The
results of the present study revealed that Bcl-2 expression was
decreased and Bax was increased in a time-dependent manner in
khat-treated MDA-MB-231 cells, with an increase in Bax/Bcl-2 ratio.
A previous study reported that the Bax/Bcl-2 ratio may be a more
important apoptosis indicator than either promoter alone (36). The mitochondrial-mediated apoptosis
signaling pathway is regulated by the Bcl-2 family of
anti-apoptotic proteins (Bcl-2, Bcl-xl and myeloid cell leukemia 1)
and pro-apoptotic proteins (Bax, Bcl2-associated agonist of cell
death and Bcl-2 homologous antagonist/killer) (17–20).
Therefore the results in the present study suggest that khat
induces apoptosis in MDA-MB-231 cells via the
mitochondrial-mediated apoptosis pathway. Additionally, no
caspase-9 protein expression was detected in MDA-MB-231 cells,
suggesting khat-induced apoptosis is not modulated by the classical
apoptosis pathway.
In humans, oxidative stress has been identified to
be involved in various pathologies, including cancer,
arteriosclerosis, type II diabetes, ischemia/reperfusion injury,
chronic inflammatory processes and various neurodegenerative
diseases (37). Excess ROS can induce
the oxidation of macromolecules and has been identified to be
involved in ageing, mtDNA mutations and cell death (38). The generation of ROS by mitochondria
serves an essential role in the release of cytochrome c and other
pro-apoptotic proteins, which can trigger caspase activation and
apoptosis (20). In the present
study, ROS levels were higher in khat-treated breast cancer cells
compared with that of the control cells. This result further
suggests that khat-induced apoptosis is modulated by the
mitochondrial-mediated apoptosis pathway.
In conclusion, the results of the present study
suggest that khat causes apoptosis in MDA-MB-231 cells via
sustainable activation of JNK and the mitochondrial-mediated
apoptosis pathway. To the best of our knowledge, this is the first
study to demonstrate that khat may suppress the breast cancer by
inducing the apoptosis of breast cancer cells.
References
|
1
|
Toennes SW, Harder S, Schramm M, Niess C
and Kauert GF: Pharmacokinetics of cathinone, cathine and
norephedrine after the chewing of khat leaves. Br J Clin Pharmacol.
56:125–130. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Kite GC, Ismail M, Simmonds MS and
Houghton PJ: Use of doubly protonated molecules in the analysis of
cathedulins in crude extracts of khat (Catha edulis) by
liquid chromatography/serial mass spectrometry. Rapid Commun Mass
Spectrom. 17:1553–1564. 2003. View
Article : Google Scholar : PubMed/NCBI
|
|
3
|
Kalix P: Cathinone, a natural amphetamine.
Pharmacol Toxicol. 70:77–86. 1992. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Al-Motarreb A, Baker K and Broadley KJ:
Khat: Pharmacological and medical aspects and its social use in
Yemen. Phytother Res. 16:403–413. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Rothman RB, Vu N, Partilla JS, Roth BL,
Hufeisen SJ, Compton-Toth BA, Birkes J, Young R and Glennon RA: In
vitro characterization of ephedrine-related stereoisomers at
biogenic amine transporters and the receptorome reveals selective
actions as norepinephrine transporter substrates. J Pharmacol Exp
Ther. 307:138–145. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Bentur Y, Bloom-Krasik A and
Raikhlin-Eisenkraft B: Illicit cathinone (‘Hagigat’) poisoning.
Clin Toxicol (Phila). 46:206–210. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Giannini AJ, Miller NS and Turner CE:
Treatment of khat addiction. J Subst Abuse Treat. 9:379–382. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Krikorian AD: Kat and its use: An
historical perspective. J Ethnopharmacol. 12:115–178. 1984.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Luqman W and Danowski TS: The use of khat
(Catha edulis) in Yemen. Social and medical observations.
Ann Intern Med. 85:246–249. 1976. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Feyissa AM and Kelly JP: A review of the
neuropharmacological properties of khat. Prog Neuropsychopharmacol
Biol Psychiatry. 32:1147–1166. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Safhi MM, Alam MF, Hussain S, Hakeem
Siddiqui MA, Khuwaja G, Khardali Jubran IA, Al-Sanosi RM and Islam
F: Cathinone, an active principle of Catha edulis,
accelerates oxidative stress in the limbic area of swiss albino
mice. J Ethnopharmacol. 156:102–106. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Kalix P and Khan I: Khat: An
amphetamine-like plant material. Bull World Health Organ.
62:681–686. 1984.PubMed/NCBI
|
|
13
|
Soufi HE, Kameswaran M and Malatani T:
Khat and oral cancer. J Laryngol Otol. 105:643–645. 1991.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Abid MD, Chen J, Xiang M, Zhou J, Chen X
and Gong F: Khat (Catha edulis) generates reactive oxygen
species and promotes hepatic cell apoptosis via MAPK activation.
Int J Mol Med. 32:389–395. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Bras M, Queenan B and Susin SA: Programmed
cell death via mitochondria: Different modes of dying. Biochemistry
(Mosc). 70:231–239. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kroemer G, Galluzzi L, Vandenabeele P,
Abrams J, Alnemri ES, Baehrecke EH, Blagosklonny MV, El-Deiry WS,
Golstein P, Green DR, et al: Classification of cell death:
Recommendations of the nomenclature committee on cell death 2009.
Cell Death Differ. 16:3–11. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xiong S, Mu T, Wang G and Jiang X:
Mitochondria-mediated apoptosis in mammals. Protein Cell.
5:737–749. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ryningen A, Ersvaer E, Øyan AM, Kalland
KH, Vintermyr OK, Gjertsen BT and Bruserud Ø: Stress-induced in
vitro apoptosis of native human acute myelogenous leukemia (AML)
cells shows a wide variation between patients and is associated
with low BCL-2: Bax ratio and low levels of heat shock protein 70
and 90. Leuk Res. 30:1531–1540. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Del Poeta G, Venditti A, Del Principe MI,
Maurillo L, Buccisano F, Tamburini A, Cox MC, Franchi A, Bruno A,
Mazzone C, et al: Amount of spontaneous apoptosis detected by
Bax/Bcl-2 ratio predicts outcome in acute myeloid leukemia (AML).
Blood. 101:2125–2131. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Ott M, Gogvadze V, Orrenius S and
Zhivotovsky B: Mitochondria, oxidative stress and cell death.
Apoptosis. 12:913–922. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Aziz HA, Peh KK and Tan YT: Extraction and
microencapsulation of khat: Effects on sexual motivation and
estradiol level in female rats. J Sex Med. 6:682–695. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Banjaw MY and Schmidt WJ: Behavioural
sensitisation following repeated intermittent oral administration
of Catha edulis in rats. Behav Brain Res. 156:181–189. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Dimba EA, Gjertsen BT, Bredholt T, Fossan
KO, Costea DE, Francis GW, Johannessen AC and Vintermyr OK: Khat
(Catha edulis)-induced apoptosis is inhibited by antagonists
of caspase-1 and −8 in human leukaemia cells. Br J Cancer.
91:1726–1734. 2004.PubMed/NCBI
|
|
24
|
Su YT, Chang HL, Shyue SK and Hsu SL:
Emodin induces apoptosis in human lung adenocarcinoma cells through
a reactive oxygen species-dependent mitochondrial signaling
pathway. Biochem Pharmacol. 70:229–241. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Ruan H, Zhang Z, Tian L, Wang S, Hu S and
Qiao JJ: The Salmonella effector SopB prevents ROS-induced
apoptosis of epithelial cells by retarding TRAF6 recruitment to
mitochondria. Biochem Biophys Res Commun. 478:618–623. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Evan GI and Vousden KH: Proliferation,
cell cycle and apoptosis in cancer. Nature. 411:342–348. 2001.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Hengartner MO: The biochemistry of
apoptosis. Nature. 407:770–776. 2000. View
Article : Google Scholar : PubMed/NCBI
|
|
28
|
Kalix P: Pharmacological properties of the
stimulant khat. Pharmacol Ther. 48:397–416. 1990. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Abid MD, Chen J, Xiang M, Zhou J, Chen X
and Gong F: Khat (Catha edulis) generates reactive oxygen
species and promotes hepatic cell apoptosis via MAPK activation.
Int J Mol Med. 32:389–395. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Mebratu Y and Tesfaigzi Y: How ERK1/2
activation controls cell proliferation and cell death: Is
subcellular localization the answer? Cell cycle. 8:1168–1175. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Xia Z, Dickens M, Raingeaud J, Davis RJ
and Greenberg ME: Opposing effects of ERK and JNK-p38 MAP kinases
on apoptosis. Science. 270:1326–1331. 1995. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Roos WP and Kaina B: DNA damage-induced
cell death by apoptosis. Trends Mol Med. 12:440–450. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Wullaert A, Heyninck K and Beyaert R:
Mechanisms of crosstalk between TNF-induced NF-kappaB and JNK
activation in hepatocytes. Biochem Pharmacol. 72:1090–1101. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Zhu J, Ye Q, Chang L, Xiong W, He Q and Li
W: Upregulation of miR-195 enhances the radiosensitivity of breast
cancer cells through the inhibition of BCL-2. Int J Clin Exp Med.
8:9142–9148. 2015.PubMed/NCBI
|
|
35
|
Yu Y, Pei M and Li L: Baicalin induces
apoptosis in hepatic cancer cells in vitro and suppresses tumor
growth in vivo. Int J Clin Exp Med. 8:8958–8967. 2015.PubMed/NCBI
|
|
36
|
Stoetzer OJ, Nüssler V, Darsow M, Gullis
E, Pelka-Fleischer R, Scheel U and Wilmanns W: Association of
bcl-2, bax, bcl-xL and interleukin-1 beta-converting enzyme
expression with initial response to chemotherapy in acute myeloid
leukemia. Leukemia. 10:(Suppl 3). S18–S22. 1996.PubMed/NCBI
|
|
37
|
Dröge W: Free radicals in the
physiological control of cell function. Physiol Rev. 82:47–95.
2002. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Lee CH and Yu HS: Role of mitochondria,
ROS, and DNA damage in arsenic induced carcinogenesis. Front Biosci
(Schol Ed). 8:312–320. 2016. View
Article : Google Scholar : PubMed/NCBI
|