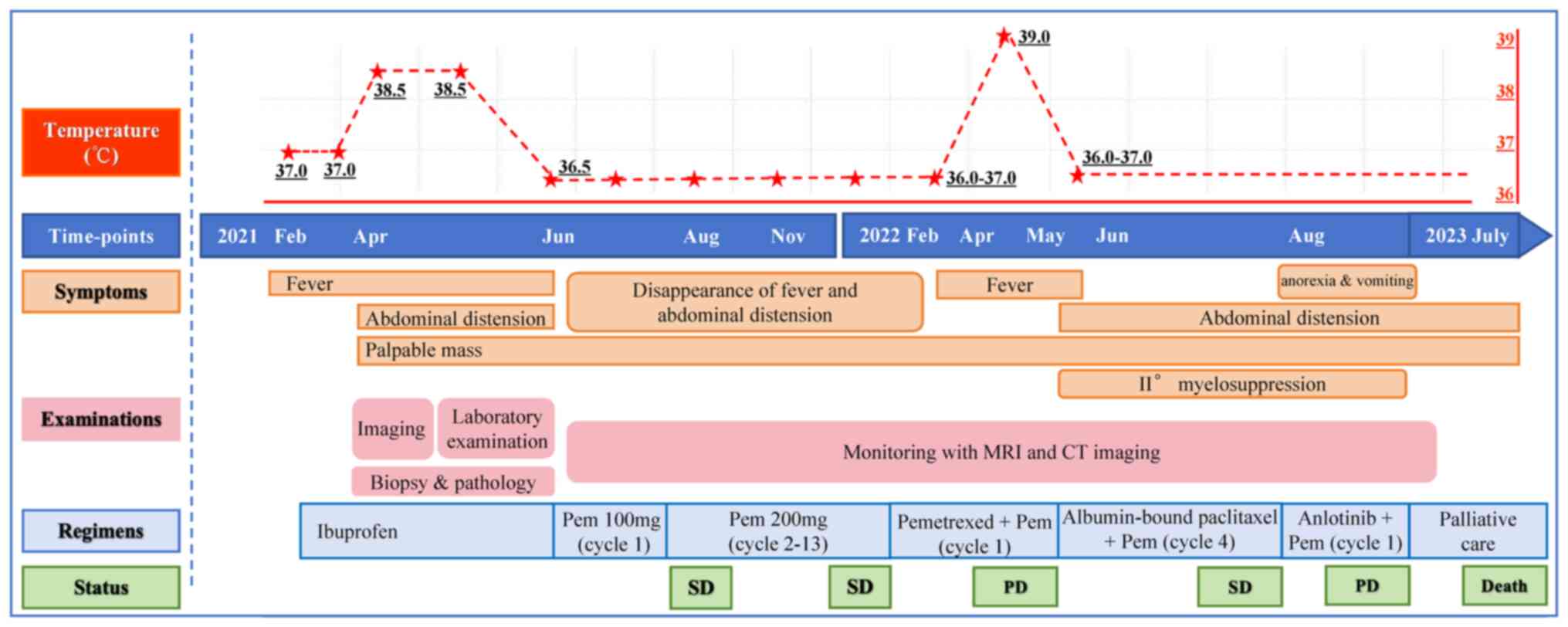
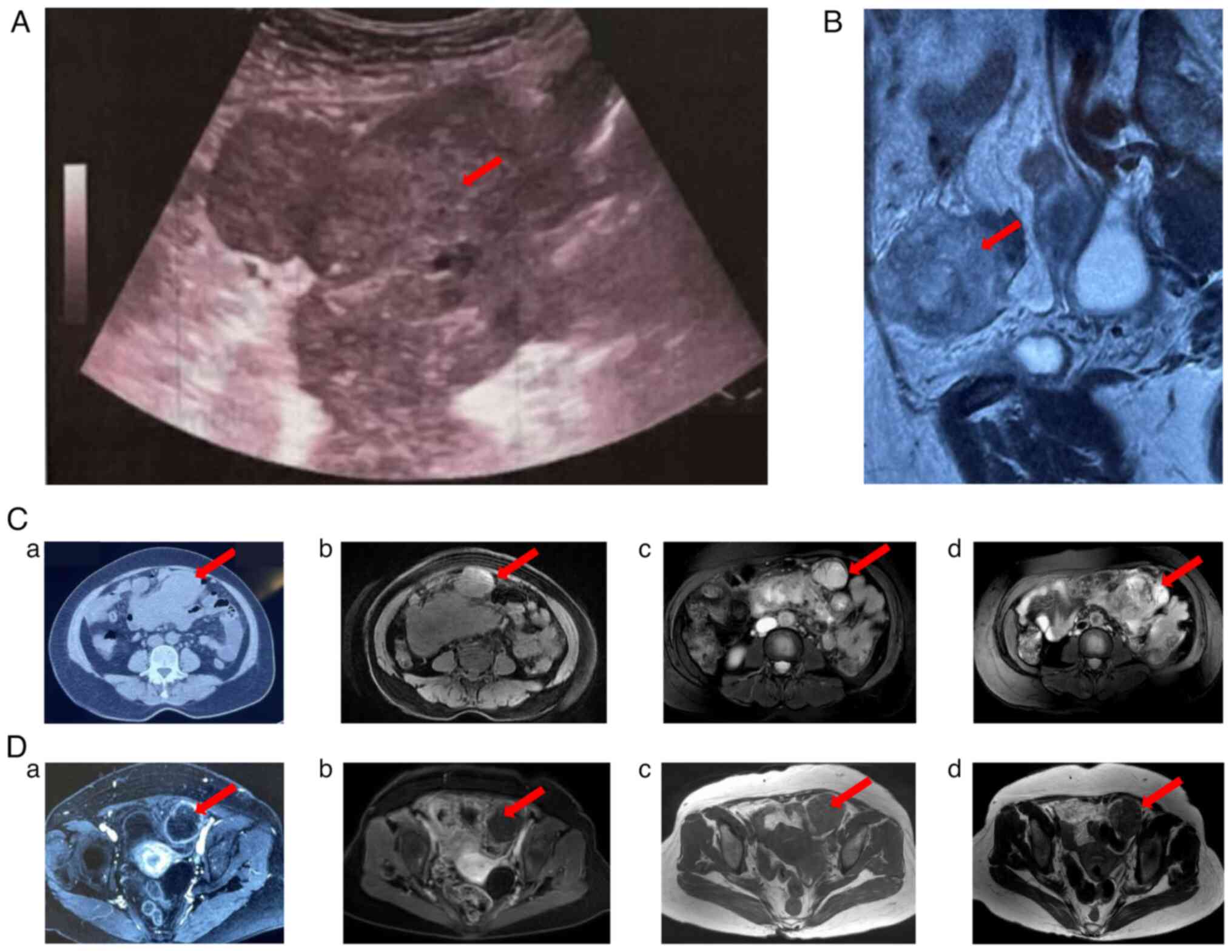
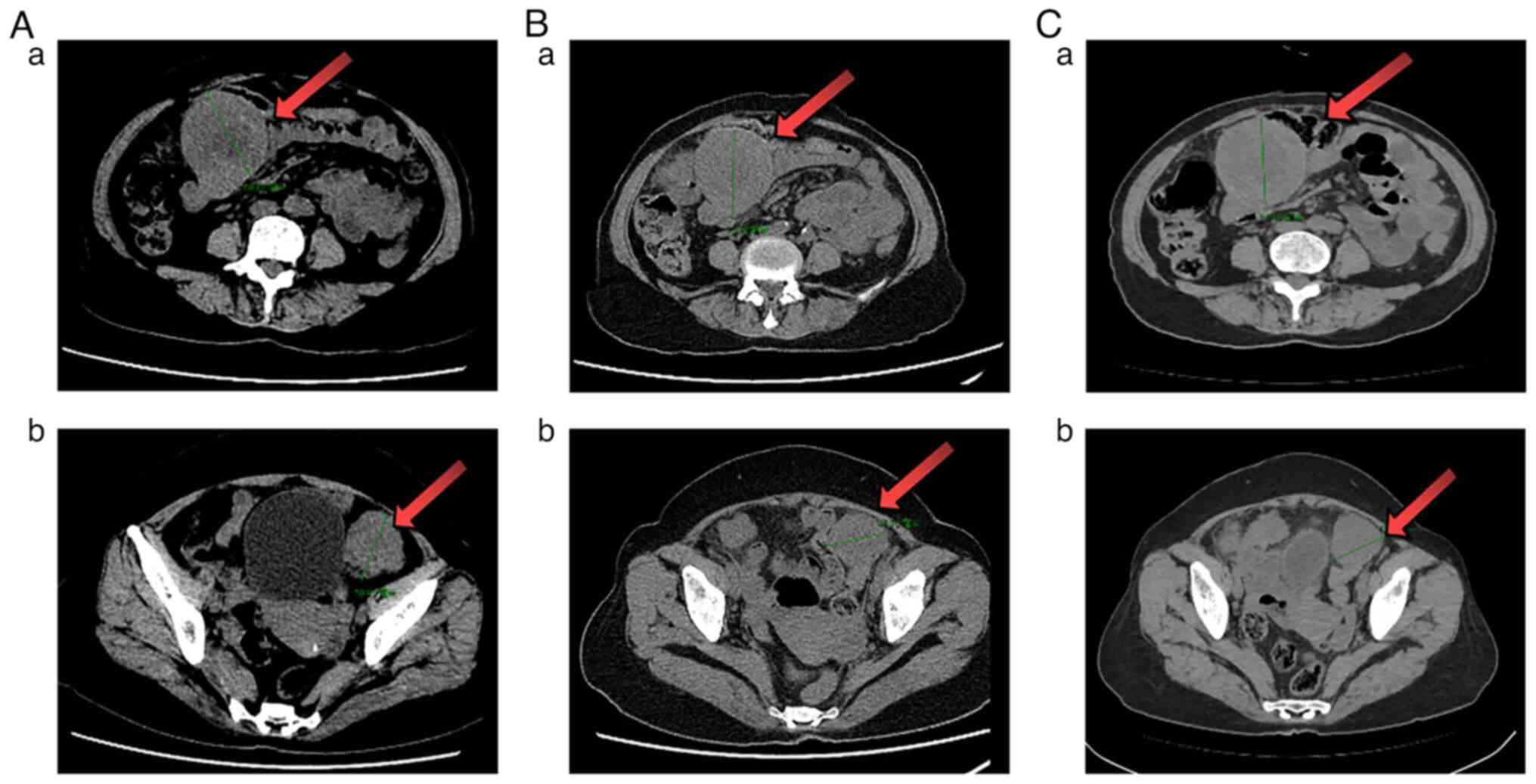
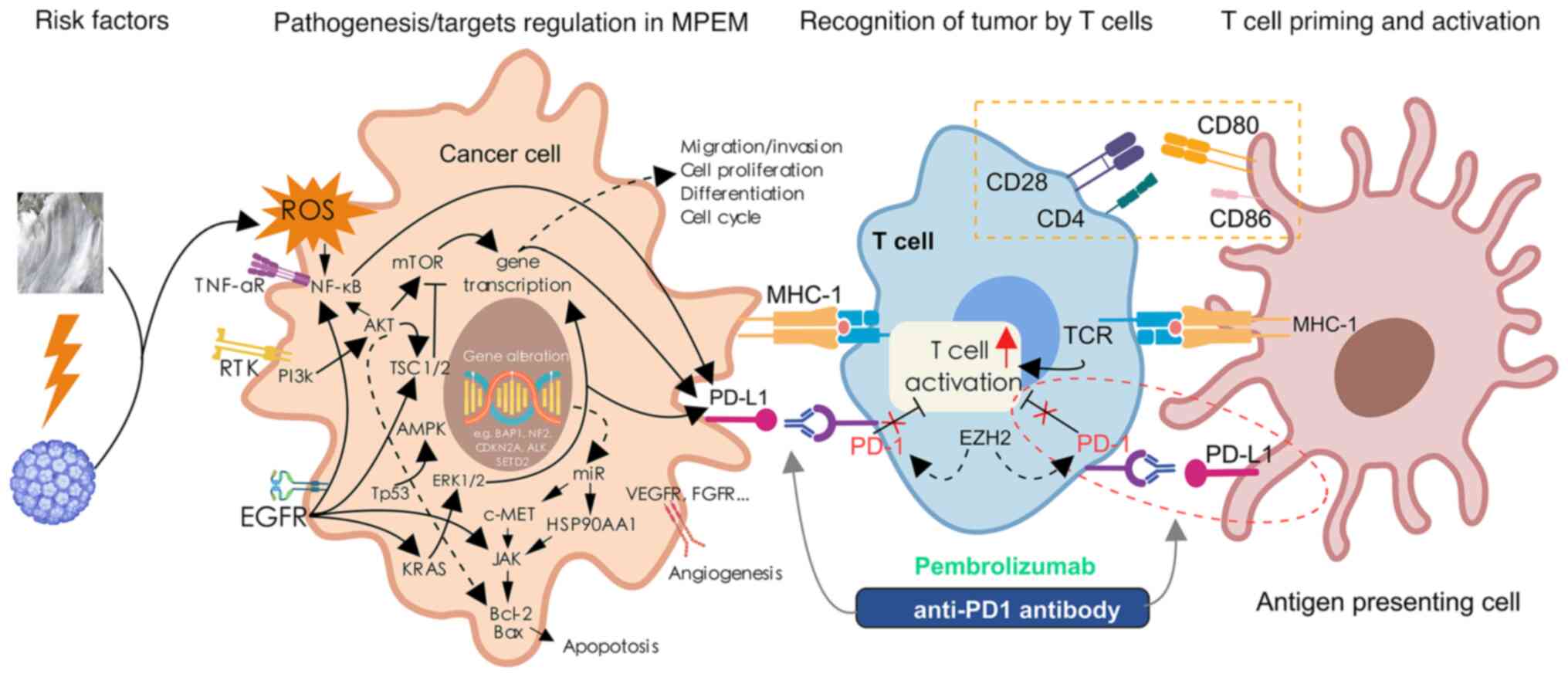
|
1
|
Kim J, Bhagwandin S and Labow DM:
Malignant peritoneal mesothelioma: A review. Ann Transl Med.
5:2362017. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Moolgavkar SH, Meza R and Turim J: Pleural
and peritoneal mesotheliomas in SEER: Age effects and temporal
trends, 1973–2005. Cancer Causes Control. 20:935–944. 2009.
View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Boffetta P: Epidemiology of peritoneal
mesothelioma: A review. Ann Oncol. 18:985–990. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Gray SG and Mutti L: Immunotherapy for
mesothelioma: A critical review of current clinical trials and
future perspectives. Transl Lung Cancer Res. 9 (Suppl 1):S100–S119.
2020. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Jacquet P and Sugarbaker PH: Clinical
research methodologies in diagnosis and staging of patients with
peritoneal carcinomatosis. Cancer Treat Res. 82:359–374. 1996.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Seymour L, Bogaerts J, Perrone A, Ford R,
Schwartz LH, Mandrekar S, Lin NU, Litière S, Dancey J, Chen A, et
al: iRECIST: Guidelines for response criteria for use in trials
testing immunotherapeutics. Lancet Oncol. 18:e143–e152. 2017.
View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Shavelle R, Vavra-Musser K, Lee J and
Brooks J: Life expectancy in pleural and peritoneal mesothelioma.
Lung Cancer Int. 2017:27825902017. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Acherman YI, Welch LS, Bromley CM and
Sugarbaker PH: Clinical presentation of peritoneal mesothelioma.
Tumori. 89:269–273. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Su Y, Liu G, Du X and Li Y: Diffuse
malignant peritoneal mesothelioma with paraneoplastic syndrome: A
case report and literature review. Cancer Res Prev Treat.
49:246–250. 2022.
|
|
10
|
Kusamura S, Torres Mesa PA, Cabras A,
Baratti D and Deraco M: The role of Ki-67 and pre-cytoreduction
parameters in selecting diffuse malignant peritoneal mesothelioma
(DMPM) patients for cytoreductive surgery (CRS) and hyperthermic
intraperitoneal chemotherapy (HIPEC). Ann Surg Oncol. 23:1468–1473.
2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Liu Y, Zheng G, Yang D, Guo X, Tian L,
Song H and Liang Y: Osteopontin, GLUT1 and Ki-67 expression in
malignant peritoneal mesothelioma: Prognostic implications. Intern
Med J. 51:896–904. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Rizzolo A, Ah-Lan KC, Nu TNT and Alcindor
T: Response to ipilimumab and nivolumab in a patient with malignant
peritoneal mesothelioma. Clin Colorectal Cancer. 21:371–374. 2022.
View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Tang LK, Li ZK, Xiang YL, Ma DY and Du GB:
Metachronous double primary malignant tumors with nasopharyngeal
carcinoma and diffuse malignant peritoneal mesothelioma accompanied
with paraneoplastic syndromes treated with nivolumab: A case
report. Medicine (Baltimore). 102:e343492023. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Deng M, Zhang X, Xu C, Luo R, Chen L, Zhou
Y and Hou Y: Clinical and pathological observation of conversion
therapy for malignant peritoneal mesothelioma: A case report and
literature review. Pathol Oncol Res. 29:16115772024. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Hung YP, Dong F, Watkins JC, Nardi V,
Bueno R, Dal Cin P, Godleski JJ, Crum CP and Chirieac LR:
Identification of ALK rearrangements in malignant peritoneal
mesothelioma. JAMA Oncol. 4:235–238. 2018. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Chapel DB, Stewart R, Furtado LV, Husain
AN, Krausz T and Deftereos G: Tumor PD-L1 expression in malignant
pleural and peritoneal mesothelioma by Dako PD-L1 22C3 pharmDx and
Dako PD-L1 28-8 pharmDx assays. Hum Pathol. 87:11–17. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Bairos Menezes M, Pedroso de Lima R,
Dunões I, Inácio M and Dinis R: A complete response to
pembrolizumab in malignant peritoneal mesothelioma: A case report.
Cureus. 16:e527162024.PubMed/NCBI
|
|
18
|
Mensi C, Mendola M, Dallari B, Sokooti M,
Tabibi R, Riboldi L and Consonni D: Differences between peritoneal
and pleural mesothelioma in Lombardy, Italy. Cancer Epidemiol.
51:68–73. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Borczuk AC, Cappellini GC, Kim HK,
Hesdorffer M, Taub RN and Powell CA: Molecular profiling of
malignant peritoneal mesothelioma identifies the
ubiquitin-proteasome pathway as a therapeutic target in poor
prognosis tumors. Oncogene. 26:610–617. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
López-Ríos F, Chuai S, Flores R, Shimizu
S, Ohno T, Wakahara K, Illei PB, Hussain S, Krug L, Zakowski MF, et
al: Global gene expression profiling of pleural mesotheliomas:
overexpression of aurora kinases and P16/CDKN2A deletion as
prognostic factors and critical evaluation of microarray-based
prognostic prediction. Cancer Res. 66:2970–2979. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Wang Q, Xu C, Wang W, Zhang Y, Li Z, Song
Z, Wang J, Yu J, Liu J, Zhang S, et al: Chinese expert consensus on
the diagnosis and treatment of malignant pleural mesothelioma.
Thorac Cancer. 14:2715–2731. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
National Comprehensive Cancer Network
(NCCN), . Mesothelioma. Peritoneal, Version 1. NCCN; Plymouth
Meeting, PA: 2021
|
|
23
|
Marmarelis ME, Wang X, Roshkovan L, Walker
S, McNulty S, Ciunci CA, Muira J, Katz SI, Cengel KA, Karakousis G
and Langer CJ: Real-world outcomes of pembrolizumab in peritoneal
mesothelioma. J Clin Oncol. 38 (15-suppl):e210942020. View Article : Google Scholar
|
|
24
|
Tanaka T, Miyamoto Y, Sakai A and Fujimoto
N: Nivolumab for malignant peritoneal mesothelioma. BMJ Case Rep.
13:e2377212020. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Khanna S, Thomas A, Abate-Daga D, Zhang J,
Morrow B, Steinberg SM, Orlandi A, Ferroni P, Schlom J, Guadagni F
and Hassan R: Malignant mesothelioma effusions are infiltrated by
CD3+ T cells highly expressing PD-L1 and the PD-L1+ tumor cells
within these effusions are susceptible to ADCC by the Anti-PD-L1
antibody avelumab. J Thorac Oncol. 11:1993–2005. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Mansfield AS, Roden AC, Peikert T, Sheinin
YM, Harrington SM, Krco CJ, Dong H and Kwon ED: B7-H1 expression in
malignant pleural mesothelioma is associated with sarcomatoid
histology and poor prognosis. J Thorac Oncol. 9:1036–1040. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Marmarelis ME, Wang X, Roshkovan L, Grady
CB, Miura JT, Ginsberg MS, Ciunci CA, Egger J, Walker S, Cercek A,
et al: Clinical outcomes associated with pembrolizumab monotherapy
among adults with diffuse malignant peritoneal mesothelioma. JAMA
Netw Open. 6:e2325262023. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Gazivoda VP, Kangas-Dick AW, Greenbaum AA,
Roshal J, Chen C, Moore DF, Langan RC, Kennedy TJ, Minerowicz C and
Alexander HR: Expression of PD-L1 in patients with malignant
peritoneal mesothelioma: A pilot study. J Surg Res. 277:131–137.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Valmary-Degano S, Colpart P, Villeneuve L,
Monnien F, M'Hamdi L, Lang Averous G, Capovilla M, Bibeau F,
Laverriere MH, Verriele-Beurrier V, et al: Immunohistochemical
evaluation of two antibodies against PD-L1 and prognostic
significance of PD-L1 expression in epithelioid peritoneal
malignant mesothelioma: A RENAPE study. Eur J Surg Oncol.
43:1915–1923. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
White MG, Schulte JJ, Xue L, Berger Y,
Schuitevoerder D, Vining CC, Kindler HL, Husain A, Turaga KK and
Eng OS: Heterogeneity in PD-L1 expression in malignant peritoneal
mesothelioma with systemic or intraperitoneal chemotherapy. Br J
Cancer. 124:564–566. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Zalcman G, Mazieres J, Margery J,
Greillier L, Audigier-Valette C, Moro-Sibilot D, Molinier O, Corre
R, Monnet I, Gounant V, et al: Bevacizumab for newly diagnosed
pleural mesothelioma in the Mesothelioma Avastin cisplatin
pemetrexed study (MAPS): A randomised, controlled, open-label,
phase 3 trial. Lancet. 387:1405–1414. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Baas P, Scherpereel A, Nowak AK, Fujimoto
N, Peters S, Tsao AS, Mansfield AS, Popat S, Jahan T, Antonia S, et
al: First-line nivolumab plus ipilimumab in unresectable malignant
pleural mesothelioma (CheckMate 743): A multicentre, randomised,
open-label, phase 3 trial. Lancet. 397:375–386. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Selleslag DL, Geraghty RJ, Ganesan TS,
Slcvin ML, Wrigley PF and Brown R: Autoimmune haemolytic anaemia
associated with malignant peritoneal mesothelioma. Acta Clin Belg.
44:199–201. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Kimura N, Ogasawara T, Asonuma S, Hama H,
Sawai T and Toyota T: Granulocyte-colony stimulating factor- and
interleukin 6-producing diffuse deciduoid peritoneal mesothelioma.
Mod Pathol. 18:446–450. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Banayan S, Hot A, Janier M, Ninet J,
Zurlinden O and Billotey C: Malignant mesothelioma of the
peritoneum as the cause of a paraneoplastic syndrome: Detection by
18F-FDG PET. Eur J Nucl Med Mol Imaging. 33:7512006. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Socola F, Loaiza-Bonilla A,
Bustinza-Linares E, Correa R and Rosenblatt JD: Recurrent
thrombotic thrombocytopenic purpura-like syndrome as a
paraneoplastic phenomenon in malignant peritoneal mesothelioma: A
case report and review of the literature. Case Rep Oncol Med.
2012:6193482012.PubMed/NCBI
|
|
37
|
Thakral B and Loghavi S: Marked
paraneoplastic leukemoid reaction in a patient with mesothelioma
mimicking a myeloid neoplasm. Blood. 135:4572020. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chen LY, Huang LX, Wang J, Qian Y and Fang
LZ: Malignant peritoneal mesothelioma presenting with persistent
high fever. J Zhejiang Univ Sci B. 12:381–384. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Hermann J, Bajko G, Stajgis M, Szmeja J,
Kościński T and Drews M: Fever of unknown origin: A clinical mask
of malignant peritoneal mesothelioma. Contemp Oncol (Pozn).
16:596–599. 2012.PubMed/NCBI
|
|
40
|
Ishizuka K, Uehara T, Arai M, Ikeda J,
Hirose Y and Ikusaka M: Medical-type peritoneal mesothelioma
leading to death two months after onset of fever of unknown origin.
Radiol Case Rep. 17:540–543. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Calabrò L, Morra A, Fonsatti E, Cutaia O,
Amato G, Giannarelli D, Di Giacomo AM, Danielli R, Altomonte M,
Mutti L and Maio M: Tremelimumab for patients with
chemotherapy-resistant advanced malignant mesothelioma: An
open-label, single-arm, phase 2 trial. Lancet Oncol. 14:1104–1111.
2013. View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Calabrò L, Morra A, Fonsatti E, Cutaia O,
Fazio C, Annesi D, Lenoci M, Amato G, Danielli R, Altomonte M, et
al: Efficacy and safety of an intensified schedule of tremelimumab
for chemotherapy-resistant malignant mesothelioma: an open-label,
single-arm, phase 2 study. Lancet Respir Med. 3:301–309. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
43
|
Maio M, Scherpereel A, Calabrò L, Aerts J,
Perez SC, Bearz A, Nackaerts K, Fennell DA, Kowalski D, Tsao AS, et
al: Tremelimumab as second-line or third-line treatment in relapsed
malignant mesothelioma (DETERMINE): A multicentre, international,
randomised, double-blind, placebo-controlled phase 2b trial. Lancet
Oncol. 18:1261–1273. 2017. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Desai A, Karrison T, Rose B, Pemberton E,
Hill B, Straus CM, Tan YHC, Seiwert TY and Kindler HL: Phase II
trial of pembrolizumab (P) in patients (pts) with
previously-treated mesothelioma (MM). J Clin Oncol. 36
(15-suppl):S85652018. View Article : Google Scholar
|
|
45
|
Hassan R, Thomas A, Nemunaitis JJ, Patel
MR, Bennouna J, Chen FL, Delord JP, Dowlati A, Kochuparambil ST,
Taylor MH, et al: Efficacy and safety of avelumab treatment in
patients with advanced unresectable mesothelioma: Phase 1b results
from the JAVELIN solid tumor trial. JAMA Oncol. 5:351–357. 2019.
View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Calabrò L, Rossi G, Morra A, Rosati C,
Cutaia O, Daffinà MG, Altomonte M, Di Giacomo AM, Casula M, Fazio
C, et al: Tremelimumab plus durvalumab retreatment and 4-year
outcomes in patients with mesothelioma: A follow-up of the open
label, non-randomised, phase 2 NIBIT-MESO-1 study. Lancet Respir
Med. 9:969–976. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Ikushima H, Sakatani T, Ohara S, Takeshima
H, Horiuchi H, Morikawa T and Usui K: Cisplatin plus pemetrexed
therapy and subsequent immune checkpoint inhibitor administration
for malignant peritoneal mesothelioma without pleural lesions: Case
report. Medicine (Baltimore). 99:e199562020. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Kitadai R, Shimoi T, Sudo K, Noguchi E,
Nagata Y, Sawada R, Takashima A, Boku N and Yonemori K: Efficacy of
second-line treatment and prognostic factors in patients with
advanced malignant peritoneal mesothelioma: A retrospective study.
BMC Cancer. 21:2942021. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Raghav K, Liu S, Overman MJ, Willett AF,
Knafl M, Fu SC, Malpica A, Prasad S, Royal RE, Scally CP, et al:
Efficacy, safety, and biomarker analysis of combined PD-L1
(Atezolizumab) and VEGF (Bevacizumab) blockade in advanced
malignant peritoneal mesothelioma. Cancer Discov. 11:2738–2747.
2021. View Article : Google Scholar : PubMed/NCBI
|
|
50
|
Raghav K, Liu S, Overman M, Morani A,
Willette A, Fournier K and Varadhachary G: Clinical efficacy of
immune checkpoint inhibitors in patients with advanced malignant
peritoneal mesothelioma. JAMA Netw Open. 4:e21199342021. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Fennell DA, Ewings S, Ottensmeier C,
Califano R, Hanna GG, Hill K, Danson S, Steele N, Nye M, Johnson L,
et al: Nivolumab versus placebo in patients with relapsed malignant
mesothelioma (CONFIRM): A multicentre, double-blind, randomised,
phase 3 trial. Lancet Oncol. 22:1530–1540. 2021. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Huang X, Hong Y, Xie SY, Liao HL, Huang
HM, Liu JH and Long WJ: Malignant peritoneal mesothelioma with
massive ascites as the first symptom: A case report. World J Clin
Cases. 10:10317–10325. 2022. View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Fujiwara Y, Takahashi Y, Okada M,
Kishimoto T, Kondo S, Fujikawa K, Hayama M, Sugeno M, Ueda S,
Komuro K, et al: Phase I study of tremelimumab monotherapy or in
combination with durvalumab in japanese patients with advanced
solid tumors or malignant mesothelioma. Oncologist. 27:e703–e722.
2022. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Peng XD, You ZY, He LX and Deng Q:
Zimberelimab plus chemotherapy as the first-line treatment of
malignant peritoneal mesothelioma: A case report and review of
literature. World J Clin Cases. 11:5296–5302. 2023. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Sugarbaker PH: Response to Nivolumab
followed by complete cytoreductive surgery with HIPEC resulted in
long-term survival in a patient with sarcomatoid-predominant
biphasic peritoneal mesothelioma. A case report. Int J Surg Case
Rep. 107:1083592023. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Lal D, Misiura M, Adeel W and Tariq R:
Brain metastases with malignant peritoneal mesothelioma: Never
reported before. Cureus. 15:e437442023.PubMed/NCBI
|