Introduction
Gastric cancer is more prevalent in East Asia,
Eastern Europe and Central and South America than in other
countries. Worldwide, gastric cancer ranks second among all causes
of death from cancer, with about 7,000,000 confirmed deaths
annually (1). In Japan, it is one
of the most frequent causes of cancer mortality, despite notable
advances in diagnosis and treatment (2). Outcomes are extremely poor in patients
with unresectable gastric cancer, with the median survival ranging
from 3–5 months even with the best supportive care (3,4).
Numerous randomized controlled trials of various
treatment regimens have previously been reported, including
5-fluorouracil, doxorubicin and mitomycin (5), epirubicin and cisplatin (CDDP) in
combination with continuous infusion of 5-fluorouracil (6) and 5-fluorouracil and cisplatin
(7). However, the trials produced
median survival rates of less than 1 year. Recently, two randomized
controlled trials were reported from Japan (8,9). One
was the JCOG9912 trial, which showed S-1 to be non-inferior to
continuous infusion of 5-fluorouracil with respect to overall
survival (OS). The other was the SPIRITS trial, which noted that
S-1 plus CDDP has been identified as the standard chemotherapy
regimen for advanced or recurrent gastric cancer in Japan (8).
Docetaxel has shown promising activity in gastric
cancer, both as a monotherapy (10)
and in combination with other agents (11–13).
Phase I and II studies of combination therapy were performed with
docetaxel and S-1 for patients with advanced or recurrent gastric
cancer (14,15). In the phase II study, the overall
response rate was 56.3% (95% confidence interval [CI], 38–66%) and
the median survival time period was 14.3 months (95% CI, 10.27–20.3
months).
On the basis of these results, a phase III study
(JACCRO GC-03 study) comparing S-1 alone with the combination
therapy (docetaxel and S-1) was launched. This is a prospective,
multicenter (Korea and Japan), multinational, randomized study of
patients with advanced gastric cancer. In total, 638 patients were
recruited, and the final results are anticipated in 2010. Previous
studies reported that the docetaxel and S-1 combination has good
clinical efficacy with acceptable toxicity when administered as the
first-line treatment for patients with advanced gastric cancer
(16,17).
Systemic chemotherapy is the key treatment for
patients with stage IV gastric cancer. In some cases, combination
chemotherapy (CDDP plus S-1 and docetaxel plus S-1) results in
long-term survival in clinical practice. In selected cases,
additional (adjuvant) surgery may result in further long-term
survival. Surgical resection was classified as curative (no
evidence of remaining disease after surgery) or palliative
(remaining disease after surgery). In this context, adjuvant
surgery aimed to be curative, as opposed to palliative, after the
response to chemotherapy. Following adjuvant surgery, chemotherapy
is required.
This study therefore aimed to evaluate the efficacy
of adjuvant surgery after response to the chemotherapy for advanced
gastric cancer.
Materials and methods
Patients
The study included 20 advanced gastric cancer
patients treated between September 2003 and December 2008 at
Hiroshima University Hospital. Patients were diagnosed with
unresectable advanced gastric cancer and had undergone
gastrectomies following the response to chemotherapy with docetaxel
and S-1.
Treatment regimen
S-1 (80 mg/m2) was administered orally
after morning and evening meals for 2 weeks, followed by a
drug-free interval of 1 week (one cycle). Docetaxel (40
mg/m2) was diluted in 100 ml of 0.9% saline and
administered as a 1-h infusion on the morning of day 1 of each
cycle (i.e., every 3 weeks). The docetaxel infusion was started
simultaneously with the S-1 administration. Dexamethasone (8 mg)
was infused 1 h before the docetaxel administration. Responses were
classified according to the Response Evaluation Criteria in Solid
Tumors (RECIST) guidelines (18)
and the guidelines of the Japanese Gastric Cancer Association
(19). To assess responses every
4–6 weeks, the tumor area was measured using a 5-mm slice computed
tomography scan for all measurable lesions. Toxicity was graded
according to the Common Terminology Criteria for Adverse Events
(CTCAE) version 3.0 (20).
Indications for adjuvant surgery
The indications for adjuvant surgery (after the
response to chemotherapy) are that curative resection (not
palliative) is anticipated based on the response to chemotherapy.
Thus, indications such as the absence of other distant metastases,
including peritoneal dissemination, extensive lymph node metastases
or lung metastasis and feasible macroscopically complete removal of
liver deposits are considered. Other indications include curative
resection which is anticipated despite a poor response to
chemotherapy (stable disease).
Statistical analysis
OS was calculated from the date of chemotherapy
initiation to the date patients succumbed to all causes or the
latest follow-up. The median OS was estimated using the
Kaplan-Meier method. Multivariate analysis of prognostic factors
was performed by the Cox proportional method to evaluate the
effects of prognostic factors on patient survival. P<0.05 was
considered to indicate a statistically significant difference.
Results
Patient characteristics
A total of 20 advanced gastric cancer patients were
treated from September 2003 to December 2008 at Hiroshima
University Hospital. Table I shows
the patient characteristics. The median age was 58.8 years (range
25–73). All 20 patients showed a performance status of 0 or 1. The
median number of cycles administered per patient was 4.4 (range
2–11). Patients were assessable for response. No complete response
was noted, while 17 patients (85%) had partial responses (PR) and 3
(15%) had stable disease (SD). The patients had been diagnosed as
resectable following chemotherapy. However, in 9 patients,
peritoneal dissemination or tumor cells in peritoneal fluid on
cytological analysis were found during surgery, resulting in
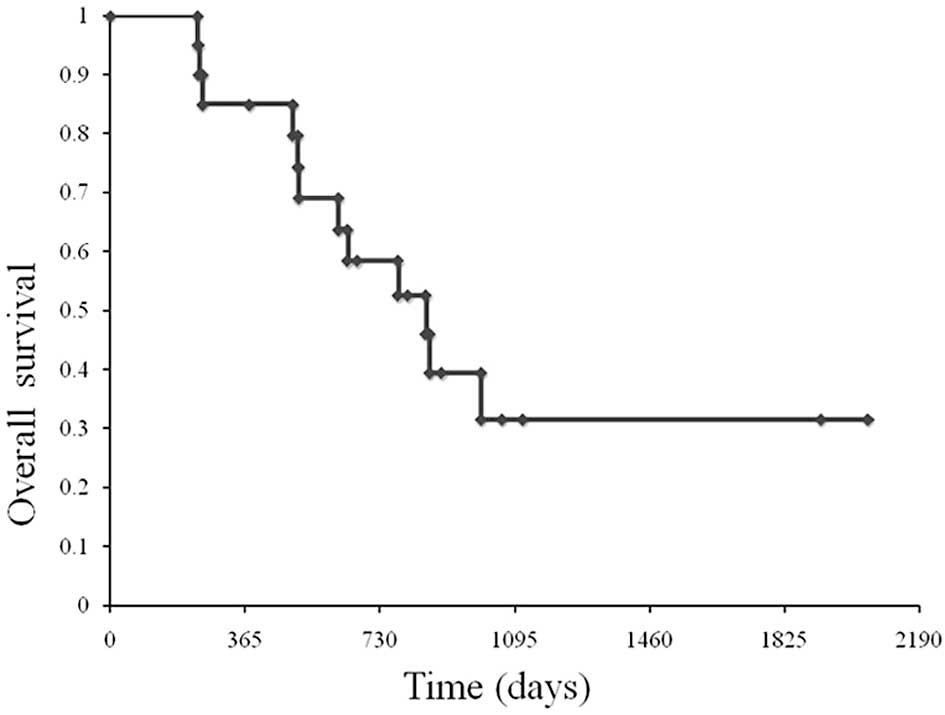
palliative surgery. At a median follow-up of 980 days, OS was 855
days (Fig. 1).
 | Table IPatient characteristics of the
adjuvant surgery for advanced gastric cancer. |
Table I
Patient characteristics of the
adjuvant surgery for advanced gastric cancer.
| Case no. | Age | Gender | Clinical stage | No. of cycles | Response | Surgical
curability |
|---|
|
|---|
| T | N | H | M | P |
|---|
| 1 | 70 | M | 3 | 1 | 1 | 0 | 0 | 3 | SD | C |
| 2 | 73 | M | 3 | 2 | 1 | 0 | 0 | 3 | SD | C |
| 3 | 64 | F | 3 | 2 | 1 | 0 | 0 | 3 | PR | B |
| 4 | 61 | F | 3 | 2 | 1 | 0 | 0 | 3 | PR | B |
| 5 | 72 | M | 3 | 2 | 1 | 0 | 0 | 4 | PR | B |
| 6 | 63 | M | 3 | 2 | 1 | 0 | 0 | 6 | PR | B |
| 7 | 65 | M | 1 | 3 | 0 | 0 | 1 | 4 | PR | C |
| 8 | 41 | M | 2 | 3 | 0 | 0 | 1 | 3 | PR | B |
| 9 | 49 | F | 3 | 1 | 0 | 0 | 1 | 6 | PR | C |
| 10 | 45 | M | 4 | 1 | 0 | 0 | 1 | 4 | PR | B |
| 11 | 27 | M | 4 | 3 | 0 | 0 | 1 | 3 | PR | C |
| 12 | 65 | M | 3 | 1 | 0 | 0 | 1 | 5 | PR | B |
| 13 | 25 | F | 3 | 3 | 0 | 0 | 0 | 3 | SD | C |
| 14 | 78 | F | 3 | 3 | 0 | 0 | 0 | 5 | PR | C |
| 15 | 75 | M | 4 | 3 | 0 | 0 | 0 | 2 | PR | B |
| 16 | 36 | M | 3 | 3 | 0 | 0 | 0 | 5 | PR | B |
| 17 | 70 | M | 3 | 3 | 0 | 1 | 0 | 5 | PR | C |
| 18 | 61 | F | 3 | 3 | 0 | 0 | 0 | 5 | PR | B |
| 19 | 67 | M | 3 | 3 | 0 | 0 | 0 | 7 | PR | B |
| 20 | 56 | M | 3 | 3 | 1 | 1 | 0 | 11 | PR | C |
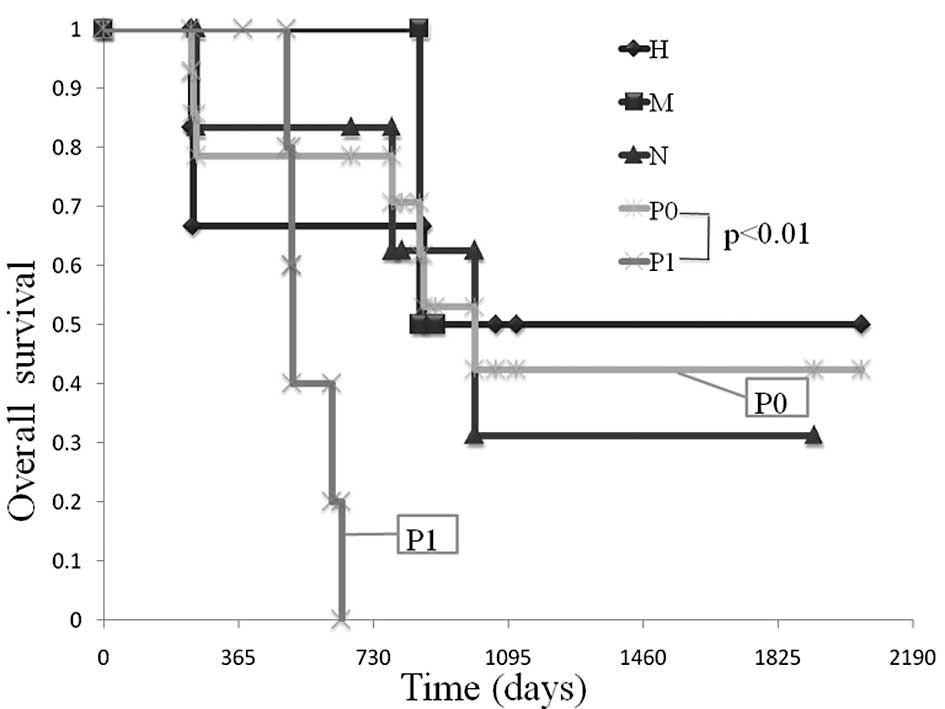
Comparison of OS in patients between the
curative resection and palliative groups
A 2- and 3-year survival was observed in 80 and
54.9% of patients, respectively, following macroscopic curative
surgery. In the palliative group, the median OS was 510 days, but a
3-year survival was not observed (Fig.
2).
Comparison of OS in patients between the
partial response and stable disease group
In the partial response group, the median OS was 865
days and a 3-year survival was observed in 37% of patients.
One-year survival was not observed in the stable disease group. OS
in the partial response group was statistically more prolonged than
the stable disease group (Fig.
3).
Comparison of OS in patients amonng the
various unresectable factors (liver metastasis, H factor;
peritoneal dissemination, P factor; lymph node metastasis, N
factor)
A comparison was made of OS among the various
unresectable factors. The median OS in patients with the H factor
was 865 days, while that in patients with the P factor was 510 days
(Fig. 4).
Discussion
Various treatment regimens have been developed
(5,6,9) and
have improved the survival of patients with advanced or recurrent
gastric cancer (21). The rationale
for the design of the combination therapy of docetaxel and S-1 was
the significant laboratory and clinical anti-tumor activity of both
docetaxel and 5-fluorouracil in gastric cancer, their synergistic
activity in vivo and the relative lack of overlapping
toxicities (22). Takahashi et
al reported increased anti-tumor activity in combination
therapy with docetaxel and S-1 using gastric cancer xenografts
(23). Wada et al reported
that docetaxel and S-1 combination therapy showed synergistic
effects by modulating the expression of the metabolic enzymes of
5-fluorouracil, including thymidylate synthase, dihydropyrimidine
dehydrogenase and orotate phosphoribosyl transferase in human
gastric cell lines (24). In a
phase II study of docetaxel and S-1 combination therapy, the median
time to tumor progression was 7.3 months (95% CI, 4.3–10.0 months)
(15).
Although progress in chemotherapy has resulted in
long-term survival, a number of patients require treatment changes
or a reduction in dose levels due to drug resistance or adverse
events. Such patients are required to change to another regimen
(second- or third-line).
When chemotherapy has produced transient tumor
regression and curative surgery may be accomplished, curative
resection in selected patients is occasionally associated with
prolonged survival. This type of surgery is referred to as adjuvant
surgery. The difference between adjuvant surgery and neoadjuvant
chemotherapy is their respective indications.
Neoadjuvant (preoperative) chemotherapy is an
investigational option. Its rationale is based on the difficulty of
performing an R0 resection in patients with locally advanced tumors
and the high risk of micrometastatic disease in these patients.
Neoadjuvant treatments aim to: i) downstage the primary tumor,
resulting in a higher R0 resection rate, and ii) simultaneously
treat micrometastases at an early stage (25–28).
Although the basic treatment in cases of advanced
gastric cancer is chemotherapy, it is insufficient. Additionally,
adjuvant surgery is preferred for patients with a favorable
response to chemotherapy.
The timing of such surgery may occur at the point
when the tumor is reduced, but prior to the appearance of drug
resistance. Empirically speaking, in gastric cancer, metastasis
occurs immediately after surgery, at the time of drug
resistance.
This study aimed to evaluate the efficacy of
adjuvant surgery after response to the chemotherapy for advanced
gastric cancer. The OS of patients in the partial response and
curative resection groups was prolonged. The survival of patients
with H or N factor was also prolonged, if they received curative
surgery. However, the survival of patients with P factor was not
prolonged. Adjuvant surgery is effective in gastric cancer patients
diagnosed as stage IV in the case of liver or distant lymph node
metastasis, but not peritoneal dissemination.
To prove the efficacy of adjuvant surgery, a
randomized controlled study is necessary. Numerous obstacles remain
to be addressed regarding the selection of combination drugs (S-1
plus CDDP, S-1 plus docetaxel and docetaxel plus CDDP plus
5-fluorouracil), the timing of adjuvant surgery and the selection
of chemotherapy after surgery.
References
|
1
|
Kamangar F, Dores GM and Anderson WF:
Patterns of cancer incidence, mortality, and prevalence across five
continents: defining priorities to reduce cancer disparities in
different geographic regions of the world. J Clin Oncol.
24:2137–2150. 2006. View Article : Google Scholar
|
|
2
|
Inoue M and Tsugane S: Epidemiology of
gastric cancer in Japan. Postgrad Med J. 81:419–424. 2005.
View Article : Google Scholar
|
|
3
|
Murad AM, Santiago FF, Petroianu A, Rocha
PR, Rodrigues MA and Rausch M: Modified therapy with
5-fluorouracil, doxorubicin, and methotrexate in advanced gastric
cancer. Cancer. 72:37–41. 1993. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Glimelius B, Ekstrom K, Hoffman K, et al:
Randomized comparison between chemotherapy plus best supportive
care with best supportive care in advanced gastric cancer. Ann
Oncol. 8:163–168. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
MacDonald JS, Schein PS, Woolley PV, et
al: 5-fluorouracil, doxorubicin, and mitomycin (fam) combination
chemotherapy for advanced gastric cancer. Ann Intern Med.
93:533–536. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Findlay M, Cunningham D, Norman A, et al:
A phase II study in advanced gastro-esophageal cancer using
epirubicin and cisplatin in combination with continuous infusion
5-fluorouracil (ECF). Ann Oncol. 5:609–616. 1994.PubMed/NCBI
|
|
7
|
Ohtsu A, Shimada Y, Shirao K, et al:
Randomized phase III trial of fluorouracil alone vs. fluorouracil
plus cisplatin vs uracil and tegafur plus mitomycin in patients
with unresectable, advanced gastric cancer: the Japan Clinical
Oncology Group Study (JCOG9205). J Clin Oncol. 21:54–59. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Koizumi W, Narahara H, Hara T, et al: S-1
plus cisplatin vs. S-1 alone for first-line treatment of advanced
gastric cancer (SPIRITS trial): A phase III trial. Lancet Oncol.
9:215–221. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Boku N, Yamamoto S, Fukuda H, et al:
Fluorouracil vs. combination of irinotecan plus cisplatin vs S-1 in
metastatic gastric cancer: a randomised phase 3 study. Lancet
Oncol. 10:1063–1069. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Einzig AI, Neuberg D, Remick SC, et al:
Phase II trial of docetaxel (taxotere) in patients with
adenocarcinoma of the upper gastrointestinal tract previously
untreated with cytotoxic chemotherapy: the Eastern Cooperative
Oncology Group (ECOG) results of protocol e1293. Med Oncol.
13:87–93. 1996. View Article : Google Scholar
|
|
11
|
Thuss-Patience PC, Kretzschmar A and
Reichardt P: Docetaxel in the treatment of gastric cancer. Future
Oncol. 2:603–620. 2006. View Article : Google Scholar
|
|
12
|
Fushida S, Fujimura T, Oyama K, Yagi Y,
Kinoshita J and Ohta T: Feasibility and efficacy of preoperative
chemotherapy with docetaxel, cisplatin and S-1 in gastric cancer
patients with para-aortic lymph node metastases. Anticancer Drugs.
20:752–756. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Zang DY, Yang DH, Kim MJ, et al:
Dose-finding study of docetaxel, oxaliplatin, and S-1 for patients
with advanced gastric cancer. Cancer Chemother Pharmacol.
64:877–883. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yoshida K, Hirabayashi N, Takiyama W, et
al: Phase I study of combination therapy with S-1 and docetaxel
(TXT) for advanced or recurrent gastric cancer. Anticancer Res.
24:1843–1851. 2004.PubMed/NCBI
|
|
15
|
Yoshida K, Ninomiya M, Takakura N, et al:
Phase II study of docetaxel and S-1 combination therapy for
advanced or recurrent gastric cancer. Clin Cancer Res.
12:3402–3407. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Suzuki T, Yoshida K, Tanabe K, et al:
Three advanced gastric cancer patients successfully treated by
combination therapy of docetaxel and TS-1. Gan To Kagaku Ryoho.
32:509–513. 2005.PubMed/NCBI
|
|
17
|
Tsutani Y, Ohara M, Suzuki T, et al:
Docetaxel and S-1 as a first-line treatment in patients with
advanced or recurrent gastric cancer. Anticancer Res. 29:2775–2779.
2009.PubMed/NCBI
|
|
18
|
Eisenhauer EA, Therasse P, Bogaerts J, et
al: New response evaluation criteria in solid tumours: revised
RECIST guideline (version 1.1). Eur J Cancer. 45:228–247. 2009.
View Article : Google Scholar
|
|
19
|
Japanese Gastric Cancer Association.
Japanese classification of gastric carcinoma - 2nd english edition.
Gastric Cancer. 1:10–24. 1998. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Trotti A, Colevas AD, Setser A, et al:
CTCAE v3.0: development of a comprehensive grading system for the
adverse effects of cancer treatment. Semin Radiat Oncol.
13:176–181. 2003. View Article : Google Scholar
|
|
21
|
Tanabe K, Yoshida K, Hamai Y, et al:
Clinical study of TS-1 for inoperative and recurrent gastric cancer
and evaluation of long survival cases. Gan To Kagaku Ryoho.
32:1145–1148. 2005.PubMed/NCBI
|
|
22
|
Van Den Neste E, de Valeriola D, Kerger J,
et al: A phase I and pharmacokinetic study of docetaxel
administered in combination with continuous intravenous infusion of
5-fluorouracil in patients with advanced solid tumors. Clin Cancer
Res. 6:64–71. 2000.PubMed/NCBI
|
|
23
|
Takahashi I, Emi Y, Kakeji Y, Uchida J,
Fukushima M and Maehara Y: Increased antitumor activity in combined
treatment TS-1 and docetaxel. A preclinical study using gastric
cancer xenografts. Oncology. 68:130–137. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wada Y, Yoshida K, Suzuki T, et al:
Synergistic effects of docetaxel and S-1 by modulating the
expression of metabolic enzymes of 5-fluorouracil in human gastric
cancer cell lines. Int J Cancer. 119:783–791. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Koizumi W, Tanabe S, Saigenji K, et al:
Phase I/II study of S-1 combined with cisplatin in patients with
advanced gastric cancer. Br J Cancer. 89:2207–2212. 2003.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Nakajima T, Ota K, Ishihara S, et al:
Combined intensive chemotherapy and radical surgery for incurable
gastric cancer. Ann Surg Oncol. 4:203–208. 1997. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Chua YJ and Cunningham D: The UK NCRI
MAGIC trial of perioperative chemotherapy in resectable gastric
cancer: implications for clinical practice. Ann Surg Oncol.
14:2687–2690. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Alexander HR, Grem JL, Pass HI, et al:
Neoadjuvant chemotherapy for locally advanced gastric
adenocarcinoma. Oncology (Williston Park). 7:37–53. 1993.PubMed/NCBI
|