Introduction
Ovarian carcinoma, the third highest morbidity among
gynecological cancers, still has the highest mortality (1), despite current strategic management
which has substantially improved the median survival rate over the
past few decades. Two thirds of ovarian cancer patients have a low
5-year survival rate (~30%) (1),
due to late diagnosis with peritoneal metastasis. The essential
steps for metastasis include proliferation, adhesion to the
peritoneum and migration into the stroma (2). Current management of ovarian cancer is
cytoreductive surgery followed by chemotherapy (3), but yields an unsatisfactory
prognosis.
Metformin has been used as an anti-hyperglycaemic
drug for type-2 diabetes (4) with a
safe tolerability profile, but also has a potential anti-tumour
effect, since a significant reduction in the risk of malignancy has
been observed in diabetic patients treated with metformin (5,6).
Metformin inhibits proliferation of breast (7), prostate (8), ovarian (9) and endometrium (10) malignant cells in vitro, which
may be mediated via upregulation of AMPK activity (11). Furthermore, metformin significantly
reduces growth of inoculated human p53-deficient breast cancer
cells in nude mice (12), and
reduces remission of breast cancer stem cells (13). However, it is still unclear if
metformin can inhibit the growth and metastasis of ovarian
cancer.
In this study, the ability of metformin to inhibit
ovarian cancer, particularly adhesion, invasion and migration was
studied in vitro was investigated. Furthermore, nude mice
inoculated with the ovarian cancer cell line were evaluated by
disease score, extent of neovascularisation and macrophage
infiltration in the inoculated region, and location and extent of
metastatic sites. These data support the clinical efficacy of
metformin therapy for ovarian cancer and raise potential new lines
of research investigating analogues of this drug for use as
anti-cancer agents.
Materials and methods
Cell culture
Human ovarian SKOV3 and HO-8910PM cell lines with
high-metastasis potential (14),
obtained from Chinese Academy of Sciences, were cultured in
RPMI-1640 medium (Gibco), supplemented with 10% FBS (Gibco) using
standard culture conditions (humidified 5% CO2 at
37°C).
Cell proliferation analysis
SKOV3 and HO-8910PM cell proliferation, following
metformin (Sigma) treatment for 24, 48 or 72 h, was assessed by
Cell Counting kit-8 (CCK-8) (Dojindo). Briefly, 3×103
cells, seeded into 96-well plates, were treated with metformin (0,
1, 5, 10, 20, 50, 100 mM). After 24, 48 and 72 h, 10 μl CCK-8 was
added to each well at 37°C for 2 h. The supernatant absorbance (450
nm) from each well was measured, using MK3 micro-plate reader
(Themo). The proliferation rate (%) was calculated by comparing the
absorbance of metformin-treated versus mock-treated cells.
Apoptosis analysis
Cell apoptosis was determined using an annexin
V-FITC kit (KeyGen). Cells (~1×105) were seeded in 6-cm
dishes and treated with metformin (0, 5, 10 and 20 mM) for 24 h
prior to analysis. The floating and adherent cells (trypsinized)
were analysed with an EPICS-XL flow cytometer (Becton-Dickinson)
after incubation with annexin V-FITC and PI at room temperature
(RT) for 15 min. Cells positive for early apoptosis (annexin V
stained only) and for late apoptosis (annexin V and PI stained)
were determined. Viable cells were negative for both annexin V and
PI.
Adhesion analysis
Matrigel (BD Biosciences) dry-coated 96-well plates
were blocked with 1% BSA for 1 h. Metformin pretreated (0, 5, 10,
20 mM for 24 h) cells (2×104) were allowed to adhere to
the coated well for 1 h at 37°C, and the non-adherent cells were
removed by 5 washings with cold PBS. The adherent cells in each
well were measured using the CCK-8 kit. The adhesion rate (%) was
calculated by comparing the adherent cell numbers following
metformin versus mock treatment.
Invasion analysis
SKOV3 and HO-8910PM cells were collected by
trypsinization and resuspended in serum-free medium
(3×105/ml). Cells (1×105/ml) in serum-free
medium were placed into the upper wells, coated with 50 μl of
Matrigel (1:6 dilution) of the Boyden chamber (8 μm pore size,
Millipore) with metformin (0, 5, 10 and 20 mM). Medium containing
20% FBS was added to the lower chamber. Cell invasion into the
Matrigel was determined following 24 h under standard culture
conditions. The membrane containing invading cells was methanol
fixed and H&E stained, after removing non-invading cells on the
upper side of the membrane with cotton swabs.
Migration analysis
Cell migration was determined using a wound-healing
motility assay (2). SKOV3 and
HO-8910PM cells (4×104) were added to a 6-well plate
with metformin (0, 5, 10 and 20 mM) and incubated for 24 h. After
scratching with a plastic cell scraper of fixed width, the
monolayer of cells was washed 3 times with PBS to remove the
detached cells. The remaining adherent cells were incubated using
standard culture conditions. Cell migration was determined by
monitoring reduction in the width of the scaped zone at various
time-points for up to 18 h, to avoid cell doubling (average
doubling time ~22 h). The migration rate (%) was calculated from
the migration distance at different dosages compared to
mock-treatment.
Metastasis assay in vivo
Five-weeks old female athymic BALB/c nude mice,
obtained from the Chinese Academy of Sciences (15), were housed under conventional
laboratory conditions with chow and water available ad
libitum. Experiments adhered to the guidelines of the Shanghai
Jiaotong University, Animal Ethics Committee. Nude mice were
inoculated on their back with 2×106 HO-8910PM cells in
100 μl of PBS, to establish a viable solid tumour. The established
tumour was dissected into 1 mm3 pieces and one piece was
implanted orthotopically on the right ovarian capsule of the
recipient nude mouse. A total of 14 female nude mice (5-weeks old)
were randomly divided into 2 groups. Two weeks post-implantation,
metformin (Glucophage, Squibb, 250 mg/kg body weight) in normal
saline (NS), was administered i.p. every other day for 4 weeks in
the metformin-treated group, whereas the mock-treated group
received NS only. The disease score was calculated by adding body
weight, activity and responsiveness, mobility, ascites and gross
tumour size together (16). The
sizes of the inoculated and metastatic tumours were determined by
mean tumour volume [MTV = XY2/2, where X is the shorter
and Y is the longer diameter (mm) of orthogonal measurements]
(15), after sacrificing the mice
at 8 weeks post-implantation. Histopathology of the original and
metastatic tumours was performed in formalin-fixed, wax-embedded
and H&E-stained sections (5 μm).
Immunohistochemistry analysis
Paraffin sections (5 μm) were incubated with rat
anti-F4/80 (WEHI) or vWF (Dako) antibodies followed by a
horseradish peroxidise-conjugated rabbit anti-rat antibody (Dako),
as described (17). All sections
were then visualized with 3,3′-diaminobenzidine (Dako), and lightly
counterstained with hematoxylin. Immunohistochemical
quantifications were performed on digitized images using image
analysis software Image-Pro Plus. The presence of
monocytes/macrophages was assessed using anti-F4/80 antibody and
the proportion of positive cells was determined by [100% × (number
of positive nuclei/total number of nuclei)].
Statistical analyses
Each experiment was done in triplicate. All data are
expressed as means ± standard error or division of the mean (SEM or
SD). Data analysis was carried out by one-way and two-way ANOVA
using GraphPad Prism 4.0. P<0.05 was considered statistically
significant.
Results
Metformin inhibits proliferation of human
ovarian cancer cells
Metformin inhibited the proliferation of SKOV3 cells
in a dose-dependent manner (0–100 mM) at 24, 48 and 72 h in
vitro (Fig. 1A). Significantly
greater inhibition of proliferation of SKOV3 cells was observed
following 48- and 72-h metformin-treatment in the range 5–50 mM,
compared to 24-h treatment. The inhibition at 48 or 72 h reached
plateau at 25 mM dose. A similar pattern was observed in HO-8910PM
cells following metformin-treatment, but a significant difference
between 24 h and 48/72 h was observed at 5 mM only (Fig. 1B). To evaluate whether the
metformin-induced reduction in proliferation was due to apoptosis,
cells were assessed for annexin V expression using flow cytometry.
No apoptosis was detected in SKOV3 cells following 24-h
metformin-treatment (0–20 mM) (Fig.
1C–F). The IC50 for metformin for SKOV3 or HO-8910PM
cells was 20.62 or 16.67 mM over 24 h, respectively
(P<0.01).
Metformin inhibits adhesion, invasion and
migration of human ovarian cancer cell lines
Metformin significantly inhibited SKOV3 and
HO-8910PM cell adhesion in a dose-dependent manner, by ~50% at the
IC50 (20 mM), compared to the mock-treated groups
(Fig. 2A). Metformin-treatment
(0–20 mM) resulted in a remarkable dose-dependent decline in the
invasive ability of the ovarian cancer cells, ~60% and ~40% in
SKOV3 and HO-8910PM cells, respectively, at the IC50 of
20 mM (Fig. 2B). Furthermore,
metformin significantly inhibited the migration in SKOV3 and
HO-8910PM cells in a dose-dependent manner (Fig. 2C), reaching ~80% inhibition at 20 mM
concentration.
Metformin inhibits ovarian cancer growth
and metastasis in vivo
All mice developed bloody ascites two weeks
post-orthotopic tumour implantation, in both the metformin (250
mg/kg) and mock-treated groups (Fig. 3A
and B). One mouse died due to extreme cachexia in the
mock-treated group at 6 weeks after implantation; whereas all 7
mice survived in the metformin-treated group. The size of the
orthotopic implanted ovarian cancer increased substantially in both
metformin and mock-treated groups (black arrow, Fig. 3C and D). No significant difference
in the size of the orthotopic tumour was observed between these two
groups [2095±768.2 mm3 (7 mice) vs 2728±987.5
mm3 (6 mice), metformin vs mock-treatment].
The extent of metastases was greater in the
mock-treated group compared to the metformin-treated group. There
were peritoneal macroscopic metastases (liver, intestine, spleen
and pancreas) (white arrows, Fig.
3D) in all mock-treated mice (6/6). The sites of metastasis
were the peritoneum (5/6), contra-lateral ovary (3/6), mesentery
(2/6), intestine (5/6), liver (4/6), kidney (1/6), spleen (1/6),
diaphragm (3/6), and mesenteric lymph nodes (1/6). By contrast, in
the metformin-treated group, fewer metastases were detected; to the
peritoneum (2/7), intestine (4/7) and mesentery (2/7). The number
and size of the intestinal metastases was noted to be fewer and
smaller than that of the mock-treated group (Fig. 3C and D).
A trend towards decreased body weight in the
mock-treatment mice was observed over 35 days, due to cachexia;
whereas there was slight body weight gain in the
metformin-treatment mice during the same period. No significant
difference in body weight was observed between the mock and
metformin-treated groups (Fig. 3E).
The disease score was >1.5-fold higher in the mock-treatment
group compared to the metformin-treated group at 8 weeks
post-orthotopic implantation (P<0.01) (Fig. 3F).
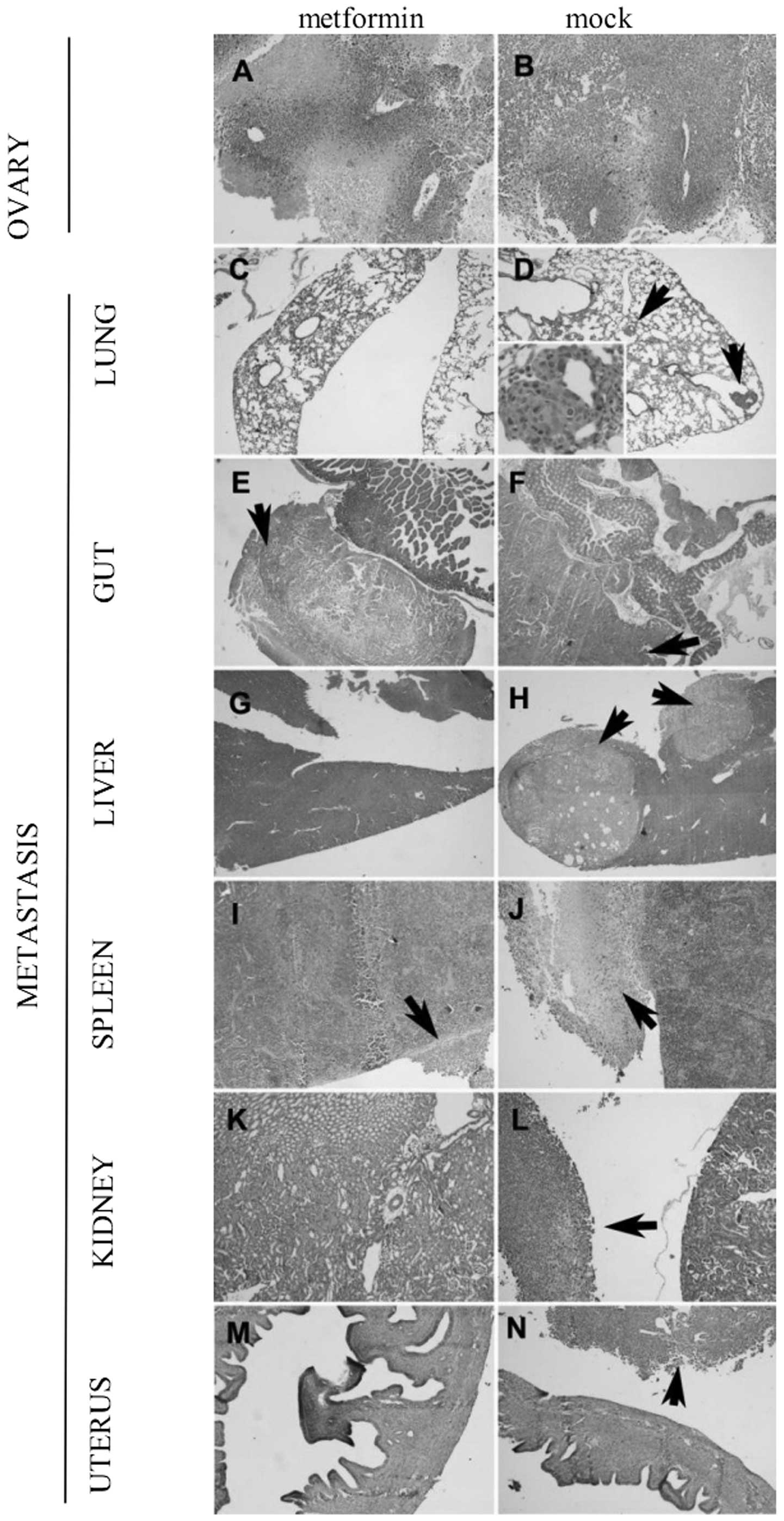
Histopathological analysis
The orthotopic tumours demonstrated similar
histopathological features to malignant papillary serous
adenocarcinoma of the ovary (Fig. 4A
and B). Microscopic, but not macroscopic, metastatic lesions
were observed in the lungs of mock-treated animals (arrow, Fig. 4D), but not in the metformin-treated
mice (Fig. 4C). There were
metastatic lesions detected in the intestine from both mock- and
metformin-treated animals (Fig. 4E and
F). Deeper invasive lesions were detected in the mock-treated
group (through the muscularis propria to the mucosa, white arrow,
Fig. 4F), compared to the
metformin-treated group (limited to the serosa, white arrow,
Fig. 4E). There were metastatic
lesions in the liver from the mock-treated mice (arrow, Fig. 4H), while no liver metastasis was
detected in the livers from the metformin-treated animals (Fig. 4G), suggesting that inability to
penetrate the muscularis propria in the metformin-treated group
inhibited portal venous metastasis. Larger capsular metastases were
observed on the spleen from the mock-treated animals (arrow,
Fig. 4J) compared to the smaller
splenic capsular metastases in the metformin-treated group
(Fig. 4I). No directly attached
metastatic lesions were observed on the kidney (Fig. 4K and L) or the uterus (Fig. 4M and N) from both mock- and
metformin-treated groups.
Metformin decreases tumour
neovascularisation and macrophage infiltration
Using immunohistochemistry, metformin was found to
inhibit neovascularisation (vWF expression), in intestinal and
hepatic metastases. vWF expression in the gut and liver from the
metformin-treated group was ~75% (P<0.001) and ~40% (P<0.05)
lower than the mock-treated group, respectively (Fig. 5A–F). Inhibition of macrophage
infiltration was also observed in the metformin group, compared to
the mock-treatment group, in the lung (~60%) and the liver (~90%)
(Fig. 5A–F).
Discussion
The rationale for our current experiment is based on
reports that metformin possesses anti-tumour capabilities (8–11). The
current standard therapy for ovarian cancer is combined
chemotherapy following surgery. However, development of
drug-resistance and other side effects compromise the effects of
chemotherapy. Metformin is an anti-hyperglycemia drug in use for
over 60 years with few adverse effects (4). Our study demonstrated that metformin
inhibited proliferation, adhesion, invasion and migration in human
epithelial ovarian cancers (HO-8910PM and SKOV3 cell lines) in
vitro, in a dose- and time-dependent manner. Furthermore,
metformin inhibited growth and metastasis of these ovarian cancers
in vivo in a nude mouse model.
Effective inhibition of SKOV3 cell proliferation
in vitro occurred at an IC50 of ~20 mM metformin,
without necrosis or apoptosis in these cells. These data suggest
that this dose has no direct cytotoxicity, suggesting that this
dose is at a therapeutic level. This is consistent with findings in
breast and prostate cancer cell lines following
metformin-treatment, where no cell death was observed, but tumour
cells were arrested at the G0/G1 stage (7,8).
Our data are the first to demonstrate that metformin
inhibits growth and metastasis of ovarian cancer in vivo, in
a nude-mouse model. Previous reports have shown that metformin
inhibited growth of inoculated breast cancer cells in nude mice,
especially in a p53-deficient- (12) and in an ERα- MDA-MB-435
human breast cancer cell line (18). Interestingly, the dosages of
metformin chosen for the breast cancer experiments were 2- and
6-fold higher, respectively, than that used in the present study.
The dosage chosen in the present study was based on the recommended
maximal therapeutic daily dose in human, i.e., 2,550 mg/60 kg. The
dosage differences may be due to the different forms of cancer,
and/or may reflect different therapeutic pathways. The precise
underlying mechanisms are worthy of further investigation.
A non-significant trend towards body-weight loss in
the mock-treated, compared to the metformin-treated, group was
observed. However, the overall condition of the mice was more
accurately quantified by the disease score, which was found to be
higher in the mock-treated group, consistent with metformin acting
as an effective anti-cancer therapy. The failure to observe a
significant difference in body-weight between the metformin- and
mock-treated groups may be due to greater ascites generation during
the growth and metastasis of ovarian cancer in the mock-treatment
group. Qualitatively, more severe cachexia was observed in the
mock-treated group.
Angiogenesis is another objective factor predicting
prognosis for overall and disease-free survival in advanced ovarian
cancer (19). We showed reduced
expression of vWF, reflecting decreased neovascularisation, in
metastatic tissues following metformin-treatment, compared to
mock-treatment. The suppressed vWF level correlates well with
reduced size and number of metastases.
Macrophages contribute to tumour dissemination by
upregulating tumour cell adhesion molecules, releasing invasive
proteases and promoting angiogenesis (20,21).
We showed metformin significantly suppressed macrophage
infiltration in metastatic tissues compared with mock-treatment,
correlating with the reduced size and number of liver and lung
metastasis. Thus, macrophage-mediated metastasis appears to play an
important role in dissemination of ovarian cancer, which may also
be a useful target for future therapeutic intervention. In
conclusion, metformin inhibits growth and metastasis of ovarian
cancer, by down-regulating pFAK and MMP-2 observed in vitro,
and by reduction of macrophage infiltration and vWF expression
observed in vivo. Future experiments will focus on the
underlying mechanism/s of the anti-tumour effects of metformin, and
its potential to move forward into clinical application for the
management of ovarian cancer.
Acknowledgements
This study was supported by grants from Hundred
Talent program, Shanghai Jiaotong University School of Medicine;
Overseas Exchange PhD Fellowship, Shanghai Jiaotong University; The
Bureau of Science and Technology; Shanghai Municipal (08XD14027);
the National Natural Science Foundation (30772306), and an Award of
Outstanding Young Academics Foundation of Shanghai Universities
(JDY08042). The authors acknowledge assistance from the
Histopathology Laboratory, The Discipline of Pathology, The
University of Sydney.
References
|
1
|
Jemal A, Siegel R, Ward E, Hao Y, Xu J and
Thun MJ: Cancer statistics, 2009. CA Cancer J Clin. 59:225–249.
2009. View Article : Google Scholar
|
|
2
|
Zhang J and Wang B: Arsenic trioxide
(As(2)O(3)) inhibits peritoneal invasion of ovarian carcinoma cells
in vitro and in vivo. Gynecol Oncol. 103:199–206. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Gubbels JA, Claussen N, Kapur AK, Connor
JP and Patankar MS: The detection, treatment, and biology of
epithelial ovarian cancer. J Ovarian Res. 3:82010. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Bailey CJ and Turner RC: Metformin. N Engl
J Med. 334:574–579. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Evans JM, Donnelly LA, Emslie-Smith AM,
Alessi DR and Morris AD: Metformin and reduced risk of cancer in
diabetic patients. BMJ. 330:1304–1305. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Libby G, Donnelly LA, Donnan PT, Alessi
DR, Morris AD and Evans JM: New users of metformin are at low risk
of incident cancer: a cohort study among people with type 2
diabetes. Diabetes Care. 32:1620–1625. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Zhuang Y and Miskimins WK: Cell cycle
arrest in Metformin treated breast cancer cells involves activation
of AMPK, downregulation of cyclin D1, and requires
p27Kip1 or p21Cip1. J Mol Signal. 3:182008.
View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Ben Sahra I, Laurent K, Loubat A, et al:
The antidiabetic drug metformin exerts an antitumoral effect in
vitro and in vivo through a decrease of cyclin D1 level. Oncogene.
27:3576–3586. 2008.PubMed/NCBI
|
|
9
|
Gotlieb WH, Saumet J, Beauchamp MC, et al:
In vitro metformin anti-neoplastic activity in epithelial ovarian
cancer. Gynecol Oncol. 110:246–250. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Cantrell LA, Zhou C, Mendivil A, Malloy
KM, Gehrig PA and Bae-Jump VL: Metformin is a potent inhibitor of
endometrial cancer cell proliferation - implications for a novel
treatment strategy. Gynecol Oncol. 116:92–98. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Zakikhani M, Dowling R, Fantus IG,
Sonenberg N and Pollak M: Metformin is an AMP kinase-dependent
growth inhibitor for breast cancer cells. Cancer Res.
66:10269–10273. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Buzzai M, Jones RG, Amaravadi RK, et al:
Systemic treatment with the antidiabetic drug metformin selectively
impairs p53-deficient tumor cell growth. Cancer Res. 67:6745–6752.
2007. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Hirsch HA, Iliopoulos D, Tsichlis PN and
Struhl K: Metformin selectively targets cancer stem cells, and acts
together with chemotherapy to block tumor growth and prolong
remission. Cancer Res. 69:7507–7511. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Shenhua X, Lijuan Q, Hanzhou N, et al:
Establishment of a highly metastatic human ovarian cancer cell line
(HO-8910PM) and its characterization. J Exp Clin Cancer Res.
18:233–239. 1999.PubMed/NCBI
|
|
15
|
Zhang S, Lin QD and Di W: Suppression of
human ovarian carcinoma metastasis by the metastasis-suppressor
gene, BRMS1. Int J Gynecol Cancer. 16:522–531. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Bao S, Carr ED, Xu YH and Hunt NH:
Gp91(phox) contributes to the development of experimental
inflammatory bowel disease. Immunol Cell Biol. 89:853–860. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Xu Y, Hunt NH and Bao S: The role of
granulocyte macrophage-colony-stimulating factor in acute
intestinal inflammation. Cell Res. 18:1220–1229. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Phoenix KN, Vumbaca F and Claffey KP:
Therapeutic metformin/AMPK activation promotes the angiogenic
phenotype in the ERalpha negative MDA-MB-435 breast cancer model.
Breast Cancer Res Treat. 113:101–111. 2009. View Article : Google Scholar
|
|
19
|
Brem S, Cotran R and Folkman J: Tumor
angiogenesis: a quantitative method for histologic grading. J Natl
Cancer Inst. 48:347–356. 1972.PubMed/NCBI
|
|
20
|
Jonjic N, Peri G, Bernasconi S, et al:
Expression of adhesion molecules and chemotactic cytokines in
cultured human mesothelial cells. J Exp Med. 176:1165–1174. 1992.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Coussens LM and Werb Z: Inflammation and
cancer. Nature. 420:860–867. 2002. View Article : Google Scholar : PubMed/NCBI
|