Introduction
Canine hepatocellular carcinoma (cHCC) accounts for
50% of primary liver tumors (1). cHCC
is classified clinically as massive, nodular, or diffuse; with ~60%
of cHCC patients categorized as massive, 30% as nodular and 10% as
diffuse (2). In addition, nodular and
diffuse cHCC exhibit a higher metastatic rate than massive tumors.
As a result of the low metastatic rate, patients with massive cHCC
demonstrate a good prognosis when treated surgically. However,
nodular and diffuse cHCC have a worse prognosis compared with
massive cHCC (3). For unresectable
massive cHCC, and for a part of nodular and diffuse cHCC cases,
transcatheter arterial embolization or transcatheter arterial
chemoembolization have been employed experimentally as minimally
invasive treatments, and partial therapeutic effects have been
reported (4). However, no curative
surgical treatment has been established for nodular and diffuse
cHCC to date.
Chemotherapy has generally been considered to have a
limited therapeutic effect in primary liver tumors. The presence of
P-glycoprotein in the tumors is suggested as a part of therapy
resistance mechanism (5). A previous
study reported that chemotherapy using mitoxantrone (MTX) resulted
in a partial response in a dog with cHCC (6). Although a response has been reported in a
dog following the empirical use of gemcitabine (GEM) (7), a retrospective study of 18 cHCC dogs with
GEM (4 massive, 10 nodular and 4 diffuse) concluded that the effect
of the therapy was worse than surgical treatment and that the
single use of GEM did not improve the survival rate in dogs with
cHCC (8). Considering these issues,
effective chemotherapy for cHCC has not yet been established.
A previous study indicated that cancer tissue is not
comprised of a single type of cells, but that cancer stem cells
(CSCs) maintain tumor function and morphology (9). CSCs are defined by their ability to
self-renew and to generate the heterogeneous lineages of cancer
cells that comprise the tumor (10).
Therefore, a novel therapeutic strategy targeted at CSCs has
recently been investigated. Similarly, a side population of
hepatocellular carcinoma stem cells (HCSCs) has been reported
(11) to be marked by cluster of
differentiation (CD) 133-positive (12,13),
CD90-positive (14), and CD44-positive
(15) cells. However, there are a
small number of reports for canine HCSCs (cHCSCs), such as CD90-
and CD44-positive (16,17).
Rhodamine 123 is a low-toxic fluorescent dye for
staining mitochondria and is used to determine mitochondrial
activity by flow cytometry (18). In
human medicine, rhodamine 123 is considered one of the markers that
identify hematopoietic stem cells (HPCs) (19) and renal carcinoma stem cells (20). However, to the best of our knowledge,
no previous studies have reported that rhodamine 123 characterizes
cHCSCs.
The aim of the current study was to determine cHCSCs
in a cHCC cell line using rhodamine 123 and flow cytometry.
Additionally, the various biological characteristics and
chemoresistance were compared between subpopulations of stem cells
with higher (RhoHi) and lower (RhoLo)
rhodamine expression.
Materials and methods
Cell line and culture
The CHCC cell line (AZACH) was purchased from Cosmo
Bio Co., Ltd., (Tokyo, Japan) and maintained in Eagle's minimum
essential medium (EMEM; Wako Pure Chemical Industries, Ltd., Osaka,
Japan) supplemented with Sigma-Aldrich 5% fetal bovine serum (FBS;
Merck KGaA, Darmstadt, Germany), amino acid supplement (GlutaMAX;
Thermo Fisher Scientific, Inc., Waltham, MA, USA), and
antibiotic-antimycotic agents (PSM; 100 U/ml penicillin, 100 µg/ml
streptomycin and 0.25 µg/ml amphotericin B, final concentrations,
all from Nacalai Tesque, Inc., Kyoto, Japan). They were
subsequently cultured for two days at 37°C in an atmosphere
containing 5% CO2.
Flow cytometry and cell sorting
The cells were enzymatically dissociated using
Accutase solution (Innovative Cell Technologies, Inc., San Diego,
CA, USA) after washing with phosphate-buffered saline (PBS). The
cells were resuspended with Dulbecco's PBS supplemented with 1% FBS
and 1 mM EDTA⋅3Na (Wako Pure Chemical Industries, Ltd.). Cells were
stained with a viability probe (Zombie NIR; BioLegend, Inc., San
Diego, CA, USA) to stain the dead cells. In addition, wells were
incubated with 0.1 µg/ml rhodamine 123 (Dojindo Molecular
Technologies, Inc., Kumamoto, Japan) at 37°C for 30 min and washed
twice with PBS. Subsequent to washing, the labeled cells were
analyzed using flow cytometry (Accuri C6; BD Biosciences, Franklin
Lakes, NJ, USA) and sorting was performed using a cell sorter
(SH800; Sony Biotechnology, Inc., Tokyo, Japan). A negative control
was run using DPBS without rhodamine 123. Data were analyzed using
FlowJo software (version 10.1; Tree Star, Inc., Ashland, OR,
USA).
Reverse transcription-quantitative
polymerase chain reaction (RT-qPCR)
Total RNA was extracted from the RhoHi
and RhoLo subpopulations incubated for 24 h after
sorting using a commercially available kit (miRNeasy Mini kit;
Qiagen, Tokyo, Japan). RT to single-strand cDNA was performed using
a commercially available kit (ReverTra Ace qPCR RT Master Mix with
gDNA Remover; Toyobo Co., Ltd., Osaka Japan) according to the
manufacturer's instructions. For qPCR, the Nanog primers (forward,
TGGAACAATCCGCTCCACAA and reverse, GATGGACTCCAGATCACCCATAGAA) and
templates were mixed with the SYBR Premix Ex TaqII (Takara Bio,
Inc., Otsu, Japan). DNA was amplified by 45 cycles of denaturation
for 5 sec at 95°C and annealing for 30 sec at 60°C using the
Thermal Cycler Dice Real-Time System II (Takara Bio, Inc.). Data
generated from each PCR reaction were analyzed using the Thermal
Cycler Dice Real-Time System version 2.10B (Takara Bio, Inc.). The
relative quantity of mRNA was normalized to that of hypoxanthine
phosphoribosyltransferase 1 (forward, GGAGCATAATCC AAAGATGGTCAA and
reverse, TCAGGTTTATAGCCA ACACTTCGAG). The data analysis was
performed using the 2−ΔΔCq method (21).
Cell proliferation assay
Cell proliferation was analyzed using WST-8 and a
Cell Counting Kit-8 (CCK-8; Dojindo Molecular Technologies, Inc.)
according to the manufacturer's instructions. Briefly, sorted
RhoHi and RhoLo subpopulations were seeded
into 96-well plates at 3×103 cells/well. Subsequently,
100 µl fresh medium containing 10 µl CCK-8 solution was added to
each well after 12, 24, 48 and 72 h. The absorbance at a wavelength
of 450 nm of each well was measured on an Epoch microplate
spectrophotometer (BioTek Instruments, Inc., Winooski, VT, USA)
followed by incubation at 37°C for 1 h. A total of six replicates
were prepared for each group.
Sphere formation assay
Sorted cells were seeded in ultra-low attachment
plates (Corning Inc., Corning, NY, USA) at a density of
1×106 cells/dish and cultured with EMEM supplemented
with 5% FBS, amino acid supplement (GlutaMAX) and
antibiotic-antimycotic agents (PSM; 100 U/ml penicillin, 100 µg/ml
streptomycin and 0.25 µg/ml amphotericin B, final concentrations)
for 3 days. Spheres were counted from 6 sites using a fluorescence
microscope (BZ-9000; Keyence Corporation, Osaka, Japan) for
quantitative analysis of sphere formation.
Chemoresistance assay
The cytotoxic effect in each subpopulation was
determined by WST-8 (CCK-8) according to the manufacturer's
instructions. Briefly, cells were seeded in 96-well plates at a
density of 3×103 cells/well with 0.1, 0.5, 1, 5, 10 and
50 µM of doxorubicin (DOX), MTX and GEM (Wako Pure Chemical
Industries, Ltd.). Following incubation for 24 h at 37°C in an
atmosphere containing 5% CO2, 100 µl fresh medium
containing 10 µl CCK-8 solution was added to each well, followed by
incubation at 37°C for 1 h. The absorbance at a wavelength of 450
nm of each well was measured on an Epoch microplate
spectrophotometer. A total of six replicates were prepared for each
group.
Statistical analysis
Statistical analysis was performed using GraphPad
Prism version 6.01 for Windows (GraphPad Software, Inc., La Jolla,
CA, USA). The results were expressed as means ± standard error.
Comparisons between two groups were performed using the independent
t-test. Multiple comparisons were performed with one-way ANOVA and
P<0.05 was considered to indicate a statistically significant
difference.
Results
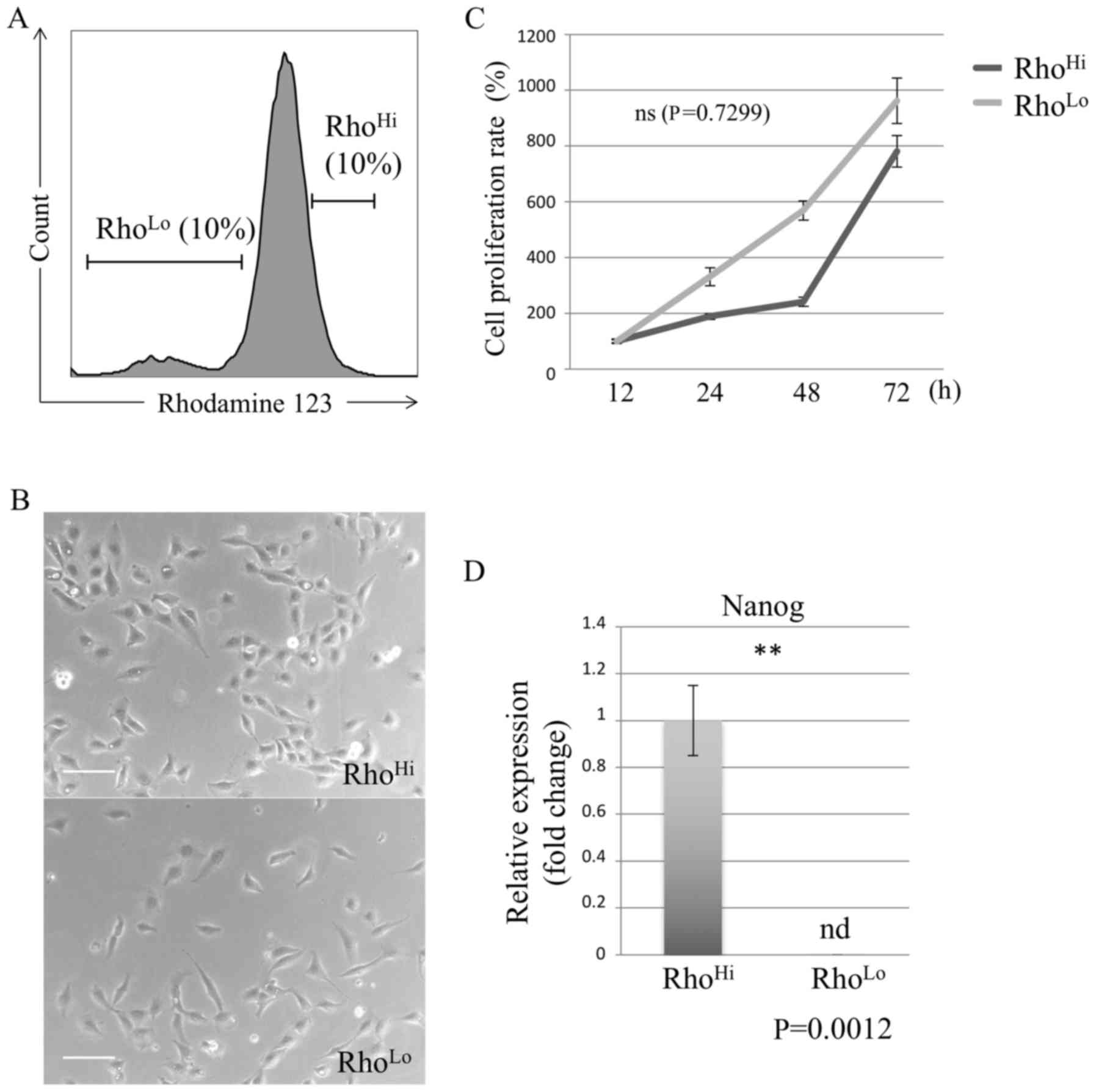
Rhodamine 123 staining and sorting of
cHCC
Flow cytometry of cultured cHCC excluding dead cells
confirmed that rhodamine was expressed. Cells labeled by rhodamine
were sorted by their expression of rhodamine into RhoHi
and RhoLo (Fig. 1A).
Subsequent to sorting, the RhoHi and RhoLo
subpopulations were incubated for 24 h, and the two subpopulations
exhibited a similar morphology (Fig.
1B).
Comparison of proliferation rate
To assess the difference in the proliferation
potential between the RhoHi and RhoLo
subpopulations, the proliferation rate was measured in each
subpopulation using the WST assay. As a result, the proliferation
potential of the RhoHi and RhoLo
subpopulations of cHCC was not identified to be significantly
different (Fig. 1C).
Comparison of Nanog expression
The gene expression of Nanog, a common type of stem
cell marker, was evaluated using RT-qPCR. The gene expression level
of Nanog in RhoHi was identified to be higher than that
in RhoLo (Fig. 1D).
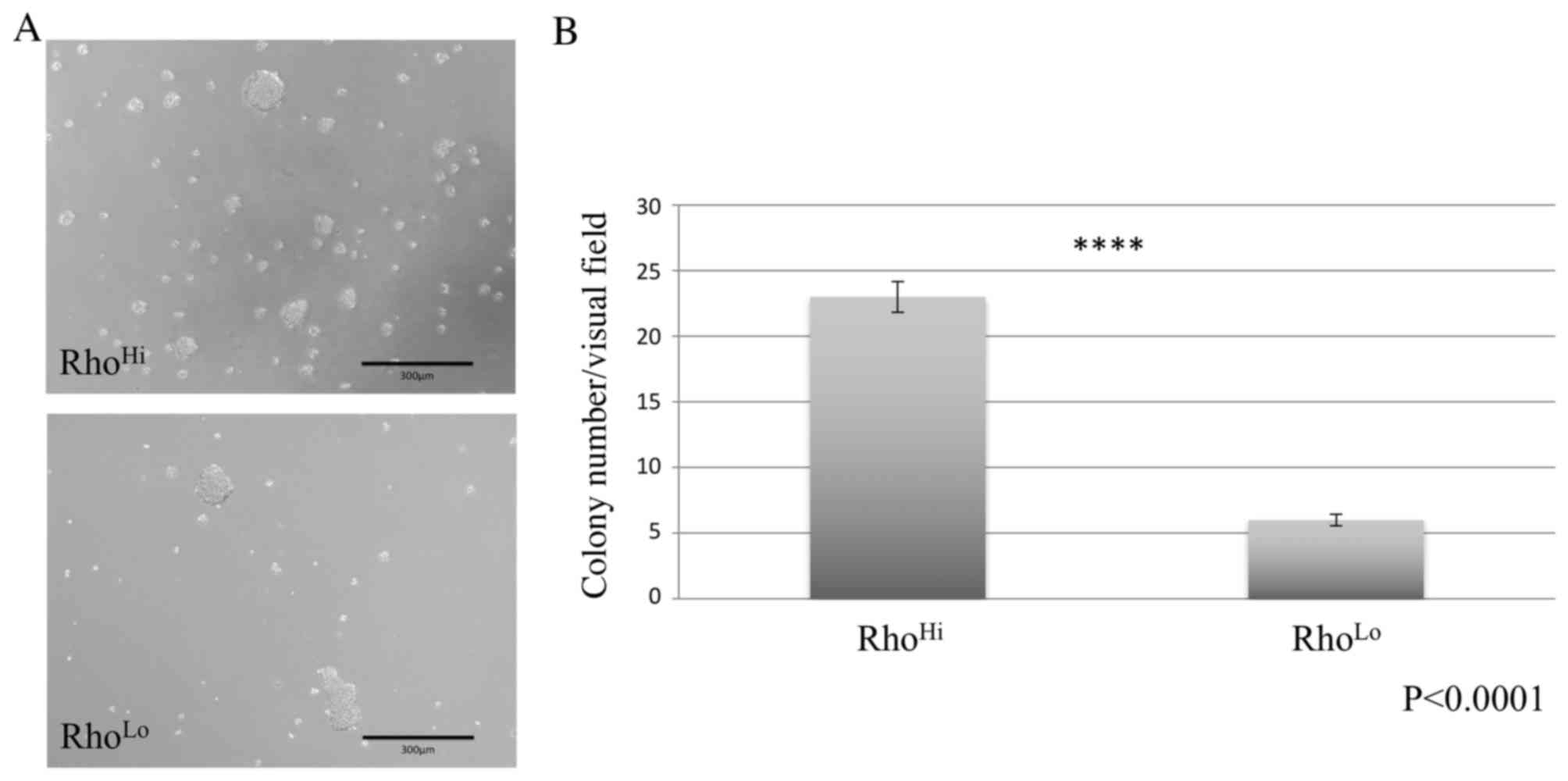
Comparison of sphere formation
A sphere formation assay was performed using
ultra-low attachment dishes and demonstrated the formation of tumor
spheres in cultures of RhoHi and RhoLo
subpopulations (Fig. 2A). However, the
RhoHi subpopulation exhibited a significantly higher
number of sphere formations per visual field than the
RhoLo subpopulation (Fig.
2B).
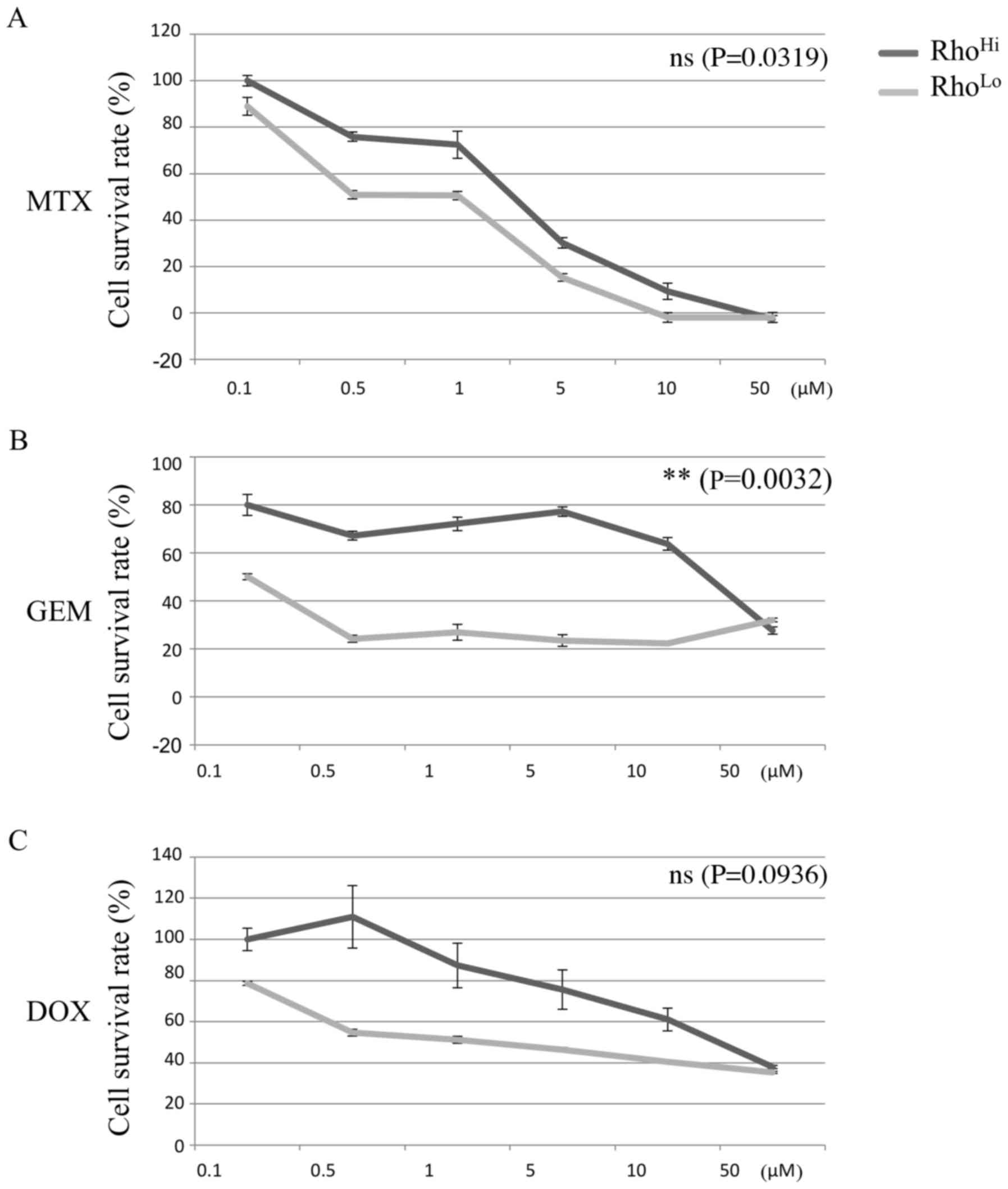
Comparison of chemoresistance
ability
Chemoresistance against MTX, GEM, and DOX was
determined for the RhoHi and RhoLo
subpopulations. RhoHi exhibited a higher survival rate
than RhoLo when GEM was administered (Fig. 3B). However, no significant difference
in the chemoresistance potential was identified between the
RhoHi and RhoLo subpopulations with MTX or
DOX administration.
Discussion
In the current study RhoHi demonstrated
higher Nanog expression level and sphere formation ability than
RhoLo. In addition, RhoHi exhibited greater
chemoresistance potential to GEM when compared with
RhoLo. However, no significant difference between
RhoHi and RhoLo regarding the proliferation
rate or chemoresistance against MTX and DOX was identified.
Rhodamine 123 is absorbed easily by living cells,
becoming concentrated in the mitochondria (22). Mitochondria are categorized as
intracellular organelles and functionally supply adenosine
triphosphate as a result of cell respiration and metabolism.
Previous studies propose that reactive oxygen species produced in
mitochondrial respiration are correlated with aging and the
formation of malignancies (23,24).
Furthermore, recent studies have revealed rhodamine to be a marker
of stem cells. However, identifying which stem cells display either
higher or lower expression levels of rhodamine is complicated.
Regarding adult stem cells, a few reports have identified HPC as
having a low expression of rhodamine 123 (19,25).
Conversely, in cancer cells, a previous study revealed that
subpopulations with a higher expression of rhodamine exhibited
higher proliferation, sphere formation, radio-resistance and tumor
differentiation potential than those with a lower expression of
rhodamine (26).
To the best of our knowledge, there is only one
report regarding HCSCs identified by rhodamine expression in humans
(27). In the present study, primary
HCCs were obtained from clinical patients; primary HCC were
compared with cells cultured with DOX and 5-fluorouracil, which
were regarded as HCSCs. HCSCs exhibited more stem cell markers,
sphere formation, and tumor differentiation and a lower level of
rhodamine expression than primary cells (27). The present study demonstrated that the
expression of rhodamine was a poor stem cell marker; however, it
did not examine chemoresistance or sphere-forming ability in the
RhoHi and RhoLo subpopulations (24). Additionally, cells from the clinical
patients cultured with DOX and 5-fluorouracil were regarded as
HCSCs. Thus, these were cells selected for their chemoresistance
and ability to proliferate well in dishes (24). As a result, RhoLo cells may
have been exhibited as being poorly differentiated.
Treatment for cHCC is limited, and poor prognoses
have been reported for cHCC with metastasis or diffusion through an
entire hepatic lobe, although massive cHCC has a good prognosis
when it is treated surgically. Thus, more effective treatments are
required for nodular and diffuse cHCC. A recent study in human
medicine have proposed a novel strategy for the treatment of cancer
targeted at CSCs, and notable results have been presented regarding
CSCs from basic and clinical trials (28). Veterinary medicine follows human
medicine, and CSCs have been reported from various types of solid
tumor and hematological malignancies (29–32). To the
best of our knowledge, the current study is the first to describe
CSCs identified by rhodamine in a canine cancer cell line, and it
may lead to basic studies regarding cHCSC-targeting treatment.
Further studies are required to reveal the mechanism by which
mitochondrial activity affects the stem cell characteristics and
chemoresistance of cHCC.
Acknowledgements
The present study was partly supported by the Japan
Society for the Promotion of Science KAKENHI (grant no.
26893172).
References
|
1
|
Withrow SJ, Vail DM and Page RL: Withrow
and MacEwen's small animal clinical oncology. 5th. Elsevier; St.
Louis, MI: 2013, View Article : Google Scholar
|
|
2
|
Patnaik AK, Hurvitz AI, Lieberman PH and
Johnson GF: Canine hepatocellular carcinoma. Vet Pathol.
18:427–438. 1981. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Liptak JM, Dernell WS, Monnet E, Powers
BE, Bachand AM, Kenney JG and Withrow SJ: Massive hepatocellular
carcinoma in dogs: 48 cases (1992–2002). J Am Vet Med Assoc.
225:1225–1230. 2004. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Weisse C, Clifford CA, Holt D and Solomon
JA: Percutaneous arterial embolization and chemoembolization for
treatment of benign and malignant tumors in three dogs and a goat.
J Am Vet Med Assoc. 221:1430–1436, 1419. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Ginn PE: Immunohistochemical detection of
P-glycoprotein in formalin-fixed and paraffin-embedded normal and
neoplastic canine tissues. Vet Pathol. 33:533–541. 1996. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Ogilvie GK, Obradovich JE, Elmslie RE,
Vail DM, Moore AS, Straw RC, Dickinson K, Cooper MF and Withrow SJ:
Efficacy of mitoxantrone against various neoplasms in dogs. J Am
Vet Med Assoc. 198:1618–1621. 1991.PubMed/NCBI
|
|
7
|
Moore AS and Kitchell BE: New chemotherapy
agents in veterinary medicine. Vet Clin North Am Small Anim Pract.
33:629–649. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Elpiner AK, Brodsky EM, Hazzah TN and Post
GS: Single-agent gemcitabine chemotherapy in dogs with
hepatocellular carcinomas. Vet Comp Oncol. 9:260–268. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Visvader JE and Lindeman GJ: Cancer stem
cells in solid tumours: Accumulating evidence and unresolved
questions. Nat Rev Cancer. 8:755–768. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Clarke MF, Dick JE, Dirks PB, Eaves CJ,
Jamieson CH, Jones DL, Visvader J, Weissman IL and Wahl GM: Cancer
stem cells - perspectives on current status and future directions:
AACR Workshop on cancer stem cells. Cancer Res. 66:9339–9344. 2006.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Haraguchi N, Utsunomiya T, Inoue H, Tanaka
F, Mimori K, Barnard GF and Mori M: Characterization of a side
population of cancer cells from human gastrointestinal system. Stem
Cells. 24:506–513. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Suetsugu A, Nagaki M, Aoki H, Motohashi T,
Kunisada T and Moriwaki H: Characterization of CD133+
hepatocellular carcinoma cells as cancer stem/progenitor cells.
Biochem Biophys Res Commun. 351:820–824. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Ma S, Lee TK, Zheng BJ, Chan KW and Guan
XY: CD133+ HCC cancer stem cells confer chemoresistance by
preferential expression of the Akt/PKB survival pathway. Oncogene.
27:1749–1758. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yang ZF, Ho DW, Ng MN, Lau CK, Yu WC, Ngai
P, Chu PW, Lam CT, Poon RT and Fan ST: Significance of CD90+ cancer
stem cells in human liver cancer. Cancer Cell. 13:153–166. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Endo K and Terada T: Protein expression of
CD44 (standard and variant isoforms) in hepatocellular carcinoma:
Relationships with tumor grade, clinicopathologic parameters, p53
expression, and patient survival. J Hepatol. 32:78–84. 2000.
View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Michishita M, Ezaki S, Ogihara K, Naya Y,
Azakami D, Nakagawa T, Sasaki N, Arai T, Shida T and Takahashi K:
Identification of tumor-initiating cells in a canine hepatocellular
carcinoma cell line. Res Vet Sci. 96:315–322. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Cogliati B, Aloia TP, Bosch RV, Alves VA,
Hernandez-Blazquez FJ and Dagli ML: Identification of hepatic
stem/progenitor cells in canine hepatocellular and
cholangiocellular carcinoma. Vet Comp Oncol. 8:112–121. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Ronot X, Benel L, Adolphe M and Mounolou
JC: Mitochondrial analysis in living cells: The use of rhodamine
123 and flow cytometry. Biol Cell. 57:1–7. 1986. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
McKenzie JL, Takenaka K, Gan OI, Doedens M
and Dick JE: Low rhodamine 123 retention identifies long-term human
hematopoietic stem cells within the LinCD34+CD38 population. Blood.
109:543–545. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Bussolati B, Bruno S, Grange C, Ferrando U
and Camussi G: Identification of a tumor-initiating stem cell
population in human renal carcinomas. FASEB J. 22:3696–3705. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Livak KJ and Schmittgen TD: Analysis of
relative gene expression data using real-time quantitative PCR and
the 2ΔΔCq method. Methods. 25:402–408. 2001. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Johnson LV, Walsh ML and Chen LB:
Localization of mitochondria in living cells with rhodamine 123.
Proc Natl Acad Sci USA. 77:pp. 990–994. 1980; View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Harman D: Aging: A theory based on free
radical and radiation chemistry. J Gerontol. 11:298–300. 1956.
View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Gatenby RA and Gillies RJ: Why do cancers
have high aerobic glycolysis? Nat Rev Cancer. 4:891–899. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Wagner-Souza K, Diamond HR, Ornellas MH,
Gomes BE, Almeida-Oliveira A, Abdelhay E, Bouzas LF and Rumjanek
VM: Rhodamine 123 efflux in human subpopulations of hematopoietic
stem cells: Comparison between bone marrow, umbilical cord blood
and mobilized peripheral blood CD34+ cells. Int J Mol Med.
22:237–242. 2008.PubMed/NCBI
|
|
26
|
Lu J, Cui Y, Zhu J, He J, Zhou G and Yue
Z: Biological characteristics of Rh123 (high) stem-like cells in a
side population of 786-O renal carcinoma cells. Oncol Lett.
5:1903–1908. 2013.PubMed/NCBI
|
|
27
|
Vu NB, Nguyen TT, Tran LC, Do CD, Nguyen
BH, Phan NK and Pham PV: Doxorubicin and 5-fluorouracil resistant
hepatic cancer cells demonstrate stem-like properties.
Cytotechnology. 65:491–503. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Xu LB and Liu C: Role of liver stem cells
in hepatocarcinogenesis. World J Stem Cells. 6:579–590. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Wilson H, Huelsmeyer M, Chun R, Young KM,
Friedrichs K and Argyle DJ: Isolation and characterisation of
cancer stem cells from canine osteosarcoma. Vet J. 175:69–75. 2008.
View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Penzo C, Ross M, Muirhead R, Else R and
Argyle DJ: Effect of recombinant feline interferon-omega alone and
in combination with chemotherapeutic agents on putative
tumour-initiating cells and daughter cells derived from canine and
feline mammary tumours. Vet Comp Oncol. 7:222–229. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
Stoica G, Lungu G, Martini-Stoica H,
Waghela S, Levine J and Smith R III: Identification of cancer stem
cells in dog glioblastoma. Vet Pathol. 46:391–406. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Liu W, Selçuk F, Rütgen BC, Moulay M,
Willenbrock S, Hammer SE, Sterenczak KA, Junghanss C,
Hewicker-Trautwein M, Nolte I, et al: Evaluation of stem cell
marker expression in canine B-cell lymphoma cell lines, B-cell
lymphoma-generated spheres and primary samples. Anticancer Res.
35:2805–2816. 2015.PubMed/NCBI
|