Introduction
The tumor cell cytoskeleton first became an
important subject in cancer biology when attempts were made to
isolate tumor revertants by cloning slowly dividing cells resistant
to chemicals that could be incorporated into DNA and poison rapidly
dividing cells (1,2). Initial observations indicated that
revertants were flat, i.e., well spread in a tissue culture dish,
which led to considerations of ‘flat revertants’ having an
extensive cytoskeletal architecture. This indeed turned out to be
verifiable with a variety of stains for the F-actin cytoskeleton
(3,4).
These results in turn led to an extensive body of work indicating
that cells with a more disorganized cytoskeleton were more
tumorigenic, particularly as determined by in vitro
parameters corresponding to a malignant phenotype (5), such as growth in soft agar; as determined
by experiments with immune-compromised mice (6). More recently, genetic techniques have
implicated specific, cytoskeletal dysfunctions as favoring
tumorigenesis, for example, mouse tumors engineered to lack
dystrophin (7,8), which links to the cytoskeleton, are more
aggressive. In addition, The Cancer Genome Atlas (TCGA) data have
revealed that cytoskeletal protein-related coding regions (CPCRs),
including extracellular matrix protein coding regions, are among
the most frequently mutated coding regions in cancer (9,10), and these
CPCR mutations have been indicated as driver mutations in certain
types of cancers (11).
However, other study has indicated that tumor cell
migration and tumor cell invasive properties are dependent on a
functioning actin cytoskeleton and the function of
cytoskeleton-dependent invadopodia (12–17). Several
cytoskeletal related proteins in particular have been implicated in
cell migration and invadopodia. For example, vinculin has been
demonstrated to be important for polarized cell motility and
metastasis (18,19); and talin-1 has been demonstrated to be
important in tissue extravasion and has been considered as a target
to prevent metastasis (20). On the
other hand, both of these proteins have been considered as
potential tumor suppressor proteins, with knock-out approaches
contradicting the idea that these two proteins are important in
their wild-type forms for cell migration or metastasis (21,22).
To help resolve the above cytoskeleton-related
contradiction, we decided to test the hypothesis that mutation
rates could define two sets of CPCRs: a CPCR-mutated set, as
previously described (9–11); and a CPCR-protected set, with a
candidate CPCR-protected set identified below for several TCGA
cancer data sets.
Materials and methods
Mutated cytoskeletal gene expression,
mutated set, protected set
The TCGA data portal (http://cancergenome.nih.gov.ezproxy.lib.usf.edu/),
under NIH/dbGAP project approval no. 6300, was used to collect
somatic mutation data for bladder urothelial carcinoma (BLCA),
breast invasive carcinoma (BRCA), cervical squamous cell carcinoma
and endocervical adenocarcinoma (CESC), head and neck squamous cell
carcinoma (HNSC), liver hepatocellular carcinoma (LIHC), prostate
adenocarcinoma (PRAD), rectum adenocarcinoma (READ), skin cutaneous
melanoma-primary (SKCM-01), skin cutaneous melanoma-metastatic
(SKCM-06), and stomach adenocarcinoma (STAD) data sets. (These data
are now available from the cBioPortal (http://www.cbioportal.org/index.do). Tumor sample
barcodes in the somatic mutation (exome) files were truncated to
contain only the following characters, TCGA-##-####. Mutation data
from the comprehensive mutation files were collected for the CPCR
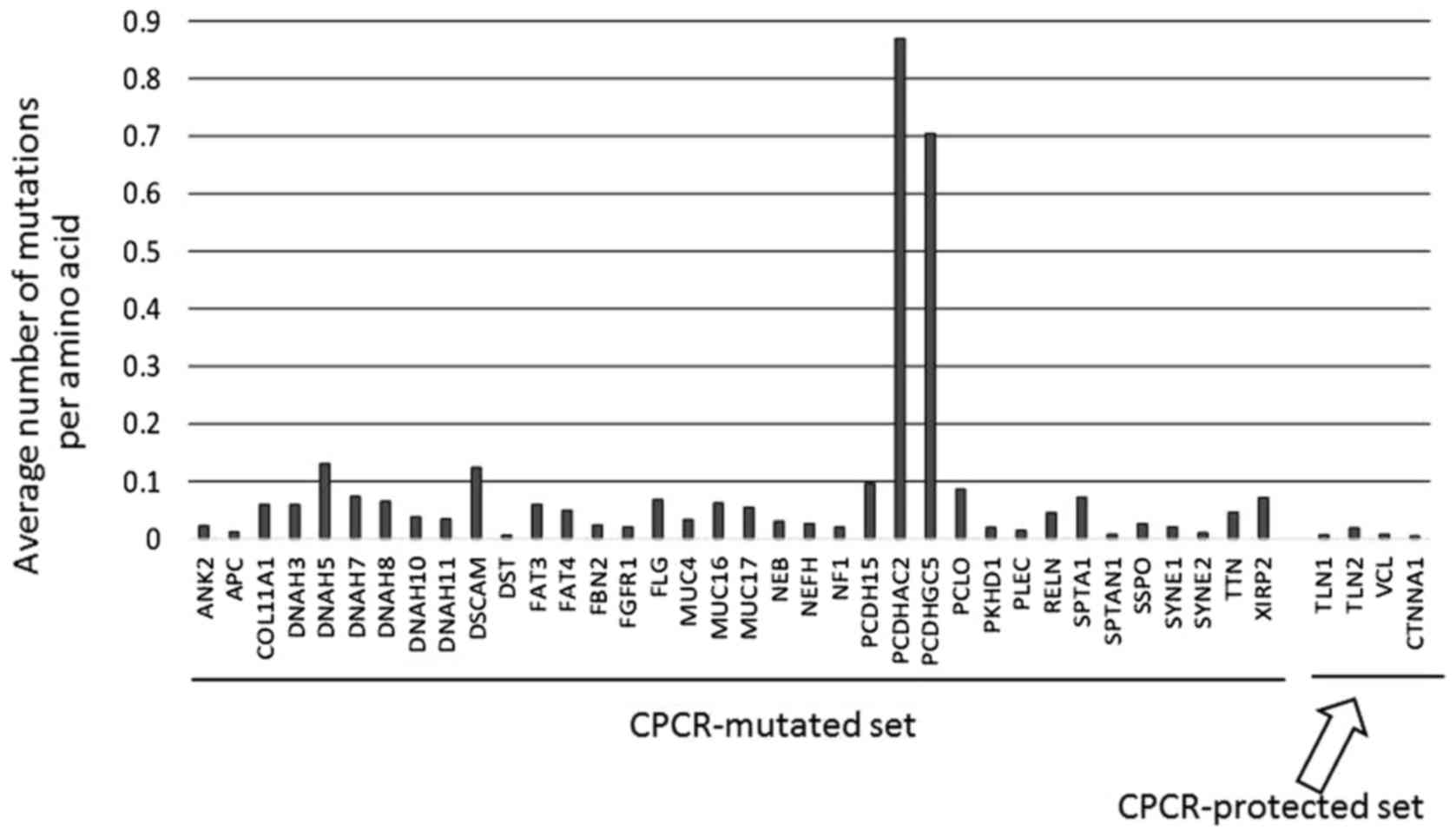
sets (HUGO symbols listed in Fig. 1)
and a Microsoft Excel COUNTIF function was used to determine the
number of mutations for each CPCR. Total mutations were then
normalized to amino acid length for respective coding regions.
Deleterious amino acid changes:
PROVEAN
The chromosome number, start position, reference
allele and tumor sequence allele data were collected for each of
the datasets. These data were then copied into PROVEAN under the
‘Human Genome Variants’ protocol. The removal of duplicates from
the ‘INPUT’ column in the PROVEAN output was used to determine the
number of deleterious amino acid changes for each dataset. A
Microsoft IF[ISERROR(MATCH)] function was then used to separate all
mutations pertaining to the protected and mutated CPCR sets. The
number of deleterious amino acid changes was then divided by the
sample size to determine the average number of deleterious amino
acid changes per barcode for each tumor set. BLCA data was further
divided into deleterious mutations belonging to No Subsequent Tumor
or New Tumor groups based on data from TCGA clinical files, as
described previously (23).
Supporting online material
The original data and the details of the
calculations in this report are available at http://www.universityseminarassociates.com/Supporting_online_material_for_scholarly_pubs.php.
Results and Discussion
To obtain an indication of whether mutation
frequencies were more prevalent in a previously defined, heavily
mutated set of CPCRs-mutated (9–11), than in a
set of cytoskeletal protein related coding regions associated with
pro-tumorigenic properties (CPCR-protected; see Materials and
methods), the authors performed the following processing steps:
Genome sequencing files from TCGA, representing 10 distinct cancer
datasets (CESC, BRCA, STAD, PRAD, HNSC, LIHC, READ, BLCA,
SKCM-primary and SKCM-metastatic), were downloaded, and total
mutations within the above gene sets were tabulated. The mutation
counts for each coding region were normalized to amino acid length
(Fig. 1). Results indicated that the
two gene groups, CPCR-mutated and CPCR-protected had significantly
different mutation rates for READ, PRAD, HNSC, LIHC, BLCA,
SKCM-primary, and SKCM-metastasis datasets, but not for the CESC,
BRCA, or STAD datasets (Fig. 2).
 | Figure 2.Average number of total mutations for
the indicated cancer datasets, for the CPCR-mutated (black) and
-protected (grey) sets, normalized to amino acid lengths. Data are
presented as the mean ± standard deviation. (READ P<0.01, PRAD
P<0.009, HNSC P<8.12 E-06, LIHC P<0.003, BLCA P<0.001,
SKCM-primary P<0.015, SKCM-metastatic P<0.012; CESC, BRCA,
STAD, left side of figure, not significant). CPCR, cytoskeletal
protein-related coding regions; READ, rectum adenocarcinoma; PRAD,
prostate adenocarcinoma; HNSC, head and neck squamous cell
carcinoma; LIHC, liver hepatocellular carcinoma; BLCA, bladder
urothelial carcinoma; SKCM, skin cutaneous melanoma; CESC, cervical
squamous cell carcinoma and endocervical adenocarcinoma; BRCA,
breast invasive carcinoma; STAD, stomach adenocarcinoma. |
Mutation data was then analyzed using the PROVEAN
web tool, using the human genome variants protocol of PROVEAN. The
output was sorted based on the PROVEAN prediction of deleterious or
neutral. Fig. 3 demonstrates the
example of SKCM-metastatic, which demonstrated significant
difference between the two CPCR sets (P<0.00035), with fewer
deleterious mutations in the CPCR-protected set. A total of four of
the ten cancer datasets (READ, BLCA, SKCM-primary and
SKCM-metastatic) also demonstrated significant differences in
average total deleterious mutations between CPCR-mutated and
CPCR-protected sets (Fig. 4). In other
words, two of the cancer datasets above, PRAD and HNSC, which
indicated a statistically significant difference between the
mutation rates for the CPCR-protected vs. CPCR-mutated sets did not
maintain that distinction using the standard of deleterious
mutations, presumably a more stringent standard.
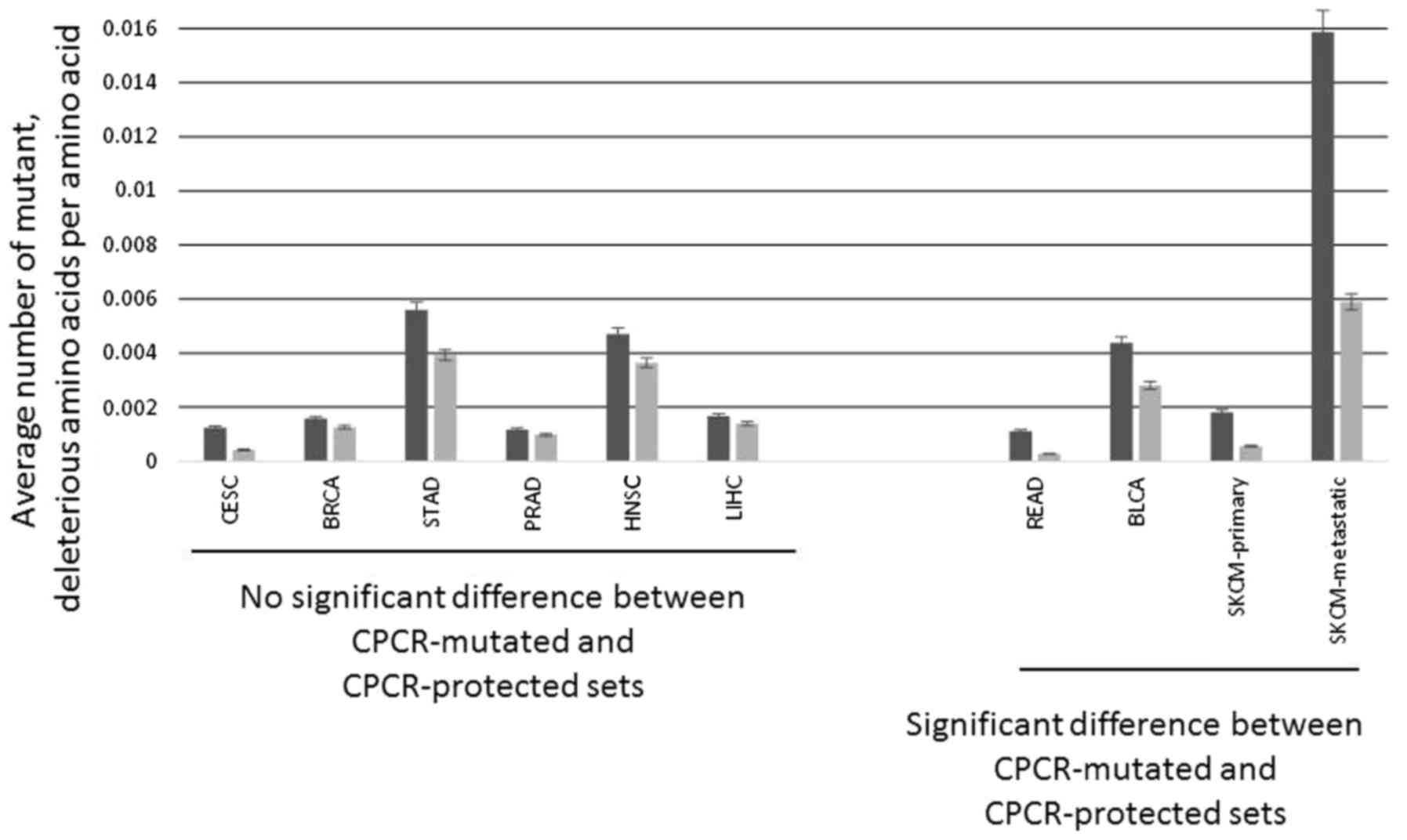 | Figure 4.Average total deleterious amino acid
substitutions for the indicated cancer datasets, normalized to
amino acid length (READ P<0.036, BLCA P<0.006, SKCM-primary
P<0.0086, SKCM-06 P<0.00035; CESC, BRCA, STAD, PRAD, HNSC,
LIHC, left side of figure, not significant). READ, rectum
adenocarcinoma; BLCA, bladder urothelial carcinoma; SKCM, skin
cutaneous melanoma; CESC, cervical squamous cell carcinoma and
endocervical adenocarcinoma; BRCA, breast invasive carcinoma; STAD,
stomach adenocarcinoma; PRAD, prostate adenocarcinoma; HNSC, head
and neck squamous cell carcinoma; LIHC, liver hepatocellular
carcinoma. |
To learn whether the CPCR-protected set could
provide a further indication of fewer mutations being important for
disease progression, the number of mutations and deleterious amino
acids per barcode in the SKCM-primary and SKCM-metastasis datasets
was determined. Results indicated that the CPCR-mutated set and the
CPCR-protected set had similar increases in mutations and
deleterious amino acids going from primary to metastatic samples
(Table I).
 | Table I.Comparison of mutations per barcode
for the CPCR-protected sets, for primary and metastatic SKCM. |
Table I.
Comparison of mutations per barcode
for the CPCR-protected sets, for primary and metastatic SKCM.
| CPCR-mutated set |
|---|
|
|---|
| Tumors | No. of mutations | No. of deleterious AA
substitutions | Total barcodes | Mutations per
barcode | Deleterious AA
substitutions per barcode |
|---|
| SKCM-primary |
1,182 | 338 | 63 | 18.76 | 5.36 |
| SKCM-metastatic | 10,055 | 2,948 | 278 | 36.16 | 10.60 |
|
| CPCR-protected
set |
|
| Tumors | No. of mutations | No. of deleterious AA
substitutions | Total barcodes | Mutations per
barcode | Deleterious AA
substitutions per barcode |
|
| SKCM-primary | 12 | 4 | 63 | 0.190 | 0.063 |
| SKCM-metastatic | 80 | 42 | 278 | 0.287 | 0.151 |
The authors further pursued this question by
analyzing two sets of BCLA barcodes available via TCGA: BLCA
barcodes where there was no known subsequent tumor and barcodes
where a new tumor was known, as detailed previously (23). Again, there was no difference in the
increase in the rate of deleterious amino acids for the two sets of
CPCRs, when no subsequent tumor and new tumor were compared
(Table II). To address this issue a
final time, the mutations per barcode were assessed for the two
different CPCR sets for PRAD-primary vs. two sets of
PRAD-metastatic barcodes, and again, there was no increase in one
of the CPCR sets that was statistically, significantly greater or
less than the other set (Table III).
In the case of PRAD, it needs to be kept in mind that there was no
evidence of a difference in the CPCR-mutated vs. CPCR-protected set
for the deleterious amino acid replacements. The PRAD distinction
for these two CPCR sets was limited to the mutations rates without
regard to amino acid changes (Fig.
2).
 | Table II.Comparison of BLCA deleterious AA
substitutions per barcode between CPCR-mutated and CPCR-protected
sets for New tumor and No subsequent tumor barcodes. |
Table II.
Comparison of BLCA deleterious AA
substitutions per barcode between CPCR-mutated and CPCR-protected
sets for New tumor and No subsequent tumor barcodes.
| CPCR-mutated set |
|---|
|
|---|
| Tumors | No. of mutations | No. of deleterious AA
substitutions | Total barcodes | Mutations per
barcode | Deleterious AA
substitutions per barcode |
|---|
| No subsequent
tumor | 217 | 66 | 27 | 8.03 | 2.44 |
| New tumor | 195 | 55 | 28 | 6.96 | 1.96 |
|
| CPCR-protected
set |
|
| Tumors | No. of mutations | No. of deleterious AA
substitutions | Total barcodes | Mutations per
barcode | Deleterious AA
substitutions per barcode |
|
| No subsequent
tumor | 4 | 1 | 27 | 0.148 | 0.037 |
| New tumor | 3 | 1 | 28 | 0.107 | 0.035 |
 | Table III.Comparison of mutations per barcode
between CPCR-mutated and CPCR-protected sets, for primary and
metastatic PRAD. |
Table III.
Comparison of mutations per barcode
between CPCR-mutated and CPCR-protected sets, for primary and
metastatic PRAD.
| CPCR-mutated set |
|---|
|
|---|
| Tumors | No. of mutations | Total barcodes | Mutations per
barcode |
|---|
| PRAD-primary | 784 | 499 | 1.57 |
| PRAD-metastatic | 77 | 61 | 1.26 |
|
Prostate-metastatic | 373 | 150 | 2.48 |
|
| CPCR-protected
set |
|
| Tumors | No. of mutations | Total barcodes | Mutations per
barcode |
|
| PRAD-primary | 18 | 499 | 0.036 |
| PRAD-metastatic | 1 | 61 | 0.016 |
|
Prostate-metastatic | 13 | 150 | 0.086 |
The above data strongly support the idea that CPCRs
can be divided into sets with statistically different rates of
mutation, including mutations that lead to deleterious amino acid
substitutions. This conclusion may serve a role in resolving the
contradictions in the literature that indicate that cytoskeletal
disorganization is a hallmark of tumorigenesis but is also an
important feature of cell migration, and presumably a feature of
metastatic cells. However, the above data do not indicate that
these distinctions are consistent with a change in tumor
aggressiveness. While this is a negative result, and cannot lead to
a final conclusion, the current data are consistent with the
possibility that CPCR-mutated and CPCR-protected sets are a
fundamental aspect of the generation of the tumor cell, rather than
representative of a distinction between aggressive and
non-aggressive cancers.
References
|
1
|
Vogel A, Risser R and Pollack R: Isolation
and characterization of revertant cell lines. 3. Isolation of
density-revertants of SV40-transformed 3T3 cells using colchicine.
J Cell Physiol. 82:181–188. 1973. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pollack RE, Green H and Todaro GJ: Growth
control in cultured cells: Selection of sublines with increased
sensitivity to contact inhibition and decreased tumor-producing
ability. Proc Natl Acad Sci USA. 60:126–133. 1968. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Verderame M, Alcorta D, Egnor M, Smith K
and Pollack R: Cytoskeletal F-actin patterns quantitated with
fluorescein isothiocyanate-phalloidin in normal and transformed
cells. Proc Natl Acad Sci USA. 77:6624–6628. 1980. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Kopelovich L, Conlon S and Pollack R:
Defective organization of actin in cultured skin fibroblasts from
patients with inherited adenocarcinoma. Proc Natl Acad Sci U S A.
74:3019–3022. 1977. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Shin SI, Freedman VH, Risser R and Pollack
R: Tumorigenicity of virus-transformed cells in nude mice is
correlated specifically with anchorage independent growth in vitro.
Proc Natl Acad Sci USA. 72:4435–4439. 1975. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Steinberg BM, Rifkin D, Shin S, Boone C
and Pollack R: Tumorigenicity of revertant from an SV40-transformed
line. J Supramol Struct. 11:539–546. 1979. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Wang Y, Marino-Enriquez A, Bennett RR, Zhu
M, Shen Y, Eilers G, Lee JC, Henze J, Fletcher BS, Gu Z, et al:
Dystrophin is a tumor suppressor in human cancers with myogenic
programs. Nat Genet. 46:601–606. 2014. View
Article : Google Scholar : PubMed/NCBI
|
|
8
|
Fernandez K, Serinagaoglu Y, Hammond S,
Martin LT and Martin PT: Mice lacking dystrophin or alpha
sarcoglycan spontaneously develop embryonal rhabdomyosarcoma with
cancer-associated p53 mutations and alternatively spliced or mutant
Mdm2 transcripts. Am J Pathol. 176:416–434. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Parry ML, Ramsamooj M and Blanck G: Big
genes are big mutagen targets: A connection to cancerous, spherical
cells? Cancer Lett. 356(2 Pt B): 1–482. 2015.
|
|
10
|
Parry ML and Blanck G: Flat cells come
full sphere: Are mutant cytoskeletal-related proteins
oncoprotein-monsters or useful immunogens? Hum Vaccin Immunother.
12:120–123. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Fawcett TJ, Parry ML and Blanck G: A Novel
Approach to Evaluating Cancer Driver Gene Mutation Densities:
Cytoskeleton-related Gene Candidates. Cancer Genomics Proteomics.
12:283–290. 2015.PubMed/NCBI
|
|
12
|
Zachary JM, Cleveland G, Kwock L, Lawrence
T, Weissman RM, Nabell L, Fried FA, Staab EV, Risinger MA and Lin
S: Actin filament organization of the Dunning R3327 rat prostatic
adenocarcinoma system: Correlation with metastatic potential.
Cancer Res. 46:926–932. 1986.PubMed/NCBI
|
|
13
|
Xu W, Mezencev R, Kim B, Wang L, McDonald
J and Sulchek T: Cell stiffness is a biomarker of the metastatic
potential of ovarian cancer cells. PLoS One. 7:e466092012.
View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Bear JE and Haugh JM: Directed migration
of mesenchymal cells: Where signaling and the cytoskeleton meet.
Curr Opin Cell Biol. 30:74–82. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Verschueren H, Van der Taelen I, Dewit J,
De Braekeleer J and De Baetselier P: Metastatic competence of
BW5147 T-lymphoma cell lines is correlated with in vitro
invasiveness, motility and F-actin content. J Leukoc Biol.
55:552–556. 1994.PubMed/NCBI
|
|
16
|
Nürnberg A, Kitzing T and Grosse R:
Nucleating actin for invasion. Nat Rev Cancer. 11:177–187. 2011.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Kim TY, Vigil D, Der CJ and Juliano RL:
Role of DLC-1, a tumor suppressor protein with RhoGAP activity, in
regulation of the cytoskeleton and cell motility. Cancer Metastasis
Rev. 28:77–83. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Carisey A, Tsang R, Greiner AM, Nijenhuis
N, Heath N, Nazgiewicz A, Kemkemer R, Derby B, Spatz J and
Ballestrem C: Vinculin regulates the recruitment and release of
core focal adhesion proteins in a force-dependent manner. Curr
Biol. 23:271–281. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Thakur RK, Yadav VK, Kumar A, Singh A, Pal
K, Hoeppner L, Saha D, Purohit G, Basundra R, Kar A, et al:
Non-metastatic 2 (NME2)-mediated suppression of lung cancer
metastasis involves transcriptional regulation of key cell adhesion
factor vinculin. Nucleic Acids Res. 42:11589–11600. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gilardi M, Bersini S, Calleja AB, Kamm RD,
Vanoni M and Moretti M: PO-12 - The key role of talin-1 in cancer
cell extravasation dissected through human vascularized 3D
microfluidic model. Thromb Res. 140 Suppl 1:S180–S181. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
21
|
Liu M, Oberg K and Zhou Y: Expression and
function of vinculin in neuroendocrine tumors. Tumour Biol.
28:196–204. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Zhang W, Mao YQ, Wang H, Yin WJ, Zhu SX
and Wang WC: MiR-124 suppresses cell motility and adhesion by
targeting talin 1 in prostate cancer cells. Cancer Cell Int.
15:492015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
Samy MD, Tong WL, Yavorski JM, Sexton WJ
and Blanck G: T cell receptor gene recombinations in human tumor
specimen exome files: Detection of T cell receptor-β VDJ
recombinations associates with a favorable oncologic outcome for
bladder cancer. Cancer Immunol Immunother. 66:403–410. 2017.
View Article : Google Scholar : PubMed/NCBI
|