Bone fracture is a health issue in patients with
bone-related disorders such as osteoporosis as it is a common event
(1). Healthy bone homeostasis
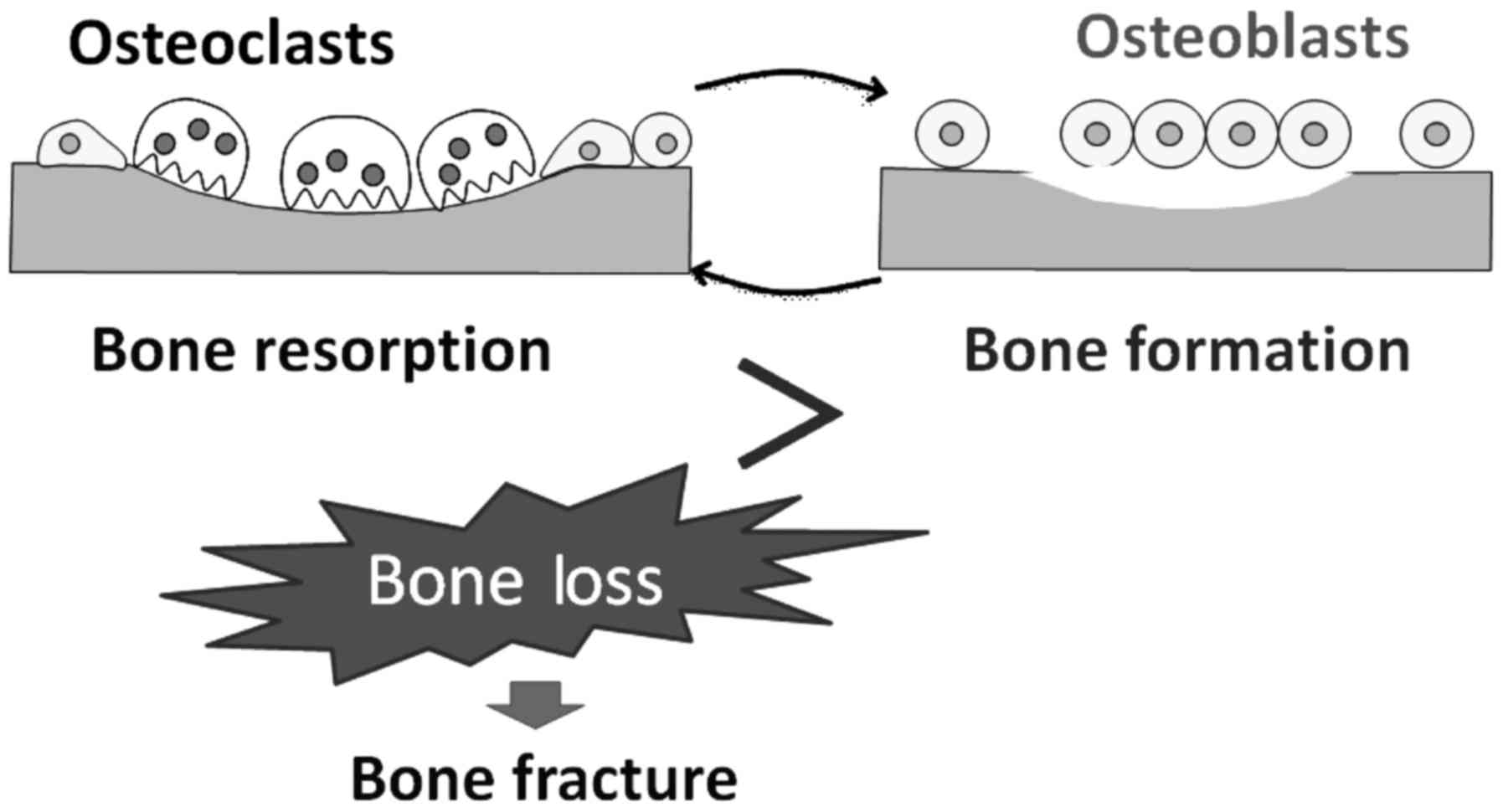
depends on a balance between osteoblastic bone formation and
osteoclastic bone resorption (2)
(Fig. 1). Bone remodeling also
requires a balance in growth, differentiation, and activity of
osteoblasts and osteoclasts. Multinucleated osteoclasts resorb
lamellar bone, and new bone is generated by osteoblasts. An
imbalance can lead to impaired bone structure and/or small bone
mass. Accordingly, osteoclasts are functionally indispensable for
supporting bone health. Hyperactivation of osteoclasts and/or their
increased number can lead to diseases characterized by bone loss,
which is a key risk factor for bone fracture (3). Bone resorption leads to degradation
of extracellular matrix which alters the environment of bone marrow
stem cell binding and differentiation. On the other hand, bone
morphogenetic proteins (BMPs), which are members of the
transforming growth factor (TGF)-β superfamily and in charge of the
development and function of different cell types, induce bone
formation (4). It has been shown
that BMP signaling in osteoblasts regulates bone mass in mice,
suggesting a role of BMP in osteoclastogenesis along with
osteoblastic action (5). In
addition, postmenopausal osteoporotic bone loss may largely result
from the stimulation of bone resorption via increased osteoclast
formation with insufficient osteoblastic bone formation (6). Clinically, medical orally available
bisphosphonates targeting osteoclasts have been widely used to
treat patients with osteoporosis and/or prevent osteoporotic
fracture. Bisphosphonates principally inhibit the activation of
osteoclasts by binding to hydroxyapatite (7); however, bisphosphonate-related side
effects including hypocalcaemia, secondary hyperparathyroidism,
renal toxicity, gastrointestinal tract problems and osteonecrosis
have recently been reported (8).
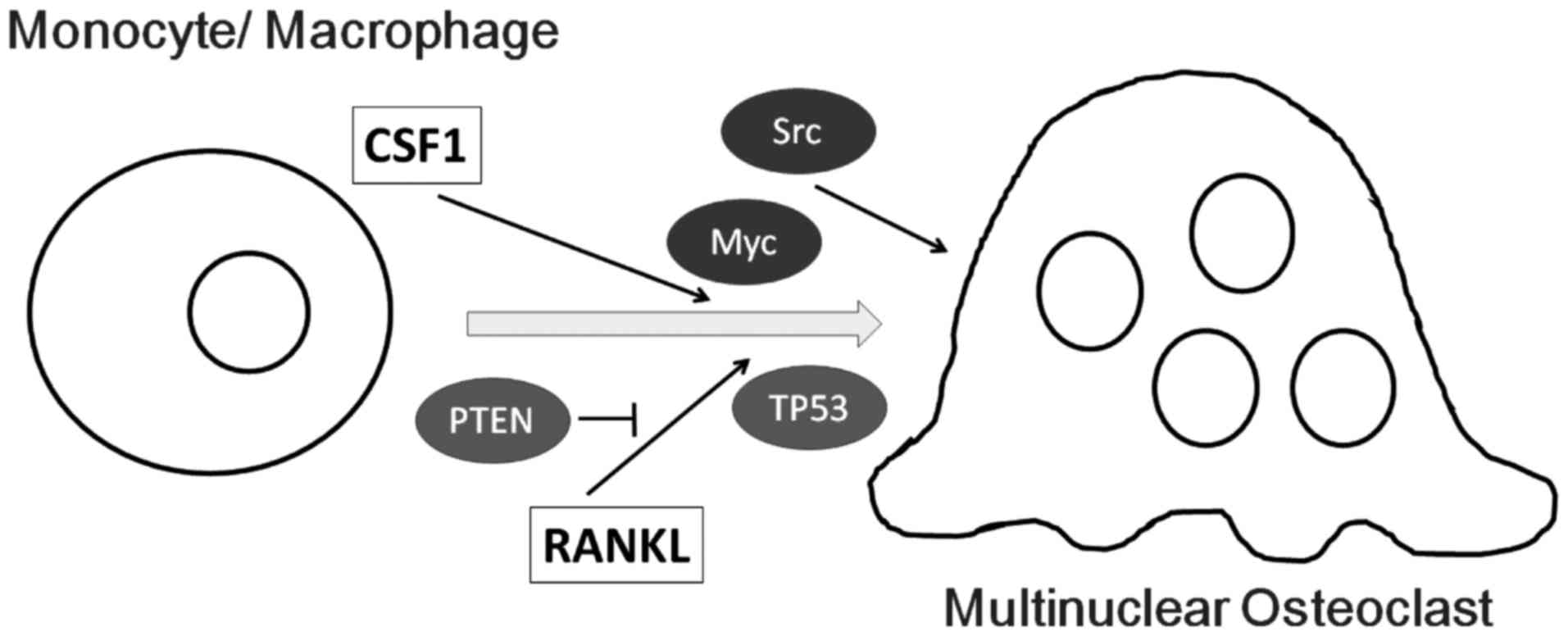
Colony-stimulating factor 1 (CSF1) is a growth factor required for
the differentiation of monocyte-macrophage precursor cells into
preosteoclasts (9). Osteoblasts
produce CSF1 and receptor activator of nuclear factor-κB ligand
(RANKL) which is essential for the early development of osteoclasts
from precursor cells derived from monocyte/macrophage lineage
originating from the liver and spleen (10). Membrane-bound RANKL invites
osteoclasts and initiates and/or activates their differentiation to
form multinucleated cells. The role of BMPs in osteoclast
differentiation has also been demonstrated, which induce RANKL
expression in osteoblasts. CSF1 is controlled by a Smad-signaling
pathway (11). In addition, the
involvement of the phosphatidylinositol 3-kinase (PI3K) and AKT
signaling pathways has been shown to be critical both in osteoblast
and in osteoclast differentiation in response to BMPs (11). Consequently, BMPs induce secretion
of CSF1 dependent on PI3K/AKT signaling. Inhibition of PI3K/AKT
signaling blocks the binding of Smads to the CSF1 BMP-responsive
element present in the CSF1 promoter, resulting in attenuation of
Smad-dependent CSF1 transcription (12).
Proto-oncogenes and tumor-suppressor genes, which
are important in the normal development of cells, are involved in
the regulation of the cell cycle and apoptosis (13). For example, the proto-oncogene
c-Src has been implicated in the development and mature
function of the nervous system (14). The protein product of the
c-Src gene is a tyrosine protein kinase that is enriched in
fetal neural tissues. Nuclear transcription of c-Src and
other proto-oncogenes such as N-ras, c-Myc and
c-Fos have been observed in proliferating and
differentiating cells (15). In
general, oncogenes are critically positioned in various growth
factor receptor signaling pathways and are relevant in cancer
development (16). TP53 is
a well-known tumor-related gene expressed ubiquitously in all cell
types as an inactive transcription factor which undertakes
activation in response to a variety of cellular stresses.
TP53 acts both as an oncogene and a tumor-suppressor gene.
The effects of TP53 are mediated by different downstream effectors
and target proteins. Among them, cyclin-dependent kinase (CDK)
inhibitors such as p21 are key mediators of TP53 action, in which
p21 may be involved in cell differentiation (17). In addition, p21 regulates cell
cycle progression, cell differentiation and senescence (18). Phosphatase and tensin homolog
(PTEN) is also a well-known tumor-suppressor gene product of the
pten gene. PTEN is a dual-specificity phosphatase that has
been shown to prevent cell proliferation and migration (19,20). The PTEN/AKT pathway appears to be
important in the regulation of inflammatory responses (21).
Osteoclasts are multinucleated cells that are formed
by the fusion of mononuclear osteoclasts, which is an essential
process in bone resorption leading to bone remodeling (22). Mechanical response is known to
regulate bone remodeling, yet the molecular events involved in the
mechanical signal transduction are poorly understood. However,
RANKL may be the most essential cytokine involved in the genesis of
osteoclasts and/or osteoclast differentiation (23). The binding of RANKL to RANK
provokes activation of signaling molecules including AKT and ERK
that later induce the activation of transcription factors such as
nuclear factor of activated T cells (NFATc1) and c-Fos to regulate
the expression of genes required for osteoclast differentiation
(24,25). The expression of c-Fos and NFATc1
is regulated by the ERK signaling pathway (26). Proto-oncogene product c-Fos is an
essential factor for the induction of NFATc1, which is a master
transcription factor that regulates the process of osteoclast
differentiation by controlling osteoclast-specific genes (27,28). NFATc1 plays a role as a
transcription factor required for regulating the expression of
osteoclast-specific genes including TRAP and c-Src (29). c-Src tyrosine kinase (Fig. 2) is also required for the
maintenance of osteoclasts and control of bone resorption through
actin cytoskeleton turnover (30,31). Selective c-Src inhibitors induce
osteoclast disruption and consequently reduce osteoclast numbers
in vivo, which induce programmed cell death in mature
osteoclasts (32,33). Caspase-3 and -9 are momentarily
activated by treatment with c-Src inhibitors probably involving
continual ERK1/2 phosphorylation (34). Both c-Fos and c-Src
are proto-oncogenes. In addition, proto-oncogene c-Myc is
strongly upregulated in RANKL-induced osteoclasts (Fig. 2), and is a transcription factor
expressed at comparatively high levels in preosteoclasts (35). Consistent with this, a
dominant-negative Myc could block RANKL-induced osteoclast
formation (35). TRAP, a typical
marker of osteoclast differentiation, is an enzyme that plays an
active role in the process of bone resorption. The specific
regulation of TRAP is performed at the transcriptional level by
Myc, suggesting that Myc may play an active role in suppressing the
transcription of mature osteoclast genes (36). In addition, Myc has a function in
the stimulation of FOXO1 which plays key roles in bone development
and remodeling by stimulating osteoclast formation (37). In addition to PI3K/AKT signaling,
small Ras GTPase also regulates osteoclast survival. Increased
activity of Ras GTPase induces the binding with PI3K, whereas
inhibition of Ras reduces PI3K-mediated osteoclast survival
(38). Actually, the
pharmacological inhibition of H-ras prevents the downstream
mechanical repression of RANKL (39). Consistently, RNA interference
(RNAi) of H-ras also retracts the mechanical repression of
RANKL (39), suggesting that the
mechanical repression of RANKL requires a specific form of Ras-GTP
activity. Spatial arrangements in the lipid raft microdomain may be
critical for downstream events in response to mechanical signals.
In general, activation of Ras and the mitogen-activated protein
kinase (MAPK) signaling pathway is known to underlie the
proliferation and differentiation of different types of cell
lineages including osteoclast progenitor cells (40).
Treatment with RANKL was found to induce an
accumulation of TP53 protein in a dose-dependent manner,
consequently activating TP53 target genes (41). TP53 is a known tumor-suppressor
molecule and also regulates osteoclast differentiation (Fig. 2). Mice with deficiency of the
TP53 gene display a high bone-mass phenotype (42). In addition, TP53-deficient
mice have an improved ability to prefer osteoclast differentiation
with increased expression of CSF1 (42,43). Therefore, TP53 acts as a regulator
of osteoclastogenesis and subsequent bone remodeling (42,43). Bone loss induced by ovariectomy
has been linked to enhanced bone turnover as a result of osteoclast
activation (44), accompanied by
increased expression of the senescence marker p16/p21 in bone
associated with a decrease in Sirt1 (44). In general, bone cell senescence is
associated with decreased Sirt1 expression and activation of TP53,
p16 and p21 (45). Sirt1 is a
candidate anti-aging gene which may suppress p16 expression through
deacetylation (45). Although the
TP53 and p16 pathways act separately to promote cellular
senescence, their contribution can be cell type-dependent depending
on the process (46). Cellular
senescence is a process of aging involving a permanent growth
arrest of mitotic cells and is different from apoptosis and/or
programmed cell death.
Potential therapeutic strategies exploit the
observations made in the critical processes required for
maintaining homeostasis of the metabolic condition characterized by
osteoblast/osteoclast balance. Accordingly, dietary regulation of
these cells is an important therapeutic strategy for preventing
and/or treating bone disorders. As mentioned above, several
oncogenes and tumor-suppressor genes are intensely involved in
osteoclastogenesis and/or osteoporosis. Thus, it is a challenge to
regulate the expression of these genes by dietary treatment. First
of all, inhibition of oncogene expression such as c-Myc and
c-H-ras by green tea and (−)-epigallocatechin gallate has
been shown in mice (52). In
addition, antioxidants such as retinoids (vitamin A), vitamin E
(such as α-tocopheryl succinate), ascorbate (vitamin C) and
carotenoids induce cell differentiation and growth inhibition in
human cells by complex mechanisms including inhibition of the
expression of c-Myc and H-ras and induction of
p21 genes (53). Moreover,
dietary calorie restriction has been associated with reduced cancer
risk, which is related to the abrogation of both Ras and PI3K
signaling (54).
Praeruptorin A is the major bioactive component
isolated from the dry root extract of Peucedanum
praeruptorum Dunn, and has several biological activities such
as anti-hypertensive activity by acting as a calcium channel
blocker (55). Praeruptorin A
attenuates the RANKL-induced phosphorylation of p38 without
affecting JNK and ERK activity. The anti-osteoclastogenic action of
praeruptorin A may be due to its potential to inhibit both the p38
and AKT signaling pathways that subsequently downregulate the
expression of c-Fos and NFATc1 (56). Honokiol, a component of the
Oriental herb Magnolia officinalis, inhibits RANKL-induced
osteoclastogenesis with nuclear factor-κB (NF-κB) activation
(57). Studies have shown that
honokiol blocks TNF-induced phosphorylation, degradation and
ubiquitination of IκBα through the inhibition of AKT (58). Expression of c-Myc is also
downregulated by honokiol (59).
Honokiol can diminish PI3K/AKT signaling by upregulation of PTEN
expression (60). Magnolol, a
honokiol isomer, has been shown to be equally active. Consumption
of a blueberry-containing diet may contribute to the prevention of
bone loss (61). Blueberries are
an admirable source of dietary polyphenols such as phenolic acids
and anthocyanins, which were found to significantly decrease the
gene expression of TP53 and p21 in human HepG2 cells
(62). Resveratrol was also found
to activate Sirt1, a member of the sirtuin family of nicotinamide
adenine dinucleotide (NAD)-dependent deacetylases (63). Stilbenes, which are related to
resveratrol, have been shown to have an inhibitory effect on the
expression of the c-Myc genes (64). In addition, resveratrol inhibits
cell proliferation and induces cell apoptosis through regulation of
TP53 expression (65). An
extract from thorns of the medicinal herb, Gleditsia
sinensis caused an increase in cell cycle arrest during the
G2/M phase, associated with increased TP53 levels (66). Treatment with an ethanol extract
of the thorns of Gleditsia sinensis was also found to be
associated with upregulation of p21 levels (67). Both TP53 and p21
mRNA levels were increased following treatment with the Chinese
herb Kanglaite, an extract from Coix seed (68). Kanglaite appears to extend the
half-life of TP53 protein (68).
A ginsenoside, one of the components of American ginseng herb, was
found to activate TP53 (69). In
addition, apoptosis induction by thymoquinone, the most abundant
component in black seed, was found to be associated with an
increase in TP53 mRNA and downstream TP53 target genes
(70,71). Treatment with an extract of
Magnolia officinalis upregulated the expression of p21 and
p27 (72). Baicalin, a
herb-derived flavonoid compound, enhanced the expression of p27
(73,74). Treatment with an extract of
Saussurea involucrate was also found to induce p21 and p27
expression, independent of the TP53 pathway (75). Treatment with triptolide, a
purified extract from the herb Tripterygium wilfordii Hook F
resulted in increased p21 expression (76,77). Curcumin, an active ingredient
derived from the root of the plant Curcuma longa, restored
PTEN expression (78). In
contrast, various components of the herb rosemary inhibited the
expression of PTEN in K562 myeloid cells (79).
Dietary intake of indole-3-carbinol was found to
upregulate PTEN in an animal model (80). Indole-3-carbinol is a promising
cancer-preventive phytochemical found in various vegetables such as
broccoli (81). In addition, PTEN
expression at the mRNA and protein levels was found to be elevated
in experimental animals fed whey protein which has been shown to
possess multiple health benefits (82). It has also been reported that DHA
and EPA raise the level of PTEN in breast cancer cells, providing a
mechanism for the beneficial effects of fish oils on breast cancer
cells (83,84). Fish oil rich in polyunsaturated
fatty acids may induce PTEN expression by activation of peroxisome
proliferator-activated receptor (PPAR) (85,86), which attenuates cellular damage
playing an important role in the activation of anti-apoptotic
signaling (87). The information
discussed here may also be useful for supporting the design of
further research concerning the prevention of osteoporosis.
It will be a challenge to elucidate how to utilize
natural compounds for the correction of critical processes required
for maintaining cellular homeostasis and metabolic conditions to
prevent osteoporosis (Fig. 3).
Identification of effective target molecules relevant for
osteoporosis allows the screening for natural products capable of
modulating targets. In addition, combination therapy using two or
more food ingredients is a promising therapeutic strategy over
traditional approaches. For example, resveratrol and curcumin
synergistically induce apoptosis by increasing the level of p21 and
decreasing the level of c-Myc (88). The information here may provide
further insight into the molecular mechanisms of special diets
underlying the daily use of certain foods as a therapeutic strategy
for osteoporosis. This may also provide the basis for the
development of rational dietary treatments against other diseases.
Future studies are required to demonstrate whether oncogenes,
tumor-suppressor genes and/or their downstream targets could be
used to modulate the cellular composition of tissue including bone,
thereby enhancing metabolic stability. Knowledge of the local
determinants of the phenotype of osteoclasts in critical lesions
and how they interact with the risk factors of bone disorders may
lead to significantly improved therapies with reduced side
effects.
This review was supported by JSPS KAKENHI (grant
nos. 26-12035 and 24240098).
|
1
|
Ward LM, Konji VN and Ma J: The management
of osteoporosis in children. Osteoporos Int. 27:2147–2179. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Pi C, Li YP, Zhou X and Gao B: The
expression and function of microRNAs in bone homeostasis. Front
Biosci (Landmark Ed). 20:119–138. 2015. View Article : Google Scholar
|
|
3
|
Horwood NJ: Macrophage polarization and
bone formation: a review. Clin Rev Allergy Immunol. 51:79–86. 2016.
View Article : Google Scholar
|
|
4
|
Sartori R and Sandri M: BMPs and the
muscle-bone connection. Bone. 80:37–42. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Paul S, Lee JC and Yeh LC: A comparative
study on BMP-induced osteoclastogenesis and osteoblastogenesis in
primary cultures of adult rat bone marrow cells. Growth Factors.
27:121–131. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Riggs BL, Khosla S and Melton LJ III: Sex
steroids and the construction and conservation of the adult
skeleton. Endocr Rev. 23:279–302. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Murphy CM, Schindeler A, Gleeson JP, Yu
NY, Cantrill LC, Mikulec K, Peacock L, O'Brien FJ and Little DG: A
collagen-hydroxyapatite scaffold allows for binding and co-delivery
of recombinant bone morphogenetic proteins and bisphosphonates.
Acta Biomater. 10:2250–2258. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
8
|
Papapetrou PD: Bisphosphonate-associated
adverse events. Hormones (Athens). 8:96–110. 2009. View Article : Google Scholar
|
|
9
|
Mandal CC, Ghosh-Choudhury G and
Ghosh-Choudhury N: Phosphatidylinositol 3 kinase/Akt signal relay
cooperates with Smad in bone morphogenetic protein-2-induced colony
stimulating factor-1 (CSF-1) expression and osteoclast
differentiation. Endocrinology. 150:4989–4998. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Lampiasi N, Russo R and Zito F: The
alternative faces of macrophage generate osteoclasts. BioMed Res
Int. 2016:90896102016. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Mandal CC, Das F, Ganapathy S, Harris SE,
Choudhury GG and Ghosh-Choudhury N: Bone morphogenetic protein-2
(BMP-2) activates NFATc1 transcription factor via an autoregulatory
loop involving Smad/Akt/Ca2+ signaling. J Biol Chem.
291:1148–1161. 2016. View Article : Google Scholar
|
|
12
|
Li W, Liu Z, Zhao C and Zhai L: Binding of
MMP-9-degraded fibronectin to β6 integrin promotes invasion via the
FAK-Src-related Erk1/2 and PI3K/Akt/Smad-1/5/8 pathways in breast
cancer. Oncol Rep. 34:1345–1352. 2015.PubMed/NCBI
|
|
13
|
Aguirre E, Renner O, Narlik-Grassow M and
Blanco-Aparicio C: Genetic modeling of PIM proteins in cancer:
proviral tagging and cooperation with oncogenes, tumor suppressor
genes, and carcinogens. Front Oncol. 4:1092014. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Yu XM and Salter MW: Src, a molecular
switch governing gain control of synaptic transmission mediated by
N-methyl-D-aspartate receptors. Proc Natl Acad Sci USA.
96:7697–7704. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Ingraham CA, Cox ME, Ward DC, Fults DW and
Maness PF: c-Src and other proto-oncogenes implicated in neuronal
differentiation. Mol Chem Neuropathol. 10:1–14. 1989. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Reichmann E: Oncogenes and epithelial cell
transformation. Semin Cancer Biol. 5:157–165. 1994.PubMed/NCBI
|
|
17
|
Kramer JL, Baltathakis I, Alcantara OS and
Boldt DH: Differentiation of functional dendritic cells and
macrophages from human peripheral blood monocyte precursors is
dependent on expression of p21 (WAF1/CIP1) and requires iron. Br J
Haematol. 117:727–734. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Grabliauskaite K, Hehl AB, Seleznik GM,
Saponara E, Schlesinger K, Zuellig RA, Dittmann A, Bain M, Reding
T, Sonda S, et al: p21WAF1/Cip1 limits senescence and
acinar-to-ductal metaplasia formation during pancreatitis. J
Pathol. 235:502–514. 2015. View Article : Google Scholar
|
|
19
|
Nakanishi A, Wada Y, Kitagishi Y and
Matsuda S: Link between PI3K/AKT/PTEN pathway and NOX proteinin
diseases. Aging Dis. 5:203–211. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Matsuda S, Nakanishi A, Minami A, Wada Y
and Kitagishi Y: Functions and characteristics of PINK1 and Parkin
in cancer. Front Biosci (Landmark Ed). 20:491–501. 2015. View Article : Google Scholar
|
|
21
|
Günzl P and Schabbauer G: Recent advances
in the genetic analysis of PTEN and PI3K innate immune properties.
Immunobiology. 213:759–765. 2008. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Appelman-Dijkstra NM and Papapoulos SE:
Modulating bone resorption and bone formation in opposite
directions in the treatment of postmenopausal osteoporosis. Drugs.
75:1049–1058. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
23
|
An J, Yang H, Zhang Q, Liu C, Zhao J,
Zhang L and Chen B: Natural products for treatment of osteoporosis:
the effects and mechanisms on promoting osteoblast-mediated bone
formation. Life Sci. 147:46–58. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Wagner EF and Eferl R: Fos/AP-1 proteins
in bone and the immune system. Immunol Rev. 208:126–140. 2005.
View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Asagiri M and Takayanagi H: The molecular
understanding of osteoclast differentiation. Bone. 40:251–264.
2007. View Article : Google Scholar
|
|
26
|
Lee MS, Kim HS, Yeon JT, Choi SW, Chun CH,
Kwak HB and Oh J: GM-CSF regulates fusion of mononuclear
osteoclasts into bone-resorbing osteoclasts by activating the
Ras/ERK pathway. J Immunol. 183:3390–3399. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Takayanagi H: The role of NFAT in
osteoclast formation. Ann NY Acad Sci. 1116:227–237. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Macián F, López-Rodríguez C and Rao A:
Partners in transcription: NFAT and AP-1. Oncogene. 20:2476–2489.
2001. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zeng XZ, He LG, Wang S, Wang K, Zhang YY,
Tao L, Li XJ and Liu SW: Aconine inhibits RANKL-induced osteoclast
differentiation in RAW264.7 cells by suppressing NF-κB and NFATc1
activation and DC-STAMP expression. Acta Pharmacol Sin. 37:255–263.
2016. View Article : Google Scholar
|
|
30
|
Lee EJ, Kim JL, Gong JH, Park SH and Kang
YH: Inhibition of osteoclast activation by phloretin through
disturbing αvβ3 integrin-c-Src pathway. Biomed Res Int.
2015:6801452015.
|
|
31
|
Fukunaga T, Zou W, Warren JT and
Teitelbaum SL: Vinculin regulates osteoclast function. J Biol Chem.
289:13554–13564. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Shakespeare W, Yang M, Bohacek R, Cerasoli
F, Stebbins K, Sundaramoorthi R, Azimioara M, Vu C, Pradeepan S,
Metcalf C III, et al: Structure-based design of an
osteoclast-selective, nonpeptide src homology 2 inhibitor with in
vivo antiresorptive activity. Proc Natl Acad Sci USA. 97:9373–9378.
2000. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Rucci N, Ricevuto E, Ficorella C, Longo M,
Perez M, Di Giacinto C, Funari A, Teti A and Migliaccio S: In vivo
bone metastases, osteoclastogenic ability, and phenotypic
characterization of human breast cancer cells. Bone. 34:697–709.
2004. View Article : Google Scholar : PubMed/NCBI
|
|
34
|
Recchia I, Rucci N, Funari A, Migliaccio
S, Taranta A, Longo M, Kneissel M, Susa M, Fabbro D and Teti A:
Reduction of c-Src activity by substituted
5,7-diphenyl-pyrrolo[2,3-d]-pyrimidines induces osteoclast
apoptosis in vivo and in vitro. Involvement of ERK1/2 pathway Bone.
34:65–79. 2004.
|
|
35
|
Battaglino R, Kim D, Fu J, Vaage B, Fu XY
and Stashenko P: c-Myc is required for osteoclast differentiation.
J Bone Miner Res. 17:763–773. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Daumer KM, Taparowsky EJ, Hall DJ and
Steinbeck MJ: Transcription from the tartrate-resistant acid
phosphatase promoter is negatively regulated by the Myc
oncoprotein. J Bone Miner Res. 17:1701–1709. 2002. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Wilhelm K, Happel K, Eelen G, Schoors S,
Oellerich MF, Lim R, Zimmermann B, Aspalter IM, Franco CA, Boettger
T, et al: FOXO1 couples metabolic activity and growth state in the
vascular endothelium. Nature. 529:216–220. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Adapala NS, Barbe MF, Tsygankov AY,
Lorenzo JA and Sanjay A: Loss of Cbl-PI3K interaction enhances
osteoclast survival due to p21-Ras mediated PI3K activation
independent of Cbl-b. J Cell Biochem. 115:1277–1289. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Rubin J, Murphy TC, Rahnert J, Song H,
Nanes MS, Greenfield EM, Jo H and Fan X: Mechanical inhibition of
RANKL expression is regulated by H-Ras-GTPase. J Biol Chem.
281:1412–1418. 2006. View Article : Google Scholar
|
|
40
|
Sharma R, Wu X, Rhodes SD, Chen S, He Y,
Yuan J, Li J, Yang X, Li X, Jiang L, et al: Hyperactive Ras/MAPK
signaling is critical for tibial nonunion fracture in
neurofibromin-deficient mice. Hum Mol Genet. 22:4818–4828. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Zauli G, Rimondi E, Corallini F, Fadda R,
Capitani S and Secchiero P: MDM2 antagonist Nutlin-3 suppresses the
proliferation and differentiation of human pre-osteoclasts through
a p53-dependent pathway. J Bone Miner Res. 22:1621–1630. 2007.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Wang X, Kua HY, Hu Y, Guo K, Zeng Q, Wu Q,
Ng HH, Karsenty G, de Crombrugghe B, Yeh J, et al: p53 functions as
a negative regulator of osteoblastogenesis, osteoblast-dependent
osteoclastogenesis, and bone remodeling. J Cell Biol. 172:115–125.
2006. View Article : Google Scholar
|
|
43
|
Zambetti GP, Horwitz EM and Schipani E:
Skeletons in the p53 tumor suppressor closet: genetic evidence that
p53 blocks bone differentiation and development. J Cell Biol.
172:795–797. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Zhang J, Lazarenko OP, Blackburn ML,
Badger TM, Ronis MJ and Chen JR: Blueberry consumption prevents
loss of collagen in bone matrix and inhibits senescence pathways in
osteoblastic cells. Age (Dordr). 35:807–820. 2013. View Article : Google Scholar
|
|
45
|
Li Y and Tollefsbol TO: p16(INK4a)
suppression by glucose restriction contributes to human cellular
lifespan extension through SIRT1-mediated epigenetic and genetic
mechanisms. PLoS One. 6:e174212011. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Campisi J and d'Adda di Fagagna F:
Cellular senescence: when bad things happen to good cells. Nat Rev
Mol Cell Biol. 8:729–740. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Wang X, Huang H and Young KH: The PTEN
tumor suppressor gene and its role in lymphoma pathogenesis. Aging
(Albany NY). 7:1032–1049. 2015. View Article : Google Scholar
|
|
48
|
Zhang LY, Ho-Fun Lee V, Wong AM, Kwong DL,
Zhu YH, Dong SS, Kong KL, Chen J, Tsao SW, Guan XY, et al:
Micro-RNA-144 promotes cell proliferation, migration and invasion
in nasopharyngeal carcinoma through repression of PTEN.
Carcinogenesis. 34:454–463. 2013. View Article : Google Scholar
|
|
49
|
Blüml S, Friedrich M, Lohmeyer T, Sahin E,
Saferding V, Brunner J, Puchner A, Mandl P, Niederreiter B, Smolen
JS, et al: Loss of phosphatase and tensin homolog (PTEN) in myeloid
cells controls inflammatory bone destruction by regulating the
osteoclastogenic potential of myeloid cells. Ann Rheum Dis.
74:227–233. 2015. View Article : Google Scholar
|
|
50
|
Jang HD, Noh JY, Shin JH, Lin JJ and Lee
SY: PTEN regulation by the Akt/GSK-3β axis during RANKL signaling.
Bone. 55:126–131. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Sugatani T, Alvarez U and Hruska KA: PTEN
regulates RANKL- and osteopontin-stimulated signal transduction
during osteoclast differentiation and cell motility. J Biol Chem.
278:5001–5008. 2003. View Article : Google Scholar
|
|
52
|
Hu G, Han C and Chen J: Inhibition of
oncogene expression by green tea and (−)-epigallocatechin gallate
in mice. Nutr Cancer. 24:203–209. 1995. View Article : Google Scholar
|
|
53
|
Prasad KN, Kumar A, Kochupillai V and Cole
WC: High doses of multiple antioxidant vitamins: essential
ingredients in improving the efficacy of standard cancer therapy. J
Am Coll Nutr. 18:13–25. 1999. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Xie L, Jiang Y, Ouyang P, Chen J, Doan H,
Herndon B, Sylvester JE, Zhang K, Molteni A, Reichle M, et al:
Effects of dietary calorie restriction or exercise on the PI3K and
Ras signaling pathways in the skin of mice. J Biol Chem.
282:28025–28035. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Huang L, Bi HC, Liu YH, Wang YT, Xue XP
and Huang M: CAR-mediated up-regulation of CYP3A4 expression in
LS174T cells by Chinese herbal compounds. Drug Metab Pharmacokinet.
26:331–340. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
56
|
Yeon JT, Kim KJ, Choi SW, Moon SH, Park
YS, Ryu BJ, Oh J, Kim MS, Erkhembaatar M, Son YJ, et al:
Anti-osteoclastogenic activity of praeruptorin A via inhibition of
p38/Akt-c-Fos-NFATc1 signaling and PLCγ-independent Ca2+
oscillation. PLoS One. 9:e889742014. View Article : Google Scholar
|
|
57
|
Hasegawa S, Yonezawa T, Ahn JY, Cha BY,
Teruya T, Takami M, Yagasaki K, Nagai K and Woo JT: Honokiol
inhibits osteoclast differentiation and function in vitro. Biol
Pharm Bull. 33:487–492. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
58
|
Li J, Shao X, Wu L, Feng T, Jin C, Fang M,
Wu N and Yao H: Honokiol: an effective inhibitor of tumor necrosis
factor-α-induced up-regulation of inflammatory cytokine and
chemokine production in human synovial fibroblasts. Acta Biochim
Biophys Sin (Shanghai). 43:380–386. 2011. View Article : Google Scholar
|
|
59
|
Hahm ER, Singh KB and Singh SV: c-Myc is a
novel target of cell cycle arrest by honokiol in prostate cancer
cells. Cell Cycle. 15:2309–2320. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
60
|
Liu H, Zang C, Emde A, Planas-Silva MD,
Rosche M, Kühnl A, Schulz CO, Elstner E, Possinger K and Eucker J:
Anti-tumor effect of honokiol alone and in combination with other
anti-cancer agents in breast cancer. Eur J Pharmacol. 591:43–51.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
61
|
Zhang J, Lazarenko OP, Blackburn ML,
Shankar K, Badger TM, Ronis MJ and Chen JR: Feeding blueberry diets
in early life prevent senescence of osteoblasts and bone loss in
ovariectomized adult female rats. PLoS One. 6:e244862011.
View Article : Google Scholar : PubMed/NCBI
|
|
62
|
Liu W, Lu X, He G, Gao X, Li M, Wu J, Li
Z, Wu J, Wang J and Luo C: Cytosolic protection against ultraviolet
induced DNA damage by blueberry anthocyanins and anthocyanidins in
hepatocarcinoma HepG2 cells. Biotechnol Lett. 35:491–498. 2013.
View Article : Google Scholar
|
|
63
|
Vetterli L, Brun T, Giovannoni L, Bosco D
and Maechler P: Resveratrol potentiates glucose-stimulated insulin
secretion in INS-1E beta-cells and human islets through a
SIRT1-dependent mechanism. J Biol Chem. 286:6049–6060. 2011.
View Article : Google Scholar
|
|
64
|
Martí-Centelles R, Falomir E, Murga J,
Carda M and Marco JA: Inhibitory effect of cytotoxic stilbenes
related to resveratrol on the expression of the VEGF, hTERT and
c-Myc genes. Eur J Med Chem. 103:488–496. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
65
|
Wang X, Wang D and Zhao Y: Effect and
mechanism of resveratrol on the apoptosis of lung adenocarcinoma
cell line A549. Cell Biochem Biophys. 73:527–531. 2015. View Article : Google Scholar
|
|
66
|
Lee SJ, Park K, Ha SD, Kim WJ and Moon SK:
Gleditsia sinensis thorn extract inhibits human colon cancer cells:
the role of ERK1/2, G2/M-phase cell cycle arrest and p53
expression. Phytother Res. 24:1870–1876. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
67
|
Lee SJ, Ryu DH, Jang LC, Cho SC, Kim WJ
and Moon SK: Suppressive effects of an ethanol extract of Gleditsia
sinensis thorns on human SNU-5 gastric cancer cells. Oncol Rep.
29:1609–1616. 2013.PubMed/NCBI
|
|
68
|
Lu Y, Li CS and Dong Q: Chinese herb
related molecules of cancer-cell-apoptosis: a minireview of
progress between Kanglaite injection and related genes. J Exp Clin
Cancer Res. 27:312008. View Article : Google Scholar : PubMed/NCBI
|
|
69
|
Li B, Zhao J, Wang CZ, Searle J, He TC,
Yuan CS and Du W: Ginsenoside Rh2 induces apoptosis and
paraptosis-like cell death in colorectal cancer cells through
activation of p53. Cancer Lett. 301:185–192. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
70
|
Gali-Muhtasib H, Kuester D, Mawrin C,
Bajbouj K, Diestel A, Ocker M, Habold C, Foltzer-Jourdainne C,
Schoenfeld P, Peters B, et al: Thymoquinone triggers inactivation
of the stress response pathway sensor CHEK1 and contributes to
apoptosis in colorectal cancer cells. Cancer Res. 68:5609–5618.
2008. View Article : Google Scholar : PubMed/NCBI
|
|
71
|
Ichwan SJ, Al-Ani IM, Bilal HG, Suriyah
WH, Taher M and Ikeda MA: Apoptotic activities of thymoquinone, an
active ingredient of black seed (Nigella sativa), in cervical
cancer cell lines. Chin J Physiol. 57:249–255. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
72
|
Lee SJ, Kim HM, Cho YH, Park K, Kim EJ,
Jung KH, Kim CH, Kim WJ and Moon SK: Aqueous extract of Magnolia
officinalis mediates proliferative capacity, p21WAF1 expression and
TNF-alpha-induced NF-kappaB activity in human urinary bladder
cancer 5637 cells; involvement of p38 MAP kinase. Oncol Rep.
18:729–736. 2007.PubMed/NCBI
|
|
73
|
Dong LH, Wen JK, Miao SB, Jia Z, Hu HJ,
Sun RH, Wu Y and Han M: Baicalin inhibits PDGF-BB-stimulated
vascular smooth muscle cell proliferation through suppressing
PDGFRβ-ERK signaling and increase in p27 accumulation and prevents
injury-induced neointimal hyperplasia. Cell Res. 20:1252–1262.
2010. View Article : Google Scholar : PubMed/NCBI
|
|
74
|
Zhang L, Pu Z and Wang J, Zhang Z, Hu D
and Wang J: Baicalin inhibits hypoxia-induced pulmonary artery
smooth muscle cell proliferation via the AKT/HIF-1α/p27-associated
pathway. Int J Mol Sci. 15:8153–8168. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
75
|
Byambaragchaa M, Dela Cruz J, Kh A and
Hwang SG: Anticancer potential of an ethanol extract of Saussurea
involucrata against hepatic cancer cells in vitro. Asian Pac J
Cancer Prev. 15:7527–7532. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
76
|
Liu J, Shen M, Yue Z, Yang Z, Wang M, Li
C, Xin C, Wang Y, Mei Q and Wang Z: Triptolide inhibits
colon-rectal cancer cells proliferation by induction of G1 phase
arrest through upregulation of p21. Phytomedicine. 19:756–762.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
77
|
Liu TE, Zhang L, Wang S, Chen C and Zheng
J: Tripterygium glycosides induce premature ovarian failure in rats
by promoting p53 phosphorylation and activating the
serine/threonine kinase 11-p53-p21 signaling pathway. Exp Ther Med.
10:12–18. 2015.PubMed/NCBI
|
|
78
|
Wong TF, Takeda T, Li B, Tsuiji K,
Kitamura M, Kondo A and Yaegashi N: Curcumin disrupts uterine
leiomyosarcoma cells through AKT-mTOR pathway inhibition. Gynecol
Oncol. 122:141–148. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
79
|
Yoshida H, Okumura N, Kitagishi Y,
Nishimura Y and Matsuda S: Ethanol extract of Rosemary repressed
PTEN expression in K562 culture cells. Int J Appl Biol Pharm
Technol. 2:316–322. 2011.
|
|
80
|
Wang X, He H, Lu Y, Ren W, Teng KY, Chiang
CL, Yang Z, Yu B, Hsu S, Jacob ST, et al: Indole-3-carbinol
inhibits tumorigenicity of hepatocellular carcinoma cells via
suppression of microRNA-21 and upregulation of phosphatase and
tensin homolog. Biochim Biophys Acta. 1853:244–253. 2015.
View Article : Google Scholar :
|
|
81
|
Royston KJ and Tollefsbol TO: The
epigenetic impact of cruciferous vegetables on cancer prevention.
Curr Pharmacol Rep. 1:46–51. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
82
|
Eason RR, Velarde MC, Chatman L Jr, Till
SR, Geng Y, Ferguson M, Badger TM and Simmen RC: Dietary exposure
to whey proteins alters rat mammary gland proliferation, apoptosis,
and gene expression during postnatal development. J Nutr.
134:3370–3377. 2004.PubMed/NCBI
|
|
83
|
Ghosh-Choudhury T, Mandal CC, Woodruff K,
St Clair P, Fernandes G, Choudhury GG and Ghosh-Choudhury N: Fish
oil targets PTEN to regulate NFkappaB for downregulation of
anti-apoptotic genes in breast tumor growth. Breast Cancer Res
Treat. 118:213–228. 2009. View Article : Google Scholar
|
|
84
|
Ishii H, Horie Y, Ohshima S, Anezaki Y,
Kinoshita N, Dohmen T, Kataoka E, Sato W, Goto T, Sasaki J, et al:
Eicosapentaenoic acid ameliorates steatohepatitis and
hepatocellular carcinoma in hepatocyte-specific Pten-deficient
mice. J Hepatol. 50:562–571. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
85
|
Rovito D, Giordano C, Vizza D, Plastina P,
Barone I, Casaburi I, Lanzino M, De Amicis F, Sisci D, Mauro L, et
al: Omega-3 PUFA ethanolamides DHEA and EPEA induce autophagy
through PPARγ activation in MCF-7 breast cancer cells. J Cell
Physiol. 228:1314–1322. 2013. View Article : Google Scholar
|
|
86
|
Kitagishi Y and Matsuda S: Diets involved
in PPAR and PI3K/AKT/PTEN pathway may contribute to neuroprotection
in a traumatic brain injury. Alzheimers Res Ther. 5:422013.
View Article : Google Scholar : PubMed/NCBI
|
|
87
|
Burdick AD, Bility MT, Girroir EE, Billin
AN, Willson TM, Gonzalez FJ and Peters JM: Ligand activation of
peroxisome proliferator-activated
receptor-beta/delta(PPARbeta/delta) inhibits cell growth of human
N/TERT-1 keratinocytes. Cell Signal. 19:1163–1171. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
88
|
Mohapatra P, Satapathy SR, Siddharth S,
Das D, Nayak A and Kundu CN: Resveratrol and curcumin
synergistically induces apoptosis in cigarette smoke condensate
transformed breast epithelial cells through a p21(Waf1/Cip1)
mediated inhibition of Hh-Gli signaling. Int J Biochem Cell Biol.
66:75–84. 2015. View Article : Google Scholar : PubMed/NCBI
|