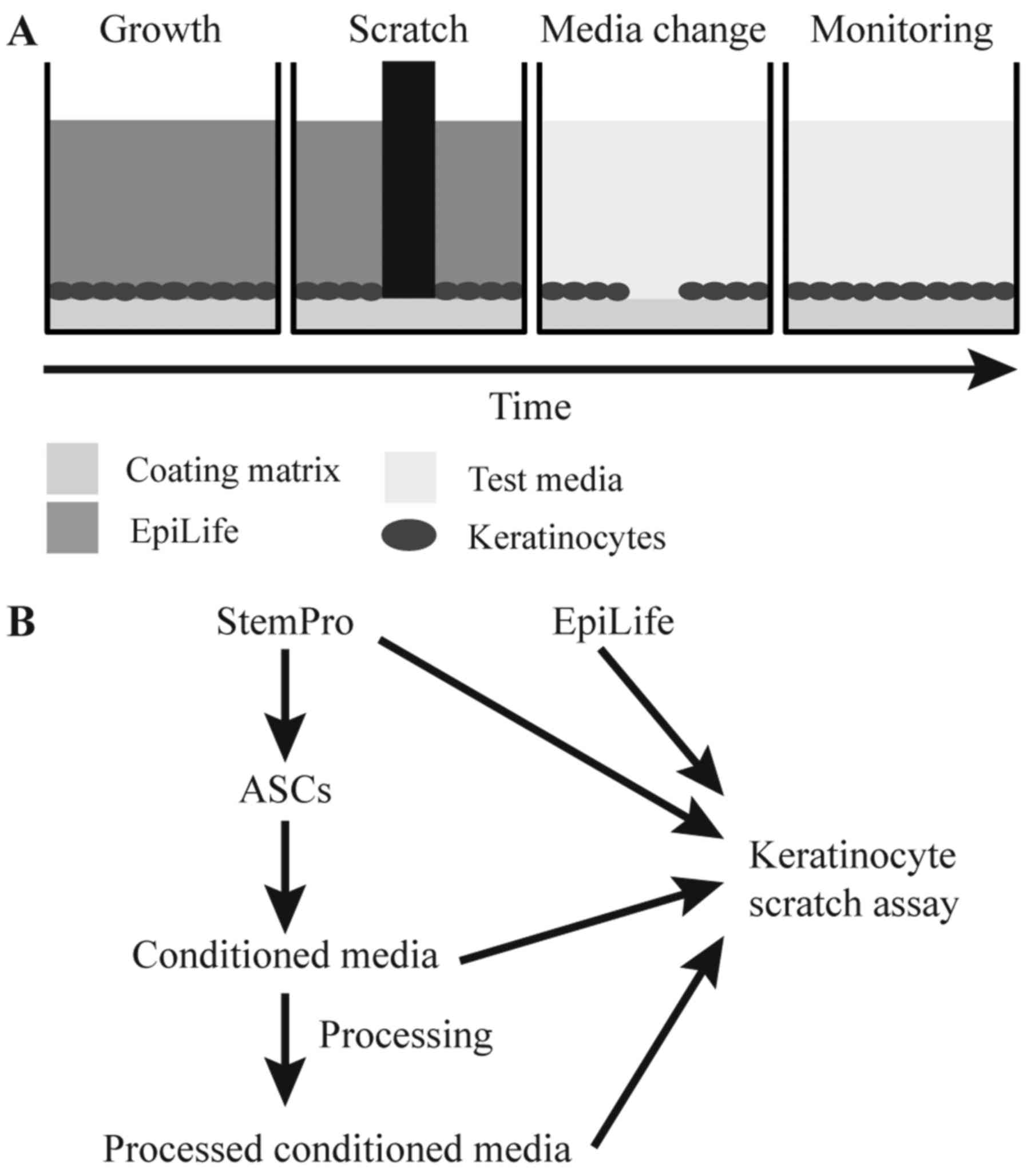
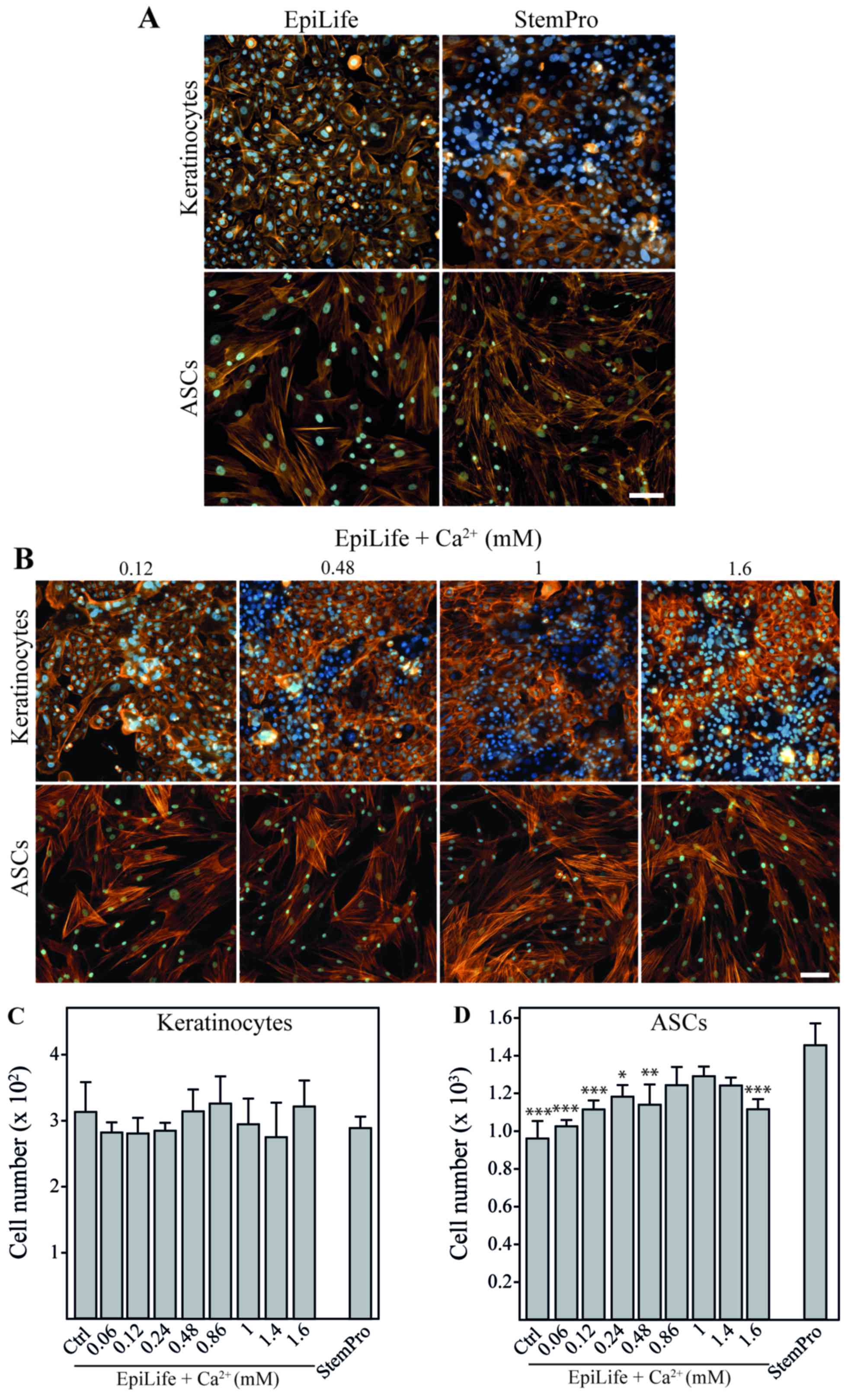
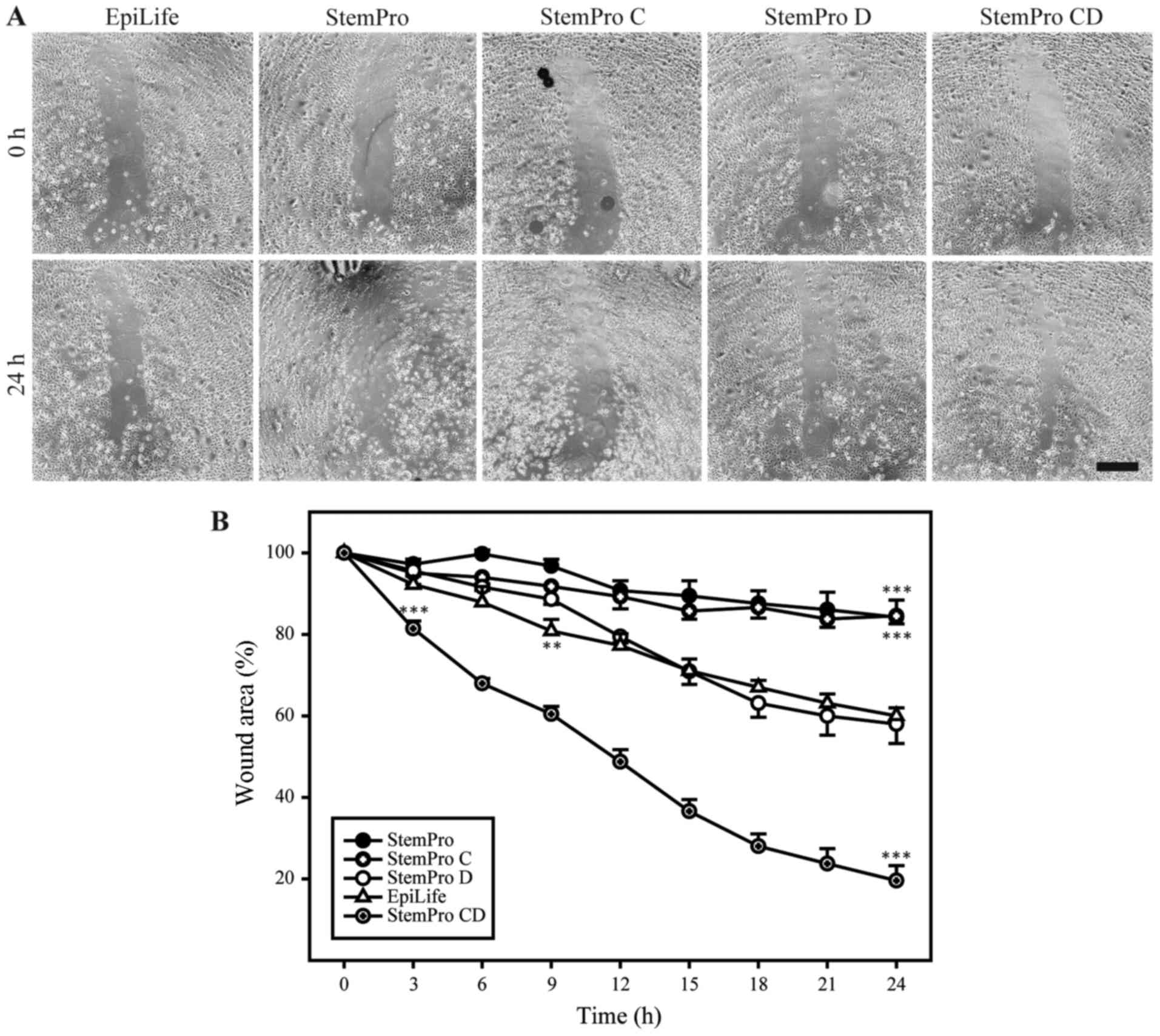
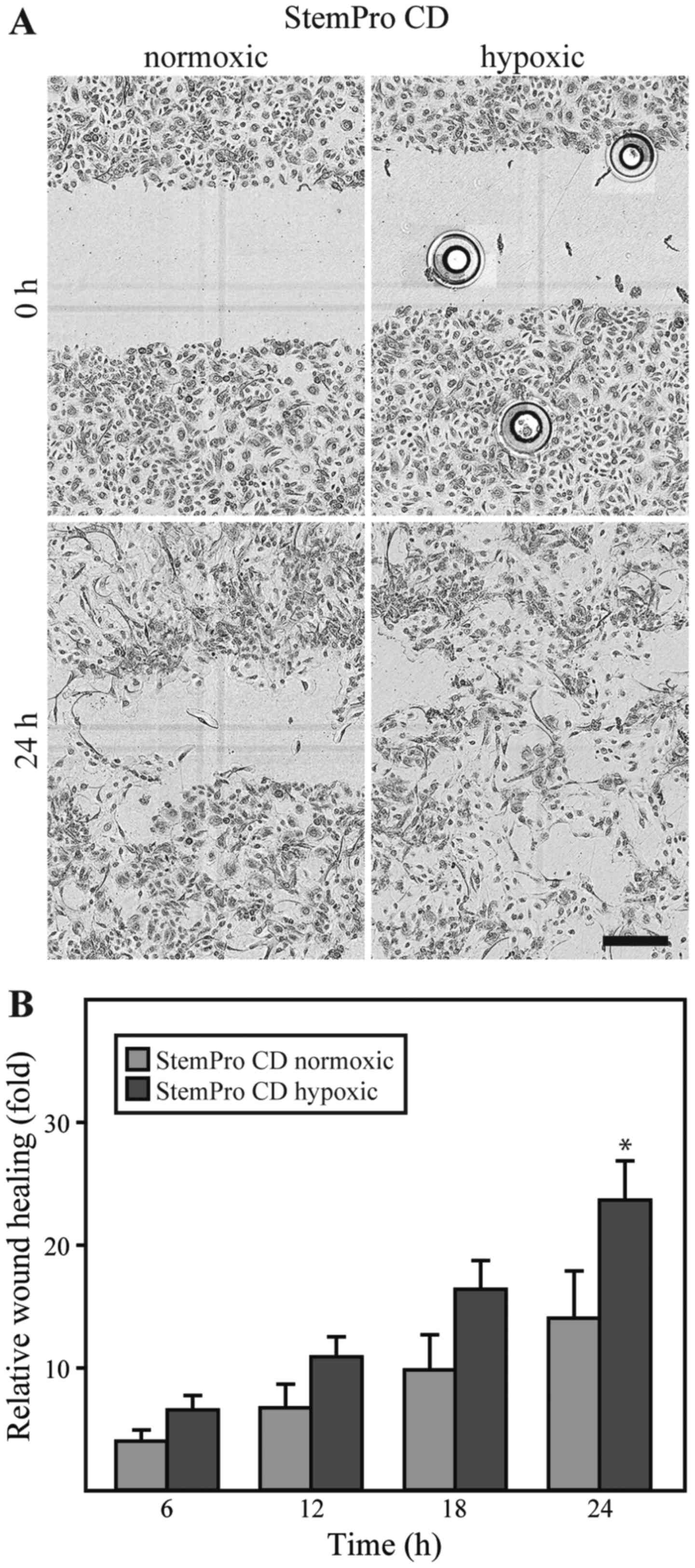
|
1
|
Hassan WU, Greiser U and Wang W: Role of
adipose-derived stem cells in wound healing. Wound Repair Regen.
22:313–325. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Rasmussen JG, Frøbert O, Pilgaard L,
Kastrup J, Simonsen U, Zachar V and Fink T: Prolonged hypoxic
culture and trypsinization increase the pro-angiogenic potential of
human adipose tissue-derived stem cells. Cytotherapy. 13:318–328.
2011. View Article : Google Scholar
|
|
3
|
Lee SH, Jin SY, Song JS, Seo KK and Cho
KH: Paracrine effects of adipose-derived stem cells on
keratinocytes and dermal fibroblasts. Ann Dermatol. 24:136–143.
2012. View Article : Google Scholar : PubMed/NCBI
|
|
4
|
Rehman J, Traktuev D, Li J, Merfeld-Clauss
S, Temm-Grove CJ, Bovenkerk JE, Pell CL, Johnstone BH, Considine RV
and March KL: Secretion of angiogenic and antiapoptotic factors by
human adipose stromal cells. Circulation. 109:1292–1298. 2004.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Hsiao ST, Lokmic Z, Peshavariya H,
Abberton KM, Dusting GJ, Lim SY and Dilley RJ: Hypoxic conditioning
enhances the angiogenic paracrine activity of human adipose-derived
stem cells. Stem Cells Dev. 22:1614–1623. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Pastar I, Stojadinovic O, Yin NC, Ramirez
H, Nusbaum AG, Sawaya A, Patel SB, Khalid L, Isseroff RR and
Tomic-Canic M: Epithelialization in Wound Healing: A comprehensive
review. Adv Wound Care (New Rochelle). 3:445–464. 2014. View Article : Google Scholar
|
|
7
|
Miranda JP, Filipe E, Fernandes AS,
Almeida JM, Martins JP, De la Fuente A, Abal M, Barcia RN, Cruz P,
Cruz H, et al: The human umbilical cord tissue-derived MSC
population UCX(®) promotes early motogenic effects on keratinocytes
and fibroblasts and G-CSF-mediated mobilization of BM-MSCs when
transplanted in vivo. Cell Transplant. 24:865–877. 2015. View Article : Google Scholar
|
|
8
|
Walter MN, Wright KT, Fuller HR, MacNeil S
and Johnson WE: Mesenchymal stem cell-conditioned medium
accelerates skin wound healing: An in vitro study of fibroblast and
keratinocyte scratch assays. Exp Cell Res. 316:1271–1281. 2010.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Hanahan D and Weinberg RA: Hallmarks of
cancer: The next generation. Cell. 144:646–674. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
10
|
Zhang J, Antonyak MA, Singh G and Cerione
RA: A mechanism for the upregulation of EGF receptor levels in
glioblastomas. Cell Reports. 3:2008–2020. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Ahmad I, Iwata T and Leung HY: Mechanisms
of FGFR-mediated carcinogenesis. Biochim Biophys Acta.
1823:850–860. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Turunen A and Syrjänen S: Extracellular
calcium regulates keratinocyte proliferation and HPV 16 E6 RNA
expression in vitro. APMIS. 122:781–789. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
13
|
Micallef L, Belaubre F, Pinon A,
Jayat-Vignoles C, Delage C, Charveron M and Simon A: Effects of
extracellular calcium on the growth-differentiation switch in
immortalized keratinocyte HaCaT cells compared with normal human
keratinocytes. Exp Dermatol. 18:143–151. 2009. View Article : Google Scholar
|
|
14
|
Conrad DR: Calcium in Cell Culture.
(Technical note). Sigma-Aldrich. http://www.sigmaaldrich.com/life-science/cell-culture/learning-center/media-expert/calcium.html.
|
|
15
|
Zachar V, Rasmussen JG and Fink T:
Isolation and growth of adipose tissue-derived stem cells. Methods
Mol Biol. 698:37–49. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Rasmussen JG, Riis SE, Frøbert O, Yang S,
Kastrup J, Zachar V, Simonsen U and Fink T: Activation of
protease-activated receptor 2 induces VEGF independently of HIF-1.
PLoS One. 7:e460872012. View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Yang S, Pilgaard L, Chase LG, Boucher S,
Vemuri MC, Fink T and Zachar V: Defined xenogeneic-free and hypoxic
environment provides superior conditions for long-term expansion of
human adipose-derived stem cells. Tissue Eng Part C Methods.
18:593–602. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Fink T, Rasmussen JG, Lund P, Pilgaard L,
Soballe K and Zachar V: Isolation and expansion of adipose-derived
stem cells for tissue engineering. Front Biosci (Elite Ed). 3. pp.
256–263. 2011, View
Article : Google Scholar
|
|
19
|
Prasad M, Zachar V, Fink T and Pennisi CP:
Moderate hypoxia influences potassium outward currents in
adipose-derived stem cells. PLoS One. 9:e1049122014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Gebäck T, Schulz MMP, Koumoutsakos P and
Detmar M: TScratch: A novel and simple software tool for automated
analysis of monolayer wound healing assays. Biotechniques.
46:265–274. 2009.PubMed/NCBI
|
|
21
|
Vespa A, Darmon AJ, Turner CE, D'Souza SJA
and Dagnino L: Ca2+-dependent localization of
integrin-linked kinase to cell junctions in differentiating
keratinocytes. J Biol Chem. 278:11528–11535. 2003. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Cerqueira MT, Pirraco RP and Marques AP:
Stem cells in skin wound healing: Are we there yet? Adv Wound Care
(New Rochelle). 5:164–175. 2016. View Article : Google Scholar
|
|
23
|
Seo BF, Kim KJ, Kim MK and Rhie JW: The
effects of human keratinocyte coculture on human adipose-derived
stem cells. Int Wound J. 13:630–635. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Bikle DD, Xie Z and Tu CL: Calcium
regulation of keratinocyte differentiation. Expert Rev Endocrinol
Metab. 7:461–472. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Moreno-Bueno G, Peinado H, Molina P,
Olmeda D, Cubillo E, Santos V, Palacios J, Portillo F and Cano A:
The morphological and molecular features of the
epithelial-to-mesenchymal transition. Nat Protoc. 4:1591–1613.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Werner S, Krieg T and Smola H:
Keratinocyte-fibroblast interactions in wound healing. J Invest
Dermatol. 127:998–1008. 2007. View Article : Google Scholar : PubMed/NCBI
|
|
27
|
Kapur SK and Katz AJ: Review of the
adipose derived stem cell secretome. Biochimie. 95:2222–2228. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Peplow PV and Chatterjee MP: A review of
the influence of growth factors and cytokines in in vitro human
keratinocyte migration. Cytokine. 62:1–21. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Zachar V, Duroux M, Emmersen J, Rasmussen
JG, Pennisi CP, Yang S and Fink T: Hypoxia and adipose-derived stem
cell-based tissue regeneration and engineering. Expert Opin Biol
Ther. 11:775–786. 2011. View Article : Google Scholar : PubMed/NCBI
|
|
30
|
Liu L, Gao J, Yuan Y, Chang Q, Liao Y and
Lu F: Hypoxia preconditioned human adipose derived mesenchymal stem
cells enhance angiogenic potential via secretion of increased VEGF
and bFGF. Cell Biol Int. 37:551–560. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
31
|
An HY, Shin HS, Choi JS, Kim HJ, Lim JY
and Kim YM: Adipose mesenchymal stem cell secretome modulated in
hypoxia for remodeling of radiation-induced salivary gland damage.
PLoS One. 10:e01418622015. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Riis S, Stensballe A, Emmersen J, Pennisi
CP, Birkelund S, Zachar V and Fink T: Mass spectrometry analysis of
adipose-derived stem cells reveals a significant effect of hypoxia
on pathways regulating extracellular matrix. Stem Cell Res Ther.
7:522016. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Riis S, Zachar V, Boucher S, Vemuri MC,
Pennisi CP and Fink T: Critical steps in the isolation and
expansion of adipose-derived stem cells for translational therapy.
Expert Rev Mol Med. 17:e112015. View Article : Google Scholar : PubMed/NCBI
|