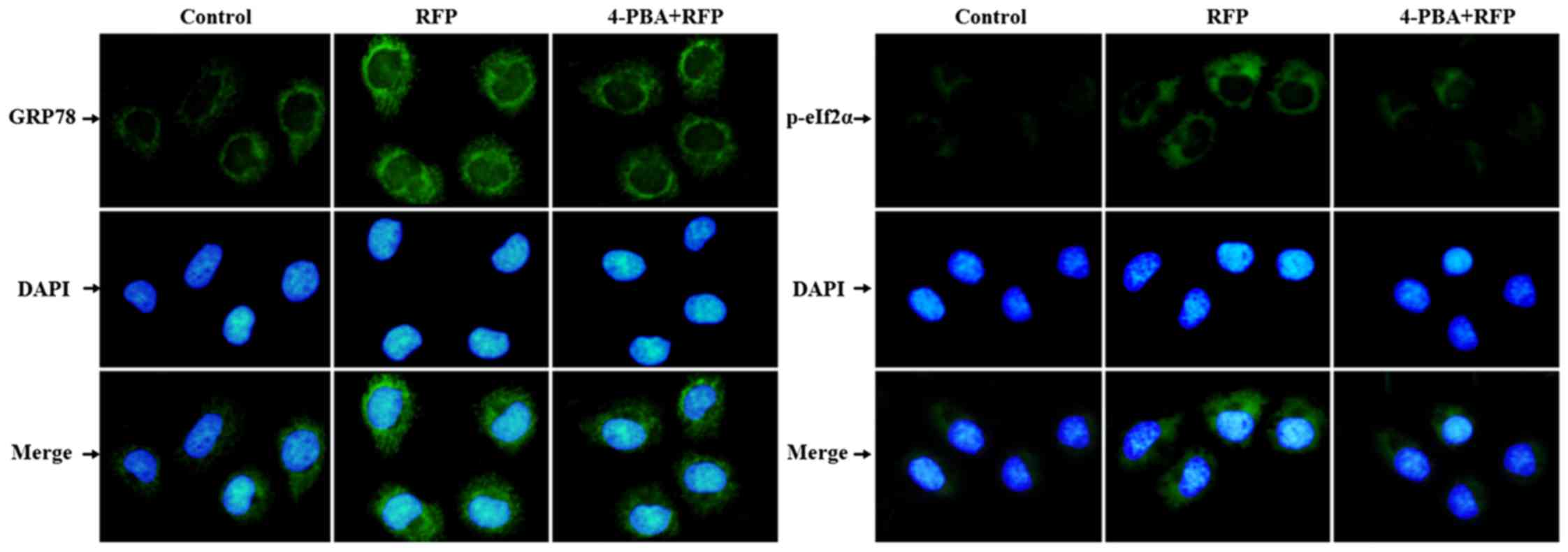
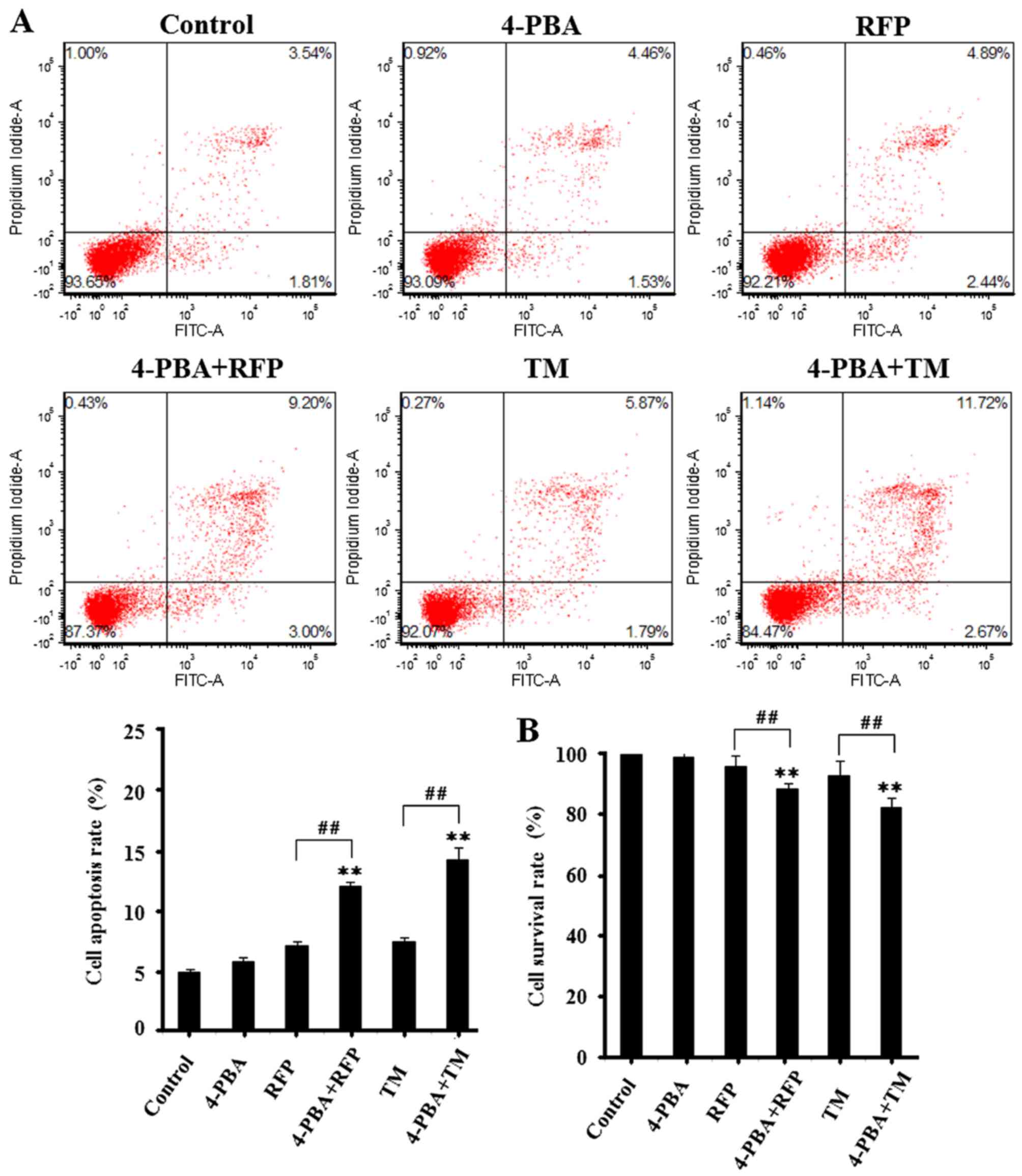
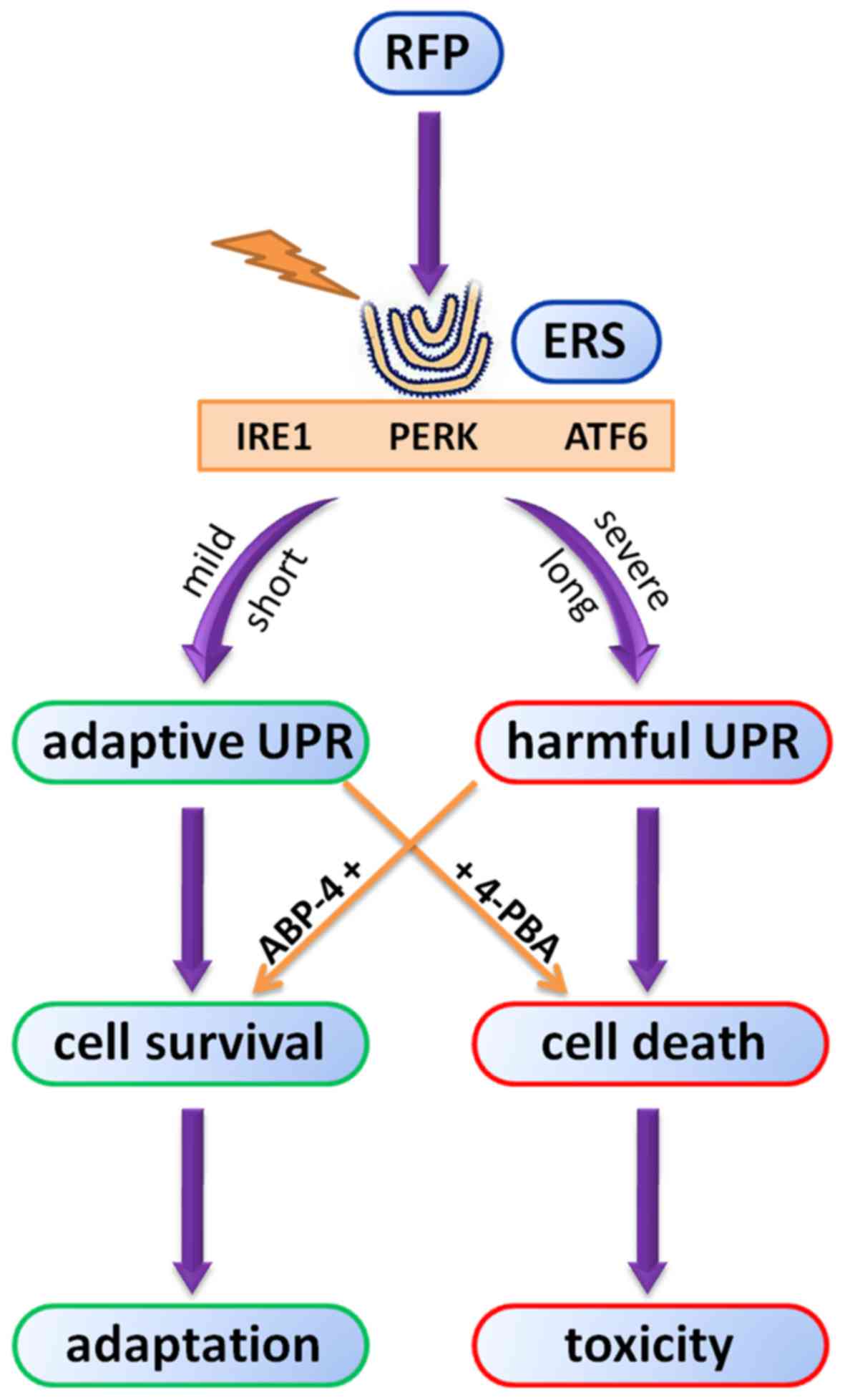
|
1
|
Perri ER, Thomas CJ, Parakh S, Spencer DM
and Atkin JD: The unfolded protein response and the role of protein
disulfide isomerase in neurodegeneration. Front Cell Dev Biol.
3:802016. View Article : Google Scholar : PubMed/NCBI
|
|
2
|
Mollereau B, Manié S and Napoletano F:
Getting the better of ER stress. J Cell Commun Signal. 8:311–321.
2014. View Article : Google Scholar : PubMed/NCBI
|
|
3
|
Takayanagi S, Fukuda R, Takeuchi Y,
Tsukada S and Yoshida K: Gene regulatory network of unfolded
protein response genes in endoplasmic reticulum stress. Cell Stress
Chaperones. 18:11–23. 2013. View Article : Google Scholar :
|
|
4
|
Sano R and Reed JC: ER stress-induced cell
death mechanisms. Biochim Biophys Acta. 1833:3460–3470. 2013.
View Article : Google Scholar : PubMed/NCBI
|
|
5
|
Lindquist SL and Kelly JW: Chemical and
biological approaches for adapting proteostasis to ameliorate
protein misfolding and aggregation diseases: Progress and
prognosis. Cold Spring Harb Perspect Biol. 3:a0045072011.
View Article : Google Scholar : PubMed/NCBI
|
|
6
|
Smith HL and Mallucci GR: The unfolded
protein response: Mechanisms and therapy of neurodegeneration.
Brain. 139:2113–2121. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
7
|
Hassler JR, Scheuner DL, Wang S, Han J,
Kodali VK, Li P, Nguyen J, George JS, Davis C, Wu SP, et al: The
IRE1α/XBP1s pathway is essential for the glucose response and
protection of β cells. PLoS Biol. 13:e10022772015. View Article : Google Scholar
|
|
8
|
Lugea A, Waldron RT and Pandol SJ:
Pancreatic adaptive responses in alcohol abuse: Role of the
unfolded protein response. Pancreatology. 15(Suppl 4): S1–S5. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
9
|
Lugea A, Tischler D, Nguyen J, Gong J,
Gukovsky I, French SW, Gorelick FS and Pandol SJ: Adaptive unfolded
protein response attenuates alcohol-induced pancreatic damage.
Gastroenterology. 140:987–997. 2011. View Article : Google Scholar :
|
|
10
|
Cano M, Wang L, Wan J, Barnett BP,
Ebrahimi K, Qian J and Handa JT: Oxidative stress induces
mitochondrial dysfunction and a protective unfolded protein
response in RPE cells. Free Radic Biol Med. 69:1–14. 2014.
View Article : Google Scholar : PubMed/NCBI
|
|
11
|
Benson EA, Eadon MT, Desta Z, Liu Y, Lin
H, Burgess KS, Segar MW, Gaedigk A and Skaar TC: Rifampin
regulation of drug transporters gene expression and the association
of MicroRNAs in human hepatocytes. Front Pharmacol. 7:1112016.
View Article : Google Scholar : PubMed/NCBI
|
|
12
|
Wang W, Ren X, Cai Y, Chen L, Zhang W and
Xu J: Rifampicin induces bicarbonate-rich choleresis in rats:
Involvement of anion exchanger 2. Dig Dis Sci. 61:126–136. 2016.
View Article : Google Scholar
|
|
13
|
Chen X, Zhang C, Wang H, Xu J, Duan ZH,
Zhang Y, Yu T, Wei W, Xu DX and Xu JM: Altered integrity and
decreased expression of hepatocyte tight junctions in
rifampicin-induced cholestasis in mice. Toxicol Appl Pharmacol.
240:26–36. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
14
|
Verma S and Kaplowitz N: Diagnosis,
management and prevention of drug-induced liver injury. Gut.
58:1555–1564. 2009. View Article : Google Scholar : PubMed/NCBI
|
|
15
|
Carlisle RE, Brimble E, Werner KE, Cruz
GL, Ask K, Ingram AJ and Dickhout JG: 4-Phenylbutyrate inhibits
tunicamycin-induced acute kidney injury via CHOP/GADD153
repression. PLoS One. 9:e846632014. View Article : Google Scholar : PubMed/NCBI
|
|
16
|
Kolb PS, Ayaub EA, Zhou W, Yum V, Dickhout
JG and Ask K: The therapeutic effects of 4-phenylbutyric acid in
maintaining proteostasis. Int J Biochem Cell Biol. 61:45–52. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
17
|
Hong D, Li LF, Gao HC, Wang X, Li CC, Luo
Y, Bai YP and Zhang GG: High-density lipoprotein prevents
endoplasmic reticulum stress-induced downregulation of liver LOX-1
expression. PLoS One. 10:e01242852015. View Article : Google Scholar : PubMed/NCBI
|
|
18
|
Rahman SM, Qadri I, Janssen RC and
Friedman JE: Fenofibrate and PBA prevent fatty acid-induced loss of
adiponectin receptor and pAMPK in human hepatoma cells and in
hepatitis C virus-induced steatosis. J Lipid Res. 50:2193–2202.
2009. View Article : Google Scholar : PubMed/NCBI
|
|
19
|
Wang L, Tang W, Jiang T, Lu P, Li Y, Sun
A, Shen Y, Chen Y, Wang H, Zong Z, et al: Endoplasmic reticulum
stress is involved in the neuroprotective effect of propofol.
Neurochem Res. 39:1741–1752. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
20
|
Douros A, Bronder E, Andersohn F, Klimpel
A, Thomae M, Sarganas G, Kreutz R and Garbe E: Drug-induced liver
injury: Results from the hospital-based Berlin Case-Control
Surveillance Study. Br J Clin Pharmacol. 79:988–999. 2015.
View Article : Google Scholar :
|
|
21
|
Takeda K, Oda M, Okada T, Yamazaki H, Ohbu
M, Watanabe M and Yokomori H: Over-the-counter drug induced
autoimmune hepatitis. Intern Med. 55:1293–1297. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
22
|
Rossi S and Navarro VJ: Herbs and liver
injury: A clinical perspective. Clin Gastroenterol Hepatol.
12:1069–1076. 2014. View Article : Google Scholar
|
|
23
|
Kaneko R, Ohishi C, Kim M, Shiina M,
Kusayanagi S, Ogawa M, Munakata K, Mizuno K and Sato Y: Two cases
of food additive-induced severe liver damage associated with
positive results on lymphocyte stimulation test and for antinuclear
anti-bodies. Clin J Gastroenterol. 5:268–274. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
24
|
Navarro VJ, Barnhart H, Bonkovsky HL,
Davern T, Fontana RJ, Grant L, Reddy KR, Seeff LB, Serrano J,
Sherker AH, et al: Liver injury from herbals and dietary
supplements in the U.S. Drug-Induced Liver Injury Network.
Hepatology. 60:1399–1408. 2014. View Article : Google Scholar : PubMed/NCBI
|
|
25
|
Chalasani N, Bonkovsky HL, Fontana R, Lee
W, Stolz A, Talwalkar J, Reddy KR, Watkins PB, Navarro V, Barnhart
H, et al United States Drug Induced Liver Injury Network: Features
and outcomes of 899 patients with drug-induced liver injury: The
DILIN prospective study. Gastroenterology. 148:1340–52e7. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
26
|
Ortega-Alonso A, Stephens C, Lucena MI and
Andrade RJ: Case characterization, clinical features and risk
factors in drug-induced liver injury. Int J Mol Sci. 17:7142016.
View Article : Google Scholar :
|
|
27
|
Chen M, Suzuki A, Borlak J, Andrade RJ and
Lucena MI: Drug-induced liver injury: Interactions between drug
properties and host factors. J Hepatol. 63:503–514. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
28
|
Björnsson ES: Hepatotoxicity by drugs: The
most common implicated agents. Int J Mol Sci. 17:2242016.
View Article : Google Scholar : PubMed/NCBI
|
|
29
|
Haque T, Sasatomi E and Hayashi PH:
Drug-induced liver injury: Pattern recognition and future
directions. Gut Liver. 10:27–36. 2016. View
Article : Google Scholar :
|
|
30
|
Devarbhavi H: Antituberculous drug-induced
liver injury: Current perspective. Trop Gastroenterol. 32:167–174.
2011.
|
|
31
|
Saukkonen JJ, Cohn DL, Jasmer RM, Schenker
S, Jereb JA, Nolan CM, Peloquin CA, Gordin FM, Nunes D, Strader DB,
et al ATS (American Thoracic Society) hepatotoxicity of
anti-tuberculosis therapy subcommittee: An official ATS statement:
Hepatotoxicity of antituberculosis therapy. Am J Respir Crit Care
Med. 174:935–952. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
32
|
Watkins PB and Seeff LB: Drug-induced
liver injury: Summary of a single topic clinical research
conference. Hepatology. 43:618–631. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
33
|
Foufelle F and Fromenty B: Role of
endoplasmic reticulum stress in drug-induced toxicity. Pharmacol
Res Perspect. 4:e002112016. View
Article : Google Scholar : PubMed/NCBI
|
|
34
|
Herbert TP and Laybutt DR: A Reevaluation
of the role of the unfolded protein response in islet dysfunction:
Maladaptation or a failure to adapt? Diabetes. 65:1472–1480. 2016.
View Article : Google Scholar : PubMed/NCBI
|
|
35
|
Su HY, Waldron RT, Gong R, Ramanujan VK,
Pandol SJ and Lugea A: The unfolded protein response plays a
predominant homeostatic role in response to mitochondrial stress in
pancreatic stellate cells. PLoS One. 11:e01489992016. View Article : Google Scholar : PubMed/NCBI
|
|
36
|
Oñate M, Catenaccio A, Martínez G,
Armentano D, Parsons G, Kerr B, Hetz C and Court FA: Activation of
the unfolded protein response promotes axonal regeneration after
peripheral nerve injury. Sci Rep. 6:217092016. View Article : Google Scholar : PubMed/NCBI
|
|
37
|
Bensellam M, Maxwell EL, Chan JY,
Luzuriaga J, West PK, Jonas JC, Gunton JE and Laybutt DR: Hypoxia
reduces ER-to-Golgi protein trafficking and increases cell death by
inhibiting the adaptive unfolded protein response in mouse beta
cells. Diabetologia. 59:1492–1502. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
38
|
Chan JY, Luzuriaga J, Bensellam M, Biden
TJ and Laybutt DR: Failure of the adaptive unfolded protein
response in islets of obese mice is linked with abnormalities in
β-cell gene expression and progression to diabetes. Diabetes.
62:1557–1568. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
39
|
Sykes EK, Mactier S and Christopherson R:
Melanoma and the unfolded protein response. Cancers (Basel).
8:E302016. View Article : Google Scholar
|
|
40
|
Luo S, Mao C, Lee B and Lee AS: GRP78/BiP
is required for cell proliferation and protecting the inner cell
mass from apoptosis during early mouse embryonic development. Mol
Cell Biol. 26:5688–5697. 2006. View Article : Google Scholar : PubMed/NCBI
|
|
41
|
Xiong Z, Jiang R, Li X, Liu Y and Guo F:
Different roles of GRP78 on cell proliferation and apoptosis in
cartilage development. Int J Mol Sci. 16:21153–21176. 2015.
View Article : Google Scholar : PubMed/NCBI
|
|
42
|
Ye R, Jung DY, Jun JY, Li J, Luo S, Ko HJ,
Kim JK and Lee AS: Grp78 heterozygosity promotes adaptive unfolded
protein response and attenuates diet-induced obesity and insulin
resistance. Diabetes. 59:6–16. 2010. View Article : Google Scholar
|
|
43
|
Fusakio ME, Willy JA, Wang Y, Mirek ET, Al
Baghdadi RJ, Adams CM, Anthony TG and Wek RC: Transcription factor
ATF4 directs basal and stress-induced gene expression in the
unfolded protein response and cholesterol metabolism in the liver.
Mol Biol Cell. 27:1536–1551. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
44
|
Aarti I, Rajesh K and Ramaiah KV:
Phosphorylation of eIF2 alpha in Sf9 cells: A stress, survival and
suicidal signal. Apoptosis. 15:679–692. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
45
|
Baird TD and Wek RC: Eukaryotic initiation
factor 2 phosphorylation and translational control in metabolism.
Adv Nutr. 3:307–321. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
46
|
Nagiah S, Phulukdaree A and Chuturgoon AA:
Lon protease and eiF2α are involved in acute, but not prolonged,
antiretroviral induced stress response in HepG2 cells. Chem Biol
Interact. 252:82–86. 2016. View Article : Google Scholar : PubMed/NCBI
|
|
47
|
Muaddi H, Majumder M, Peidis P, Papadakis
AI, Holcik M, Scheuner D, Kaufman RJ, Hatzoglou M and Koromilas AE:
Phosphorylation of eIF2α at serine 51 is an important determinant
of cell survival and adaptation to glucose deficiency. Mol Biol
Cell. 21:3220–3231. 2010. View Article : Google Scholar : PubMed/NCBI
|
|
48
|
Lee SK and Kim YS: Phosphorylation of
eIF2α attenuates statin-induced apoptosis by inhibiting the
stabilization and translocation of p53 to the mitochondria. Int J
Oncol. 42:810–816. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
49
|
Peidis P, Papadakis AI, Muaddi H, Richard
S and Koromilas AE: Doxorubicin bypasses the cytoprotective effects
of eIF2α phosphorylation and promotes PKR-mediated cell death. Cell
Death Differ. 18:145–154. 2011. View Article : Google Scholar
|
|
50
|
Yuan T, Luo BL, Wei TH, Zhang L, He BM and
Niu RC: Salubrinal protects against cigarette smoke extract-induced
HBEpC apoptosis likely via regulating the activity of PERK-eIF2α
signaling pathway. Arch Med Res. 43:522–529. 2012. View Article : Google Scholar : PubMed/NCBI
|
|
51
|
Wang H, Wang X, Ke ZJ, Comer AL, Xu M,
Frank JA, Zhang Z, Shi X and Luo J: Tunicamycin-induced unfolded
protein response in the developing mouse brain. Toxicol Appl
Pharmacol. 283:157–167. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
52
|
Zamarbide M, Martinez-Pinilla E,
Ricobaraza A, Aragón T, Franco R and Pérez-Mediavilla A: Phenyl
acyl acids attenuate the unfolded protein response in
tunicamycin-treated neuroblastoma cells. PLoS One. 8:e710822013.
View Article : Google Scholar : PubMed/NCBI
|
|
53
|
Fan L, Sun G, Ma T, Zhong F, Lei Y, Li X
and Wei W: Melatonin reverses tunicamycin-induced endoplasmic
reticulum stress in human hepatocellular carcinoma cells and
improves cytotoxic response to doxorubicin by increasing CHOP and
decreasing survivin. J Pineal Res. 55:184–194. 2013. View Article : Google Scholar : PubMed/NCBI
|
|
54
|
Quan X, Wang J, Liang C, Zheng H and Zhang
L: Melatonin inhibits tunicamycin-induced endoplasmic reticulum
stress and insulin resistance in skeletal muscle cells. Biochem
Biophys Res Commun. 463:1102–1107. 2015. View Article : Google Scholar : PubMed/NCBI
|
|
55
|
Liu SH, Yang CC, Chan DC, Wu CT, Chen LP,
Huang JW, Hung KY and Chiang CK: Chemical chaperon 4-phenylbutyrate
protects against the endoplasmic reticulum stress-mediated renal
fibrosis in vivo and in vitro. Oncotarget. 7:22116–22127.
2016.PubMed/NCBI
|
|
56
|
Vilatoba M, Eckstein C, Bilbao G, Smyth
CA, Jenkins S, Thompson JA, Eckhoff DE and Contreras JL: Sodium
4-phenylbutyrate protects against liver ischemia reperfusion injury
by inhibition of endoplasmic reticulum-stress mediated apoptosis.
Surgery. 138:342–351. 2005. View Article : Google Scholar : PubMed/NCBI
|
|
57
|
Yan S, Zhang H, Wang J, Zheng F and Dai J:
Perfluorooctanoic acid exposure induces endoplasmic reticulum
stress in the liver and its effects are ameliorated by
4-phenylbutyrate. Free Radic Biol Med. 87:300–311. 2015. View Article : Google Scholar : PubMed/NCBI
|